Analgesic Effect of Indian Gooseberry (Emblica officinalis Fruit) Extracts on Postoperative and Neuropathic Pain in Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of E. officinalis Extracts
2.2. Animals and Treatments
2.3. Plantar Incision of Postoperative Pain Rat Model
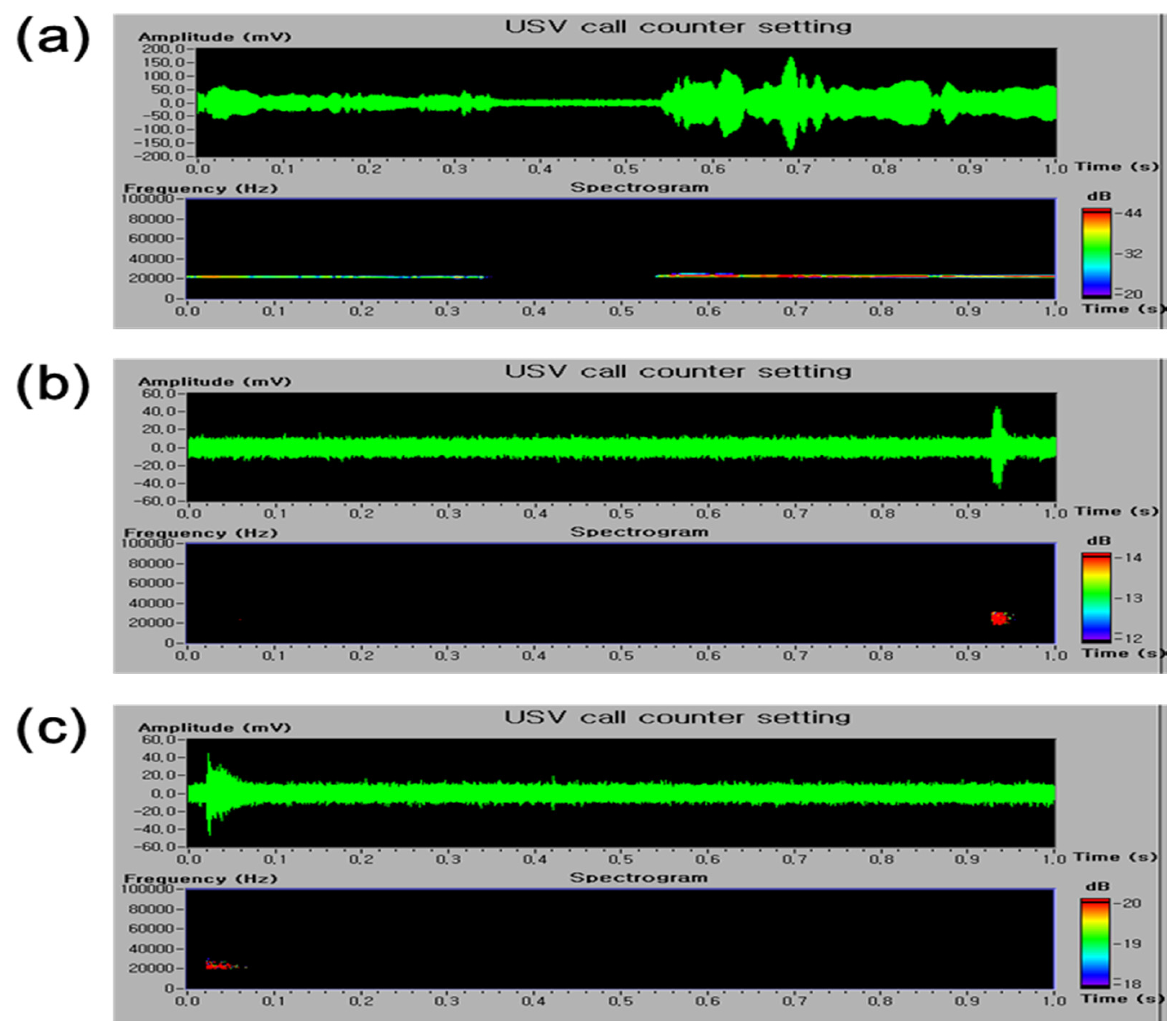
2.4. Ultrasonic Vocalization Analysis
2.5. Spared Nerve Injury of the Neuropathic Pain Rat Model
2.6. Mechanical Withdrawal Threshold Analysis
2.7. Cytokine Analysis
2.8. Statistical Analysis
3. Results
3.1. Effects of E. officinalis Extracts on Mechanical Hyperalgesia Induced by Plantar Incision
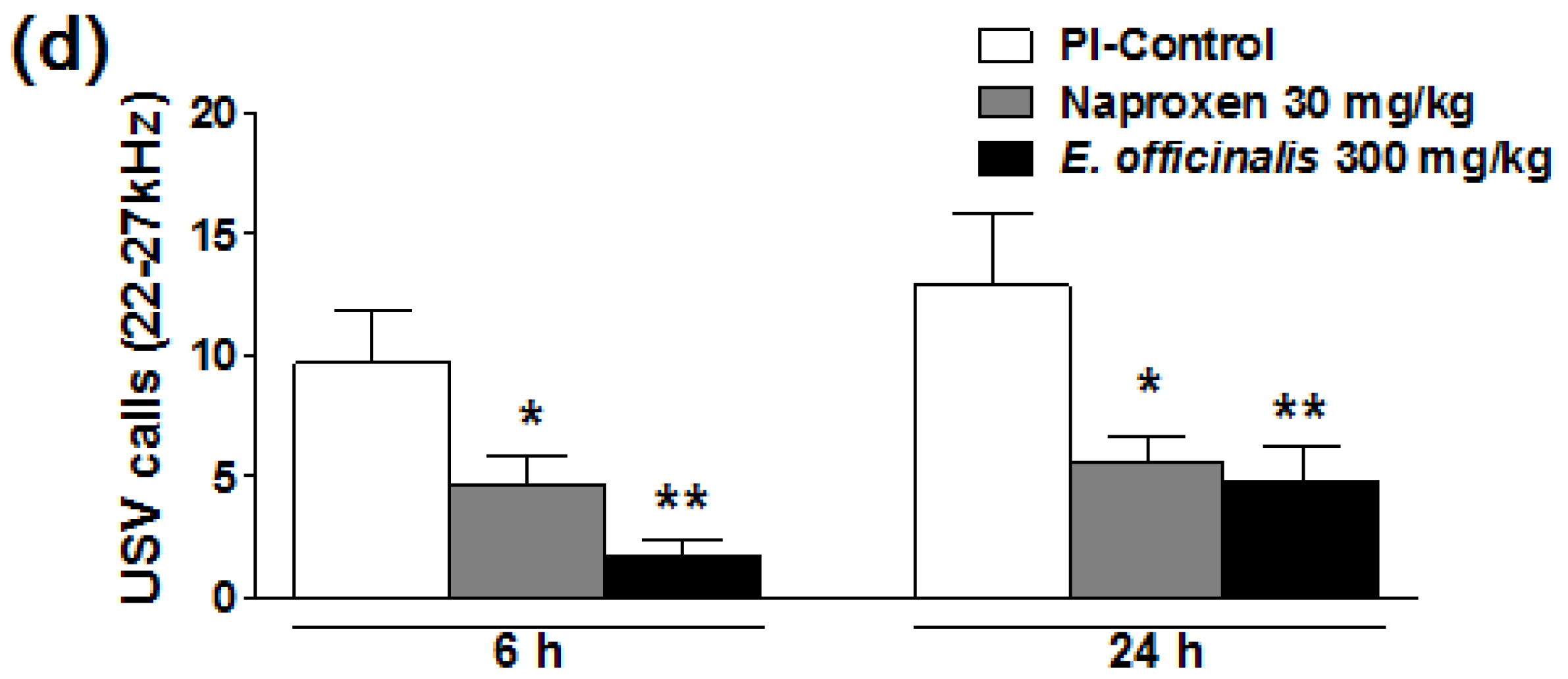
3.2. Effects of E. officinalis Extracts on USV Induced by Plantar Incision
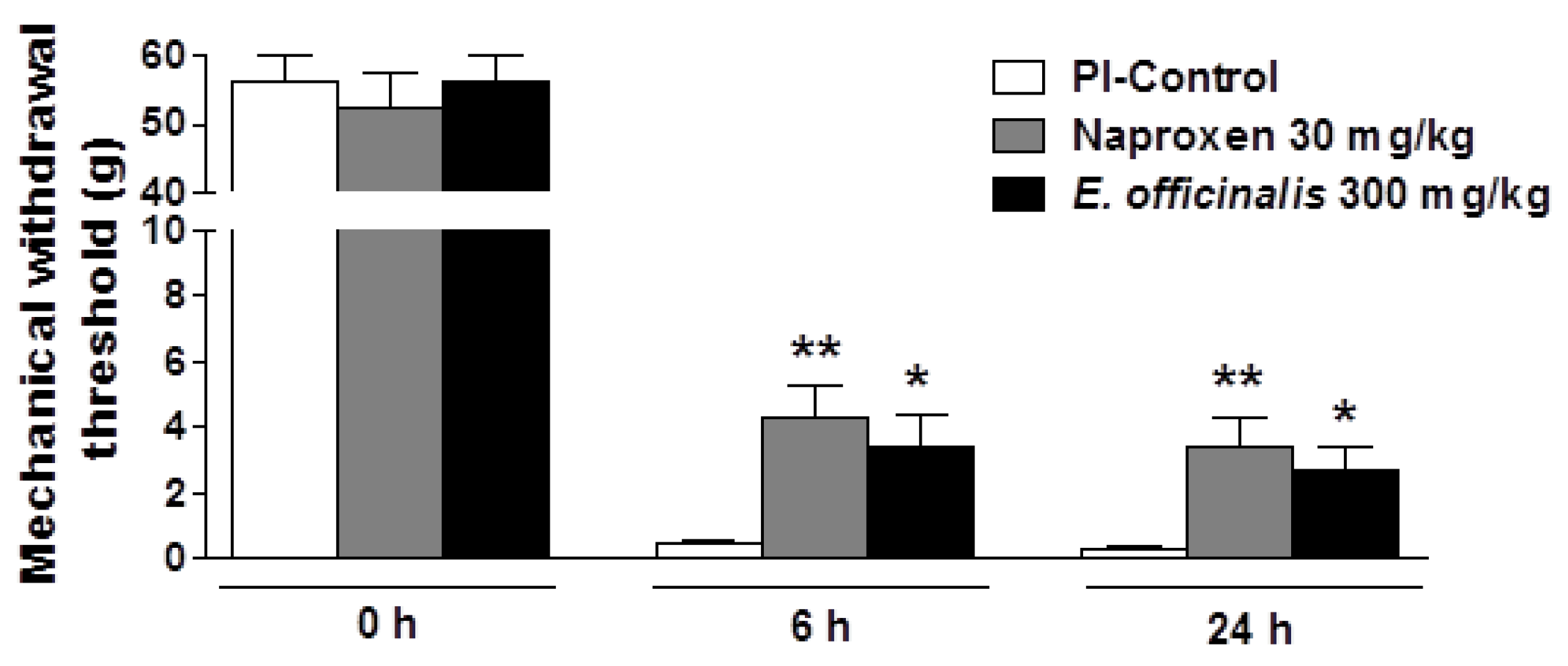
3.3. Effects of E. officinalis Extracts on Mechanical Hyperalgesia Induced by SNI
3.4. Effects of E. officinalis Extracts on the Expression of Cytokines Induced by SNI in Rat DRG
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gilron, I.; Coderre, T.J. Emerging drugs in neuropathic pain. Expert Opin. Emerg. Drugs 2007, 12, 113–126. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.; Knaus, E.E. Evolution of nonsteroidal anti-inflammatory drugs (nsaids): Cyclooxygenase (cox) inhibition and beyond. J. Pharm. Pharm. Sci. 2008, 11, 81s–110s. [Google Scholar] [CrossRef] [PubMed]
- Furlan, A.D.; Sandoval, J.A.; Mailis-Gagnon, A.; Tunks, E. Opioids for chronic noncancer pain: A meta-analysis of effectiveness and side effects. CMAJ 2006, 174, 1589–1594. [Google Scholar] [CrossRef] [PubMed]
- Brower, V. New paths to pain relief. Nat. Biotechnol. 2000, 18, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, K.A.; Manjavachi, M.N.; Paszcuk, A.F.; Pivatto, M.; Viegas, C., Jr.; Bolzani, V.S.; Calixto, J.B. Plant derived alkaloid (−)-cassine induces anti-inflammatory and anti-hyperalgesics effects in both acute and chronic inflammatory and neuropathic pain models. Neuropharmacology 2012, 62, 967–977. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Zhou, L.; Chen, Z.; Hu, C. Analgesic effect of iridoid glycosides from paederia scandens (lour.) merrill (rubiaceae) on spared nerve injury rat model of neuropathic pain. Pharmacol. Biochem. Behav. 2012, 102, 465–470. [Google Scholar] [CrossRef] [PubMed]
- Yimam, M.; Brownell, L.; Hodges, M.; Jia, Q. Analgesic effects of a standardized bioflavonoid composition from scutellaria baicalensis and acacia catechu. J. Diet. Suppl. 2012, 9, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.K.; Khurdiya, D.S. Vitamin C enrichment of fruit juice based ready-to-serve beverages through blending of indian gooseberry (Emblica officinalis gaertn.) juice. Plant Foods Hum. Nutr. 2004, 59, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Govindarajan, R.; Singh, D.P.; Rawat, A.K. High-performance liquid chromatographic method for the quantification of phenolics in ’chyavanprash’ a potent ayurvedic drug. J. Pharm. Biomed. Anal. 2007, 43, 527–532. [Google Scholar] [CrossRef] [PubMed]
- Poltanov, E.A.; Shikov, A.N.; Dorman, H.J.; Pozharitskaya, O.N.; Makarov, V.G.; Tikhonov, V.P.; Hiltunen, R. Chemical and antioxidant evaluation of indian gooseberry (Emblica officinalis gaertn., syn. Phyllanthus emblica L.) supplements. Phytother. Res. 2009, 23, 1309–1315. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.J.; Tanaka, T.; Iwamoto, Y.; Yang, C.R.; Kouno, I. Novel norsesquiterpenoids from the roots of Phyllanthus emblica. J. Nat. Prod. 2000, 63, 1507–1510. [Google Scholar] [CrossRef] [PubMed]
- Anila, L.; Vijayalakshmi, N.R. Flavonoids from Emblica officinalis and mangifera indica—Effectiveness for dyslipidemia. J. Ethnopharmacol. 2002, 79, 81–87. [Google Scholar] [CrossRef]
- Kim, H.J.; Yokozawa, T.; Kim, H.Y.; Tohda, C.; Rao, T.P.; Juneja, L.R. Influence of amla (Emblica officinalis gaertn.) on hypercholesterolemia and lipid peroxidation in cholesterol-fed rats. J. Nutr. Sci. Vitaminol. 2005, 51, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Sato, R.; Buesa, L.M.; Nerurkar, P.V. Anti-obesity effects of Emblica officinalis (amla) are associated with inhibition of nuclear transcription factor, peroxisome proliferator-activated receptor gamma (ppar gamma). FASEB J. 2010, 24, 661–664. [Google Scholar]
- Rao, T.P.; Sakaguchi, N.; Juneja, L.R.; Wada, E.; Yokozawa, T. Amla (Emblica officinalis gaertn.) extracts reduce oxidative stress in streptozotocin-induced diabetic rats. J. Med. Food 2005, 8, 362–368. [Google Scholar] [CrossRef] [PubMed]
- De, A.; De, A.; Papasian, C.; Hentges, S.; Banerjee, S.; Haque, I.; Banerjee, S.K. Emblica officinalis extract induces autophagy and inhibits human ovarian cancer cell proliferation, angiogenesis, growth of mouse xenograft tumors. PLoS ONE 2013, 8, e72748. [Google Scholar] [CrossRef] [PubMed]
- Jose, J.K.; Kuttan, R. Hepatoprotective activity of Emblica officinalis and chyavanaprash. J. Ethnopharmacol. 2000, 72, 135–140. [Google Scholar] [CrossRef]
- Bhattacharya, A.; Chatterjee, A.; Ghosal, S.; Bhattacharya, S.K. Antioxidant activity of active tannoid principles of Emblica officinalis (amla). Indian J. Exp. Biol. 1999, 37, 676–680. [Google Scholar] [PubMed]
- Antony, B.; Benny, M.; Kaimal, T.N. A pilot clinical study to evaluate the effect of Emblica officinalis extract (amlamax) on markers of systemic inflammation and dyslipidemia. Indian J. Clin. Biochem. 2008, 23, 378–381. [Google Scholar] [CrossRef] [PubMed]
- Perianayagam, J.B.; Sharma, S.K.; Joseph, A.; Christina, A.J. Evaluation of anti-pyretic and analgesic activity of Emblica officinalis gaertn. J. Ethnopharmacol. 2004, 95, 83–85. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.P.; Annamalai, A.R.; Thakur, R.S. Antinociceptive property of Emblica officinalis gaertn. (amla) in high fat diet-fed/low dose streptozotocin induced diabetic neuropathy in rats. Indian J. Exp. Biol. 2009, 47, 737–742. [Google Scholar] [PubMed]
- Brennan, T.J.; Vandermeulen, E.P.; Gebhart, G.F. Characterization of a rat model of incisional pain. Pain 1996, 64, 493–501. [Google Scholar] [CrossRef]
- Bourquin, A.F.; Suveges, M.; Pertin, M.; Gilliard, N.; Sardy, S.; Davison, A.C.; Spahn, D.R.; Decosterd, I. Assessment and analysis of mechanical allodynia-like behavior induced by spared nerve injury (SNI) in the mouse. Pain 2006, 122, 14.e11–14.e14. [Google Scholar] [CrossRef] [PubMed]
- Swiergiel, A.H.; Zhou, Y.; Dunn, A.J. Effects of chronic footshock, restraint and corticotropin-releasing factor on freezing, ultrasonic vocalization and forced swim behavior in rats. Behav. Brain Res. 2007, 183, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Lim, D.W.; Kim, J.G.; Han, D.; Kim, Y.T. Analgesic effect of harpagophytum procumbens on postoperative and neuropathic pain in rats. Molecules 2014, 19, 1060–1068. [Google Scholar] [CrossRef] [PubMed]
- Decosterd, I.; Woolf, C.J. Spared nerve injury: An animal model of persistent peripheral neuropathic pain. Pain 2000, 87, 149–158. [Google Scholar] [CrossRef]
- Whiteside, G.T.; Harrison, J.; Boulet, J.; Mark, L.; Pearson, M.; Gottshall, S.; Walker, K. Pharmacological characterisation of a rat model of incisional pain. Br. J. Pharm. 2004, 141, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.G.; Lim, D.W.; Cho, S.; Han, D.; Kim, Y.T. The edible brown seaweed ecklonia cava reduces hypersensitivity in postoperative and neuropathic pain models in rats. Molecules 2014, 19, 7669–7678. [Google Scholar] [CrossRef] [PubMed]
- Litvin, Y.; Blanchard, D.C.; Blanchard, R.J. Rat 22kHz ultrasonic vocalizations as alarm cries. Behav. Brain Res. 2007, 182, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Knutson, B.; Burgdorf, J.; Panksepp, J. Anticipation of play elicits high-frequency ultrasonic vocalizations in young rats. J. Comp. Psychol. 1998, 112, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Knutson, B.; Burgdorf, J.; Panksepp, J. Ultrasonic vocalizations as indices of affective states in rats. Psychol. Bull. 2002, 128, 961–977. [Google Scholar] [CrossRef] [PubMed]
- Brudzynski, S.M.; Chiu, E.M. Behavioural responses of laboratory rats to playback of 22 kHz ultrasonic calls. Physiol. Behav. 1995, 57, 1039–1044. [Google Scholar] [CrossRef]
- Miczek, K.A.; Weerts, E.M.; Vivian, J.A.; Barros, H.M. Aggression, anxiety and vocalizations in animals: Gabaa and 5-ht anxiolytics. Psychopharmacology 1995, 121, 38–56. [Google Scholar] [CrossRef] [PubMed]
- Portfors, C.V. Types and functions of ultrasonic vocalizations in laboratory rats and mice. J. Am. Assoc. Lab. Anim. Sci. 2007, 46, 28–34. [Google Scholar] [PubMed]
- Richner, M.; Bjerrum, O.J.; Nykjaer, A.; Vaegter, C.B. The spared nerve injury (SNI) model of induced mechanical allodynia in mice. J. Vis. Exp. 2011, 54, e3092. [Google Scholar] [CrossRef] [PubMed]
- Costigan, M.; Scholz, J.; Woolf, C.J. Neuropathic pain: A maladaptive response of the nervous system to damage. Annu. Rev. Neurosci. 2009, 32, 1–32. [Google Scholar] [CrossRef] [PubMed]
- Wieseler-Frank, J.; Maier, S.F.; Watkins, L.R. Central proinflammatory cytokines and pain enhancement. NeuroSignals 2005, 14, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Wagner, R.; Janjigian, M.; Myers, R.R. Anti-inflammatory interleukin-10 therapy in cci neuropathy decreases thermal hyperalgesia, macrophage recruitment, and endoneurial tnf-alpha expression. Pain 1998, 74, 35–42. [Google Scholar] [CrossRef]
- Padi, S.S.; Kulkarni, S.K. Differential effects of naproxen and rofecoxib on the development of hypersensitivity following nerve injury in rats. Pharmacol. Biochem. Behav. 2004, 79, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Rijsdijk, M.; van Wijck, A.J.M.; Kalkman, C.J.; Yaksh, T.L. The effects of glucocorticoids on neuropathic pain: A review with emphasis on intrathecal methylprednisolone acetate delivery. Anesth. Analg. 2014, 118, 1097–1112. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Xie, W.; Strong, J.A.; Zhang, J.M. Systemic antiinflammatory corticosteroid reduces mechanical pain behavior, sympathetic sprouting, and elevation of proinflammatory cytokines in a rat model of neuropathic pain. Anesthesiology 2007, 107, 469–477. [Google Scholar] [CrossRef] [PubMed]
- De Bosscher, K.; Vanden Berghe, W.; Haegeman, G. The interplay between the glucocorticoid receptor and nuclear factor-kappa b or activator protein-1: Molecular mechanisms for gene repression. Endocr. Rev. 2003, 24, 488–522. [Google Scholar] [CrossRef] [PubMed]
- Rhen, T.; Cidlowski, J.A. Antiinflammatory action of glucocorticoids—New mechanisms for old drugs. N. Engl. J. Med. 2005, 353, 1711–1723. [Google Scholar] [CrossRef] [PubMed]
- Gouwy, M.; Struyf, S.; Proost, P.; Van Damme, J. Synergy in cytokine and chemokine networks amplifies the inflammatory response. Cytokine Growth Factor Rev. 2005, 16, 561–580. [Google Scholar] [CrossRef] [PubMed]
- Baliga, M.S.; Dsouza, J.J. Amla (Emblica officinalis gaertn.), a wonder berry in the treatment and prevention of cancer. Eur. J. Cancer Prev. 2011, 20, 225–239. [Google Scholar] [CrossRef] [PubMed]
- Gaire, B.P.; Subedi, L. Phytochemistry, pharmacology and medicinal properties of Phyllanthus emblica linn. Chin. J. Integr. Med. 2014. [Google Scholar] [CrossRef] [PubMed]
- Dang, G.K.; Parekar, R.R.; Kamat, S.K.; Scindia, A.M.; Rege, N.N. Antiinflammatory activity of Phyllanthus emblica, Plumbago zeylanica and Cyperus rotundus in acute models of inflammation. Phytother. Res. 2011, 25, 904–908. [Google Scholar] [CrossRef] [PubMed]
- Muthuraman, A.; Sood, S.; Singla, S.K. The antiinflammatory potential of phenolic compounds from Emblica officinalis L. in rat. Inflammopharmacology 2011, 19, 327–334. [Google Scholar] [CrossRef] [PubMed]

| Cytokines (pg/mg Protein) | SNI-Sham | SNI-Control | Naproxen | E. officinalis |
|---|---|---|---|---|
| IL-1β | 1.64 ± 0.09 | 4.43 ± 0.34 a | 1.69 ± 0.28 b | 2.23 ± 0.27 b |
| IL-2 | 7.16 ± 0.34 | 12.55 ± 0.72 a | 7.15 ± 0.23 b | 7.20 ± 0.33 b |
| IL-6 | 48.21 ± 1.01 | 80.20 ± 3.57 a | 49.32 ± 2.66 b | 53.07 ± 3.03 b |
| IL-10 | 6.28 ± 0.77 | 3.59 ± 0.17 | 7.36 ± 0.55 c | 7.52 ± 1.15 c |
| IL-12 | 21.31 ± 1.11 | 40.56 ± 2.10 a | 22.05 ± 1.62 b | 22.31 ± 0.53 b |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lim, D.W.; Kim, J.G.; Kim, Y.T. Analgesic Effect of Indian Gooseberry (Emblica officinalis Fruit) Extracts on Postoperative and Neuropathic Pain in Rats. Nutrients 2016, 8, 760. https://doi.org/10.3390/nu8120760
Lim DW, Kim JG, Kim YT. Analgesic Effect of Indian Gooseberry (Emblica officinalis Fruit) Extracts on Postoperative and Neuropathic Pain in Rats. Nutrients. 2016; 8(12):760. https://doi.org/10.3390/nu8120760
Chicago/Turabian StyleLim, Dong Wook, Jae Goo Kim, and Yun Tai Kim. 2016. "Analgesic Effect of Indian Gooseberry (Emblica officinalis Fruit) Extracts on Postoperative and Neuropathic Pain in Rats" Nutrients 8, no. 12: 760. https://doi.org/10.3390/nu8120760





