Influence of Tryptophan and Serotonin on Mood and Cognition with a Possible Role of the Gut-Brain Axis
Abstract
:1. Introduction
2. Serotonin and Kynurenine
Serotonin and Its Receptors
3. Serotonin and Mood
4. Serotonin and Cognition
5. Tryptophan Depletion
6. Tryptophan Depletion, Serotonin and Mood
6.1. Clinical Studies
6.2. Preclinical Studies
7. Tryptophan Depletion, Serotonin and Cognition
7.1. Clinical Studies
7.2. Preclinical Studies
8. Tryptophan Supplementation and Cognition
9. Tryptophan, Sleep, Mood and Cognition
10. Tryptophan, Serotonin and the Brain-Gut Axis
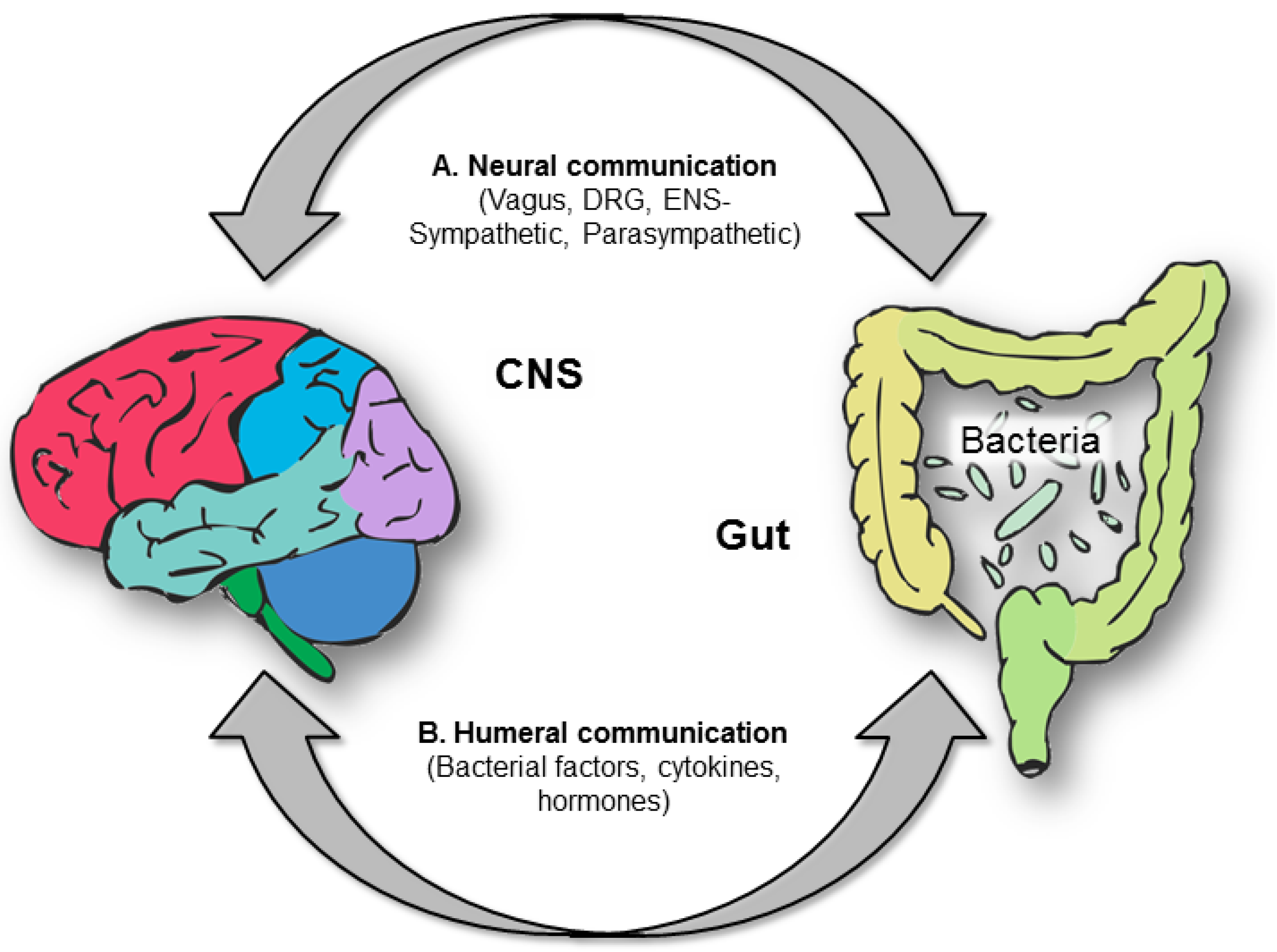
10.1. Tryptophan and the Gut Microbiota
10.2. Behaviour and the Gut Microbiome
10.3. Tryptophan Depletion and the Gut-Brain Axis
11. Concluding Remarks
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Friedman, M.; Levin, C.E. Nutritional and medicinal aspects of d-amino acids. Amino Acids 2012, 42, 1553–1582. [Google Scholar] [CrossRef] [PubMed]
- Herrera, C.P.; Smith, K.; Atkinson, F.; Ruell, P.; Chow, C.M.; O’Connor, H.; Brand-Miller, J. High-glycaemic index and -glycaemic load meals increase the availability of tryptophan in healthy volunteers. Br. J. Nutr. 2011, 105, 1601–1606. [Google Scholar] [CrossRef] [PubMed]
- Young, V.R.; Hussein, M.A.; Murray, E.; Scrimshaw, N.S. Plasma tryptophan response curve and its relation to tryptophan requirements in young adult men. J. Nutr. 1971, 101, 45–59. [Google Scholar] [PubMed]
- Richard, D.M.; Dawes, M.A.; Mathias, C.W.; Acheson, A.; Hill-Kapturczak, N.; Dougherty, D.M. l-tryptophan: Basic metabolic functions, behavioral research and therapeutic indications. Int. J. Tryptophan Res. IJTR 2009, 2, 45–60. [Google Scholar] [PubMed]
- Stone, T.W.; Darlington, L.G. Endogenous kynurenines as targets for drug discovery and development. Nat. Rev. Drug Discov. 2002, 1, 609–620. [Google Scholar] [CrossRef] [PubMed]
- Stone, T.W.; Darlington, L.G. The kynurenine pathway as a therapeutic target in cognitive and neurodegenerative disorders. Br. J. Pharmacol. 2013, 169, 1211–1227. [Google Scholar] [CrossRef] [PubMed]
- Crockett, M.J.; Clark, L.; Roiser, J.P.; Robinson, O.J.; Cools, R.; Chase, H.W.; den Ouden, H.; Apergis-Schoute, A.; Campbell-Meikeljohn, D.; Seymour, B.; et al. Converging evidence for central 5-HT effects in acute tryptophan depletion. Mol. Psychiatr. 2012, 17, 121–123. [Google Scholar] [CrossRef] [PubMed]
- Hughes, M.M.; Carballedo, A.; McLoughlin, D.M.; Amico, F.; Harkin, A.; Frodl, T.; Connor, T.J. Tryptophan depletion in depressed patients occurs independent of kynurenine pathway activation. Brain Behav. Immun. 2012, 26, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Van Donkelaar, E.L.; Blokland, A.; Ferrington, L.; Kelly, P.A.; Steinbusch, H.W.; Prickaerts, J. Mechanism of acute tryptophan depletion: Is it only serotonin? Mol. Psychiatr. 2011, 16, 695–713. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, C.L.; van Swearingen, A.E.D.; Arrant, A.E.; Biskup, C.S.; Kuhn, C.M.; Zepf, F.D. Simplified dietary acute tryptophan depletion: Effects of a novel amino acid mixture on the neurochemistry of C57BL/6J mice. Food Nutr. Res. 2015, 59. [Google Scholar] [CrossRef] [PubMed]
- Fernstrom, J.D.; Fernstrom, M.H. Exercise, serum free tryptophan, and central fatigue. J. Nutr. 2006, 136, 553S–559S. [Google Scholar] [PubMed]
- Berger, M.; Gray, J.A.; Roth, B.L. The expanded biology of serotonin. Ann. Rev. Med. 2009, 60, 355–366. [Google Scholar] [CrossRef] [PubMed]
- Hoyer, D.; Clarke, D.E.; Fozard, J.R.; Hartig, P.R.; Martin, G.R.; Mylecharane, E.J.; Saxena, P.R.; Humphrey, P.P. International union of pharmacology classification of receptors for 5-hydroxytryptamine (serotonin). Pharmacol. Rev. 1994, 46, 157–203. [Google Scholar] [PubMed]
- Lesch, K.P.; Waider, J. Serotonin in the modulation of neural plasticity and networks: Implications for neurodevelopmental disorders. Neuron 2012, 76, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Waraich, P.; Goldner, E.M.; Somers, J.M.; Hsu, L. Prevalence and incidence studies of mood disorders: A systematic review of the literature. Can. J. Psychiatry 2004, 49, 124–138. [Google Scholar] [PubMed]
- Cleare, A.; Pariante, C.M.; Young, A.H.; Anderson, I.M.; Christmas, D.; Cowen, P.J.; Dickens, C.; Ferrier, I.N.; Geddes, J.; Gilbody, S.; et al. Evidence-based guidelines for treating depressive disorders with antidepressants: A revision of the 2008 British Association for Psychopharmacology guidelines. J. Psychopharmacol. 2015, 29, 459–525. [Google Scholar] [CrossRef] [PubMed]
- Morrissette, D.A.; Stahl, S.M. Modulating the serotonin system in the treatment of major depressive disorder. Cns Spectr. 2014, 19, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Mulinari, S. Monoamine theories of depression: Historical impact on biomedical research. J. Hist. Neurosci. 2012, 21, 366–392. [Google Scholar] [CrossRef] [PubMed]
- Melander, H.; Salmonson, T.; Abadie, E.; van Zwieten-Boot, B. A regulatory apologia—A review of placebo-controlled studies in regulatory submissions of new-generation antidepressants. Eur. Neuropsychopharmacol. 2008, 18, 623–627. [Google Scholar] [CrossRef] [PubMed]
- Walsh, B.T.; Seidman, S.N.; Sysko, R.; Gould, M. Placebo response in studies of major depression: Variable, substantial, and growing. Jama 2002, 287, 1840–1847. [Google Scholar] [CrossRef] [PubMed]
- Hindmarch, I. Beyond the monoamine hypothesis: Mechanisms, molecules and methods. Eur. Psychiatry 2002, 17, 294s–299s. [Google Scholar] [CrossRef]
- Owens, M.J. Selectivity of antidepressants: From the monoamine hypothesis of depression to the SSRI revolution and beyond. J. Clin. Psychiatry 2004, 65, 5–10. [Google Scholar] [PubMed]
- Young, S.N. Acute tryptophan depletion in humans: A review of theoretical, practical and ethical aspects. J. Psychiatry Neurosci. JPN 2013, 38, 294–305. [Google Scholar] [CrossRef] [PubMed]
- Meneses, A. 5-HT system and cognition. Neurosci. Biobehav. R. 1999, 23, 1111–1125. [Google Scholar] [CrossRef]
- Buhot, M.C.; Martin, S.; Segu, L. Role of serotonin in memory impairment. Ann. Med. 2000, 32, 210–221. [Google Scholar] [CrossRef] [PubMed]
- Ogren, S.O.; Eriksson, T.M.; Elvander-Tottie, E.; D’Addario, C.; Ekstrom, J.C.; Svenningsson, P.; Meister, B.; Kehr, J.; Stiedl, O. The role of 5-HT(1A) receptors in learning and memory. Behav. Brain Res. 2008, 195, 54–77. [Google Scholar] [CrossRef] [PubMed]
- Sharma, T.; Mockler, D. The cognitive efficacy of atypical antipsychotics in schizophrenia. J. Clin. Psychopharm. 1998, 18, 12s–19s. [Google Scholar] [CrossRef]
- Bockaert, J.; Claeysen, S.; Compan, V.; Dumuis, A. 5-HT4 receptors, a place in the sun: Act two. Curr. Opin. Pharmacol. 2011, 11, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Wolf, H. Preclinical and clinical pharmacology of the 5-HT3 receptor antagonists. Scand. J. Rheumatol. 2000, 29, 37–45. [Google Scholar] [CrossRef]
- Cowen, P.; Sherwood, A.C. The role of serotonin in cognitive function: Evidence from recent studies and implications for understanding depression. J. Psychopharmacol. 2013, 27, 575–583. [Google Scholar] [CrossRef] [PubMed]
- Shopsin, B.; Friedman, E.; Gershon, S. Parachlorophenylalanine reversal of tranylcypromine effects in depressed patients. Arch. Gen. Psychiatry 1976, 33, 811–819. [Google Scholar] [CrossRef] [PubMed]
- Biskup, C.S.; Sanchez, C.L.; Arrant, A.; van Swearingen, A.E.; Kuhn, C.; Zepf, F.D. Effects of acute tryptophan depletion on brain serotonin function and concentrations of dopamine and norepinephrine in C57BL/6J and BALB/cJ mice. PLoS ONE 2012, 7, e35916. [Google Scholar] [CrossRef] [PubMed]
- Ardis, T.C.; Cahir, M.; Elliott, J.J.; Bell, R.; Reynolds, G.P.; Cooper, S.J. Effect of acute tryptophan depletion on noradrenaline and dopamine in the rat brain. J. Psychopharmacol. 2009, 23, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Jans, L.A.W.; Korte-Bouws, G.A.H.; Korte, S.M.; Blokland, A. The effects of acute tryptophan depletion on affective behaviour and cognition in brown norway and sprague dawley rats. J. Psychopharmacol. 2010, 24, 605–614. [Google Scholar] [CrossRef] [PubMed]
- Lieben, C.K.; Blokland, A.; Westerink, B.; Deutz, N.E. Acute tryptophan and serotonin depletion using an optimized tryptophan-free protein-carbohydrate mixture in the adult rat. Neurochem. Int. 2004, 44, 9–16. [Google Scholar] [CrossRef]
- Young, S.N.; Ervin, F.R.; Pihl, R.O.; Finn, P. Biochemical aspects of tryptophan depletion in primates. Psychopharmacology 1989, 98, 508–511. [Google Scholar] [CrossRef] [PubMed]
- Young, S.N.; Smith, S.E.; Pihl, R.O.; Ervin, F.R. Tryptophan depletion causes a rapid lowering of mood in normal males. Psychopharmacology 1985, 87, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Riedel, W.J.; Klaassen, T.; Deutz, N.E.; van Someren, A.; van Praag, H.M. Tryptophan depletion in normal volunteers produces selective impairment in memory consolidation. Psychopharmacology 1999, 141, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Cahir, M.; Ardis, T.; Reynolds, G.P.; Cooper, S.J. Acute and chronic tryptophan depletion differentially regulate central 5-HT 1A and 5-HT 2A receptor binding in the rat. Psychopharmacology 2007, 190, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Nishizawa, S.; Benkelfat, C.; Young, S.N.; Leyton, M.; Mzengeza, S.; de Montigny, C.; Blier, P.; Diksic, M. Differences between males and females in rates of serotonin synthesis in human brain. Proc. Natl. Acad. Sci. USA 1997, 94, 5308–5313. [Google Scholar] [CrossRef] [PubMed]
- Williams, W.A.; Shoaf, S.E.; Hommer, D.; Rawlings, R.; Linnoila, M. Effects of acute tryptophan depletion on plasma and cerebrospinal fluid tryptophan and 5-hydroxyindoleacetic acid in normal volunteers. J. Neurochem. 1999, 72, 1641–1647. [Google Scholar] [CrossRef] [PubMed]
- Moreno, F.A.; Parkinson, D.; Palmer, C.; Castro, W.L.; Misiaszek, J.; el Khoury, A.; Mathe, A.A.; Wright, R.; Delgado, P.L. CSF neurochemicals during tryptophan depletion in individuals with remitted depression and healthy controls. Eur. Neuropsychopharmacol. 2010, 20, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Young, S.N.; Leyton, M. The role of serotonin in human mood and social interaction. Insight from altered tryptophan levels. Pharmacol. Biochem. Behav. 2002, 71, 857–865. [Google Scholar] [CrossRef]
- Toker, L.; Amar, S.; Bersudsky, Y.; Benjamin, J.; Klein, E.; Agam, G. The biology of tryptophan depletion and mood disorders. Israel J. Psychiatry Relat. Sci. 2010, 47, 46–55. [Google Scholar]
- Mace, J.L.; Porter, R.J.; Dalrymple-Alford, J.C.; Wesnes, K.A.; Anderson, T.J. The effects of acute tryptophan depletion on neuropsychological function, mood and movement in the healthy elderly. J. Psychopharmacol. 2011, 25, 1337–1343. [Google Scholar] [CrossRef] [PubMed]
- Hughes, J.H.; Gallagher, P.; Stewart, M.E.; Matthews, D.; Kelly, T.P.; Young, A.H. The effects of acute tryptophan depletion on neuropsychological function. J. Psychopharmacol. 2003, 17, 300–309. [Google Scholar] [CrossRef] [PubMed]
- Ellenbogen, M.A.; Young, S.N.; Dean, P.; Palmour, R.M.; Benkelfat, C. Mood response to acute tryptophan depletion in healthy volunteers: Sex differences and temporal stability. Neuropsychopharmacology 1996, 15, 465–474. [Google Scholar] [CrossRef]
- Feder, A.; Skipper, J.; Blair, J.R.; Buchholz, K.; Mathew, S.J.; Schwarz, M.; Doucette, J.T.; Alonso, A.; Collins, K.A.; Neumeister, A.; et al. Tryptophan depletion and emotional processing in healthy volunteers at high risk for depression. Biol. Psychiatry 2011, 69, 804–807. [Google Scholar] [CrossRef] [PubMed]
- Van der Veen, F.M.; Evers, E.A.T.; Deutz, N.E.P.; Schmitt, J.A.J. Effects of acute tryptophan depletion on mood and facial emotion perception related brain activation and performance in healthy women with and without a family history of depression. Neuropsychopharmacology 2007, 32, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.A.; Fairburn, C.G.; Cowen, P.J. Relapse of depression after rapid depletion of tryptophan. Lancet 1997, 349, 915–919. [Google Scholar] [CrossRef]
- Moreno, F.A.; Gelenberg, A.J.; Heninger, G.R.; Potter, R.L.; McKnight, K.M.; Allen, J.; Phillips, A.P.; Delgado, P.L. Tryptophan depletion and depressive vulnerability. Biol. Psychiatry 1999, 46, 498–505. [Google Scholar] [CrossRef]
- Booij, L.; van der Does, A.J.W.; Haffmans, P.M.J.; Riedel, W.J.; Fekkes, D.; Blom, M.J.B. The effects of high-dose and low-dose tryptophan depletion on mood and cognitive functions of remitted depressed patients. J. Psychopharmacol. 2005, 19, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Booij, L.; van der Does, A.J.; Haffmans, P.M.; Riedel, W.J. Acute tryptophan depletion in depressed patients treated with a selective serotonin-noradrenalin reuptake inhibitor: Augmentation of antidepressant response? J. Affect. Disord. 2005, 86, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Delgado, P.L.; Price, L.H.; Miller, H.L.; Salomon, R.M.; Licinio, J.; Krystal, J.H.; Heninger, G.R.; Charney, D.S. Rapid serotonin depletion as a provocative challenge test for patients with major depression: Relevance to antidepressant action and the neurobiology of depression. Psychopharmacol. Bull. 1991, 27, 321–330. [Google Scholar] [PubMed]
- Zhang, L.M.; Guadarrama, L.; Corona-Morales, A.A.; Vega-Gonzalez, A.; Rocha, L.; Escobar, A. Rats subjected to extended l-tryptophan restriction during early postnatal stage exhibit anxious-depressive features and structural changes. J. Neuropathol. Exp. Neur. 2006, 65, 562–570. [Google Scholar] [CrossRef]
- Uchida, S.; Kitamoto, A.; Umeeda, H.; Nakagawa, N.; Masushige, S.; Kida, S. Chronic reduction in dietary tryptophan leads to changes in the emotional response to stress in mice. J. Nutr. Sci. Vitaminol. 2005, 51, 175–181. [Google Scholar] [CrossRef] [PubMed]
- van Donkelaar, E.L.; Blokland, A.; Lieben, C.K.J.; Kenis, G.; Ferrington, L.; Kelly, P.A.T.; Steinbusch, H.W.M.; Prickaerts, J. Acute tryptophan depletion in C57BL/6 mice does not induce central serotonin reduction or affective behavioural changes. Neurochem. Int. 2010, 56, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Mendelsohn, D.; Riedel, W.J.; Sambeth, A. Effects of acute tryptophan depletion on memory, attention and executive functions: A systematic review. Neurosci. Biobehav. R. 2009, 33, 926–952. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, J.A.; Jorissen, B.L.; Sobczak, S.; van Boxtel, M.P.; Hogervorst, E.; Deutz, N.E.; Riedel, W.J. Tryptophan depletion impairs memory consolidation but improves focussed attention in healthy young volunteers. J. Psychopharmacol. 2000, 14, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Booij, L.; van der Does, A.J.W. Emotional processing as a predictor of symptom change: An acute tryptophan depletion study in depressed patients. Eur. Neuropsychopharm. 2011, 21, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Beacher, F.D.C.C.; Gray, M.A.; Minati, L.; Whale, R.; Harrison, N.A.; Critchley, H.D. Acute tryptophan depletion attenuates conscious appraisal of social emotional signals in healthy female volunteers. Psychopharmacology 2011, 213, 603–613. [Google Scholar] [CrossRef] [PubMed]
- Epperson, C.N.; Amin, Z.; Ruparel, K.; Gur, R.; Loughead, J. Interactive effects of estrogen and serotonin on brain activation during working memory and affective processing in menopausal women. Psychoneuroendocrino 2012, 37, 372–382. [Google Scholar] [CrossRef] [PubMed]
- Mace, J.L.; Porter, R.J.; Dalrymple-Alford, J.C.; Wesnes, K.A.; Anderson, T.J. Effects of acute tryptophan depletion on neuropsychological and motor function in parkinson’s disease. J. Psychopharmacol. 2010, 24, 1465–1472. [Google Scholar] [CrossRef] [PubMed]
- Zepf, F.D.; Landgraf, M.; Biskup, C.S.; Dahmen, B.; Poustka, F.; Wockel, L.; Stadler, C. No effect of acute tryptophan depletion on verbal declarative memory in young persons with adhd. Acta Psychiatr. Scand. 2013, 128, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Crean, J.; Richards, J.B.; de Wit, H. Effect of tryptophan depletion on impulsive behavior in men with or without a family history of alcoholism. Behav. Brain Res. 2002, 136, 349–357. [Google Scholar] [CrossRef]
- Porter, R.J.; Lunn, B.S.; O’Brien, J.T. Effects of acute tryptophan depletion on cognitive function in Alzheimer’s disease and in the healthy elderly. Psychol. Med. 2003, 33, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, P.J.; Clarke, G.; O’Neill, A.; Groeger, J.A.; Quigley, E.M.; Shanahan, F.; Cryan, J.F.; Dinan, T.G. Cognitive performance in irritable bowel syndrome: Evidence of a stress-related impairment in visuospatial memory. Psychol. Med. 2014, 44, 1553–1566. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, P.J.; Allen, A.P.; O’Neill, A.; Quigley, E.M.; Cryan, J.F.; Dinan, T.G.; Clarke, G. Acute tryptophan depletion reduces kynurenine levels: Implications for treatment of impaired visuospatial memory performance in irritable bowel syndrome. Psychopharmacology 2015, 232, 1357–1371. [Google Scholar] [CrossRef] [PubMed]
- Von Ah, D.; Skaar, T.; Unverzagt, F.; Yu, M.G.; Wu, J.W.; Schneider, B.; Storniolo, A.M.; Moser, L.; Ryker, K.; Milata, J.; et al. Evaluating the role of serotonin on neuropsychological function after breast cancer using acute tryptophan depletion. Biol. Res. Nurs. 2012, 14, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Blokland, A.; Lieben, C.; Deutz, N.E.P. Anxiogenic and depressive-like effects, but no cognitive deficits, after repeated moderate tryptophan depletion in the rat. J. Psychopharmacol. 2002, 16, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Lieben, C.K.; van Oorsouw, K.; Deutz, N.E.; Blokland, A. Acute tryptophan depletion induced by a gelatin-based mixture impairs object memory but not affective behavior and spatial learning in the rat. Behav. Brain Res. 2004, 151, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Rutten, K.; Lieben, C.; Smits, L.; Blokland, A. The PDE4 inhibitor rolipram reverses object memory impairment induced by acute tryptophan depletion in the rat. Psychopharmacology 2007, 192, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Van Donkelaar, E.L.; Rutten, K.; Blokland, A.; Akkerman, S.; Steinbusch, H.W.; Prickaerts, J. Phosphodiesterase 2 and 5 inhibition attenuates the object memory deficit induced by acute tryptophan depletion. Eur. J. Pharmacol. 2008, 600, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.P.; Zhou, J.; Fang, L.; Liu, Z.; Fan, S.H.; Xie, P. Acute tryptophan depletion reduces nitric oxide synthase in the rat hippocampus. Neurochem. Res. 2013, 38, 2595–2603. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, T.A.; Elliott, J.J.; Ardis, T.C.; Cahir, M.; Reynolds, G.P.; Bell, R.; Cooper, S.J. Tryptophan depletion impairs object-recognition memory in the rat: Reversal by risperidone. Behav. Brain Res. 2010, 208, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Uchida, S.; Umeeda, H.; Kitamoto, A.; Masushige, S.; Kida, S. Chronic reduction in dietary tryptophan leads to a selective impairment of contextual fear memory in mice. Brain Res. 2007, 1149, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Bortolato, M.; Frau, R.; Orru, M.; Collu, M.; Mereu, G.; Carta, M.; Fadda, F.; Stancampiano, R. Effects of tryptophan deficiency on prepulse inhibition of the acoustic startle in rats. Psychopharmacology 2008, 198, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Carta, M.; Fadda, F.; Stancampiano, R. Tryptophan-deficient diet increases the neurochemical and behavioral response to amphetamine. Brain Res. 2006, 1094, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Mohajeri, M.H.; Wittwer, J.; Vargas, K.; Hogan, E.; Holmes, A.; Rogers, P.J.; Goralczyk, R.; Gibson, E.L. Chronic treatment with a tryptophan-rich protein hydrolysate improves emotional processing, mental energy levels and reaction time in middle-aged women. Br. J. Nutr. 2015, 113, 350–365. [Google Scholar] [CrossRef] [PubMed]
- Booij, L.; Merens, W.; Markus, C.R.; van der Does, A.J.W. Diet rich in alpha-lactalbumin improves memory in unmedicated recovered depressed patients and matched controls. J. Psychopharmacol. 2006, 20, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Murphy, S.E.; Longhitano, C.; Ayres, R.E.; Cowen, P.J.; Harmer, C.J. Tryptophan supplementation induces a positive bias in the processing of emotional material in healthy female volunteers. Psychopharmacology 2006, 187, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Rondanelli, M.; Opizzi, A.; Faliva, M.; Mozzoni, M.; Antoniello, N.; Cazzola, R.; Savare, R.; Cerutti, R.; Grossi, E.; Cestaro, B. Effects of a diet integration with an oily emulsion of DHA-phospholipids containing melatonin and tryptophan in elderly patients suffering from mild cognitive impairment. Nutr. Neurosci. 2012, 15, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, E. Effects of l-tryptophan on sleepiness and on sleep. J. Psychiatr. Res. 1982, 17, 107–113. [Google Scholar] [CrossRef]
- Silber, B.Y.; Schmitt, J.A. Effects of tryptophan loading on human cognition, mood, and sleep. Neurosci. Biobehav. Rev. 2010, 34, 387–407. [Google Scholar] [CrossRef] [PubMed]
- Markus, C.R.; Jonkman, L.M.; Lammers, J.H.; Deutz, N.E.; Messer, M.H.; Rigtering, N. Evening intake of alpha-lactalbumin increases plasma tryptophan availability and improves morning alertness and brain measures of attention. Am. J. Clin. Nutr. 2005, 81, 1026–1033. [Google Scholar] [PubMed]
- Bhatti, T.; Gillin, J.C.; Seifritz, E.; Moore, P.; Clark, C.; Golshan, S.; Stahl, S.; Rapaport, M.; Kelsoe, J. Effects of a tryptophan-free amino acid drink challenge on normal human sleep electroencephalogram and mood. Biol. Psychiatry 1998, 43, 52–59. [Google Scholar] [CrossRef]
- Carhart-Harris, R.L.; Nutt, D.J.; Munafo, M.R.; Christmas, D.M.; Wilson, S.J. Equivalent effects of acute tryptophan depletion on rem sleep in ecstasy users and controls. Psychopharmacology 2009, 206, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Nakamaru-Ogiso, E.; Miyamoto, H.; Hamada, K.; Tsukada, K.; Takai, K. Novel biochemical manipulation of brain serotonin reveals a role of serotonin in the circadian rhythm of sleep-wake cycles. Eur. J. Neurosci. 2012, 35, 1762–1770. [Google Scholar] [CrossRef] [PubMed]
- Tsuno, N.; Besset, A.; Ritchie, K. Sleep and depression. J. Clin. Psychiatry 2005, 66, 1254–1269. [Google Scholar] [CrossRef] [PubMed]
- Beasley, C.M.; Sayler, M.E.; Weiss, A.M.; Potvin, J.H. Fluoxetine-activating and sedating effects at multiple fixed doses. J. Clin. Psychopharm. 1992, 12, 328–333. [Google Scholar] [CrossRef]
- Moore, P.; Gillin, J.C.; Bhatti, T.; DeModena, A.; Seifritz, E.; Clark, C.; Stahl, S.; Rapaport, M.; Kelsoe, J. Rapid tryptophan depletion, sleep electroencephalogram, and mood in men with remitted depression on serotonin reuptake inhibitors. Arch. Gen. Psychiatry 1998, 55, 534–539. [Google Scholar] [CrossRef] [PubMed]
- Landolt, H.P.; Kelsoe, J.R.; Rapaport, M.H.; Gillin, J.C. Rapid tryptophan depletion reverses phenelzine-induced suppression of rem sleep. J. Sleep Res. 2003, 12, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Voderholzer, U.; Riemann, D.; Huwig-Poppe, C.; Kuelz, A.K.; Kordon, A.; Bruestle, K.; Berger, M.; Hohagen, F. Sleep in obsessive compulsive disorder-polysomnographic studies under baseline conditions and after experimentally induced serotonin deficiency. Eur. Arch. Psychiatry Clin. Neurosci. 2007, 257, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Sansone, R.A.; Sansone, L.A. SSRIs: Bad to the bone? Innov. Clin. Neurosci. 2012, 9, 42–47. [Google Scholar] [PubMed]
- Crane, J.D.; Palanivel, R.; Mottillo, E.P.; Bujak, A.L.; Wang, H.; Ford, R.J.; Collins, A.; Blumer, R.M.; Fullerton, M.D.; Yabut, J.M.; et al. Inhibiting peripheral serotonin synthesis reduces obesity and metabolic dysfunction by promoting brown adipose tissue thermogenesis. Nat. Med. 2015, 21, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Gershon, M.D. Review article: Roles played by 5-hydroxytryptamine in the physiology of the bowel. Aliment. Pharm. Ther. 1999, 13, 15–30. [Google Scholar] [CrossRef]
- Amireault, P.; Sibon, D.; Cote, F. Life without peripheral serotonin: Insights from tryptophan hydroxylase 1 knockout mice reveal the existence of paracrine/autocrine serotonergic networks. ACS Chem. Neurosci. 2013, 4, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Sakakibara, Y.; Katoh, M.; Kawayanagi, T.; Nadai, M. Species and tissue differences in serotonin glucuronidation. Xenobiotica 2015, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Jandhyala, S.M.; Talukdar, R.; Subramanyam, C.; Vuyyuru, H.; Sasikala, M.; Reddy, D.N. Role of the normal gut microbiota. World J. Gastroenterol. 2015, 21, 8787–8803. [Google Scholar] [CrossRef] [PubMed]
- La Rosa, P.S.; Warner, B.B.; Zhou, Y.; Weinstock, G.M.; Sodergren, E.; Hall-Moore, C.M.; Stevens, H.J.; Bennett, W.E., Jr.; Shaikh, N.; Linneman, L.A.; et al. Patterned progression of bacterial populations in the premature infant gut. Proc. Natl. Acad. Sci. USA 2014, 111, 12522–12527. [Google Scholar] [CrossRef] [PubMed]
- O’Mahony, S.M.; Clarke, G.; Borre, Y.E.; Dinan, T.G.; Cryan, J.F. Serotonin, tryptophan metabolism and the brain-gut-microbiome axis. Behav. Brain Res. 2015, 277, 32–48. [Google Scholar] [CrossRef] [PubMed]
- Clarke, G.; Grenham, S.; Scully, P.; Fitzgerald, P.; Moloney, R.D.; Shanahan, F.; Dinan, T.G.; Cryan, J.F. The microbiome-gut-brain axis during early life regulates the hippocampal serotonergic system in a sex-dependent manner. Mol. Psychiatry 2013, 18, 666–673. [Google Scholar] [CrossRef] [PubMed]
- Wikoff, W.R.; Anfora, A.T.; Liu, J.; Schultz, P.G.; Lesley, S.A.; Peters, E.C.; Siuzdak, G. Metabolomics analysis reveals large effects of gut microflora on mammalian blood metabolites. Proc. Natl. Acad. Sci. USA 2009, 106, 3698–3703. [Google Scholar] [CrossRef] [PubMed]
- Diaz Heijtz, R.; Wang, S.; Anuar, F.; Qian, Y.; Bjorkholm, B.; Samuelsson, A.; Hibberd, M.L.; Forssberg, H.; Pettersson, S. Normal gut microbiota modulates brain development and behavior. Proc. Natl. Acad. Sci. USA 2011, 108, 3047–3052. [Google Scholar] [CrossRef] [PubMed]
- Spiller, R. Serotonin and GI clinical disorders. Neuropharmacology 2008, 55, 1072–1080. [Google Scholar] [CrossRef] [PubMed]
- Nakai, A.; Kumakura, Y.; Boivin, M.; Rosa, P.; Diksic, M.; D’Souza, D.; Kersey, K. Sex differences of brain serotonin synthesis in patients with irritable bowel syndrome using alpha-[11c]methyl-l-tryptophan, positron emission tomography and statistical parametric mapping. Can. J. Gastroenterol. 2003, 17, 191–196. [Google Scholar] [PubMed]
- Brint, E.K.; MacSharry, J.; Fanning, A.; Shanahan, F.; Quigley, E.M.M. Differential expression of toll-like receptors in patients with irritable bowel syndrome. Am. J. Gastroenterol. 2011, 106, 329–336. [Google Scholar] [CrossRef] [PubMed]
- McKernan, D.P.; Gaszner, G.; Quigley, E.M.; Cryan, J.F.; Dinan, T.G. Altered peripheral toll-like receptor responses in the irritable bowel syndrome. Aliment. Pharm. Ther. 2011, 33, 1045–1052. [Google Scholar] [CrossRef] [PubMed]
- Reigstad, C.S.; Salmonson, C.E.; Rainey, J.F.; Szurszewski, J.H.; Linden, D.R.; Sonnenburg, J.L.; Farrugia, G.; Kashyap, P.C. Gut microbes promote colonic serotonin production through an effect of short-chain fatty acids on enterochromaffin cells. FASEB J. 2015, 29, 1395–1403. [Google Scholar] [CrossRef] [PubMed]
- Neufeld, K.M.; Kang, N.; Bienenstock, J.; Foster, J.A. Reduced anxiety-like behavior and central neurochemical change in germ-free mice. Neurogastroenterol. Motil. 2011, 23, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Bravo, J.A.; Forsythe, P.; Chew, M.V.; Escaravage, E.; Savignac, H.M.; Dinan, T.G.; Bienenstock, J.; Cryan, J.F. Ingestion of lactobacillus strain regulates emotional behavior and central gaba receptor expression in a mouse via the vagus nerve. Proc. Natl. Acad. Sci. USA 2011, 108, 16050–16055. [Google Scholar] [CrossRef] [PubMed]
- O’Mahony, S.M.; Marchesi, J.R.; Scully, P.; Codling, C.; Ceolho, A.M.; Quigley, E.M.M.; Cryan, J.F.; Dinan, T.G. Early life stress alters behavior, immunity, and microbiota in rats: Implications for irritable bowel syndrome and psychiatric illnesses. Biol. Psychiatry 2009, 65, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Park, A.J.; Collins, J.; Blennerhassett, P.A.; Ghia, J.E.; Verdu, E.F.; Bercik, P.; Collins, S.M. Altered colonic function and microbiota profile in a mouse model of chronic depression. Neurogastroenterol. Motil. 2013, 25. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.Y.; Ling, Z.X.; Zhang, Y.H.; Mao, H.J.; Ma, Z.P.; Yin, Y.; Wang, W.H.; Tang, W.X.; Tan, Z.L.; Shi, J.F.; et al. Altered fecal microbiota composition in patients with major depressive disorder. Brain Behav. Immun. 2015, 48, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Gareau, M.G.; Wine, E.; Rodrigues, D.M.; Cho, J.H.; Whary, M.T.; Philpott, D.J.; MacQueen, G.; Sherman, P.M. Bacterial infection causes stress-induced memory dysfunction in mice. Gut 2011, 60, 307–317. [Google Scholar] [CrossRef] [PubMed]
- Desbonnet, L.; Clarke, G.; Shanahan, F.; Dinan, T.G.; Cryan, J.F. Microbiota is essential for social development in the mouse. Mol. Psychiatry 2014, 19, 146–148. [Google Scholar] [CrossRef] [PubMed]
- Bekkering, P.; Jafri, I.; van Overveld, F.J.; Rijkers, G.T. The intricate association between gut microbiota and development of type 1, type 2 and type 3 diabetes. Expert Rev. Clin. Immun. 2013, 9, 1031–1041. [Google Scholar] [CrossRef] [PubMed]
- Naseer, M.I.; Bibi, F.; Alqahtani, M.H.; Chaudhary, A.G.; Azhar, E.I.; Kamal, M.A.; Yasir, M. Role of gut microbiota in obesity, type 2 diabetes and Alzheimer’s disease. CNS Neurol. Disord. Drug 2014, 13, 305–311. [Google Scholar] [CrossRef]
- Labus, J.S.; Mayer, E.A.; Jarcho, J.; Kilpatrick, L.A.; Kilkens, T.O.C.; Evers, E.A.T.; Backes, W.H.; Brummer, R.J.M.; van Nieuwenhoven, M.A. Acute tryptophan depletion alters the effective connectivity of emotional arousal circuitry during visceral stimuli in healthy women. Gut 2011, 60, 1196–1203. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.M. Acute tryptophan depletion and functional brain imaging in irritable bowel syndrome. J. Neurogastroenterol. 2012, 18, 106–108. [Google Scholar] [CrossRef] [PubMed]
- Kilkens, T.O.; Honig, A.; van Nieuwenhoven, M.A.; Riedel, W.J.; Brummer, R.J. Acute tryptophan depletion affects brain-gut responses in irritable bowel syndrome patients and controls. Gut 2004, 53, 1794–1800. [Google Scholar] [CrossRef] [PubMed]
- Keszthelyi, D.; Troost, F.J.; Jonkers, D.M.; van Donkelaar, E.L.; Dekker, J.; Buurman, W.A.; Masclee, A.A. Does acute tryptophan depletion affect peripheral serotonin metabolism in the intestine? Am. J. Clin. Nutr. 2012, 95, 603–608. [Google Scholar] [CrossRef] [PubMed]
- Geeraerts, B.; van Oudenhove, L.; Boesmans, W.; Vos, R.; Vanden Berghe, P.; Tack, J. Influence of acute tryptophan depletion on gastric sensorimotor function in humans. Am. J. Physiol. 2011, 300, G228–G235. [Google Scholar] [CrossRef] [PubMed]
- Van Nieuwenhoven, M.A.; Kilkens, T.O. The effect of acute serotonergic modulation on rectal motor function in diarrhea-predominant irritable bowel syndrome and healthy controls. Eur. J. Gastroenterol. Hepatol. 2012, 24, 1259–1265. [Google Scholar] [CrossRef] [PubMed]
- Jun, S.E.; Kohen, R.; Cain, K.C.; Jarrett, M.E.; Heitkemper, M.M. Tph gene polymorphisms are associated with disease perception and quality of life in women with irritable bowel syndrome. Biol. Res. Nurs. 2014, 16, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Mayer, E.A.; Knight, R.; Mazmanian, S.K.; Cryan, J.F.; Tillisch, K. Gut microbes and the brain: Paradigm shift in neuroscience. J. Neurosci. 2014, 34, 15490–15496. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jenkins, T.A.; Nguyen, J.C.D.; Polglaze, K.E.; Bertrand, P.P. Influence of Tryptophan and Serotonin on Mood and Cognition with a Possible Role of the Gut-Brain Axis. Nutrients 2016, 8, 56. https://doi.org/10.3390/nu8010056
Jenkins TA, Nguyen JCD, Polglaze KE, Bertrand PP. Influence of Tryptophan and Serotonin on Mood and Cognition with a Possible Role of the Gut-Brain Axis. Nutrients. 2016; 8(1):56. https://doi.org/10.3390/nu8010056
Chicago/Turabian StyleJenkins, Trisha A., Jason C. D. Nguyen, Kate E. Polglaze, and Paul P. Bertrand. 2016. "Influence of Tryptophan and Serotonin on Mood and Cognition with a Possible Role of the Gut-Brain Axis" Nutrients 8, no. 1: 56. https://doi.org/10.3390/nu8010056
APA StyleJenkins, T. A., Nguyen, J. C. D., Polglaze, K. E., & Bertrand, P. P. (2016). Influence of Tryptophan and Serotonin on Mood and Cognition with a Possible Role of the Gut-Brain Axis. Nutrients, 8(1), 56. https://doi.org/10.3390/nu8010056