Immune-Enhancing Effects of a High Molecular Weight Fraction of Cynanchum wilfordii Hemsley in Macrophages and Immunosuppressed Mice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Preparation of a High Molecular Weight Fraction of Cynanchum wilfordii
2.3. Extraction of a Crude Polysaccharide
2.4. Chemical Analyses
2.5. Monosaccharide Composition and Molecular Weight Distribution
2.6. Protease Treatment and Periodate Oxidation of HMFO
2.7. Cell Culture
2.8. Cell Viability Assay
2.9. Measurement of Nitric Oxide (NO)
2.10. Determination of TNF-α, (IL-6) and IL-1β Production
2.11. Phagocytosis Assay in RAW 264.7 Cells
2.12. Western Blot Analysis
2.13. Animals and Treatments
2.14. Preparation of Mouse Splenocytes
2.15. Splenocyte Proliferation Assay
2.16. Flow Cytometric Analysis
2.17. NK Cell Activity
2.18. Statistical Analysis
3. Results
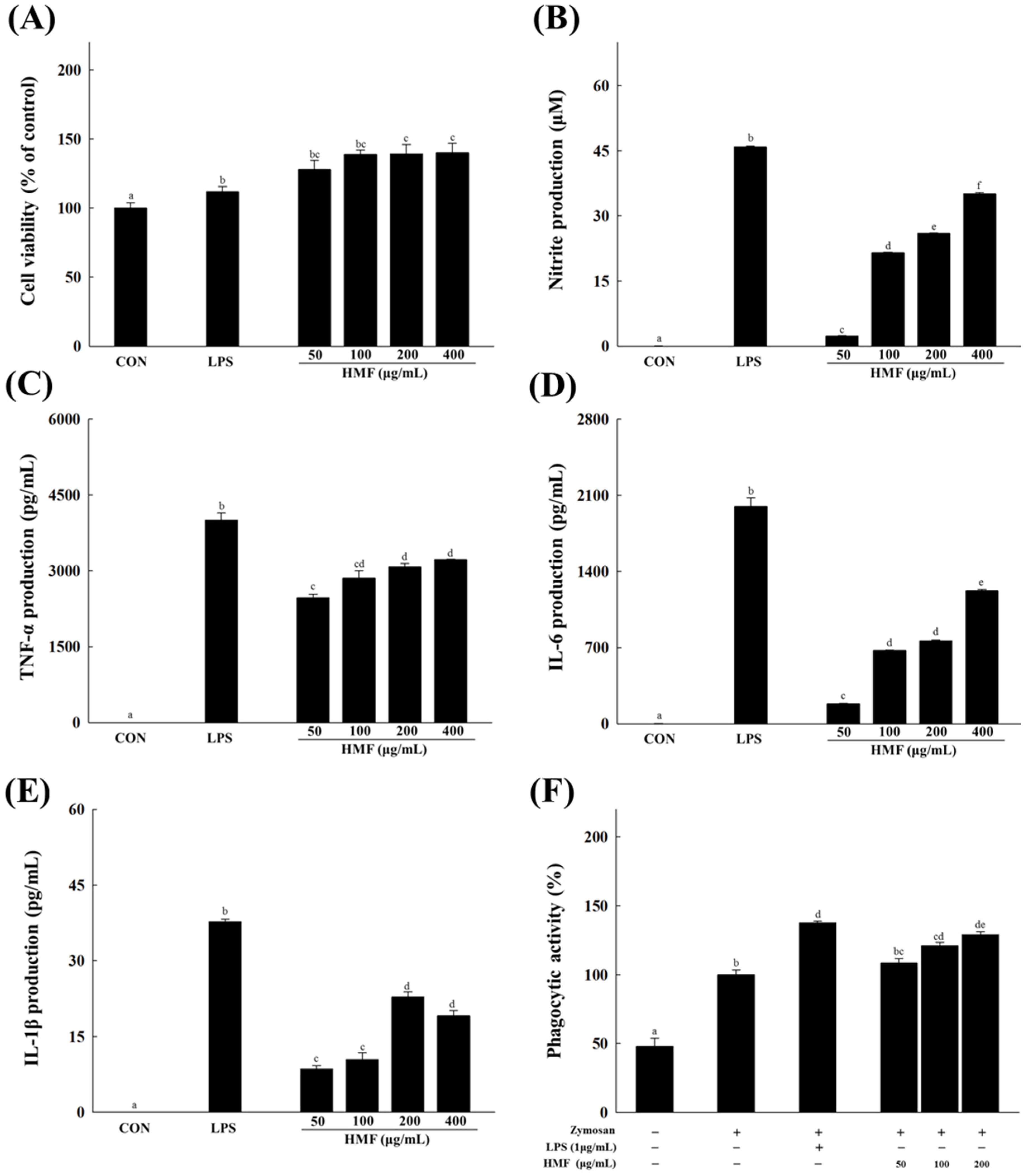
3.1. Effects of HMF on NO Production and Cytokine and Phagocytic Activity
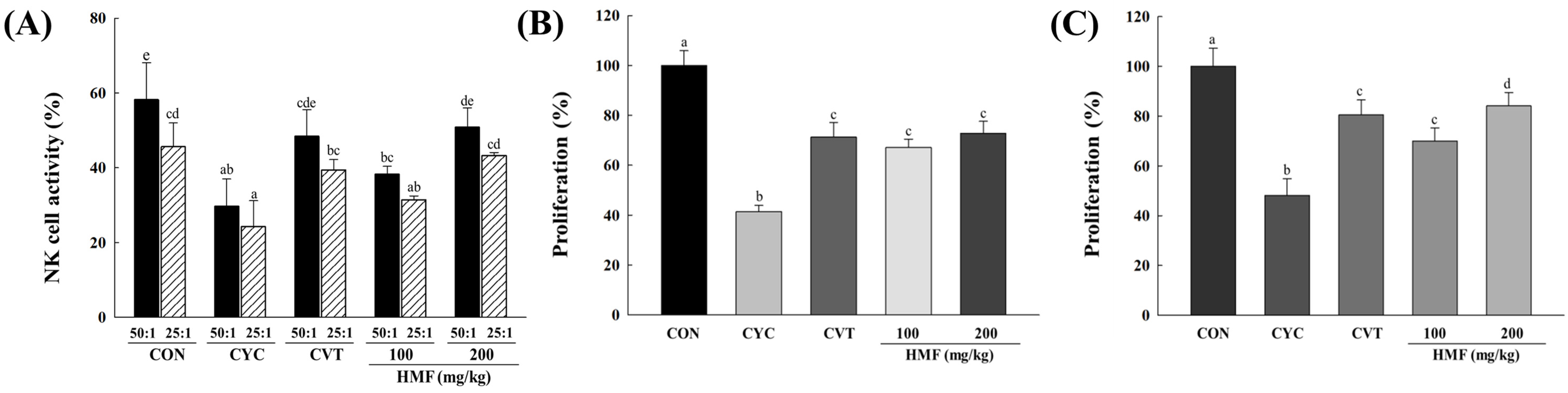
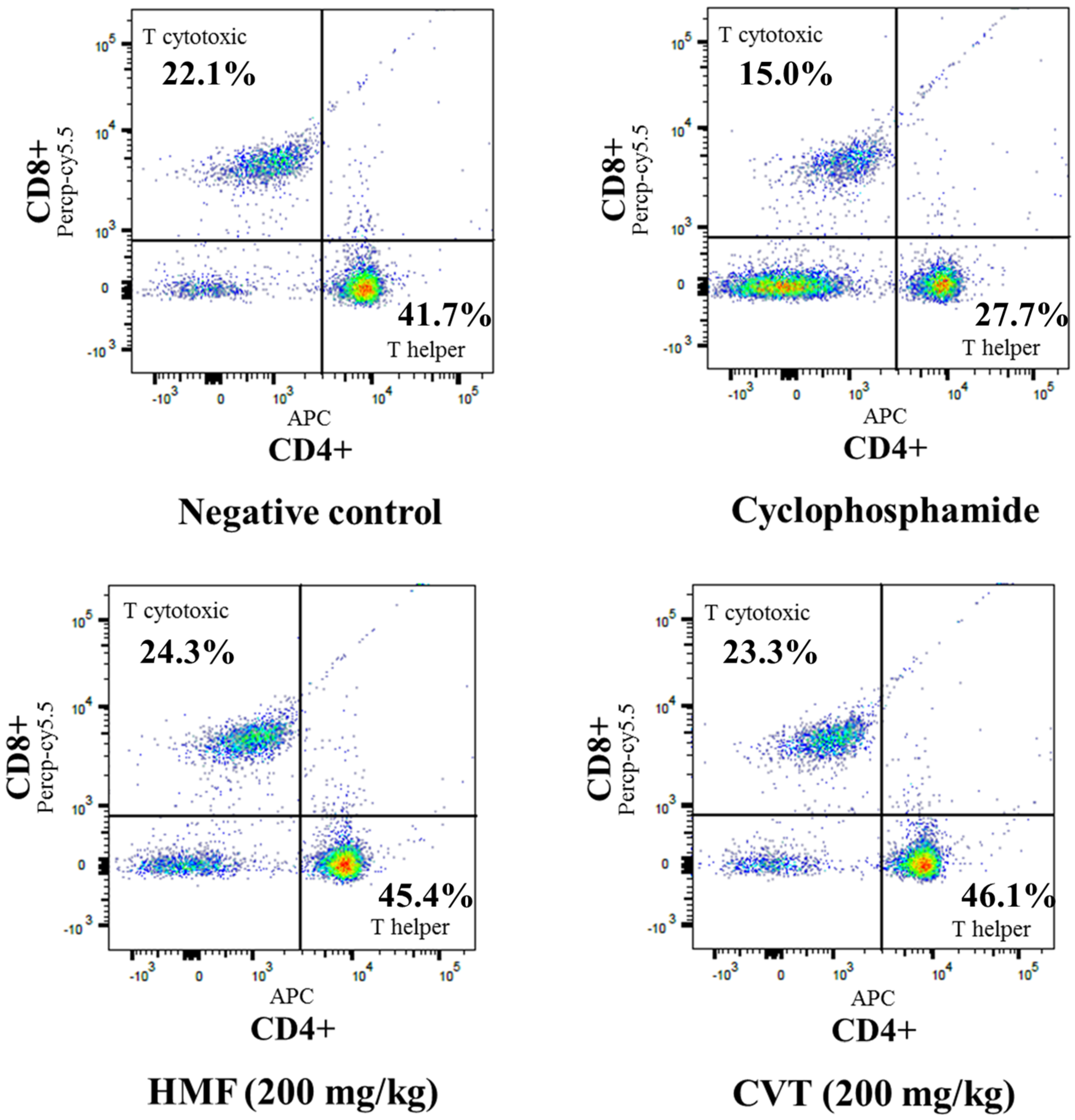
3.2. Effects of HMF on Decreased Immune Function in CYC-Treated Mice
3.3. Effects of a Crude Polysaccharide of HMF on the Immunostimulatory Activity in RAW 264.7 Macrophages
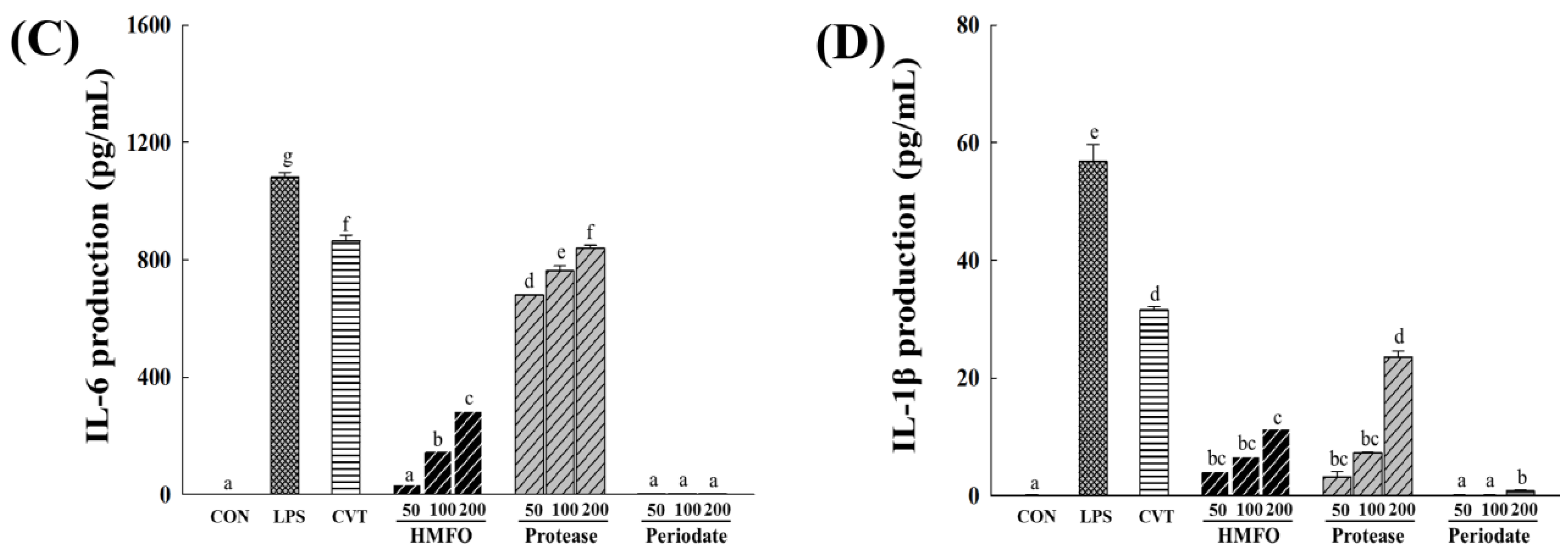
3.3.1. Effects of Chemical and Enzymatic Treatments of HMFO on Immunostimulatory Activity in RAW 264.7 Macrophages
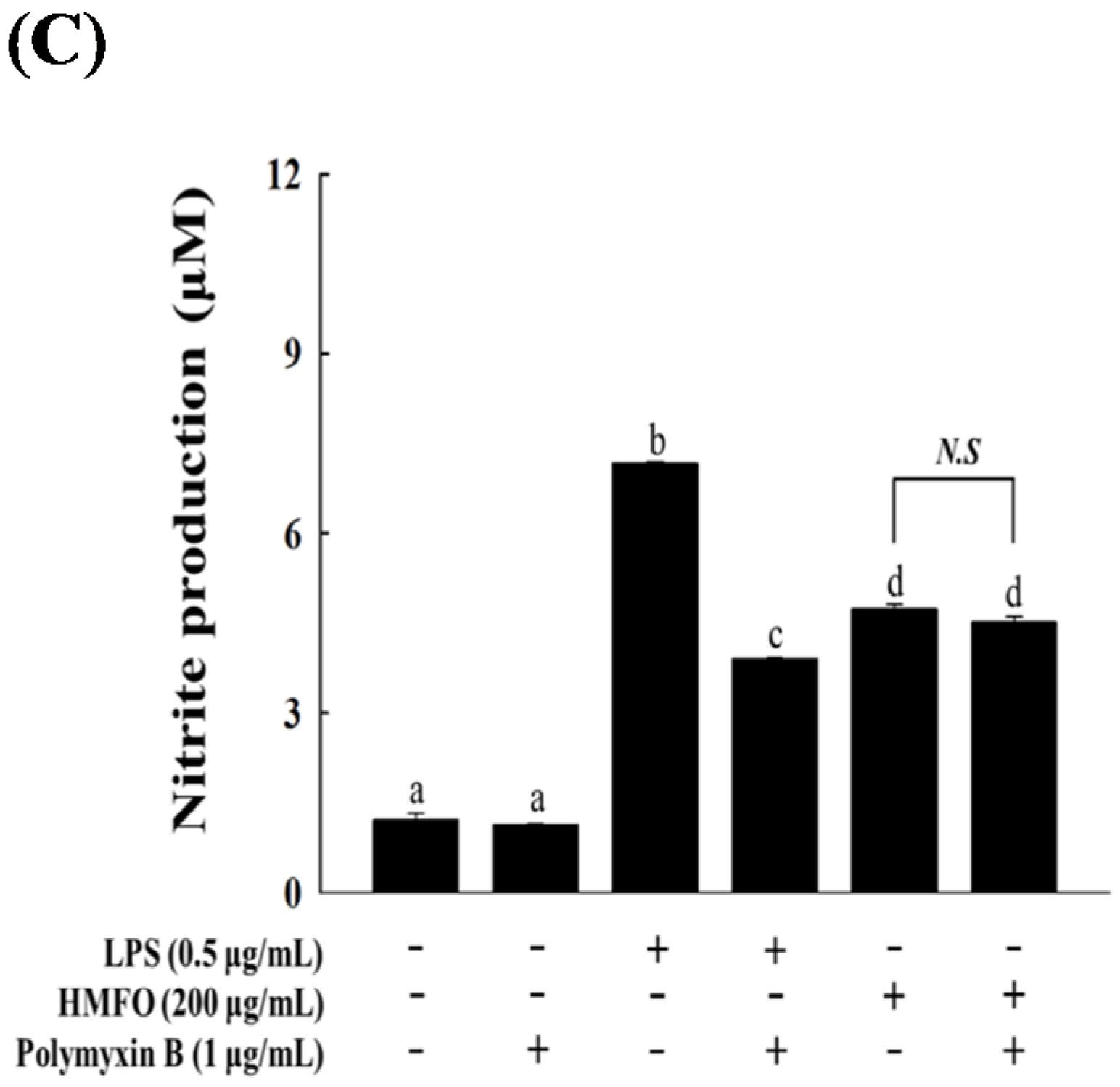
3.3.2. Effects of HMFO on NO Production and iNOS Expression in RAW 264.7 Macrophages
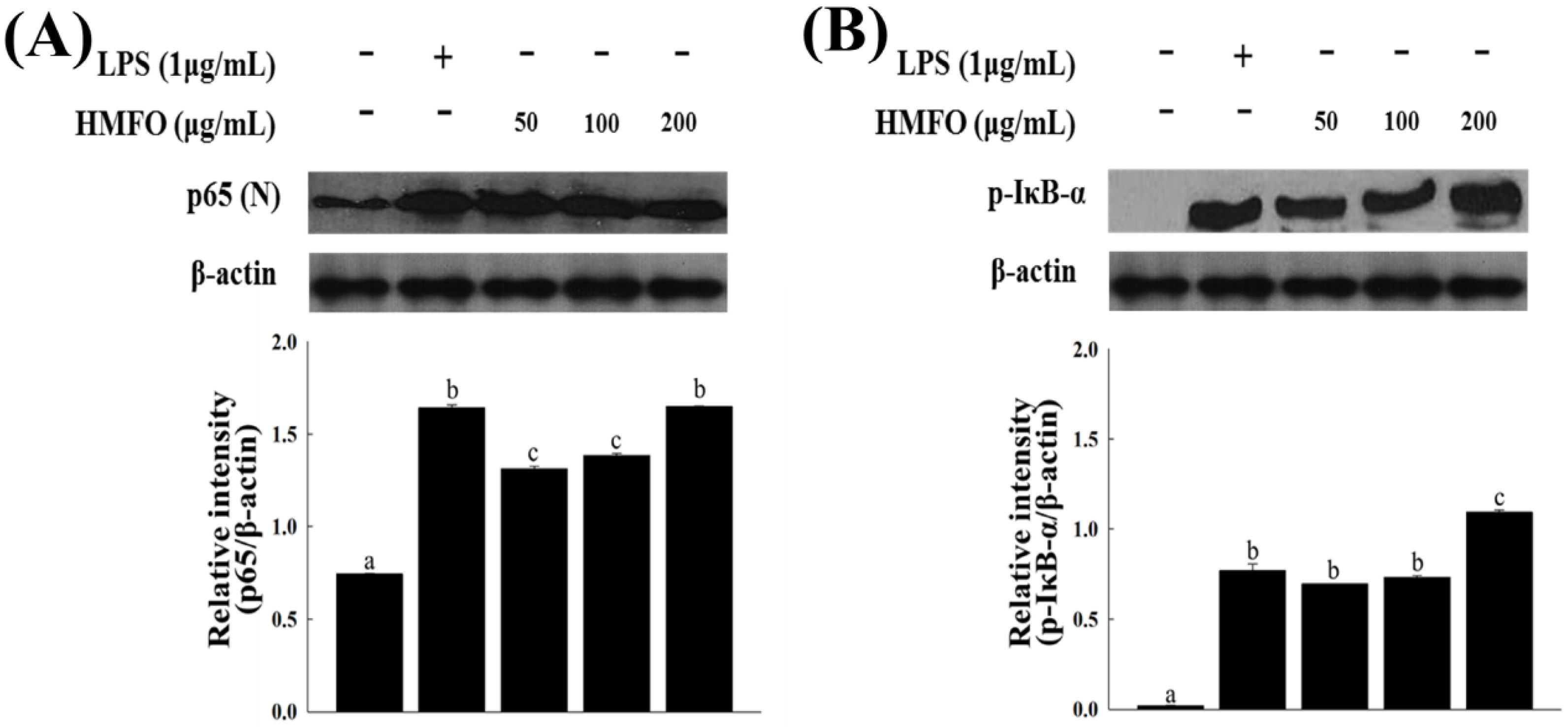
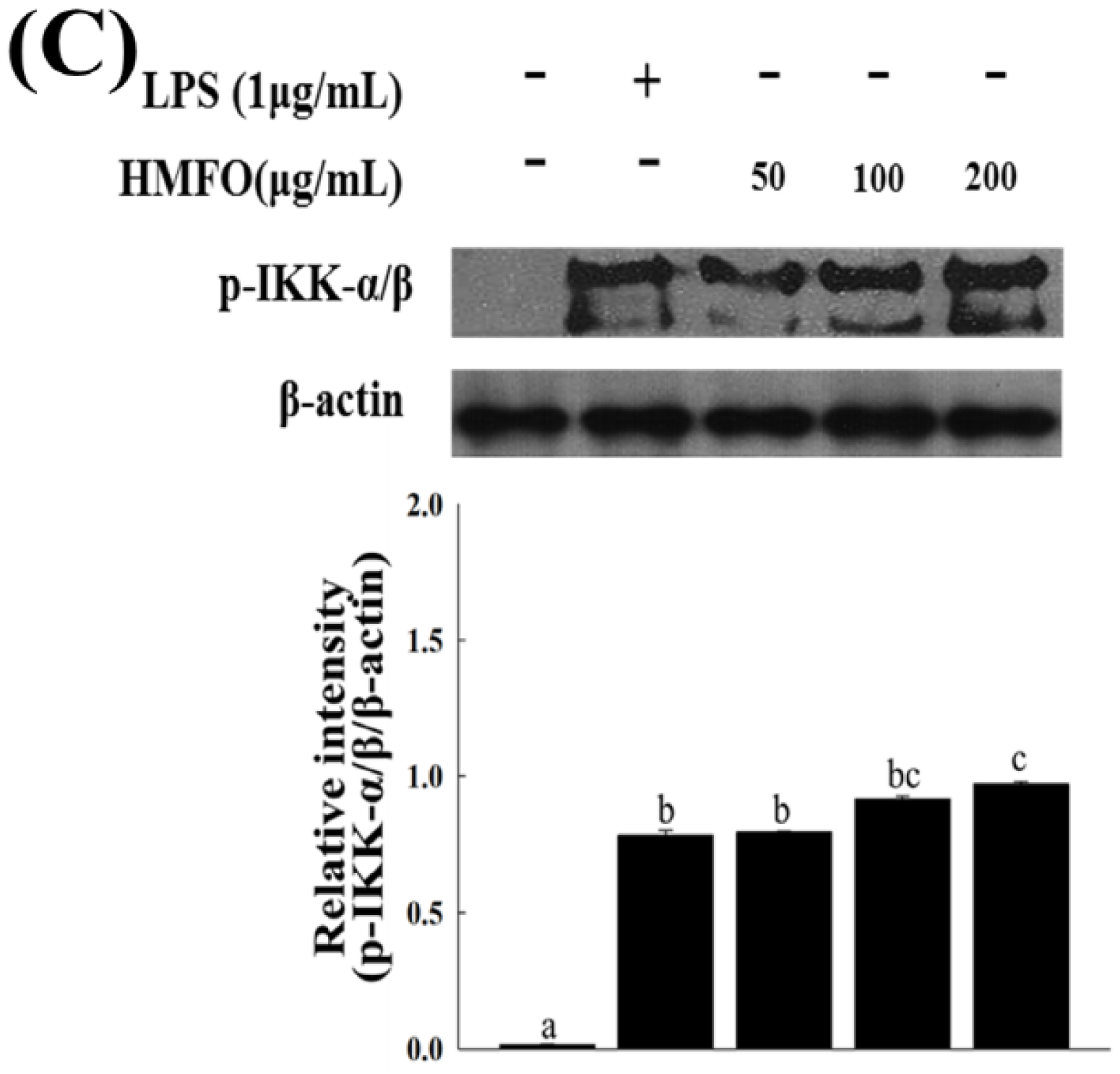
3.3.3. Effects of HMFO on the Phosphorylation of IκB-α and IKK-α/β in RAW 264.7 Macrophages
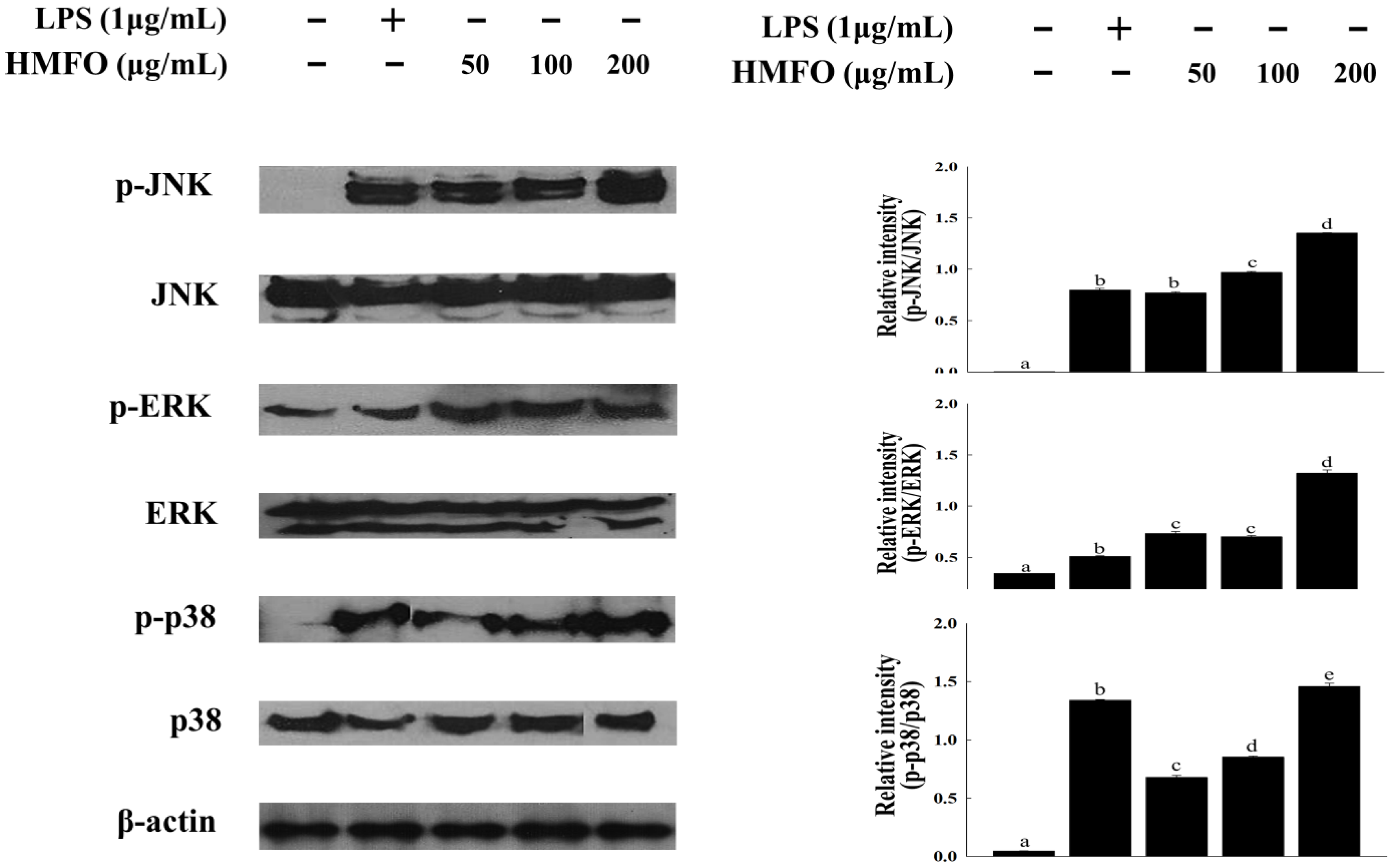
3.3.4. Effects of HMFO on Mitogen-Activated Protein Kinase (MAPK) Phosphorylation in RAW 264.7 Macrophages
3.4. Chemical Compositions and Molecular Weight Distribution
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Parham, P. The Immune System, 3rd ed.; Garland Science: Milton Park, UK, 2009; pp. 1–2. [Google Scholar]
- Mishra, K.P.; Padwad, Y.S.; Jain, M.; Karan, D.; Ganju, L.; Sawhney, R.C. Aqueous extract of Rhodiola imbricata rhizome stimulates proinflammatory mediators via phosphorylated IkappaB and transcription factor nuclear factor-kappaB. Immunopharmacol. Immunotoxicol. 2006, 28, 201–212. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Jiao, F.; Qiu, Y.; Li, W.; Qu, Y.; Tian, C.; Li, Y.; Bai, R.; Lao, F.; Zhao, Y.; et al. Immunostimulatory properties and enhanced TNF-alpha mediated cellular immunity for tumor therapy by C60(OH)20 nanoparticles. Nanotechnology 2009, 20, 41. [Google Scholar]
- Riahi, B.; Rafatpanah, H.; Mahmoudi, M.; Memar, B.; Brook, A.; Tabasi, N.; Karimi, G. Immunotoxicity of paraquat after subacute exposure to mice. Food. Chem. Toxicol. 2010, 48, 1627–1631. [Google Scholar] [CrossRef] [PubMed]
- Riahi, B.; Rafatpanah, H.; Mahmoudi, M.; Memar, B.; Fakhr, A.; Tabasi, N.; Karimi, G. Evaluation of suppressive effects of paraquat on innate immunity in Balb/c mice. J. Immunotoxicol. 2011, 8, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Wahab, S.; Hussain, A.; Ahmad, M.P.; Hussain, M.S.; Rizvi, A.; Ahmad, M.F.; Ansari, N.H.; Farooqui, A.H.A. The ameliorative effects of Averroha carambola on humoral response to sheep erythrocytes in non-treated and cyclophosphamide-immunocompromised mice. J. Acute Dis. 2014, 3, 115–123. [Google Scholar] [CrossRef]
- Block, K.I.; Mead, M.N. Immune system effects of echinacea, ginseng, and astragalus: A review. Integr. Cancer Ther. 2003, 2, 247–267. [Google Scholar] [CrossRef] [PubMed]
- Korea Food and Drug Administration (KFDA). Available online: http://www.mfds.go.kr/herbmed/index.do?nMenuCode=7&code=KHP-N126&includeUrl=/herbmed/view.jsp (accessed on 14 July 2016).
- Shan, L.; Liu, R.H.; Shen, Y.H.; Zhang, W.D.; Zhang, C.; Wu, D.Z.; Min, L.; Su, J.; Xu, X.K. Gastroprotective effect of a traditional Chinese herbal drug ‘Baishouwu’ on experimental gastric lesions in rats. J. Ethnopharmacol. 2006, 107, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.H.; Lee, Y.J.; Oh, H.C.; Cui, Y.L.; Kim, J.S.; Kang, D.G.; Lee, H.S. Improved endothelial dysfunction by Cynanchum wilfordii in apolipoprotein E(−/−) mice fed a high fat/cholesterol diet. J. Med. Food. 2012, 15, 169–179. [Google Scholar] [CrossRef] [PubMed]
- Hwang, B.Y.; Kim, S.E.; Kim, Y.H.; Kim, H.S.; Hong, Y.S.; Ro, J.S.; Lee, K.S.; Lee, J.J. Pregnane glycoside multidrug-resistance modulators from Cynanchum wilfordii. J. Nat. Prod. 1999, 62, 640–643. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Choi, J.H.; Kim, Y.E.; Kim, I.H.; Kim, B.M.; Lee, C.H. Effects of Cynanchum wilfordii extract on serum lipid components and enzyme activities in hyperlipidemic and streptozotocin-induced diabetic rats. Prev. Nutr. Food Sci. 2013, 18, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Niu, J.Z.; Ye, B.K.; Wang, D.F. Observation of the protection effect of Baishouwu to the liver of the high serum cholesterol mouse. Ji Sheng Chong Yu Yi Xue Kun Chong Xue Bao 1998, 3, 266–268. (In Chinese) [Google Scholar]
- Shan, L.; Zhang, W.D.; Zhang, C.; Liu, R.H.; Su, J.; Zhou, Y. Antitumor activity of crude extract and fractions from root tuber of Cynanchum auriculatum Royle ex Wight. Phytother. Res. 2005, 19, 259–261. [Google Scholar] [CrossRef] [PubMed]
- Yoon, M.Y.; Choi, N.H.; Min, B.S.; Choi, G.J.; Choi, Y.H.; Jang, K.S.; Han, S.S.; Cha, B.C.; Kim, J.C. Potent in vivo antifungal activity against powdery mildews of pregnane glycosides from the roots of Cynanchum wilfordii. J. Agric. Food Chem. 2011, 59, 12210–12216. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.K.; Yeo, H.; Kim, J.; Markelonis, G.J.; Oh, T.H.; Kim, Y.C. Cynandione A from Cynanchum wilfordii protects cultured cortical neurons from toxicity induced by H2O2, l-glutamate, and kainate. J. Neurosci. Res. 2000, 59, 259–264. [Google Scholar] [CrossRef]
- Lee, M.K.; Yeo, H.; Kim, J.; Kim, Y.C. Protection of rat hepatocytes exposed to CCl4 in vitro by cynandione A, a biacetophenone from Cynanchum wilfordii. J. Pharm. Pharmacol. 2000, 52, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Biochem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Taylor, K.A.; Buchanan-Smith, J.G. A colorimetric method for the quantitation of uronic acids and a specific assay for galacturonic acid. Anal. Biochem. 1992, 201, 190–196. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Karkhanis, Y.D.; Zeltner, J.Y.; Jackson, J.J.; Carlo, D.J. A new and improved microassay to determine 2-keto-3-deoxyoctonate in lipopolysaccharide of gram-negative bacteria. Anal. Biochem. 1978, 85, 595–601. [Google Scholar] [CrossRef]
- Biondo, P.D.; Goruk, S.; Ruth, M.R.; O’Connell, E.; Field, C.J. Effect of CVT-E002™ (COLD-fX®) versus a ginsenoside extract on systemic and gut-associated immune function. Int. Immunopharmacol. 2008, 8, 1134–1142. [Google Scholar] [CrossRef] [PubMed]
- Sepp, A.; Binns, R.M.; Lechler, R.I. Improved protocol for colorimetric detection of complement-mediated cytotoxicity based on the measurement of cytoplasmic lactate dehydrogenase activity. J. Immunol. Methods 1996, 196, 175–180. [Google Scholar] [CrossRef]
- Belardelli, F. Role of interferons and other cytokines in the regulation of the immune response. Acta Pathol. Microbiol. Immunol. Scand. 1995, 103, 161–179. [Google Scholar] [CrossRef]
- Moncada, S.; Palmer, R.M.; Higgs, E.A. Nitric oxide: Physiology, pathophysiology, and pharmacology. Pharmacol. Rev. 1991, 43, 109–142. [Google Scholar] [PubMed]
- Bai, Y.; Zhang, P.; Chen, G.; Cao, J.; Huang, T.; Chen, K. Macrophage immunomodulatory activity of extracellular polysaccharide (PEP) of Antarctic bacterium Pseudoaltermonas sp.S-5. Int. Immunopharmacol. 2012, 12, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Jiao, L.; Li, X.; Li, T.; Jiang, P.; Zhang, L.; Wu, M.; Zhang, L. Characterization and anti-tumor activity of alkali-extracted polysaccharide from Enteromorpha intestinalis. Int. Immunopharmacol. 2009, 9, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Tang, J.; Wang, X.; Sun, F.; Liang, S. An immunostimulatory polysaccharide (SCP-IIa) from the fruit of Schisandra chinensis (Turcz.) Baill. Int. J. Biol. Macromol. 2012, 50, 844–848. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Nie, W.; Fan, S.; Zhang, J.; Wang, Y.; Lu, J.; Jin, L. A polysaccharide from Sargassum fusiforme protects against immunosuppression in cyclophosphamide-treated mice. Carbohydr. Polym. 2012, 90, 1114–1119. [Google Scholar] [CrossRef] [PubMed]
- Pang, X.; Chen, Z.; Gao, X.; Liu, W.; Slavin, M.; Yao, W.; Yu, L.L. Potential of a novel polysaccharide preparation (GLPP) from Anhui-grown Ganoderma lucidum in tumor treatment and immunostimulation. J. Food Sci. 2007, 72, S435–S442. [Google Scholar] [CrossRef] [PubMed]
- Herberman, R.B.; Nunn, M.E.; Lavrin, D.H. Natural cytotoxic reactivity of mouse lymphoid cells against syngeneic acid allogeneic tumors. I. Distribution of reactivity and specificity. Int. J. Cancer 1975, 16, 216–229. [Google Scholar] [CrossRef] [PubMed]
- Kiessling, R.; Klein, E.; Wigzell, H. “Natural” killer cells in the mouse. I. Cytotoxic cells with specificity for mouse Moloney leukemia cells. Specificity and distribution according to genotype. Eur. J. Immunol. 1975, 5, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Smyth, M.J.; Hayakawa, Y.; Takeda, K.; Yagita, H. New aspects of natural-killer-cell surveillance and therapy of cancer. Nat. Rev. Cancer 2002, 2, 850–861. [Google Scholar] [CrossRef] [PubMed]
- Vesely, M.D.; Kershaw, M.H.; Schreiber, R.D.; Smyth, M.J. Natural innate and adaptive immunity to cancer. Annu. Rev. Immunol. 2011, 29, 235–271. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Li, M.; Luo, Y.; Wu, W. Isolation and structural characterization of an immunostimulating polysaccharide from fuzi, Aconitum carmichaeli. Carbohydr. Res. 2006, 341, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Lee, G.H.; Kwon, Y.K.; Park, J.H.; Shin, S.W. Water extracted Evodiae fructus possesses immunomodulatory activities on cyclophosphamide induced immunesuppression. Korean J. Orient. Physiol. Pathol. 2007, 21, 485–490. [Google Scholar]
- Rehermann, B. Hepatitis C virus versus innate and adaptive immune responses: A tale of coevolution and coexistence. J. Clin. Investig. 2009, 119, 1745–1754. [Google Scholar] [CrossRef] [PubMed]
- Parel, Y.; Chizzolini, C. Presence of CD4+CD8+ double-positive (DP) T cells in health and disease. Autoimmun. Rev. 2004, 3, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Dalmo, R.A.; Bøgwald, J. β-Glucans as conductors of immune symphonies. Fish Shellfish Immunol. 2008, 25, 384–396. [Google Scholar] [CrossRef] [PubMed]
- Ramberg, J.E.; Nelson, E.D.; Sinnott, R.A. Immunomodulatory dietary polysaccharides: A systematic review of the literature. Nutr. J. 2010, 9, 54. [Google Scholar] [CrossRef] [PubMed]
- Schepetkin, I.A.; Quinn, M.T. Botanical polysaccharides: Macrophage immunomodulation and therapeutic potential. Int. Immunopharmacol. 2006, 6, 317–333. [Google Scholar] [CrossRef] [PubMed]
- Sharon, N. Complex Carbohydrates: Their Chemistry, Biosynthesis, and Functions; Addison-Wesley Publishers Inc.: Boston, MA, USA, 1975; pp. 92–97. [Google Scholar]
- Etemadi, A.H. Membrane asymmetry a survey and critical appraisal of the methodology. I. Methods for assessing the asymmetric orientation and distribution of proteins. Biochim. Biophys. Acta 1980, 604, 347–422. [Google Scholar] [PubMed]
- Karin, M.; Ben-Neriah, Y. Phosphorylation meets ubiquitination: The control of NF-(kappa)B activity. Annu. Rev. Immunol. 2000, 18, 621–663. [Google Scholar] [CrossRef] [PubMed]
- Perkins, N.D. Integrating cell-signalling pathways with NF-kappaB and IKK function. Nat. Rev. Mol. Cell Biol. 2007, 8, 49–62. [Google Scholar] [CrossRef] [PubMed]
- Sekine, Y.; Yumioka, T.; Yamamoto, T.; Muromoto, R.; Imoto, S.; Sugiyma, K.; Oritani, K.; Shimoda, K.; Minoguchi, M.; Akira, S.; et al. Modulation of TLR4 signaling by a novel adaptor protein signal-transducing adaptor protein-2 in macrophages. J. Immunol. 2006, 176, 380–389. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Akira, S. Roles of Toll-like receptors in innate immune responses. Gene Cells 2001, 6, 733–742. [Google Scholar] [CrossRef]
- Cho, C.W.; Han, C.J.; Rhee, Y.K.; Lee, Y.C.; Shin, K.S.; Hong, H.D. Immunostimulatory effects of polysaccharides isolated from Makgeolli (traditional Korean rice wine). Molecules 2014, 19, 5266–5277. [Google Scholar] [CrossRef] [PubMed]
- Engelsen, S.B.; Cros, S.; Mackie, W.; Perez, S. A molecular builder for carbohydrates: Application to polysaccharides and complex carbohydrates. Biopolymers 1996, 39, 417–433. [Google Scholar] [CrossRef]
- Yamada, H. Contribution of pectins on health care. In Pectins and Pectinases; Visser, J., Voragen, A.G.J., Eds.; Elsevier Science B.V.: Amsterdam, The Netherlands, 1996; pp. 173–190. [Google Scholar]


| Group | Initial Body Weight (g) | Terminal Body Weight (g) | Spleen Weight | Thymus Weight | ||
|---|---|---|---|---|---|---|
| Absolute (g) | Relative (%) | Absolute (g) | Relative (%) | |||
| Negative control | 19.11 ± 0.75 a | 20.03 ± 0.65 c | 0.103 ± 0.009 a | 0.512 ± 0.027 ab | 0.047 ± 0.004 a | 0.233 ± 0.017 a |
| CYC | 18.82 ± 0.94 a | 18.10 ± 0.57 a | 0.085 ± 0.005 b | 0.471 ± 0.022 a | 0.041 ± 0.005 a | 0.225 ± 0.023 a |
| HMF (100 mg/kg) | 18.38 ± 0.52 a | 18.41 ± 0.53 ab | 0.100 ± 0.002 a | 0.544 ± 0.014 b | 0.042 ± 0.003 a | 0.230 ± 0.012 b |
| HMF (200 mg/kg) | 19.11 ± 0.34 a | 19.52 ± 0.63 bc | 0.108 ± 0.007 a | 0.554 ± 0.021 b | 0.045 ± 0.003 a | 0.232 ± 0.009 b |
| Positive control | 19.32 ± 0.64 a | 19.69 ± 0.85 c | 0.100 ± 0.008 a | 0.510 ± 0.037 ab | 0.047 ± 0.005 a | 0.238 ± 0.022 b |
| HMFO | |
|---|---|
| Chemical composition (%) | |
| Neutral sugar 1 | 63.8 ± 0.9 |
| Uronic acid 2 | 33.1 ± 1.9 |
| KDO 3 | 0.3 ± 0.1 |
| Protein 4 | 2.9 ± 0.2 |
| Component monosaccharide (mg/g) | |
| Arabinose | 86.0 ± 1.6 |
| Galactose | 76.5 ± 2.9 |
| Rhamnose | 23.0 ± 0.4 |
| Xylose | 2.2 ± 0.6 |
| Glucose | 241.2 ± 6.5 |
| Mannose | 9.0 ± 1.6 |
| Fucose | 5.7 ± 0.2 |
| Fructose | ND 5 |
| Galacturonic acid | 178.8 ± 2.5 |
| Glucuronic acid | 4.9 ± 0.2 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jang, M.; Lim, T.-G.; Ahn, S.; Hong, H.-D.; Rhee, Y.K.; Kim, K.-T.; Lee, E.; Lee, J.H.; Lee, Y.J.; Jung, C.S.; et al. Immune-Enhancing Effects of a High Molecular Weight Fraction of Cynanchum wilfordii Hemsley in Macrophages and Immunosuppressed Mice. Nutrients 2016, 8, 600. https://doi.org/10.3390/nu8100600
Jang M, Lim T-G, Ahn S, Hong H-D, Rhee YK, Kim K-T, Lee E, Lee JH, Lee YJ, Jung CS, et al. Immune-Enhancing Effects of a High Molecular Weight Fraction of Cynanchum wilfordii Hemsley in Macrophages and Immunosuppressed Mice. Nutrients. 2016; 8(10):600. https://doi.org/10.3390/nu8100600
Chicago/Turabian StyleJang, Mi, Tae-Gyu Lim, Sungeun Ahn, Hee-Do Hong, Young Kyoung Rhee, Kyung-Tack Kim, Eunjung Lee, Jeong Hoon Lee, Yun Ji Lee, Chan Sik Jung, and et al. 2016. "Immune-Enhancing Effects of a High Molecular Weight Fraction of Cynanchum wilfordii Hemsley in Macrophages and Immunosuppressed Mice" Nutrients 8, no. 10: 600. https://doi.org/10.3390/nu8100600
APA StyleJang, M., Lim, T.-G., Ahn, S., Hong, H.-D., Rhee, Y. K., Kim, K.-T., Lee, E., Lee, J. H., Lee, Y. J., Jung, C. S., Lee, D. Y., & Cho, C.-W. (2016). Immune-Enhancing Effects of a High Molecular Weight Fraction of Cynanchum wilfordii Hemsley in Macrophages and Immunosuppressed Mice. Nutrients, 8(10), 600. https://doi.org/10.3390/nu8100600





