Cocoa Bioactive Compounds: Significance and Potential for the Maintenance of Skin Health
Abstract
:1. Introduction
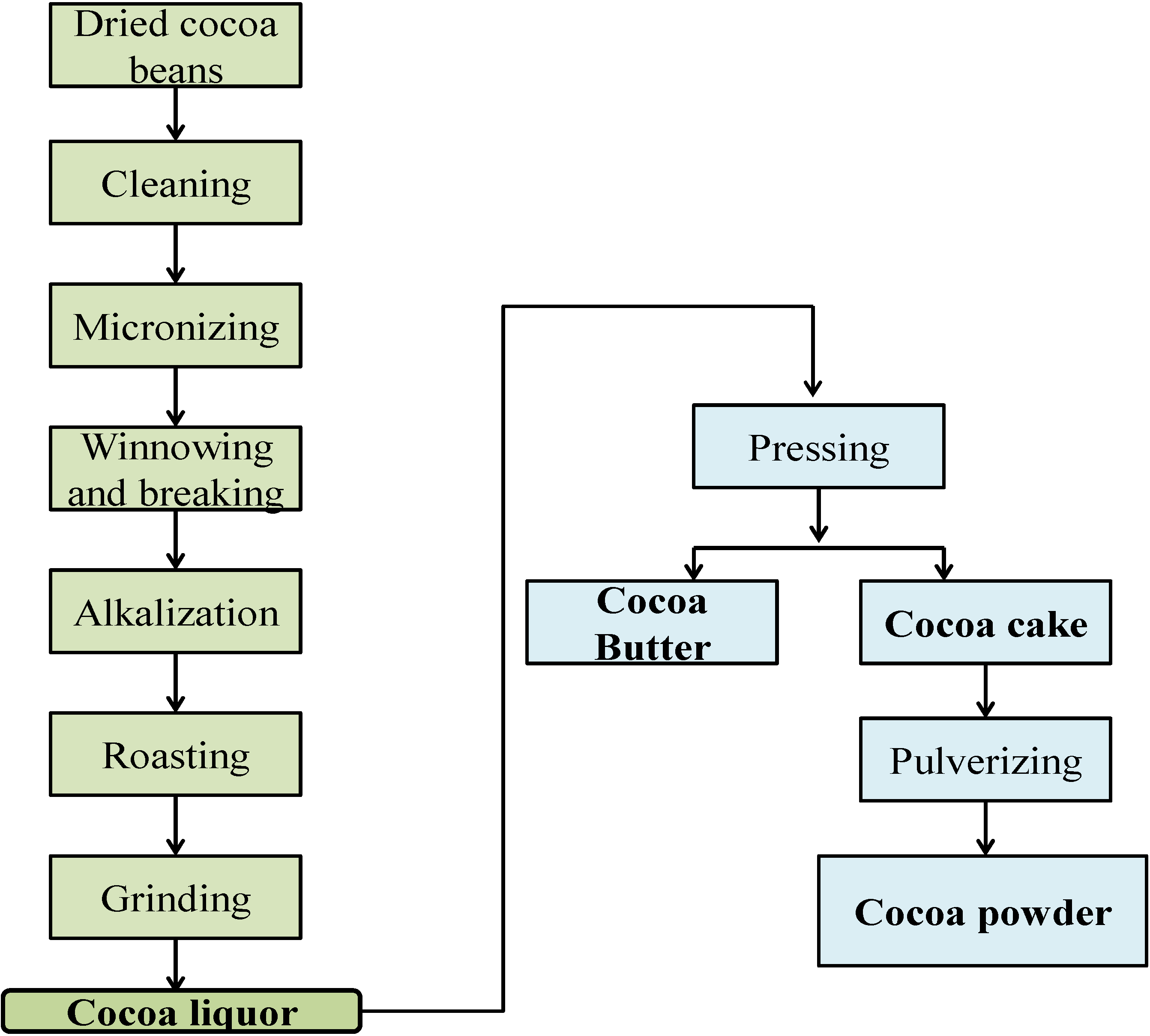
2. Cocoa Bioactive Compounds and Minerals
2.1. Polyphenols
2.2. Theobromine
2.3. Minerals
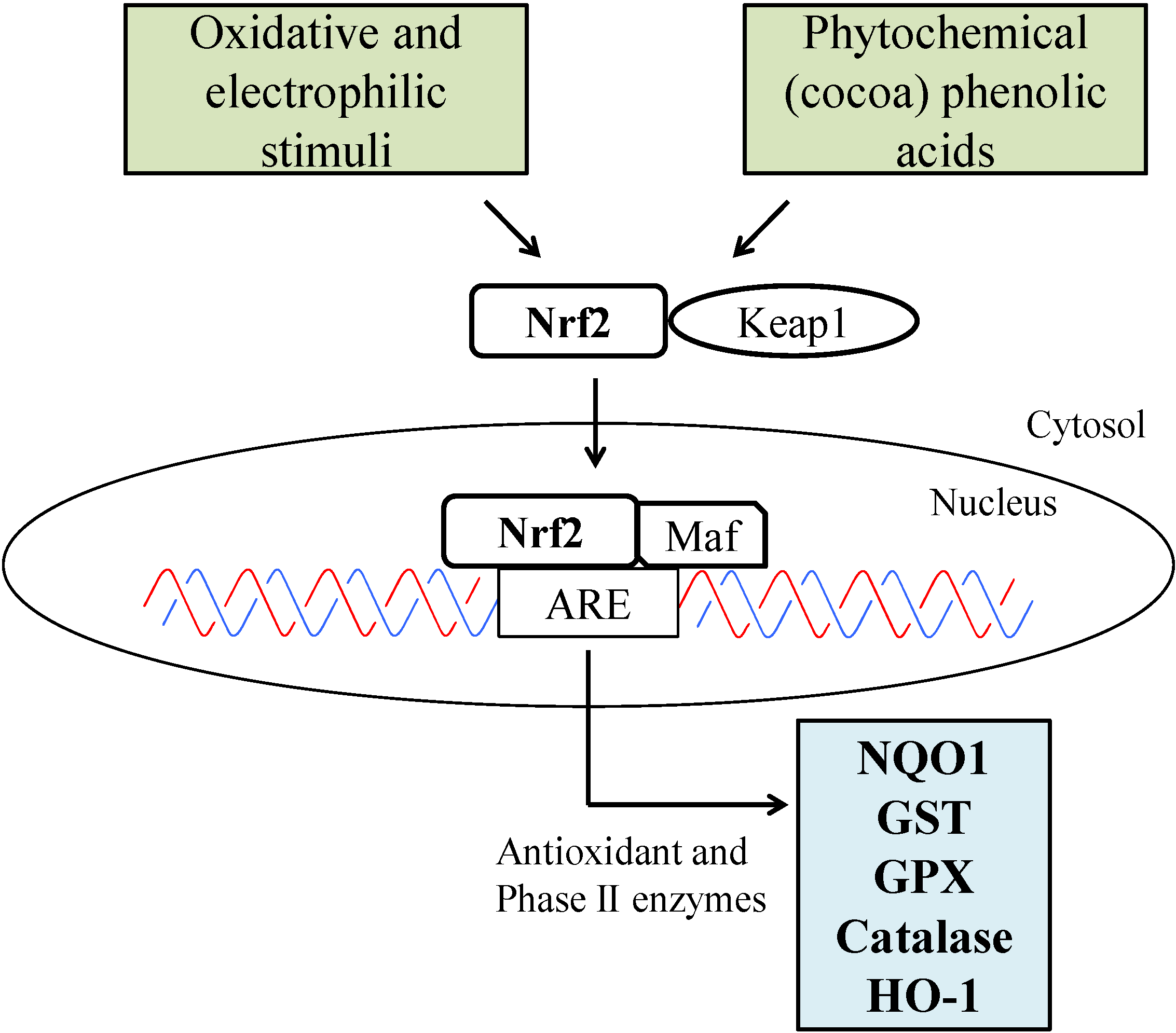
3. Molecular Mechanisms Relevant to Skin Health
4. Role of Cocoa Polyphenols in the Skin: Anti-Aging Effects
5. Conclusions
Author Contributions
Conflict of Interest
References
- Dillinger, T.L.; Barriga, P.; Escárcega, S.; Jimenez, M.; Salazar Lowe, D.; Grivetti, L.E. Food of the gods: Cure for humanity? A cultural history of the medicinal and ritual use of chocolate. J. Nutr. 2000, 130, 2057S–2072S. [Google Scholar]
- Tomaru, M.; Takano, H.; Osakabe, N.; Yasuda, A.; Inoue, K.; Yanagisawa, R.; Ohwatari, T.; Uematsu, H. Dietary supplementation with cacao liquor proanthocyanidins prevents elevation of blood glucose levels in diabetic obese mice. Nutrition 2007, 23, 351–355. [Google Scholar] [CrossRef]
- Hurst, W.J.; Krake, S.H.; Bergmeier, S.C.; Payne, M.J.; Miller, K.B.; Stuart, D.A. Impact of fermentation, drying, roasting and Dutch processing on flavan-3-ol stereochemistry in cacao beans and cocoa ingredients. Chem. Cent. J. 2011, 5, 53. [Google Scholar] [CrossRef]
- Miller, K.B.; Hurst, W.J.; Payne, M.J.; Stuart, D.A.; Apgar, J.; Sweigart, D.S.; Ou, B. Impact of alkalization on the antioxidant and flavanol content of commercial cocoa powders. J. Agric. Food Chem. 2008, 56, 8527–8533. [Google Scholar] [CrossRef]
- Ellam, S.; Williamson, G. Cocoa and human health. Annu. Rev. Nutr. 2013, 33, 105–128. [Google Scholar] [CrossRef]
- Solà, R.; Valls, R.M.; Godàs, G.; Perez-Busquets, G.; Ribalta, J.; Girona, J.; Heras, M.; Cabré, A.; Castro, A.; Domenech, G.; et al. Cocoa, hazelnuts, sterols and soluble fiber cream reduces lipids and inflammation biomarkers in hypertensive patients: A randomized controlled trial. PLoS One 2012, 7, e31103. [Google Scholar] [CrossRef]
- Corti, R.; Flammer, A.J.; Hollenberg, N.K.; Lüscher, T.F. Cocoa and cardiovascular health. Circulation 2009, 119, 1433–1441. [Google Scholar] [CrossRef]
- Lee, K.W.; Kim, Y.J.; Lee, H.J.; Lee, C.Y. Cocoa has more phenolic phytochemicals and a higher antioxidant capacity than teas and red wine. J. Agric. Food Chem. 2003, 51, 7292–7295. [Google Scholar] [CrossRef]
- Katz, D.L.; Doughty, K.; Ali, A. Cocoa and chocolate in human health and disease. Antioxid. Redox Signal. 2011, 15, 2779–2811. [Google Scholar] [CrossRef]
- Kim, J.; Kim, J.; Shim, J.; Lee, C.Y.; Lee, K.W.; Lee, H.J. Cocoa phytochemicals: Recent advances in molecular mechanisms on health. Crit. Rev. Food Sci. Nutr. 2014, 54, 1458–1472. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar]
- Harborne, J.B.; Williams, C.A. Advances in flavonoid research since 1992. Phytochemistry 2000, 55, 481–504. [Google Scholar] [CrossRef]
- Bravo, L. Polyphenols: Chemistry, Dietary Sources, Metabolism, and Nutritional Significance. Nutr. Rev. 1998, 56, 317–333. [Google Scholar] [CrossRef]
- Cheynier, V. Polyphenols in foods are more complex than often thought. Am. J. Clin. Nutr. 2005, 81, 223S–229S. [Google Scholar]
- Tsao, R. Chemistry and biochemistry of dietary polyphenols. Nutrients 2010, 2, 1231–1246. [Google Scholar] [CrossRef]
- Ortega, N.; Romero, M.P.; Macià, A.; Reguant, J.; Anglès, N.; Morelló, J.R.; Motilva, M.J. Obtention and characterization of phenolic extracts from different cocoa sources. J. Agric. Food Chem. 2008, 56, 9621–9627. [Google Scholar] [CrossRef]
- Ramiro-Puig, E.; Castell, M. Cocoa: Antioxidant and immunomodulator. Br. J. Nutr. 2009, 101, 931–940. [Google Scholar] [CrossRef]
- Jalil, A.M.; Ismail, A. Polyphenols in cocoa and cocoa products: Is there a link between antioxidant properties and health? Molecules 2008, 13, 2190–2219. [Google Scholar] [CrossRef]
- Heinrich, U.; Neukam, K.; Tronnier, H.; Sies, H.; Stahl, W. Long-term ingestion of high flavanol cocoa provides photoprotection against UV-induced erythema and improves skin condition in women. J. Nutr. 2006, 136, 1565–1569. [Google Scholar]
- Neukam, K.; Stahl, W.; Tronnier, H.; Sies, H.; Heinrich, U. Consumption of flavanol-rich cocoa acutely increases microcirculation in human skin. Eur. J. Nutr. 2007, 46, 53–66. [Google Scholar] [CrossRef]
- Ashihara, H.; Kato, M.; Crozier, A. Distribution, biosynthesis and catabolism of methylxanthines in plants. Handb. Exp. Pharmacol. 2011, 200, 11–31. [Google Scholar] [CrossRef]
- Smit, H.J.; Blackburn, R.J. Reinforcing effects of caffeine and theobromine as found in chocolate. Psychopharmacology (Berl) 2005, 18, 101–106. [Google Scholar] [CrossRef]
- Smit, H.J.; Gaffan, E.A.; Rogers, P.J. Methylxanthines are the psycho-pharmacologically active constituents of chocolate. Psychopharmacology (Berl) 2004, 176, 412–419. [Google Scholar] [CrossRef]
- Neufingerl, N.; Zebregs, Y.E.; Schuring, E.A.; Trautwein, E.A. Effect of cocoa and theobromine consumption on serum HDL-cholesterol concentrations: A randomized controlled trial. Am. J. Clin. Nutr. 2013, 97, 1201–1209. [Google Scholar] [CrossRef]
- Blinks, J.R.; Olson, C.B.; Jewell, B.R.; Bravený, P. Influence of caffeine and other methylxanthines on mechanical properties of isolated mammalian heart muscle. Evidence for a dual mechanism of action. Circ. Res. 1972, 30, 367–392. [Google Scholar] [CrossRef]
- Shively, C.A.; Tarka, S.M., Jr. Methylxanthine composition and consumption patterns of cocoa and chocolate products. Prog. Clin. Biol. Res. 1984, 158, 149–178. [Google Scholar]
- Sprügel, W.; Mitznegg, P.; Heim, F. The influence of caffeine and theobromine on locomotive activity and the brain cGMP/cAMP ratio in white mice. Biochem. Pharmacol. 1977, 26, 1723–1724. [Google Scholar] [CrossRef]
- Scapagnini, G.; Davinelli, S.; Drago, F.; de Lorenzo, A.; Oriani, G. Antioxidants as antidepressants: Fact or fiction? CNS Drugs 2012, 26, 477–490. [Google Scholar] [CrossRef]
- Judelson, D.A.; Preston, A.G.; Miller, D.L.; Muñoz, C.X.; Kellogg, M.D.; Lieberman, H.R. Effects of theobromine and caffeine on mood and vigilance. J. Clin. Psychopharmacol. 2013, 33, 499–506. [Google Scholar] [CrossRef]
- Scholey, A.; Owen, L. Effects of chocolate on cognitive function and mood: A systematic review. Nutr. Rev. 2013, 71, 665–681. [Google Scholar] [CrossRef]
- F’guyer, S.; Afaq, F.; Mukhtar, H. Photochemoprevention of skin cancer by botanical agents. Photodermatol. Photoimmunol. Photomed. 2003, 19, 56–72. [Google Scholar] [CrossRef]
- Steinberg, F.M.; Bearden, M.M.; Keen, C.L. Cocoa and chocolate flavonoids: implications for cardiovascular health. J. Am. Diet. Assoc. 2003, 103, 215–223. [Google Scholar] [CrossRef]
- Gums, J.G. Magnesium in cardiovascular and other disorders. Am. J. Health Syst. Pharm. 2004, 61, 1569–1576. [Google Scholar]
- Ueshima, K. Magnesium and ischemic heart disease: A review of epidemiological, experimental, and clinical evidences. Magnes. Res. 2005, 18, 275–284. [Google Scholar]
- Olivares, M.; Uauy, R. Copper as an essential nutrient. Am. J. Clin. Nutr. 1996, 63, 791S–796S. [Google Scholar]
- Kris-Etherton, P.M.; Keen, C.L. Evidence that the antioxidant flavonoids in tea and cocoa are beneficial for cardiovascular health. Curr. Opin. Lipidol. 2002, 13, 41–49. [Google Scholar] [CrossRef]
- Serafini, M.; Bugianesi, R.; Maiani, G.; Valtuena, S.; de Santis, S.; Crozier, A. Plasma antioxidants from chocolate. Nature 2003, 424, 1013. [Google Scholar] [CrossRef]
- Engler, M.B.; Engler, M.M. The emerging role of flavonoid-rich cocoa and chocolate in cardiovascular health and disease. Nutr. Rev. 2006, 64, 109–118. [Google Scholar] [CrossRef]
- Scapagnini, G.; Vasto, S.; Abraham, N.G.; Caruso, C.; Zella, D.; Fabio, G. Modulation of Nrf2/ARE pathway by food polyphenols: A nutritional neuroprotective strategy for cognitive and neurodegenerative disorders. Mol. Neurobiol. 2011, 44, 192–201. [Google Scholar] [CrossRef] [Green Version]
- Shah, Z.A.; Li, R.C.; Ahmad, A.S.; Kensler, T.W.; Yamamoto, M.; Biswal, S.; Doré, S. The flavanol (−)-epicatechin prevents stroke damage through the Nrf2/HO1 pathway. J. Cereb. Blood Flow Metab. 2010, 30, 1951–1961. [Google Scholar] [CrossRef]
- Schroeter, H.; Heiss, C.; Balzer, J.; Kleinbongard, P.; Keen, C.L.; Hollenberg, N.K.; Sies, H.; Kwik-Uribe, C.; Schmitz, H.H.; Kelm, M. (−)-Epicatechin mediates beneficial effects of flavanol-rich cocoa on vascular function in humans. Proc. Natl. Acad. Sci. USA 2006, 103, 1024–1029. [Google Scholar] [CrossRef]
- Sies, H.; Schewe, T.; Heiss, C.; Kelm, M. Cocoa polyphenols and inflammatory mediators. Am. J. Clin. Nutr. 2005, 81, 304S–312S. [Google Scholar]
- Lee, K.W.; Kang, N.J.; Oak, M.H.; Hwang, M.K.; Kim, J.H.; Schini-Kerth, V.B.; Lee, H.J. Cocoa procyanidins inhibit expression and activation of MMP-2 in vascular smooth muscle cells by direct inhibition of MEK and MT1-MMP activities. Cardiovasc. Res. 2008, 79, 34–41. [Google Scholar] [CrossRef]
- Ramiro, E.; Franch, A.; Castellote, C.; Pérez-Cano, F.; Permanyer, J.; Izquierdo-Pulido, M.; Castell, M. Flavonoids from Theobroma cacao down-regulate inflammatory mediators. J. Agric. Food Chem. 2005, 53, 8506–8611. [Google Scholar] [CrossRef]
- Selmi, C.; Mao, T.K.; Keen, C.L.; Schmitz, H.H.; Eric Gershwin, M. The anti-inflammatory properties of cocoa flavanols. J. Cardiovasc. Pharmacol. 2006, 47, S163–S171. [Google Scholar] [CrossRef]
- Afaq, F.; Adhami, V.M.; Mukhtar, H. Photochemoprevention of ultraviolet B signaling and photocarcinogenesis. Mutat. Res. 2005, 571, 153–173. [Google Scholar] [CrossRef]
- Urbach, F. Incidence of nonmelanoma skin cancer. Dermatol. Clin. 1991, 9, 751–755. [Google Scholar]
- Nichols, J.A.; Katiyar, S.K. Skin photoprotection by natural polyphenols: Anti-inflammatory, antioxidant and DNA repair mechanisms. Arch. Dermatol. Res. 2010, 302, 71–83. [Google Scholar] [CrossRef]
- Lee, K.W.; Kundu, J.K.; Kim, S.O.; Chun, K.S.; Lee, H.J.; Surh, Y.J. Cocoa polyphenols inhibit phorbol ester-induced superoxide anion formation in cultured HL-60 cells and expression of cyclooxygenase-2 and activation of NF-kappaB and MAPKs in mouse skin in vivo. J. Nutr. 2006, 136, 1150–1155. [Google Scholar]
- Kim, J.E.; Son, J.E.; Jung, S.K.; Kang, N.J.; Lee, C.Y.; Lee, K.W.; Lee, H.J. Cocoa polyphenols suppress TNF-α-induced vascular endothelial growth factor expression by inhibiting phosphoinositide 3-kinase (PI3K) and mitogen-activated protein kinase kinase-1 (MEK1) activities in mouse epidermal cells. Br. J. Nutr. 2010, 104, 957–964. [Google Scholar] [CrossRef]
- Gasser, P.; Lati, E.; Peno-Mazzarino, L.; Bouzoud, D.; Allegaert, L.; Bernaert, H. Cocoa polyphenols and their influence on parameters involved in ex vivo skin restructuring. Int. J. Cosmet. Sci. 2008, 30, 339–345. [Google Scholar] [CrossRef]
- Mitani, H.; Ryu, A.; Suzuki, T.; Yamashita, M.; Arakane, K.; Koide, C. Topical application of plant extracts containing xanthine derivatives can prevent UV-induced wrinkle formation in hairless mice. Photodermatol. Photoimmunol. Photomed. 2007, 23, 86–94. [Google Scholar] [CrossRef]
- Braverman, I.M. The cutaneous microcirculation. J. Investig. Dermatol. Symp. Proc. 2000, 5, 3–9. [Google Scholar] [CrossRef]
- Williams, S.; Tamburic, S.; Lally, C. Eating chocolate can significantly protect the skin from UV light. J. Cosmet. Dermatol. 2009, 8, 169–173. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Scapagnini, G.; Davinelli, S.; Di Renzo, L.; De Lorenzo, A.; Olarte, H.H.; Micali, G.; Cicero, A.F.; Gonzalez, S. Cocoa Bioactive Compounds: Significance and Potential for the Maintenance of Skin Health. Nutrients 2014, 6, 3202-3213. https://doi.org/10.3390/nu6083202
Scapagnini G, Davinelli S, Di Renzo L, De Lorenzo A, Olarte HH, Micali G, Cicero AF, Gonzalez S. Cocoa Bioactive Compounds: Significance and Potential for the Maintenance of Skin Health. Nutrients. 2014; 6(8):3202-3213. https://doi.org/10.3390/nu6083202
Chicago/Turabian StyleScapagnini, Giovanni, Sergio Davinelli, Laura Di Renzo, Antonino De Lorenzo, Hector Hugo Olarte, Giuseppe Micali, Arrigo F. Cicero, and Salvador Gonzalez. 2014. "Cocoa Bioactive Compounds: Significance and Potential for the Maintenance of Skin Health" Nutrients 6, no. 8: 3202-3213. https://doi.org/10.3390/nu6083202
APA StyleScapagnini, G., Davinelli, S., Di Renzo, L., De Lorenzo, A., Olarte, H. H., Micali, G., Cicero, A. F., & Gonzalez, S. (2014). Cocoa Bioactive Compounds: Significance and Potential for the Maintenance of Skin Health. Nutrients, 6(8), 3202-3213. https://doi.org/10.3390/nu6083202






