Colostrum and Mature Human Milk of Women from London, Moscow, and Verona: Determinants of Immune Composition
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Setting, Eligibility Criteria, and Ethics
2.2. Medical Records and Interview
2.3. Human Milk Sampling
2.4. Electro-Chemiluminescence
2.5. Protein Analysis
2.6. Statistical Analysis
3. Results
3.1. Study Population
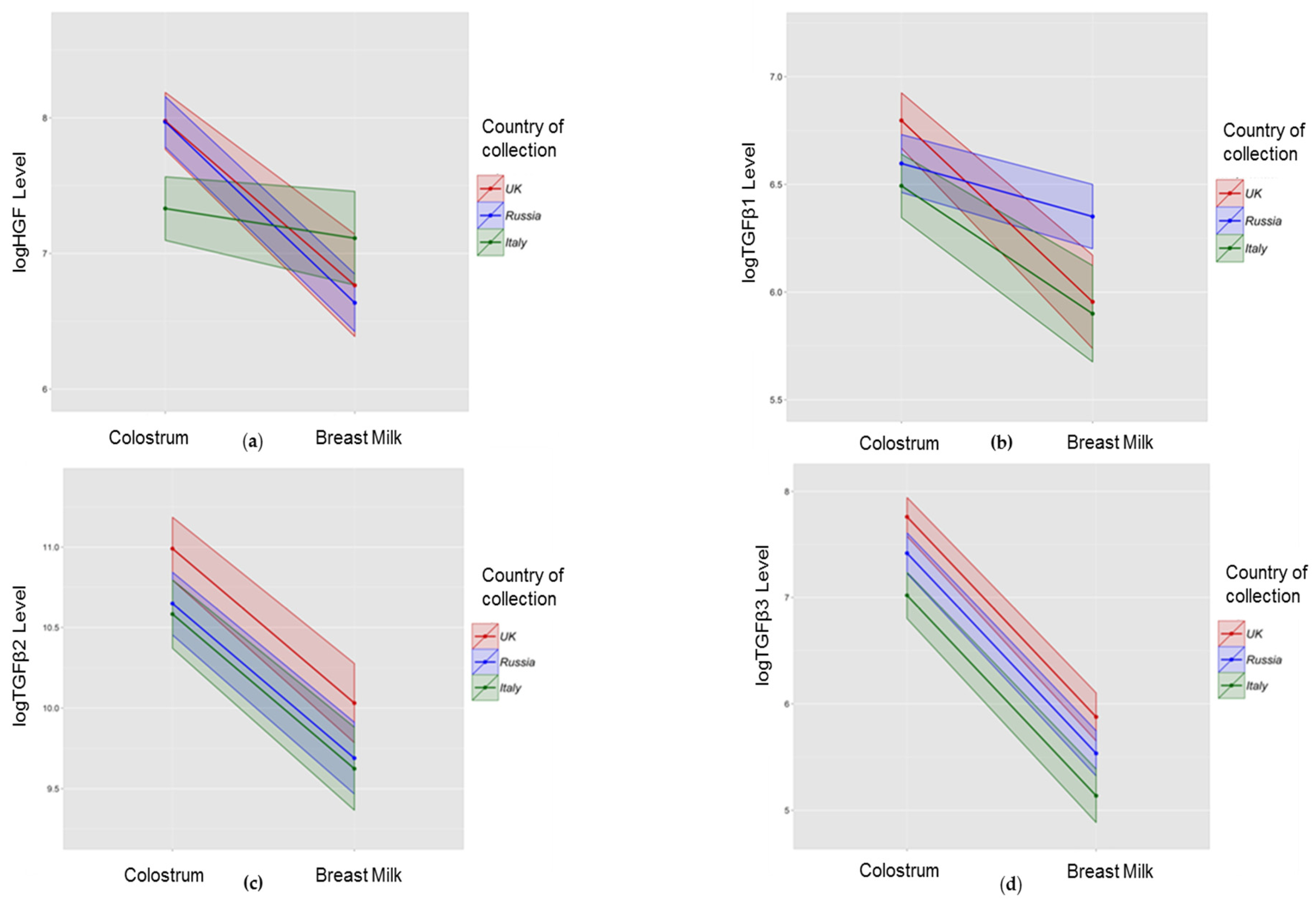
3.2. Association between Collection Time and Colostrum/Breast Milk Composition
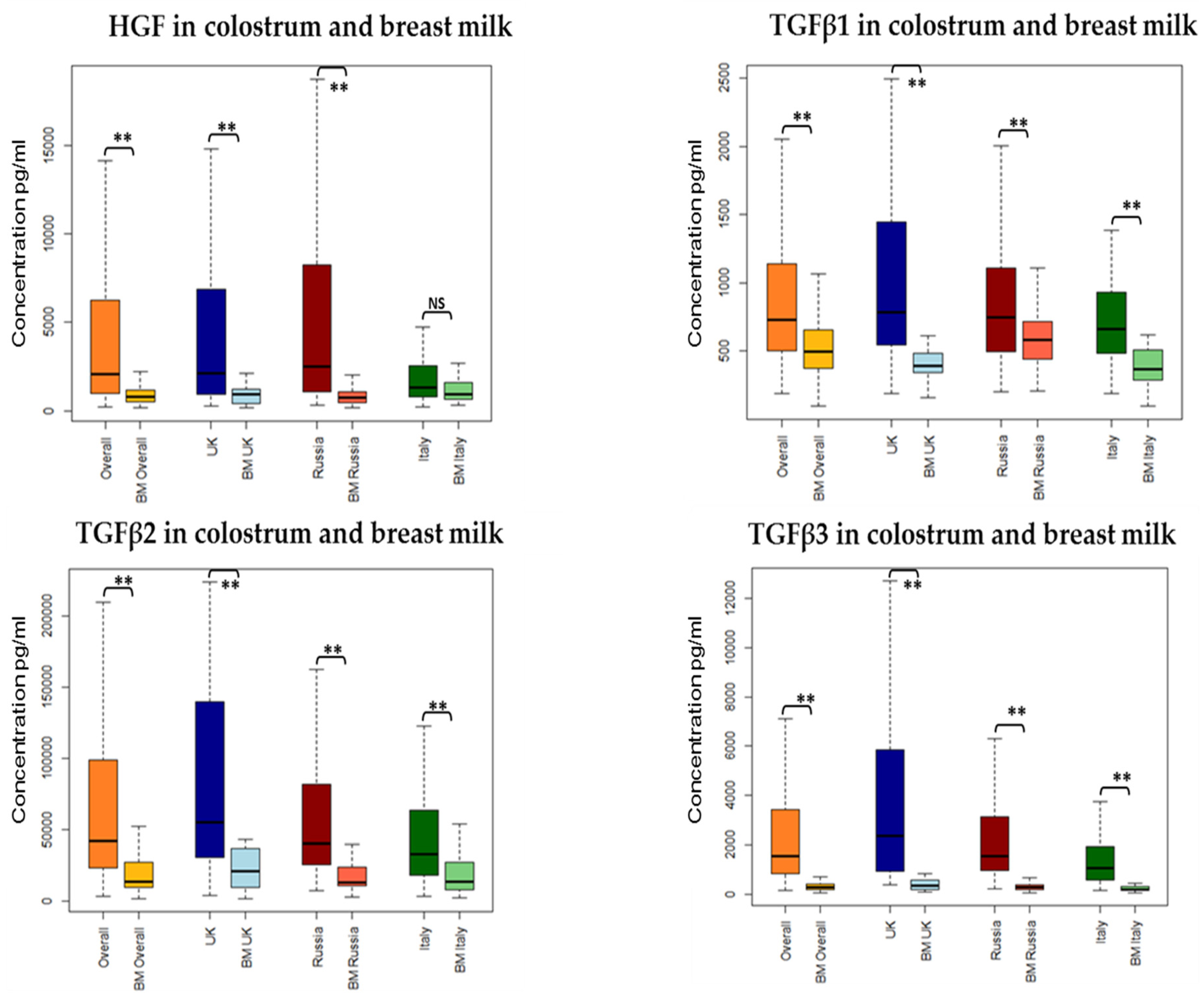
3.3. Association between Collection Site and Colostrum/Breast Milk Composition
3.3.1. Growth Factors
3.3.2. Cytokines
3.3.3. Protein and Sodium in Colostrum
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
| Immune Active Molecule | Median Lower Limit of Detection | Median (IQR) Colostrum | Median (IQR) HM |
|---|---|---|---|
| IFN-γ | 3.49 | 1.23 (0.51–3.49) | 3.49 (1.54–7.8) |
| IL2 | 2.06 | 0.6 (0.29–1.47) | 0.98 (0.23–2.06) |
| IL4 | 1.83 | 1.83 (0.24–1.83) | 1.83 (0.50–1.83) |
| IL5 | 2.89 | 1.44 (0.57–2.85) | 1.57 (0.33–2.89) |
| IL10 | 1.50 | 2.67 (1.01–9.04) | 1.5 (0.74–2.66) |
| IL12 | 3.50 | 1.85 (0.34–3.5) | 2.13 (0.32–3.5) |
| IL13 | 4.60 | 4.6 (1.85–4.69) | 4.6 (3.52–6.76) |
| HGF | 73.00 | 2055.31 (964–6239) | 784.041 (508–1189) |
| TGFβ1 | 8.73 | 731.534 (505–1142) | 493.514 (375–653) |
| TGFβ2 | 265.00 | 42,209.88 (23,847–98,597) | 14,040.62 (10,080–27,262) |
| TGFβ3 | 8.37 | 1535.081 (847–3395) | 279.41 (183–395) |
| Growth Factor Concentration (pg/mL)/Time of Collection | Raw Concentration (pg/mL) | Growth Factor/Protein Ratio |
|---|---|---|
| HGF | r = −0.39, p < 0.001 ↓ | r = −0.19, p = 0.003 ↓ |
| TGFβ1 | r = −0.21, p < 0.001 ↓ | r = 0.25, p < 0.001 ↑ |
| TGFβ2 | r = −0.16, p = 0.01 ↓ | r = 0.20, p = 0.004 ↑ |
| TGFβ3 | r = −0.35, p < 0.001 ↓ | r = −0.06, p = 0.34 ↓ |
References
- Matheson, M.C.; Allen, K.J.; Tang, M.L. Understanding the evidence for and against the role of breastfeeding in allergy prevention. Clin. Exp. Allergy 2012, 42, 827–851. [Google Scholar] [CrossRef] [PubMed]
- Hoppu, U.; Isolauri, E.; Laakso, P.; Matomaki, J.; Laitinen, K. Probiotics and dietary counselling targeting maternal dietary fat intake modifies breast milk fatty acids and cytokines. Eur. J. Nutr. 2012, 51, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Urwin, H.J.; Miles, E.A.; Noakes, P.S.; Kremmyda, L.S.; Vlachava, M.; Diaper, N.D.; Perez-Cano, F.J.; Godfrey, K.M.; Calder, P.C.; Yaqoob, P. Salmon consumption during pregnancy alters fatty acid composition and secretory IgA concentration in human breast milk. J. Nutr. 2012, 142, 1603–1610. [Google Scholar] [CrossRef] [PubMed]
- Amoudruz, P.; Holmlund, U.; Schollin, J.; Sverremark-Ekstrom, E.; Montgomery, S.M. Maternal country of birth and previous pregnancies are associated with breast milk characteristics. Pediatr. Allergy Immunol. 2009, 20, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Holmlund, U.; Amoudruz, P.; Johansson, M.A.; Haileselassie, Y.; Ongoiba, A.; Kayentao, K.; Traore, B.; Doumbo, S.; Schollin, J.; Doumbo, O.; et al. Maternal country of origin, breast milk characteristics and potential influences on immunity in offspring. Clin. Exp. Immunol. 2010, 162, 500–509. [Google Scholar] [CrossRef] [PubMed]
- Peroni, D.G.; Pescollderungg, L.; Piacentini, G.L.; Rigotti, E.; Maselli, M.; Watschinger, K.; Piazza, M.; Pigozzi, R.; Boner, A.L. Immune regulatory cytokines in the milk of lactating women from farming and urban environments. Pediatr. Allergy Immunol. 2010, 21, 977–982. [Google Scholar] [CrossRef] [PubMed]
- Tomicic, S.; Johansson, G.; Voor, T.; Bjorksten, B.; Bottcher, M.F.; Jenmalm, M.C. Breast milk cytokine and IgA composition differ in Estonian and Swedish mothers-relationship to microbial pressure and infant allergy. Pediatr. Res. 2010, 68, 330–334. [Google Scholar] [CrossRef] [PubMed]
- Striker, G.A.; Casanova, L.D.; Nagao, A.T. Influence of type of delivery on A, G and M immunoglobulin concentration in maternal colostrum. J. Pediatr. 2004, 80, 123–128. [Google Scholar] [CrossRef]
- Bottcher, M.F.; Jenmalm, M.C.; Garofalo, R.P.; Bjorksten, B. Cytokines in breast milk from allergic and nonallergic mothers. Pediatr. Res. 2000, 47, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Laiho, K.; Lampi, A.M.; Hamalainen, M.; Moilanen, E.; Piironen, V.; Arvola, T.; Syrjanen, S.; Isolauri, E. Breast milk fatty acids, eicosanoids, and cytokines in mothers with and without allergic disease. Pediatr. Res. 2003, 53, 642–647. [Google Scholar] [CrossRef] [PubMed]
- Prokesova, L.; Lodinova-Zadnikova, R.; Zizka, J.; Kocourkova, I.; Novotna, O.; Petraskova, P.; Sterzl, I. Cytokine levels in healthy and allergic mothers and their children during the first year of life. Pediatr. Allergy Immunol. 2006, 17, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Rigotti, E.; Piacentini, G.L.; Ress, M.; Pigozzi, R.; Boner, A.L.; Peroni, D.G. Transforming growth factor-beta and interleukin-10 in breast milk and development of atopic diseases in infants. Clin. Exp. Allergy 2006, 36, 614–618. [Google Scholar] [CrossRef] [PubMed]
- Marek, A.; Zagierski, M.; Liberek, A.; Aleksandrowicz, E.; Korzon, M.; Krzykowski, G.; Kaminska, B.; Szlagatys-Sidorkiewicz, A. TGF-beta(1), IL-10 and IL-4 in colostrum of allergic and nonallergic mothers. Acta Biochim. Pol. 2009, 56, 411–414. [Google Scholar] [PubMed]
- Sidor, K.; Jarmolowska, B.; Kaczmarski, M.; Kostyra, E.; Iwan, M.; Kostyra, H. Content of beta-casomorphins in milk of women with a history of allergy. Pediatr. Allergy Immunol. 2008, 19, 587–591. [Google Scholar] [CrossRef] [PubMed]
- Snijders, B.E.; Damoiseaux, J.G.; Penders, J.; Kummeling, I.; Stelma, F.F.; van Ree, R.; van den Brandt, P.A.; Thijs, C. Cytokines and soluble CD14 in breast milk in relation with atopic manifestations in mother and infant (KOALA study). Clin. Exp. Allergy 2006, 36, 1609–1615. [Google Scholar] [CrossRef] [PubMed]
- Bottcher, M.F.; Abrahamsson, T.R.; Fredriksson, M.; Jakobsson, T.; Bjorksten, B. Low breast milk tgf-beta2 is induced by lactobacillus reuteri supplementation and associates with reduced risk of sensitization during infancy. Pediatr. Allergy Immunol. 2008, 19, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Boyle, R.J.; Ismail, I.H.; Kivivuori, S.; Licciardi, P.V.; Robins-Browne, R.M.; Mah, L.J.; Axelrad, C.; Moore, S.; Donath, S.; Carlin, J.B.; et al. Lactobacillus GG treatment during pregnancy for the prevention of eczema: A randomized controlled trial. Allergy 2011, 66, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Prescott, S.L.; Wickens, K.; Westcott, L.; Jung, W.; Currie, H.; Black, P.N.; Stanley, T.V.; Mitchell, E.A.; Fitzharris, P.; Siebers, R.; et al. Supplementation with Lactobacillus Rhamnosus or Bifidobacterium Lactis probiotics in pregnancy increases cord blood interferon-gamma and breast milk transforming growth factor-beta and immunoglobin a detection. Clin. Exp. Allergy 2008, 38, 1606–1614. [Google Scholar] [CrossRef] [PubMed]
- Rautava, S.; Kalliomaki, M.; Isolauri, E. Probiotics during pregnancy and breast-feeding might confer immunomodulatory protection against atopic disease in the infant. J. Allergy Clin. Immunol. 2002, 109, 119–121. [Google Scholar] [CrossRef] [PubMed]
- Kuitunen, M.; Kukkonen, A.K.; Savilahti, E. Impact of maternal allergy and use of probiotics during pregnancy on breast milk cytokines and food antibodies and development of allergy in children until 5 years. Int. Arch. Allergy Immunol. 2012, 159, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Zanardo, V.; Nicolussi, S.; Cavallin, S.; Trevisanuto, D.; Barbato, A.; Faggian, D.; Favaro, F.; Plebani, M. Effect of maternal smoking on breast milk interleukin-1alpha, beta-endorphin, and leptin concentrations and leptin concentrations. Environ. Health Perspect. 2005, 113, 1410–1413. [Google Scholar] [CrossRef] [PubMed]
- Burch, J.; Karmaus, W.; Gangur, V.; Soto-Ramirez, N.; Yousefi, M.; Goetzl, L.M. Pre- and perinatal characteristics and breast milk immune markers. Pediatr. Res. 2013, 74, 615–621. [Google Scholar] [CrossRef] [PubMed]
- Min, J.K.; Lee, Y.M.; Kim, J.H.; Kim, Y.M.; Kim, S.W.; Lee, S.Y.; Gho, Y.S.; Oh, G.T.; Kwon, Y.G. Hepatocyte growth factor suppresses vascular endothelial growth factor-induced expression of endothelial ICAM-1 and VCAM-1 by inhibiting the nuclear factor-kappaB pathway. Circ. Res. 2005, 96, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Kobata, R.; Tsukahara, H.; Ohshima, Y.; Ohta, N.; Tokuriki, S.; Tamura, S.; Mayumi, M. High levels of growth factors in human breast milk. Early Hum. Dev. 2008, 84, 67–69. [Google Scholar] [CrossRef] [PubMed]
- Horibe, N.; Okamoto, T.; Itakura, A.; Nakanishi, T.; Suzuki, T.; Kazeto, S.; Tomoda, Y. Levels of hepatocyte growth factor in maternal serum and amniotic fluid. Am. J. Obstet. Gynecol. 1995, 173, 937–942. [Google Scholar] [CrossRef]
- Yamada, Y.; Saito, S.; Morikawa, H. Hepatocyte growth factor in human breast milk. Am. J. Reprod. Immunol. 1998, 40, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Strachan, D.P. Family size, infection and atopy: The first decade of the “hygiene hypothesis”. Thorax 2000, 55 (Suppl. 1), S2–S10. [Google Scholar] [CrossRef] [PubMed]
- Strachan, D.P. Hay fever, hygiene, and household size. BMJ 1989, 299, 1259–1260. [Google Scholar] [CrossRef] [PubMed]
- Munblit, D.; Boyle, R.J.; Warner, J.O. Factors affecting breast milk composition and potential consequences for development of the allergic phenotype. Clin. Exp. Allergy 2015, 45, 583–601. [Google Scholar] [CrossRef] [PubMed]
- Ramirez-Santana, C.; Perez-Cano, F.J.; Audi, C.; Castell, M.; Moretones, M.G.; Lopez-Sabater, M.C.; Castellote, C.; Franch, A. Effects of cooling and freezing storage on the stability of bioactive factors in human colostrum. J. Dairy Sci. 2012, 95, 2319–2325. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.A.; Holloway, J.A.; Popplewell, E.J.; Diaper, N.D.; Holloway, J.W.; Vance, G.H.; Warner, J.A.; Warner, J.O. Reduced soluble CD14 levels in amniotic fluid and breast milk are associated with the subsequent development of atopy, eczema, or both. J. Allergy Clin. Immunol. 2002, 109, 858–866. [Google Scholar] [CrossRef] [PubMed]
- Orivuori, L.; Loss, G.; Roduit, C.; Dalphin, J.C.; Depner, M.; Genuneit, J.; Lauener, R.; Pekkanen, J.; Pfefferle, P.; Riedler, J.; et al. Soluble immunoglobulin A in breast milk is inversely associated with atopic dermatitis at early age: The PASTURE cohort study. Clin. Exp. Allergy 2014, 44, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Savilahti, E.; Siltanen, M.; Kajosaari, M.; Vaarala, O.; Saarinen, K.M. IgA antibodies, TGF-beta1 and -beta2, and soluble CD14 in the colostrum and development of atopy by age 4. Pediatr. Res. 2005, 58, 1300–1305. [Google Scholar] [CrossRef] [PubMed]
- Kondo, N.; Suda, Y.; Nakao, A.; Oh-Oka, K.; Suzuki, K.; Ishimaru, K.; Sato, M.; Tanaka, T.; Nagai, A.; Yamagata, Z. Maternal psychosocial factors determining the concentrations of transforming growth factor-beta in breast milk. Pediatr. Allergy Immunol. 2011, 22, 853–861. [Google Scholar] [CrossRef] [PubMed]
- Ismail, I.H.; Licciardi, P.V.; Oppedisano, F.; Boyle, R.J.; Tang, M.L. Relationship between breast milk sCD14, Tgf-beta1 and total IgA in the first month and development of eczema during infancy. Pediatr. Allergy Immunol. 2013, 24, 352–360. [Google Scholar] [CrossRef] [PubMed]
- Groer, M.W.; Shelton, M.M. Exercise is associated with elevated proinflammatory cytokines in human milk. J. Obstet. Gynecol. Neonatal Nurs. 2009, 38, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Walter, J.; Kuhn, L.; Ghosh, M.K.; Kankasa, C.; Semrau, K.; Sinkala, M.; Mwiya, M.; Thea, D.M.; Aldrovandi, G.M. Low and undetectable breast milk interleukin-7 concentrations are associated with reduced risk of postnatal HIV transmission. J. Acquir. Immune Defic. Syndr. 2007, 46, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Hawkes, J.S.; Bryan, D.L.; Neumann, M.A.; Makrides, M.; Gibson, R.A. Transforming growth factor beta in human milk does not change in response to modest intakes of docosahexaenoic acid. Lipids 2001, 36, 1179–1181. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Kang, M.J.; Kwon, J.W.; Park, K.S.; Hong, S.J. Breastfeeding might have protective effects on atopy in children with the CD14C-159T CT/CC genotype. Allergy Asthma Immunol. Res. 2013, 5, 239–241. [Google Scholar] [CrossRef] [PubMed]
- Takahata, Y.; Takada, H.; Nomura, A.; Ohshima, K.; Nakayama, H.; Tsuda, T.; Nakano, H.; Hara, T. Interleukin-18 in human milk. Pediatr. Res. 2001, 50, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Ustundag, B.; Yilmaz, E.; Dogan, Y.; Akarsu, S.; Canatan, H.; Halifeoglu, I.; Cikim, G.; Aygun, A.D. Levels of cytokines (IL-1beta, IL-2, IL-6, IL-8, TNF-alpha) and trace elements (Zn, Cu) in breast milk from mothers of preterm and term infants. Mediat. Inflamm. 2005, 2005, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Soto-Ramirez, N.; Karmaus, W.; Yousefi, M.; Zhang, H.; Liu, J.; Gangur, V. Maternal immune markers in serum during gestation and in breast milk and the risk of asthma-like symptoms at ages 6 and 12 months: A longitudinal study. Allergy Asthma Clin. Immunol. 2012, 8, 11. [Google Scholar] [CrossRef] [PubMed]
- Laverty, H.G.; Wakefield, L.M.; Occleston, N.L.; O’Kane, S.; Ferguson, M.W. TGF-β3 and cancer: A review. Cytokine Growth Factor Rev. 2009, 20, 305–317. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, A.V.; Pollard, J.W. Transforming growth factor beta3 induces cell death during the first stage of mammary gland involution. Development 2000, 127, 3107–3118. [Google Scholar] [PubMed]
- Ogawa, J.; Sasahara, A.; Yoshida, T.; Sira, M.M.; Futatani, T.; Kanegane, H.; Miyawaki, T. Role of transforming growth factor-beta in breast milk for initiation of IgA production in newborn infants. Early Hum. Dev. 2004, 77, 67–75. [Google Scholar] [CrossRef] [PubMed]

| UK | Russia | Italy | p-Value (Three Countries) | |
|---|---|---|---|---|
| Maternal allergic sensitisation * | 35/94 (37) | 22/156 (14) | 9/40 (23) | <0.01 a |
| Maternal Age (years) | 32.8 (4.78) | 29.8 (4.45) | 37.4 (5.38) | <0.01 b |
| Vaginal Delivery | 70/101 (69) | 188/219 (86) | 62/76 (82) | <0.01 a |
| Male sex | 54/101 (53) | 118/216 (55) | 41/76 (54) | 0.98 a |
| Birth Weight (grams) | 3527 (535.37) | 3526 (438.97) | 3328 (476.95) | 0.05 b |
| Primiparous women | 55/100 (55) | 93/216 (43) | 29/75 (39) | 0.06 a |
| Household tobacco smoke exposure | 30/99 (30) | 135/218 (62) | 25/76 (33) | <0.01 a |
| Antenatal Infections † | 16/100 (16) | 61/211 (29) | 29/76 (38) | <0.01 a |
| Time of colostrum collection (hours) | 58.61 (33.2) | 50.03 (14.34) | 57.84 (26.52) | <0.01 b |
| Immune Modulator | Colostrum Composition | Paired Differences between HM and Colostrum of Colostrum and Breast Milk Composition | ||||
|---|---|---|---|---|---|---|
| Change over Time | β | p-Value | Difference between Colostrum and HM over Time | β | p-Value | |
| HGF | Lower | −0.01 | <0.001 ** | Higher in colostrum | −1.35 | <0.001 ** |
| TGFβ1 | Lower | −0.003 | 0.01 * | Higher in colostrum | −0.93 | <0.001 ** |
| TGFβ2 | No change | −0.003 | 0.12 | Higher in colostrum | −1.12 | <0.001 ** |
| TGFβ3 | Lower | −0.01 | <0.001 ** | Higher in colostrum | −2.03 | <0.001 ** |
| IL2 | Lower | −0.02 | 0.02 * | No difference | 0.32 | 0.30 |
| IL4 | No change | −0.01 | 0.22 | Higher in HM | 0.72 | 0.04 * |
| IL5 | Lower | −0.03 | <0.001 ** | No difference | −0.54 | 0.09 |
| IL10 | Lower | −0.02 | <0.001 ** | Higher in colostrum | −1.66 | <0.001 ** |
| IFNγ | Lower | −0.01 | 0.04 * | Higher in HM | 1.20 | <0.001 ** |
| IL12 | No change | −0.01 | 0.12 | No difference | −0.11 | 0.73 |
| IL13 | No change | −0.01 | 0.09 | No difference | 0.09 | 0.72 |
| Median (IQR) pg/mL | Important Growth Factor Level Difference between the Groups | ||
|---|---|---|---|
| Colostrum | |||
| HGF | 2055.31 (964–6239) | UK and Russia higher than in Italy | p < 0.001 |
| Primipara higher than Multipara | p = 0.05 | ||
| TGFβ1 | 731.534 (505–1142) | UK higher than Italy | p = 0.01 |
| Fish consumption Less than once a week higher than At least once a week | p = 0.04 | ||
| TGFβ2 | 42,209.88 (23,847–98,597) | UK higher than Russia and Italy | p < 0.05 |
| TGFβ3 | 1535.081 (847–3395) | UK higher than Russia higher than Italy | p < 0.05 |
| Breast Milk | |||
| HGF | 784.041 (508–1189) | Primiparous higher than Multigravida | p = 0.05 |
| TGFβ1 | 493.514 (375–653) | Russia higher than UK and Italy | p < 0.05 |
| TGFβ2 | 14,040.62 (10,080–27,262) | UK higher than Russia and Italy | p < 0.05 |
| TGFβ3 | 279.41 (183–395) | UK higher than Russia higher than Italy | p < 0.05 |
| Colostrum Detectable | Human Milk Detectable | Factors Associated with Cytokines Detectability | |
|---|---|---|---|
| IL2 | 49/342 (14%) | 38/190 (20%) | NA |
| IL4 | 35/342 (10%) | 30/190 (16%) | NA |
| IL5 | 77/342 (23%) | 27/190 (14%) | Antenatal infections OR 0.49 (95% CI 0.25–0.98) |
| IL10 | 225/342 (66%) | 69/190 (36%) | NA |
| IFNγ | 66/342 (19%) | 92/190 (48%) | NA |
| IL12 | 63/342 (18%) | 31/190 (16%) | NA |
| IL13 | 86/342 (25%) | 58/190 (31%) | NA |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Munblit, D.; Treneva, M.; Peroni, D.G.; Colicino, S.; Chow, L.; Dissanayeke, S.; Abrol, P.; Sheth, S.; Pampura, A.; Boner, A.L.; et al. Colostrum and Mature Human Milk of Women from London, Moscow, and Verona: Determinants of Immune Composition. Nutrients 2016, 8, 695. https://doi.org/10.3390/nu8110695
Munblit D, Treneva M, Peroni DG, Colicino S, Chow L, Dissanayeke S, Abrol P, Sheth S, Pampura A, Boner AL, et al. Colostrum and Mature Human Milk of Women from London, Moscow, and Verona: Determinants of Immune Composition. Nutrients. 2016; 8(11):695. https://doi.org/10.3390/nu8110695
Chicago/Turabian StyleMunblit, Daniel, Marina Treneva, Diego G. Peroni, Silvia Colicino, LiYan Chow, Shobana Dissanayeke, Priya Abrol, Shreya Sheth, Alexander Pampura, Attilio L. Boner, and et al. 2016. "Colostrum and Mature Human Milk of Women from London, Moscow, and Verona: Determinants of Immune Composition" Nutrients 8, no. 11: 695. https://doi.org/10.3390/nu8110695
APA StyleMunblit, D., Treneva, M., Peroni, D. G., Colicino, S., Chow, L., Dissanayeke, S., Abrol, P., Sheth, S., Pampura, A., Boner, A. L., Geddes, D. T., Boyle, R. J., & Warner, J. O. (2016). Colostrum and Mature Human Milk of Women from London, Moscow, and Verona: Determinants of Immune Composition. Nutrients, 8(11), 695. https://doi.org/10.3390/nu8110695






