The Association between Dietary Vitamin A and Carotenes and the Risk of Primary Liver Cancer: A Case–Control Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Data Collection
2.3. Dietary Assessment
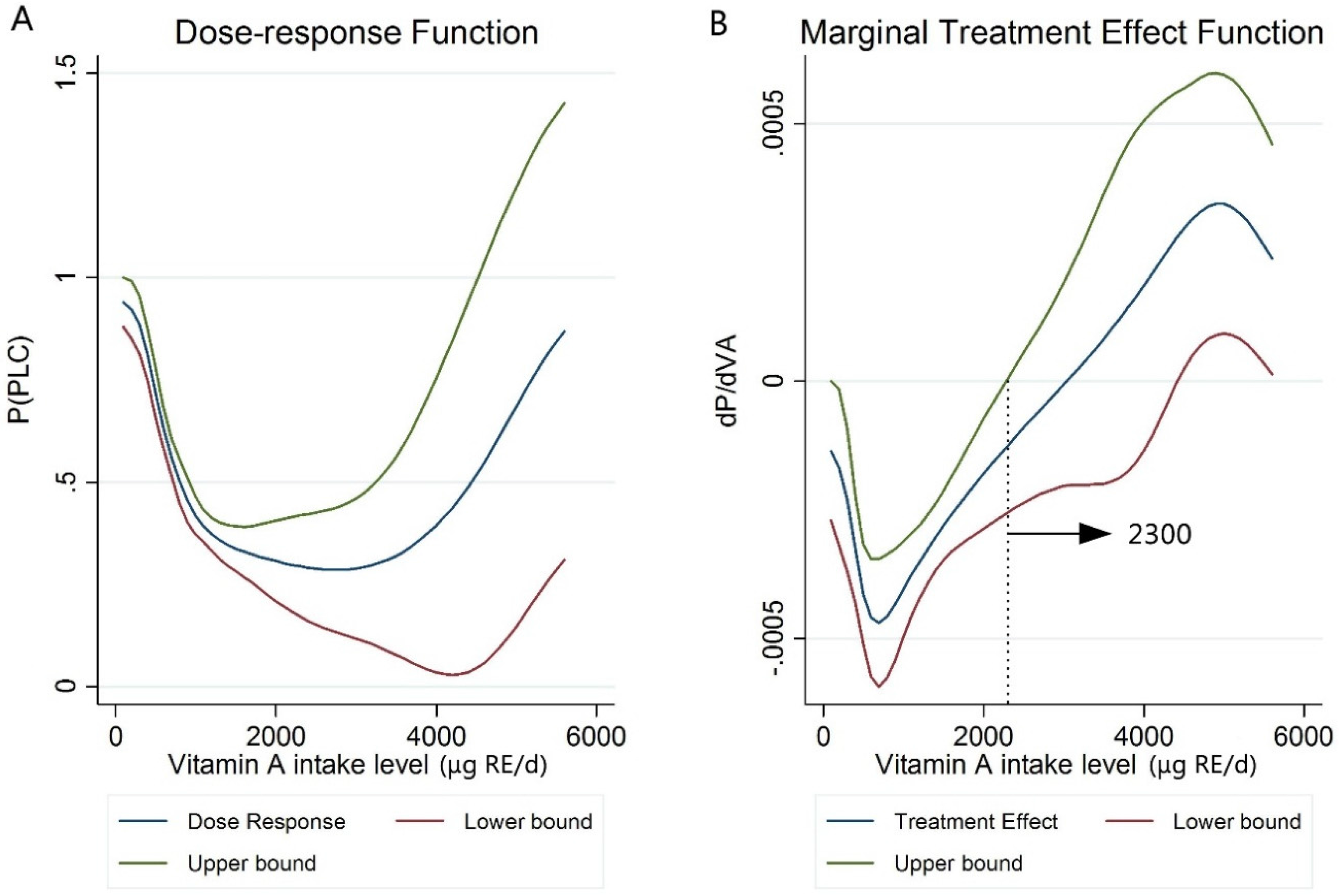
2.4. Generalized Propensity Score
2.5. Statistical Analysis
3. Results
3.1. Participant Characteristics
3.2. Dietary Intakes
3.3. Associations between Dietary Intakes of Vitamin A, Carotenes, and Retinol and PLC Risk
3.4. Stratified Analyses
3.5. Generalized Propensity Score (GPS)
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| PLC | primary liver cancer |
| HBV | hepatitis B virus |
| HCV | hepatitis C virus |
| VA | Vitamin A |
| HCC | hepatocellular carcinoma |
| ATBC | the Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study |
| CARET | Beta-Carotene and Retinol Efficacy Trial |
| GPS | generalized propensity score |
| NCCN | National Comprehensive Cancer Network |
| FFQ | food frequency questionnaire |
| OR | odds ratios |
| RE | retinol equivalent |
| SELECT | the Selenium and Vitamin E Cancer Prevention Trial |
| ROS | reactive oxygen species |
| RNS | reactive nitrogen species |
| 8-OhdG | 8-hydroxydeoxyguanosine |
| HSC | hepatic stellate cells |
| MET | metabolic equivalent |
| WHR | waist-to-hip ratio retinol |
| BMI | Body mass index |
References
- Teo, E.K.; Fock, K.M. Hepatocellular carcinoma: An Asian perspective. Dig. Dis. 2001, 19, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Bamia, C.; Lagiou, P.; Jenab, M.; Aleksandrova, K.; Fedirko, V.; Trichopoulos, D.; Overvad, K.; Tjonneland, A.; Olsen, A.; Clavel-Chapelon, F.; et al. Fruit and vegetable consumption in relation to hepatocellular carcinoma in a multi-centre, European cohort study. Br. J. Cancer 2015, 112, 1273–1282. [Google Scholar] [CrossRef] [PubMed]
- Duarte-Salles, T.; Fedirko, V.; Stepien, M.; Trichopoulou, A.; Bamia, C.; Lagiou, P.; Lukanova, A.; Trepo, E.; Overvad, K.; Tjonneland, A.; et al. Dairy products and risk of hepatocellular carcinoma: The European prospective investigation into cancer and nutrition. Int. J. Cancer 2014, 135, 1662–1672. [Google Scholar] [CrossRef] [PubMed]
- Fedirko, V.; Trichopolou, A.; Bamia, C.; Duarte-Salles, T.; Trepo, E.; Aleksandrova, K.; Nothlings, U.; Lukanova, A.; Lagiou, P.; Boffetta, P.; et al. Consumption of fish and meats and risk of hepatocellular carcinoma: The European prospective investigation into cancer and nutrition (epic). Ann. Oncol. 2013, 24, 2166–2173. [Google Scholar] [CrossRef] [PubMed]
- Chaves, G.V.; Peres, W.A.; Goncalves, J.C.; Ramalho, A. Vitamin a and retinol-binding protein deficiency among chronic liver disease patients. Nutrition 2015, 31, 664–668. [Google Scholar] [CrossRef] [PubMed]
- Cooke, M.S.; Evans, M.D.; Dizdaroglu, M.; Lunec, J. Oxidative DNA damage: Mechanisms, mutation, and disease. FASEB J. 2003, 17, 1195–1214. [Google Scholar] [CrossRef] [PubMed]
- Ramirez-Tortosa, C.; Andersen, O.M.; Cabrita, L.; Gardner, P.T.; Morrice, P.C.; Wood, S.G.; Duthie, S.J.; Collins, A.R.; Duthie, G.G. Anthocyanin-rich extract decreases indices of lipid peroxidation and DNA damage in vitamin e-depleted rats. Free Radic Biol. Med. 2001, 31, 1033–1037. [Google Scholar] [CrossRef]
- Yuan, J.M.; Gao, Y.T.; Ong, C.N.; Ross, R.K.; Yu, M.C. Prediagnostic level of serum retinol in relation to reduced risk of hepatocellular carcinoma. J. Natl. Cancer Inst. 2006, 98, 482–490. [Google Scholar] [CrossRef] [PubMed]
- Lai, G.Y.; Weinstein, S.J.; Albanes, D.; Taylor, P.R.; Virtamo, J.; McGlynn, K.A.; Freedman, N.D. Association of serum alpha-tocopherol, beta-carotene, and retinol with liver cancer incidence and chronic liver disease mortality. Br. J. Cancer 2014, 111, 2163–2171. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.H.; Wang, C.Y.; Huang, S.M.; Yeh, S.Y.; Lin, W.G.; Lin, D.I.; Liaw, Y.F. Vitamin A, vitamin E or beta-carotene status and hepatitis B-related hepatocellular carcinoma. Ann. Epidemiol. 1993, 3, 217–224. [Google Scholar] [CrossRef]
- Clemente, C.; Elba, S.; Buongiorno, G.; Berloco, P.; Guerra, V.; Di Leo, A. Serum retinol and risk of hepatocellular carcinoma in patients with child-pugh class a cirrhosis. Cancer Lett. 2002, 178, 123–129. [Google Scholar] [CrossRef]
- Yu, M.W.; Hsieh, H.H.; Pan, W.H.; Yang, C.S.; Chen, C.J. Vegetable consumption, serum retinol level, and risk of hepatocellular carcinoma. Cancer Res. 1995, 55, 1301–1305. [Google Scholar] [PubMed]
- Yamamoto, Y.; Yamashita, S.; Fujisawa, A.; Kokura, S.; Yoshikawa, T. Oxidative stress in patients with hepatitis, cirrhosis, and hepatoma evaluated by plasma antioxidants. Biochem. Biophys. Res. Commun. 1998, 247, 166–170. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Shu, X.O.; Li, H.; Yang, G.; Cai, H.; Ji, B.T.; Gao, J.; Gao, Y.T.; Zheng, W.; Xiang, Y.B. Vitamin intake and liver cancer risk: A report from two cohort studies in China. J. Natl. Cancer Inst. 2012, 104, 1173–1181. [Google Scholar] [CrossRef] [PubMed]
- Lai, G.Y.; Weinstein, S.J.; Taylor, P.R.; McGlynn, K.A.; Virtamo, J.; Gail, M.H.; Albanes, D.; Freedman, N.D. Effects of alpha-tocopherol and beta-carotene supplementation on liver cancer incidence and chronic liver disease mortality in the ATBC study. Br. J. Cancer 2014, 111, 2220–2223. [Google Scholar] [CrossRef] [PubMed]
- Heinonen, O.P.; Albanes, D. The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers. N. Engl. J. Med. 1994, 330, 1029–1035. [Google Scholar]
- Omenn, G.S.; Goodman, G.E.; Thornquist, M.D.; Balmes, J.; Cullen, M.R.; Glass, A.; Keogh, J.P.; Meyskens, F.L.; Valanis, B.; Williams, J.H.; et al. Effects of a combination of beta carotene and vitamin A on lung cancer and cardiovascular disease. N. Engl. J. Med. 1996, 334, 1150–1155. [Google Scholar] [CrossRef] [PubMed]
- Klein, E.A.; Thompson, I.M., Jr.; Tangen, C.M.; Crowley, J.J.; Lucia, M.S.; Goodman, P.J.; Minasian, L.M.; Ford, L.G.; Parnes, H.L.; Gaziano, J.M.; et al. Vitamin E and the risk of prostate cancer: The selenium and vitamin E cancer prevention trial(SELECT). JAMA 2011, 306, 1549–1556. [Google Scholar] [CrossRef] [PubMed]
- Le Gal, K.; Ibrahim, M.X.; Wiel, C.; Sayin, V.I.; Akula, M.K.; Karlsson, C.; Dalin, M.G.; Akyurek, L.M.; Lindahl, P.; Nilsson, J.; et al. Antioxidants can increase melanoma metastasis in mice. Sci. Transl. Med. 2015, 7, 308. [Google Scholar] [CrossRef] [PubMed]
- Sayin, V.I.; Ibrahim, M.X.; Larsson, E.; Nilsson, J.A.; Lindahl, P.; Bergo, M.O. Antioxidants accelerate lung cancer progression in mice. Sci. Transl. Med. 2014, 6, 221ra15. [Google Scholar] [CrossRef] [PubMed]
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Hepatobiliary Cancers. National Comprehensive Cancer Network, 2009. Available online: http://www.nccn.org (accessed on 25 September 2009).
- Yang, Y.X.; Wang, G.Y.; Pan, X.C. China Food Composition; Peking University Medical Press: Beijing, China, 2004. [Google Scholar]
- Zhang, C.X.; Ho, S.C. Validity and reproducibility of a food frequency questionnaire among Chinese women in Guangdong province. Asia Pac. J. Clin. Nutr. 2009, 18, 240–250. [Google Scholar] [PubMed]
- Kreif, N.; Grieve, R.; Diaz, I.; Harrison, D. Evaluation of the effect of a continuous treatment: A machine learning approach with an application to treatment for traumatic brain injury. Health Econ. 2015, 24, 1213–1228. [Google Scholar] [CrossRef] [PubMed]
- Austin, P.C. The performance of different propensity-score methods for estimating differences in proportions (risk differences or absolute risk reductions) in observational studies. Stat. Med. 2010, 29, 2137–2148. [Google Scholar] [CrossRef] [PubMed]
- Willett, W.; Stampfer, M.J. Total energy intake: Implications for epidemiologic analyses. Am. J. Epidemiol. 1986, 124, 17–27. [Google Scholar] [PubMed]
- Michaud, D.S.; Feskanich, D.; Rimm, E.B.; Colditz, G.A.; Speizer, F.E.; Willett, W.C.; Giovannucci, E. Intake of specific carotenoids and risk of lung cancer in 2 prospective us cohorts. Am. J. Clin. Nutr. 2000, 72, 990–997. [Google Scholar] [PubMed]
- Tang, G. Bioconversion of dietary provitamin A carotenoids to vitamin A in humans. Am. J. Clin. Nutr. 2010, 91, 1468S–1473S. [Google Scholar] [CrossRef] [PubMed]
- Morre, D.M.; Kloppel, T.M.; Rosenthal, A.L.; Fink, P.C. Chemoprevention of tumor development and metastasis of transplantable hepatocellular carcinomas in rats by vitamin A. J. Nutr. 1980, 110, 1629–1634. [Google Scholar] [PubMed]
- Tanvetyanon, T.; Bepler, G. Beta-carotene in multivitamins and the possible risk of lung cancer among smokers versus former smokers: A meta-analysis and evaluation of national brands. Cancer 2008, 113, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Wiseman, H.; Halliwell, B. Damage to DNA by reactive oxygen and nitrogen species: Role in inflammatory disease and progression to cancer. Biochem. J. 1996, 313, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Altekruse, S.F.; McGlynn, K.A.; Dickie, L.A.; Kleiner, D.E. Hepatocellular carcinoma confirmation, treatment, and survival in surveillance, epidemiology, and end results registries, 1992–2008. Hepatology 2012, 55, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Rocchi, E.; Casalgrandi, G.; Ronzoni, A.; Rosa, M.C.; Cioni, G.; Marazzi, A.; Manenti, A.; Marchini, S.; Ventura, E. Antioxidant liposoluble vitamins and carotenoids in chronic hepatitis. Eur. J. Intern. Med. 2001, 12, 116–121. [Google Scholar] [CrossRef]
- Vidali, M.; Tripodi, M.F.; Ivaldi, A.; Zampino, R.; Occhino, G.; Restivo, L.; Sutti, S.; Marrone, A.; Ruggiero, G.; Albano, E.; et al. Interplay between oxidative stress and hepatic steatosis in the progression of chronic hepatitis c. J. Hepatol. 2008, 48, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Hagen, T.M.; Huang, S.; Curnutte, J.; Fowler, P.; Martinez, V.; Wehr, C.M.; Ames, B.N.; Chisari, F.V. Extensive oxidative DNA damage in hepatocytes of transgenic mice with chronic active hepatitis destined to develop hepatocellular carcinoma. Proc. Natl. Acad. Sci. USA 1994, 91, 12808–12812. [Google Scholar] [CrossRef] [PubMed]
- Shimoda, R.; Nagashima, M.; Sakamoto, M.; Yamaguchi, N.; Hirohashi, S.; Yokota, J.; Kasai, H. Increased formation of oxidative DNA damage, 8-hydroxydeoxyguanosine, in human livers with chronic hepatitis. Cancer Res. 1994, 54, 3171–3172. [Google Scholar] [PubMed]
- Lee, U.E.; Friedman, S.L. Mechanisms of hepatic fibrogenesis. Best Pract. Res. Clin. Gastroenterol. 2011, 25, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Faouzi, S.; Lepreux, S.; Bedin, C.; Dubuisson, L.; Balabaud, C.; Bioulac-Sage, P.; Desmouliere, A.; Rosenbaum, J. Activation of cultured rat hepatic stellate cells by tumoral hepatocytes. Lab. Investig. 1999, 79, 485–493. [Google Scholar] [PubMed]
- Arantes Ferreira Peres, W.; Villaca Chaves, G.; Saraiva Goncalves, J.C.; Ramalho, A.; Moraes Coelho, H.S. Assessment of the relative dose–response test as indicators of hepatic vitamin A stores in various stages of chronic liver disease. Nutr. Clin. Pract. 2013, 28, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Newsome, P.N.; Beldon, I.; Moussa, Y.; Delahooke, T.E.; Poulopoulos, G.; Hayes, P.C.; Plevris, J.N. Low serum retinol levels are associated with hepatocellular carcinoma in patients with chronic liver disease. Aliment. Pharmacol. Ther. 2000, 14, 1295–1301. [Google Scholar] [CrossRef] [PubMed]
- Peres, W.A.; Chaves, G.V.; Goncalves, J.C.; Ramalho, A.; Coelho, H.S. Vitamin A deficiency in patients with hepatitis C virus-related chronic liver disease. Br. J. Nutr. 2011, 106, 1724–1731. [Google Scholar] [CrossRef] [PubMed]
- Moodie, E.E.; Stephens, D.A. Estimation of dose–response functions for longitudinal data using the generalised propensity score. Stat. Methods Med. Res. 2012, 21, 149–166. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.Y.; Wu, K.S.; Zeng, Y.; Tang, W.R.; Du, P.L.; Xu, Z.X.; Xu, X.L.; Luo, J.Y.; Lin, K. Liver cancer mortality characteristics and trends in China from 1991 to 2012. Asian Pac. J. Cancer Prev. 2015, 16, 1959–1964. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.W.; Zheng, R.S.; Li, N.; Zeng, H.M.; Dai, Z.; Zou, X.N.; Chen, W.Q. Analysis and prediction of liver cancer incidence in China. Chin. J. Prev. Med. 2012, 46, 587–592. [Google Scholar]
- Huang, B.X.; Zhu, Y.Y.; Tan, X.Y.; Lan, Q.Y.; Li, C.L.; Chen, Y.M.; Zhu, H.L. Serum betaine is inversely associated with low lean mass mainly in men in a Chinese middle-aged and elderly community-dwelling population. Br. J. Nutr. 2016, 115, 2181–2188. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.L.; Wu, B.H.; Xue, W.Q.; He, M.G.; Fan, F.; Ouyang, W.F.; Tu, S.L.; Zhu, H.L.; Chen, Y.M. Greater intake of fruit and vegetables is associated with a lower risk of osteoporotic hip fractures in elderly Chinese: A 1:1 matched case–control study. Osteoporos. Int. 2013, 24, 2827–2836. [Google Scholar] [CrossRef] [PubMed]
- Zeng, F.F.; Wu, B.H.; Fan, F.; Xie, H.L.; Xue, W.Q.; Zhu, H.L.; Chen, Y.M. Dietary patterns and the risk of hip fractures in elderly Chinese: A matched case–control study. J. Clin. Endocrinol. Metab. 2013, 98, 2347–2355. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | PLC (n = 644) | Controls (n = 644) | p |
|---|---|---|---|
| Age (years) 1 | 54.33 (10.34) | 54.36 (10.23) | 0.961 * |
| Gender (%) | |||
| Male | 559 (86.8) | 559 (86.8) | 1 # |
| Female | 85 (13.2) | 85 (13.2) | |
| BMI (kg/m2) 1 | 22.84 (3.33) | 23.34 (3.16) | 0.005 * |
| WHR 1 | 0.91 (0.07) | 0.93 (0.06) | 0.001 * |
| Physical activity (MET h/per day) 1 | 32.91 (13.29) | 37.46 (9.61) | <0.001 * |
| Education level, n (%) | |||
| Secondary school or below | 384 (59.6) | 399 (62.0) | 0.424 # |
| High school or above | 260 (40.4) | 245 (38.0) | |
| Household income (Yuan/month), n (%) | |||
| ≤2000 | 230 (35.7) | 254 (39.4) | 0.094 + |
| 2001–6000 | 333 (51.7) | 325 (50.5) | |
| >6000 | 81 (12.6) | 65 (10.1) | |
| Smoking, n (%) | 345 (53.6) | 283 (43.9) | 0.001 # |
| Alcohol user, n (%) | 208 (32.3) | 125 (19.4) | <0.001 # |
| Tea Drinker, n (%) | 353 (54.8) | 398 (61.8) | 0.011 # |
| Energy intake (kcal/day) 2,4 | 1523 (1224, 1865) | 1635 (1255, 1938) | 0.018 + |
| VA (μg RE/day) 3,4 | 824 (577, 1150) | 1024 (753, 1421) | <0.001 + |
| Carotenes (μg/day) 3,4 | 3486 (2276, 5152) | 4556 (3123, 6356) | <0.001 + |
| Retinol (μg/day) 3,4 | 150 (87, 262) | 191 (125, 308) | <0.001 + |
| Food Sources | Proportion (%) | |
|---|---|---|
| Cases | Controls | |
| Retinol | ||
| Animal offal | 52.8 | 54.0 |
| Eggs | 20.9 | 20.3 |
| Dairy products | 3.9 | 5.7 |
| Lean hogs | 4.8 | 3.9 |
| Freshwater fish | 3.7 | 3.6 |
| Sub-total | 86.1 | 87.5 |
| Carotenes | ||
| Leafy green vegetables | 70.5 | 73.1 |
| Carrots | 15.7 | 13.3 |
| Melon and fruit vegetables | 4.3 | 3.9 |
| Tomatoes | 2.3 | 2.7 |
| Citrus fruits | 1.8 | 1.7 |
| Sub-total | 94.6 | 94.7 |
| Amount | n (Cases/Controls) | Crude OR (95% CI) | Adjusted OR (95% CI) # | |
|---|---|---|---|---|
| Quartile of VA (μg RE/day) | ||||
| Q1 | ≤753 | 276/161 | 1.00 | 1.00 |
| Q2 | 753–1024 | 149/161 | 0.54 (0.40–0.73) *** | 0.53 (0.39–0.73) *** |
| Q3 | 1024–1421 | 122/161 | 0.44 (0.33–0.60) *** | 0.44 (0.32–0.60) *** |
| Q4 | >1421 | 97/161 | 0.35 (0.26–0.48) *** | 0.34 (0.24–0.48) *** |
| P-trend | <0.001 | <0.001 | ||
| Quartile of Carotenes (μg/day) | ||||
| Q1 | ≤3123 | 272/161 | 1.00 | 1.00 |
| Q2 | 3123–4556 | 163/161 | 0.60 (0.45–0.80) ** | 0.56 (0.41–0.77) *** |
| Q3 | 4556–6356 | 110/161 | 0.40 (0.30–0.55) *** | 0.41 (0.29–0.57) *** |
| Q4 | >6356 | 99/161 | 0.36 (0.27–0.50) *** | 0.35 (0.25–0.49) *** |
| P-trend | <0.001 | <0.001 | ||
| Quartile of Retinol (μg/day) | ||||
| Q1 | ≤125 | 266/161 | 1.00 | 1.00 |
| Q2 | 125–191 | 146/161 | 0.55 (0.41–0.74) *** | 0.49 (0.36–0.68) *** |
| Q3 | 191–308 | 108/161 | 0.41 (0.30–0.56) *** | 0.34 (0.24–0.48) *** |
| Q4 | >308 | 124/161 | 0.47 (0.34–0.63) *** | 0.37 (0.27–0.52) *** |
| P-trend | <0.001 | <0.001 | ||
| Q1 | Q2 | Q3 | Q4 | P–trend | ||
|---|---|---|---|---|---|---|
| VA | ||||||
| Smoking | ||||||
| Yes | n (cases/controls) | 149/70 | 85/72 | 63/70 | 48/71 | |
| OR (95% CI) 1 | 1.00 | 0.55 (0.35–0.86) * | 0.37 (0.23–0.60) *** | 0.32 (0.20–0.53) *** | <0.001 | |
| No | n (cases/controls) | 121/90 | 72/91 | 55/90 | 51/90 | |
| OR (95% CI) 1 | 1.00 | 0.54 (0.34–0.85) ** | 0.42 (0.26–0.67) *** | 0.37 (0.23–0.59) *** | <0.001 | |
| P–interaction | 0.657 | |||||
| Alcohol use | ||||||
| Yes | n (cases/controls) | 98/31 | 61/32 | 29/31 | 20/31 | |
| OR (95% CI) 1 | 1.00 | 0.57 (0.30–1.1) + | 0.30 (0.15–0.60) ** | 0.22 (0.10–0.47) *** | <0.001 | |
| No | n (cases/controls) | 178/129 | 100/131 | 83/130 | 75/129 | |
| OR (95% CI) 1 | 1.00 | 0.54 (0.38–0.78) ** | 0.46 (0.31–0.67) *** | 0.39 (0.27–0.58) *** | <0.001 | |
| P–interaction | 0.062 | |||||
| Tea drinking | ||||||
| Yes | n (cases/controls) | 144/100 | 86/99 | 70/99 | 53/100 | |
| OR (95% CI) 1 | 1.00 | 0.60 (0.39–0.92) * | 0.52 (0.34–0.81) ** | 0.40 (0.25–0.63) *** | <0.001 | |
| No | n (cases/controls) | 132/62 | 66/61 | 47/61 | 46/62 | |
| OR (95% CI) 1 | 1.00 | 0.41 (0.25–0.68) *** | 0.33 (0.19–0.55) *** | 0.34 (0.20–0.58) *** | <0.001 | |
| P–interaction | 0.343 | |||||
| Carotenes | ||||||
| Smoking | ||||||
| Yes | n (cases/controls) | 142/71 | 102/71 | 56/70 | 45/71 | |
| OR (95% CI) 1 | 1.00 | 0.64 (0.41–1.00) + | 0.36 (0.22–0.58) *** | 0.31 (0.19–0.52) *** | <0.001 | |
| No | n (cases/controls) | 122/90 | 69/90 | 54/91 | 54/90 | |
| OR (95% CI) 1 | 1.00 | 0.56 (0.35–0.88) * | 0.43 (0.27–0.69) *** | 0.40 (0.25–0.65) *** | <0.001 | |
| P–interaction | 0.375 | |||||
| Alcohol use | ||||||
| Yes | n (cases/controls) | 91/31 | 68/32 | 30/31 | 19/31 | |
| OR (95% CI) 1 | 1.00 | 0.54 (0.28–1.04) + | 0.29 (0.14–0.59) ** | 0.18 (0.08–0.39) *** | <0.001 | |
| No | n (cases/controls) | 176/129 | 105/131 | 76/129 | 79/130 | |
| OR (95% CI) 1 | 1.00 | 0.57 (0.39–0.81) ** | 0.43 (0.29–0.64) ** | 0.41 (0.28–0.61) *** | <0.001 | |
| P–interaction | 0.069 | |||||
| Tea drinking | ||||||
| Yes | n (cases/controls) | 147/100 | 89/99 | 64/99 | 53/100 | |
| OR (95% CI) 1 | 1.00 | 0.57 (0.37–0.88) * | 0.49 (0.31–0.76) ** | 0.38 (0.24–0.60) *** | <0.001 | |
| No | n (cases/controls) | 129/62 | 70/61 | 46/61 | 46/62 | |
| OR (95% CI) 1 | 1.00 | 0.46 (0.28–0.75) ** | 0.34 (0.20–0.57) *** | 0.34 (0.20–0.57) *** | <0.001 | |
| P–interaction | 0.070 | |||||
| Retinol | ||||||
| Smoking | ||||||
| Yes | n (cases/controls) | 125/70 | 79/71 | 70/71 | 71/71 | |
| OR (95% CI) 1 | 1.00 | 0.56 (0.35–0.90) * | 0.42 (0.25–0.69) ** | 0.43 (0.27–0.70) ** | <0.001 | |
| No | n (cases/controls) | 142/90 | 66/91 | 38/90 | 53/90 | |
| OR (95% CI) 1 | 1.00 | 0.41 (0.26–0.64) *** | 0.24 (0.15–0.41) *** | 0.30 (0.19–0.49) *** | <0.001 | |
| P–interaction | 0.484 | |||||
| Alcohol use | ||||||
| Yes | n (cases/controls) | 83/31 | 58/32 | 31/31 | 36/31 | |
| OR (95% CI) 1 | 1.00 | 0.61 (0.32–1.20) | 0.27 (0.13-0.57) ** | 0.43 (0.21-0.87) * | 0.003 | |
| No | n (cases/controls) | 182/130 | 104/129 | 64/130 | 86/130 | |
| OR (95% CI) 1 | 1.00 | 0.52 (0.36–0.76) ** | 0.33 (0.22–0.50) *** | 0.40 (0.27–0.59) *** | <0.001 | |
| P–interaction | 0.912 | |||||
| Tea drinking | ||||||
| Yes | n (cases/controls) | 148/100 | 79/99 | 56/99 | 70/100 | |
| OR (95% CI) 1 | 1.00 | 0.58 (0.37–0.89) * | 0.40 (0.25–0.64) *** | 0.43 (0.27–0.67) *** | <0.001 | |
| No | n (cases/controls) | 113/62 | 75/61 | 46/61 | 57/62 | |
| OR (95% CI) 1 | 1.00 | 0.58 (0.35–0.95) * | 0.30 (0.17–0.53) *** | 0.42 (0.25–0.71) ** | <0.001 | |
| P–interaction | 0.366 | |||||
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lan, Q.-Y.; Zhang, Y.-J.; Liao, G.-C.; Zhou, R.-F.; Zhou, Z.-G.; Chen, Y.-M.; Zhu, H.-L. The Association between Dietary Vitamin A and Carotenes and the Risk of Primary Liver Cancer: A Case–Control Study. Nutrients 2016, 8, 624. https://doi.org/10.3390/nu8100624
Lan Q-Y, Zhang Y-J, Liao G-C, Zhou R-F, Zhou Z-G, Chen Y-M, Zhu H-L. The Association between Dietary Vitamin A and Carotenes and the Risk of Primary Liver Cancer: A Case–Control Study. Nutrients. 2016; 8(10):624. https://doi.org/10.3390/nu8100624
Chicago/Turabian StyleLan, Qiu-Ye, Yao-Jun Zhang, Gong-Cheng Liao, Rui-Fen Zhou, Zhong-Guo Zhou, Yu-Ming Chen, and Hui-Lian Zhu. 2016. "The Association between Dietary Vitamin A and Carotenes and the Risk of Primary Liver Cancer: A Case–Control Study" Nutrients 8, no. 10: 624. https://doi.org/10.3390/nu8100624
APA StyleLan, Q.-Y., Zhang, Y.-J., Liao, G.-C., Zhou, R.-F., Zhou, Z.-G., Chen, Y.-M., & Zhu, H.-L. (2016). The Association between Dietary Vitamin A and Carotenes and the Risk of Primary Liver Cancer: A Case–Control Study. Nutrients, 8(10), 624. https://doi.org/10.3390/nu8100624







