Impact of Leucine Supplementation on Exercise Training Induced Anti-Cardiac Remodeling Effect in Heart Failure Mice
Abstract
:1. Introduction
2. Material and Methods
2.1. Animals’ Care
2.2. Leucine Supplementation and Food Intake Measurement
2.3. Exercise Training
2.4. Cardiovascular Measurement
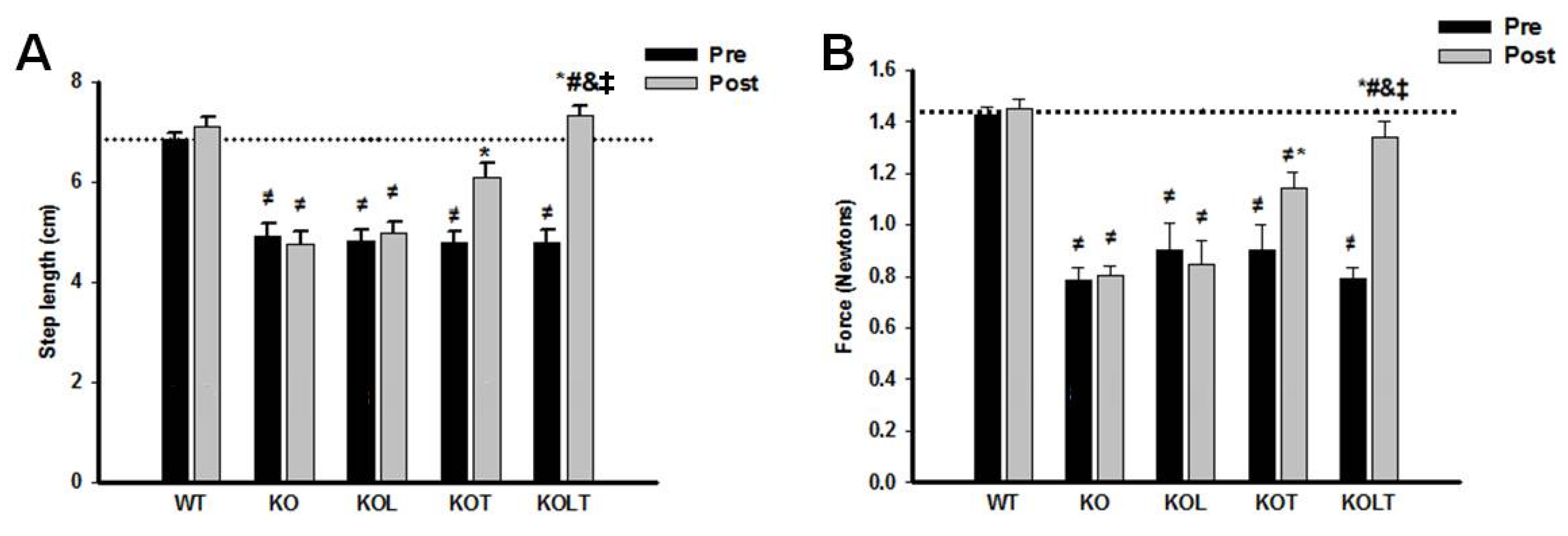
2.5. Skeletal Muscle Functional Assessment
2.6. Renal Parameters
2.7. Structural Analysis
2.8. Western Blot
2.9. Slot Blot
2.10. Assay of 26S Proteasome Activity
2.11. Statistical Analysis
3. Results
3.1. Physiologic Parameters
| Variable | WT | KO | KOL | KOT | KOLT |
|---|---|---|---|---|---|
| Body weight (g) | |||||
| Initial | 28.29 ± 1.62 (11) | 27.92 ± 0.82 (9) | 29.31 ± 0.87 (11) | 27.87 ± 1.49 (12) | 28.23 ± 1.25 (12) |
| Final | 30.31 ± 1.81 (11) | 27.52 ± 0.80 (9) ≠ | 27.43 ± 0.88 (11) ≠ | 27.63 ± 1.70 (12) ≠ | 28.24 ± 1.02 (12) ≠ |
| Food intake (g/g * 100) | |||||
| Initial | 8.79 ± 1.46 (8) | 8.47 ± 1.05 (8) | 8.50 ± 0.64 (8) | 8.92 ± 0.51 (8) | 8.86 ± 0.82 (8) |
| Final | 8.74 ± 0.92 (8) | 8.70 ± 0.67 (8) | 8.73 ± 0.81 (8) | 9.22 ± 0.84 (8) | 8.87 ± 0.47 (8) |
| Lung wet:dry ratio | 2.32 ± 1.32 (9) | 7.94 ± 1.82 (9) ≠ | 7.88 ± 2.00 (9) ≠ | 2.82 ± 1.30 (10) #& | 3.23 ± 1.14 (11) #& |
| Liver wet:dry ratio | 1.62 ± 0.24 (9) | 3.37 ± 0.41 (9) ≠ | 3.02 ± 0.70 (9) ≠ | 2.24 ± 0.40 (10) ≠#& | 2.24 ± 0.42 (11) ≠#& |
| RV (mg/mm * 100) | 1.53 ± 0.23 (9) | 1.54 ± 0.31 (9) | 1.40 ± 0.12 (9) | 1.37 ± 0.18 (10) | 1.30 ± 0.28 (11) |
| LV (mg/mm * 100) | 4.97 ± 0.46 (9) | 5.28 ± 0.68 (9) | 5.34 ± 0.56 (9) | 4.98 ± 0.61 (10) | 4.80 ± 1.14 (11) |
| Cardiac mass (mg/mm * 100) | 6.92 ± 0.61 (9) | 7.43 ± 0.85 (9) | 6.71 ± 0.69 (9) | 6.66 ± 1.37 (10) | 6.89 ± 0.56 (11) |
| Kidney mass (mg/mm * 100) | 0.17 ± 0.01 (8) | 0.17 ± 0.01 (8) | 0.18 ± 0.02 (9) | 0.18 ± 0.03 (8) | 0.19 ± 0.03 (8) |
| Blood urea nitrogen (mg/dL) | 35.91 ± 1.81 (8) | 40.74 ± 7.49 (8) | 39.54 ± 9.30 (8) | 36.00 ± 7.96 (8) | 36.05 ± 9.28 (8) |
| Urinary creatinine (mg/dL) | 3.98 ± 0.93 (8) | 3.27 ± 0.49 (8) | 3.12 ± 0.51 (8) | 4.44 ± 1.16 (8) #& | 4.52 ± 0.64 (8) #& |
| Blood creatinine (mg/dL) | 0.34 ± 0.06 (8) | 0.38 ± 0.02 (8) | 0.38 ± 0.05 (8) | 0.36 ± 0.04 (8) | 0.37 ± 0.08 (8) |
| Creatinine clearance (μL/min/kg) | 9.71 ± 1.81(8) | 12.27 ± 3.91 (8) | 9.20 ± 2.34 (8) | 18.04 ± 3.93 (8) ≠#& | 17.62 ± 6.55(8) ≠#& |
| Protein urinary (mg/24 h) | 2.68 ± 1.22 (8) | 10.32 ± 2.50 (8) ≠ | 9.06 ± 1.47 (8) ≠ | 6.07 ± 3.43 (8) ≠#& | 5.01 ± 1.67 (8) ≠#& |
| Lipid hydroperoxides (nmol/mg protein) | 224.36 ± 39.20 (7) | 468.49 ± 42.50 (7) ≠ | 472.40 ± 37.56 (7) ≠ | 312.14 ± 36.18 (7) #& | 308.73 ± 33.26 (7) #& |
3.2. Exercise Tolerance and HR
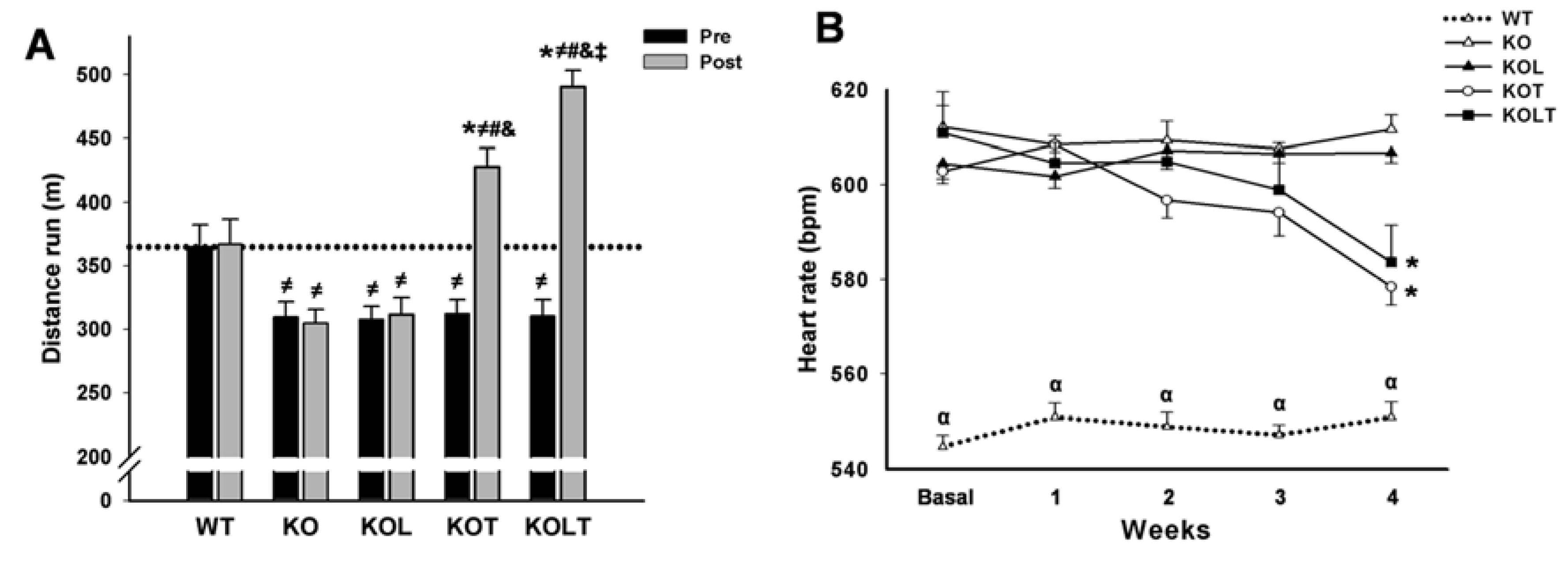
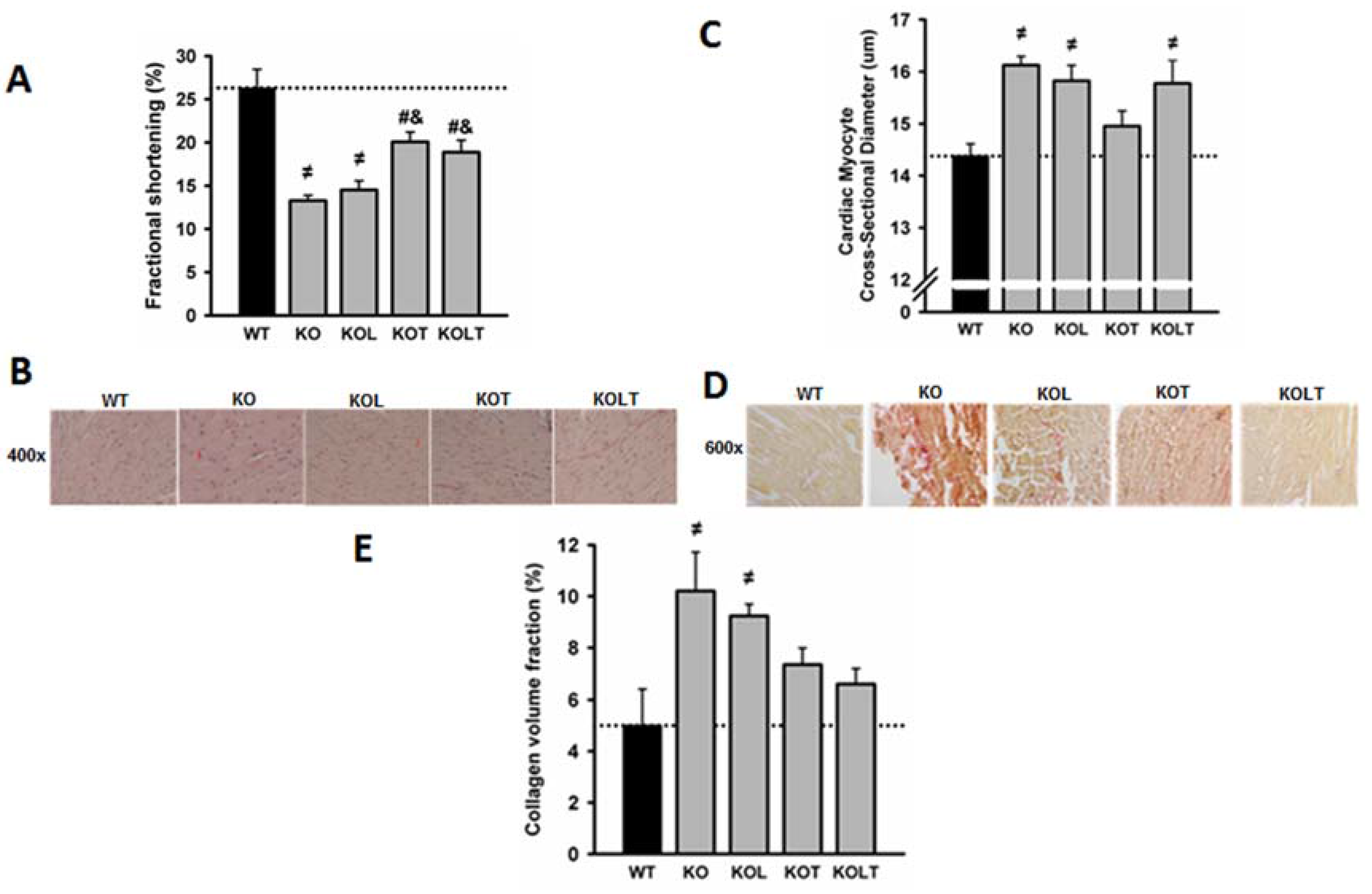
3.3. Systolic Function and Cardiac Morphology
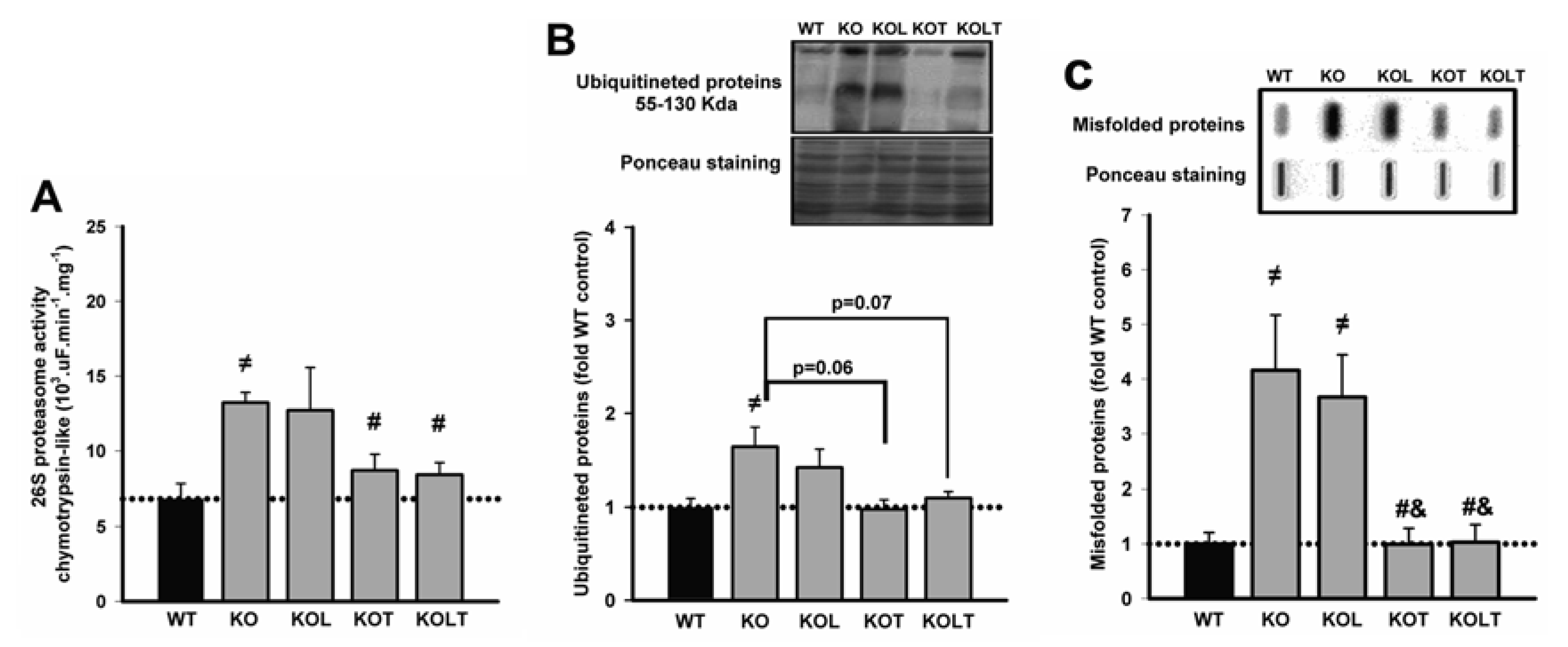
3.4. 26S-Proteasome Activity, Ubiquitinated and Misfolded Protein
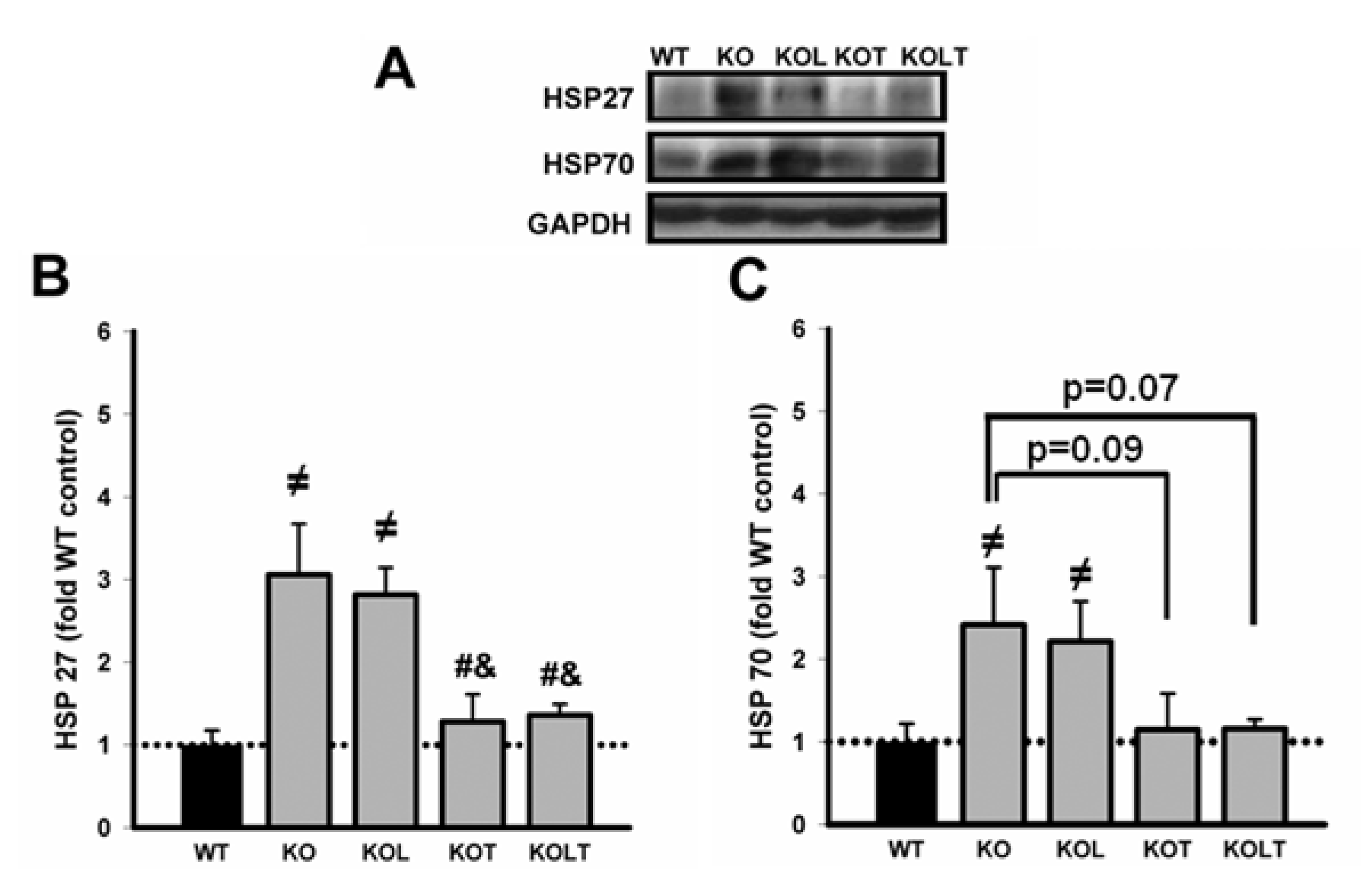
3.5. Heat Shock Protein Expression
3.6. Skeletal Muscle Functional
4. Discussion
5. Conclusions
Acknowledgements
Financial Support
Author Contributions
Conflicts of Interest
References
- Mann, D.L.; Bristow, M.R. Mechanisms and models in heart failure: The biomechanical model and beyond. Circulation 2005, 111, 2837–2849. [Google Scholar] [CrossRef]
- Gaudron, P.; Eilles, C.; Kugler, I.; Ertl, G. Progressive left ventricular dysfunction and remodeling after myocardial infarction. Potential mechanisms and early predictors. Circulation 1993, 87, 755–763. [Google Scholar]
- Predmore, J.M.; Wang, P.; Davis, F.; Bartolone, S.; Westfall, M.V.; Dyke, D.B.; Pagani, F.; Powell, S.R.; Day, S.M. Ubiquitin proteasome dysfunction in human hypertrophic and dilated cardiomyopathies. Circulation 2010, 121, 997–1004. [Google Scholar] [CrossRef] [PubMed]
- Tsukamoto, O.; Minamino, T.; Okada, K.; Shintani, Y.; Takashima, S.; Kato, H.; Liao, Y.; Okazaki, H.; Asai, M.; Hirata, A.; et al. Depression of proteasome activities during the progression of cardiac dysfunction in pressure-overloaded heart of mice. Biochem. Biophys. Res. Commun. 2006, 340, 1125–1133. [Google Scholar]
- Campos, J.C.; Queliconi, B.B.; Dourado, P.M.; Cunha, T.F.; Zambelli, V.O.; Bechara, L.R.; Kowaltowski, A.J.; Brum, P.C.; Mochly-Rosen, D.; Ferreira, J.C. Exercise training restores cardiac protein quality control in heart failure. PLoS ONE 2012, 7, e52764. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rolim, N.P.; Medeiros, A.; Rosa, K.T.; Mattos, K.C.; Irigoyen, M.C.; Krieger, E.M.; Krieger, J.E.; Negrao, C.E.; Brum, P.C. Exercise training improves the net balance of cardiac Ca2+ handling protein expression in heart failure. Physiol. Genomics 2007, 29, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.G.; Ferreira, J.C.; Bueno, C.R., Jr.; Mattos, K.C.; Rosa, K.T.; Irigoyen, M.C.; Oliveira, E.M.; Krieger, J.E.; Brum, P.C. Exercise training reduces cardiac angiotensin II levels and prevents cardiac dysfunction in a genetic model of sympathetic hyperactivity-induced heart failure in mice. Eur. J. Appl. Physiol. 2009, 105, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Brum, P.C.; Kosek, J.; Patterson, A.; Bernstein, D.; Kobilka, B. Abnormal cardiac function associated with sympathetic nervous system hyperactivity in mice. Am. J. Physiol. Heart Circ. Physiol. 2002, 283, H1838–H1845. [Google Scholar] [CrossRef] [PubMed]
- Cunha, T.F.; Moreira, J.B.; Paixao, N.A.; Campos, J.C.; Monteiro, A.W.; Bacurau, A.V.; Bueno, C.R., Jr.; Ferreira, J.C.; Brum, P.C. Aerobic exercise training upregulates skeletal muscle calpain and ubiquitin-proteasome systems in healthy mice. J. Appl. Physiol. 2012, 112, 1839–1846. [Google Scholar] [CrossRef] [PubMed]
- Cunha, T.F.; Bacurau, A.V.; Moreira, J.B.; Paixao, N.A.; Campos, J.C.; Ferreira, J.C.; Leal, M.L.; Negrao, C.E.; Moriscot, A.S.; Wisloff, U.; et al. Exercise training prevents oxidative stress and ubiquitin-proteasome system overactivity and reverse skeletal muscle atrophy in heart failure. PLoS ONE 2012, 7, e41701. [Google Scholar]
- Moreira, J.B.; Bechara, L.R.; Bozi, L.H.; Jannig, P.R.; Monteiro, A.W.; Dourado, P.M.; Wisloff, U.; Brum, P.C. High- versus moderate-intensity aerobic exercise training effects on skeletal muscle of infarcted rats. J. Appl. Physiol. 2013, 114, 1029–1041. [Google Scholar] [CrossRef] [PubMed]
- Salomao, E.M.; Toneto, A.T.; Silva, G.O.; Gomes-Marcondes, M.C. Physical exercise and a leucine-rich diet modulate the muscle protein metabolism in Walker tumor-bearing rats. Nutr. Cancer 2010, 62, 1095–1104. [Google Scholar] [CrossRef] [PubMed]
- D’Antona, G.; Ragni, M.; Cardile, A.; Tedesco, L.; Dossena, M.; Bruttini, F.; Caliaro, F.; Corsetti, G.; Bottinelli, R.; Carruba, M.O.; et al. Branched-chain amino acid supplementation promotes survival and supports cardiac and skeletal muscle mitochondrial biogenesis in middle-aged mice. Cell Metab. 2010, 12, 362–372. [Google Scholar]
- Scognamiglio, R.; Testa, A.; Aquilani, R.; Dioguardi, F.S.; Pasini, E. Impairment in walking capacity and myocardial function in the elderly: Is there a role for nonpharmacologic therapy with nutritional amino acid supplements? Am. J. Cardiol. 2008, 101, 78E–81E. [Google Scholar] [CrossRef] [PubMed]
- Rozentryt, P.; von Haehling, S.; Lainscak, M.; Nowak, J.U.; Kalantar-Zadeh, K.; Polonski, L.; Anker, S.D. The effects of a high-caloric protein-rich oral nutritional supplement in patients with chronic heart failure and cachexia on quality of life, body composition, and inflammation markers: a randomized, double-blind pilot study. J. Cachexia Sarcopenia Muscle 2010, 1, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Bacurau, A.V.; Jardim, M.A.; Ferreira, J.C.; Bechara, L.R.; Bueno, C.R., Jr.; Alba-Loureiro, T.C.; Negrao, C.E.; Casarini, D.E.; Curi, R.; Ramires, P.R.; et al. Sympathetic hyperactivity differentially affects skeletal muscle mass in developing heart failure: Role of exercise training. J. Appl. Physiol. 2009, 106, 1631–1640. [Google Scholar]
- Bartholomeu, J.B.; Vanzelli, A.S.; Rolim, N.P.; Ferreira, J.C.; Bechara, L.R.; Tanaka, L.Y.; Rosa, K.T.; Alves, M.M.; Medeiros, A.; Mattos, K.C.; et al. Intracellular mechanisms of specific beta-adrenoceptor antagonists involved in improved cardiac function and survival in a genetic model of heart failure. J. Mol. Cell. Cardiol. 2008, 45, 240–249. [Google Scholar]
- Vanzelli, A.S.; Medeiros, A.; Rolim, N.; Bartholomeu, J.B.; Cunha, T.F.; Bechara, L.R.; Gomes, E.R.; Mattos, K.C.; Sirvente, R.; Salemi, V.M.; et al. Integrative effect of carvedilol and aerobic exercise training therapies on improving cardiac contractility and remodeling in heart failure mice. PLoS ONE 2013, 8, e62452. [Google Scholar]
- Crozier, S.J.; Kimball, S.R.; Emmert, S.W.; Anthony, J.C.; Jefferson, L.S. Oral leucine administration stimulates protein synthesis in rat skeletal muscle. J. Nutr. 2005, 135, 376–382. [Google Scholar] [PubMed]
- Ferreira, J.C.; Rolim, N.P.; Bartholomeu, J.B.; Gobatto, C.A.; Kokubun, E.; Brum, P.C. Maximal lactate steady state in running mice: effect of exercise training. Clin. Exp. Pharmacol. Physiol. 2007, 34, 760–765. [Google Scholar] [CrossRef] [PubMed]
- Vanzelli, A.S.; Medeiros, A.; Sirvente Rde, A.; Salemi, V.M.; Mady, C.; Brum, P.C. Association of physical training with beta-blockers in heart failure in mice. Arq. Bras. Cardiol. 2010, 95, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Medeiros, A.; Vanzelli, A.S.; Rosa, K.T.; Irigoyen, M.C.; Brum, P.C. Effect of exercise training and carvedilol treatment on cardiac function and structure in mice with sympathetic hyperactivity-induced heart failure. Braz. J. Med. Biol. Res. 2008, 41, 812–817. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vieira, N.M.; Bueno, C.R., Jr.; Brandalise, V.; Moraes, L.V.; Zucconi, E.; Secco, M.; Suzuki, M.F.; Camargo, M.M.; Bartolini, P.; Brum, P.C.; et al. SJL dystrophic mice express a significant amount of human muscle proteins following systemic delivery of human adipose-derived stromal cells without immunosuppression. Stem Cells 2008, 26, 2391–2398. [Google Scholar]
- Anderson, K.D.; Abdul, M.; Steward, O. Quantitative assessment of deficits and recovery of forelimb motor function after cervical spinal cord injury in mice. Exp. Neurol. 2004, 190, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Nourooz-Zadeh, J.; Tajaddini-Sarmadi, J.; Wolff, S.P. Measurement of plasma hydroperoxide concentrations by the ferrous oxidation-xylenol orange assay in conjunction with triphenylphosphine. Anal. Biochem. 1994, 220, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Wang, X. The ubiquitin-proteasome system in cardiac proteinopathy: A quality control perspective. Cardiovasc. Res. 2010, 85, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.; Li, J.; Huang, W.; Su, H.; Liang, Q.; Tian, Z.; Horak, K.M.; Molkentin, J.D.; Wang, X. Proteasome functional insufficiency activates the calcineurin-NFAT pathway in cardiomyocytes and promotes maladaptive remodelling of stressed mouse hearts. Cardiovasc. Res. 2010, 88, 424–433. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Chen, Y.; Yang, Y.; Chen, B.; Liu, D.; Xiong, Z.; Zhang, C.; Dong, Y. Proteasome inhibition attenuates heart failure during the late stages of pressure overload through alterations in collagen expression. Biochem. Pharmacol. 2013, 85, 223–233. [Google Scholar] [CrossRef] [PubMed]
- De Andrade, L.H.; de Moraes, W.M.; Matsuo Junior, E.H.; de Moura, E.D.O.C.; Antunes, H.K.; Montemor, J.; Antonio, E.L.; Bocalini, D.S.; Serra, A.J.; Tucci, P.J.; et al. Aerobic exercise training improves oxidative stress and ubiquitin proteasome system activity in heart of spontaneously hypertensive rats. Mol. Cell. Biochem. 2015, 402, 193–202. [Google Scholar]
- Depre, C.; Wang, Q.; Yan, L.; Hedhli, N.; Peter, P.; Chen, L.; Hong, C.; Hittinger, L.; Ghaleh, B.; Sadoshima, J.; et al. Activation of the cardiac proteasome during pressure overload promotes ventricular hypertrophy. Circulation 2006, 114, 1821–1828. [Google Scholar]
- Knowlton, A.A.; Kapadia, S.; Torre-Amione, G.; Durand, J.B.; Bies, R.; Young, J.; Mann, D.L. Differential expression of heat shock proteins in normal and failing human hearts. J. Mol. Cell. Cardiol. 1998, 30, 811–818. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.S.; Ferreira, J.C.; Gomes, E.R.; Paixao, N.A.; Rolim, N.P.; Medeiros, A.; Guatimosim, S.; Brum, P.C. Cardiac anti-remodelling effect of aerobic training is associated with a reduction in the calcineurin/NFAT signalling pathway in heart failure mice. J. Physiol. 2009, 587, 3899–3910. [Google Scholar] [CrossRef] [PubMed]
- Medeiros, A.; Rolim, N.P.; Oliveira, R.S.; Rosa, K.T.; Mattos, K.C.; Casarini, D.E.; Irigoyen, M.C.; Krieger, E.M.; Krieger, J.E.; Negrao, C.E.; et al. Exercise training delays cardiac dysfunction and prevents calcium handling abnormalities in sympathetic hyperactivity-induced heart failure mice. J. Appl. Physiol. 2008, 104, 103–109. [Google Scholar]
- Mitrega, K.; Zorniak, M.; Varghese, B.; Lange, D.; Nozynski, J.; Porc, M.; Bialka, S.; Krzeminski, T.F. Beneficial effects of l-leucine and l-valine on arrhythmias, hemodynamics and myocardial morphology in rats. Pharmacol. Res. 2011, 64, 218–225. [Google Scholar] [CrossRef] [PubMed]
- Witham, W.G.; Yester, K.A.; McGaffin, K.R. A high leucine diet mitigates cardiac injury and improves survival after acute myocardial infarction. Metabolism 2013, 62, 290–302. [Google Scholar] [CrossRef] [PubMed]
- Lu, G.; Ren, S.; Korge, P.; Choi, J.; Dong, Y.; Weiss, J.; Koehler, C.; Chen, J.N.; Wang, Y. A novel mitochondrial matrix serine/threonine protein phosphatase regulates the mitochondria permeability transition pore and is essential for cellular survival and development. Genes Dev. 2007, 21, 784–796. [Google Scholar] [CrossRef] [PubMed]
- Tischler, M.E.; Desautels, M.; Goldberg, A.L. Does leucine, leucyl-tRNA, or some metabolite of leucine regulate protein synthesis and degradation in skeletal and cardiac muscle? J. Biol. Chem. 1982, 257, 1613–1621. [Google Scholar] [PubMed]
- Harrington, D.; Anker, S.D.; Chua, T.P.; Webb-Peploe, K.M.; Ponikowski, P.P.; Poole-Wilson, P.A.; Coats, A.J. Skeletal muscle function and its relation to exercise tolerance in chronic heart failure. J. Am. Coll. Cardiol. 1997, 30, 1758–1764. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Lu, G.; Gao, C.; Wang, Y.; Sun, H. Tissue-specific and nutrient regulation of the branched-chain alpha-keto acid dehydrogenase phosphatase, protein phosphatase 2Cm (PP2Cm). J. Biol. Chem. 2012, 287, 23397–23406. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.G.; Silva, M.T.; Carlassara, E.O.; Goncalves, D.A.; Abrahamsohn, P.A.; Kettelhut, I.C.; Moriscot, A.S.; Aoki, M.S.; Miyabara, E.H. Leucine supplementation accelerates connective tissue repair of injured tibialis anterior muscle. Nutrients 2014, 6, 3981–4001. [Google Scholar] [CrossRef] [PubMed]
- Anker, S.D.; Chua, T.P.; Ponikowski, P.; Harrington, D.; Swan, J.W.; Kox, W.J.; Poole-Wilson, P.A.; Coats, A.J. Hormonal changes and catabolic/anabolic imbalance in chronic heart failure and their importance for cardiac cachexia. Circulation 1997, 96, 526–534. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Moraes, W.M.A.M.; Melara, T.P.; De Souza, P.R.M.; De Salvi Guimarães, F.; Bozi, L.H.M.; Brum, P.C.; Medeiros, A. Impact of Leucine Supplementation on Exercise Training Induced Anti-Cardiac Remodeling Effect in Heart Failure Mice. Nutrients 2015, 7, 3751-3766. https://doi.org/10.3390/nu7053751
De Moraes WMAM, Melara TP, De Souza PRM, De Salvi Guimarães F, Bozi LHM, Brum PC, Medeiros A. Impact of Leucine Supplementation on Exercise Training Induced Anti-Cardiac Remodeling Effect in Heart Failure Mice. Nutrients. 2015; 7(5):3751-3766. https://doi.org/10.3390/nu7053751
Chicago/Turabian StyleDe Moraes, Wilson Max Almeida Monteiro, Thaís Plasti Melara, Pamella Ramona Moraes De Souza, Fabiana De Salvi Guimarães, Luiz Henrique Marchesi Bozi, Patricia Chakur Brum, and Alessandra Medeiros. 2015. "Impact of Leucine Supplementation on Exercise Training Induced Anti-Cardiac Remodeling Effect in Heart Failure Mice" Nutrients 7, no. 5: 3751-3766. https://doi.org/10.3390/nu7053751




