Phenolic Lipids Affect the Activity and Conformation of Acetylcholinesterase from Electrophorus electricus (Electric eel)
Abstract
:1. Introduction
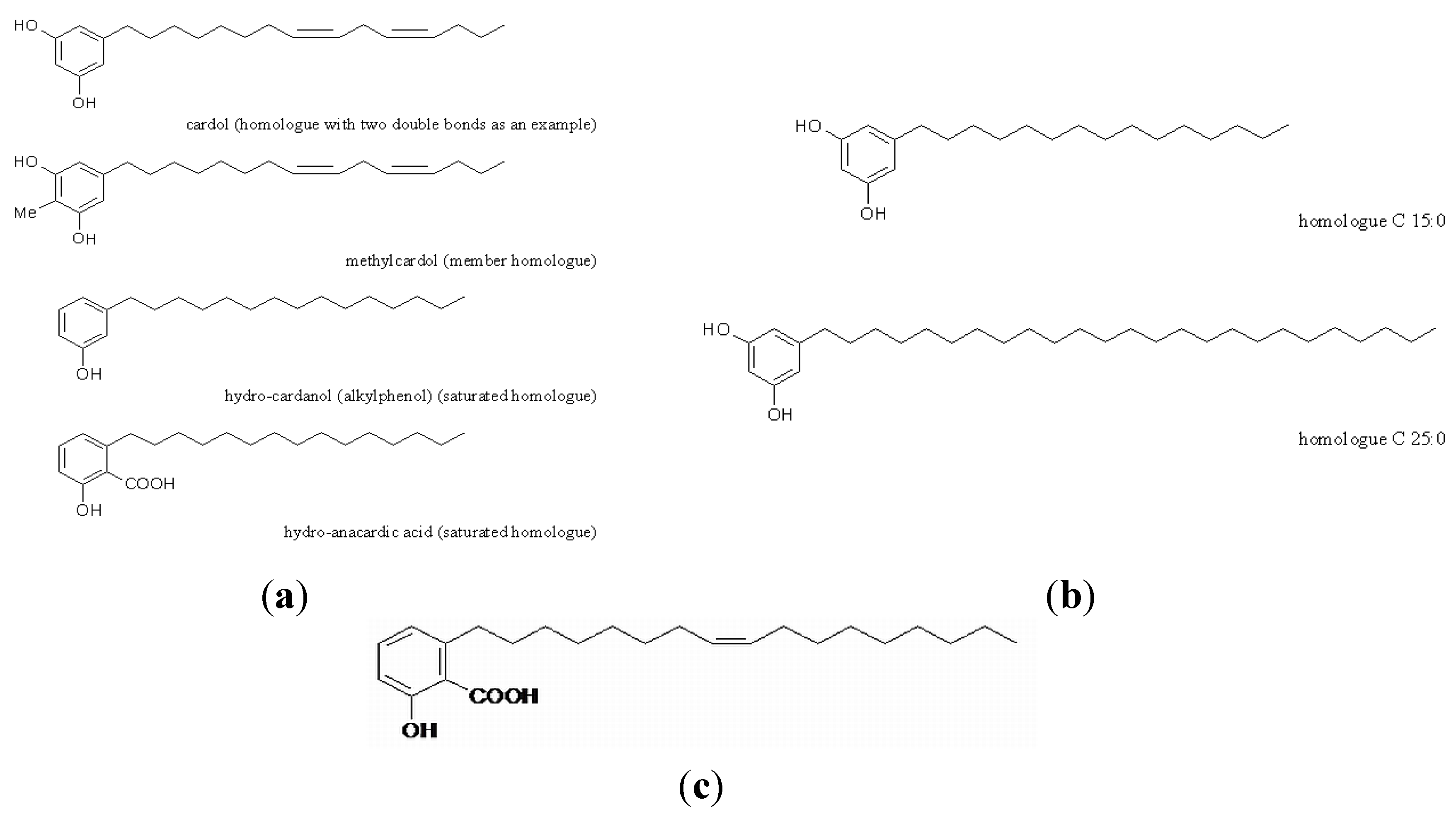
2. Materials and Methods
2.1. Chemicals
2.2. Estimation of AChE Activity
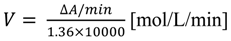
2.3. Estimation of the Effect of Phenolic Lipids on Intrinsic Fluorescence of Trp Residues of AChE
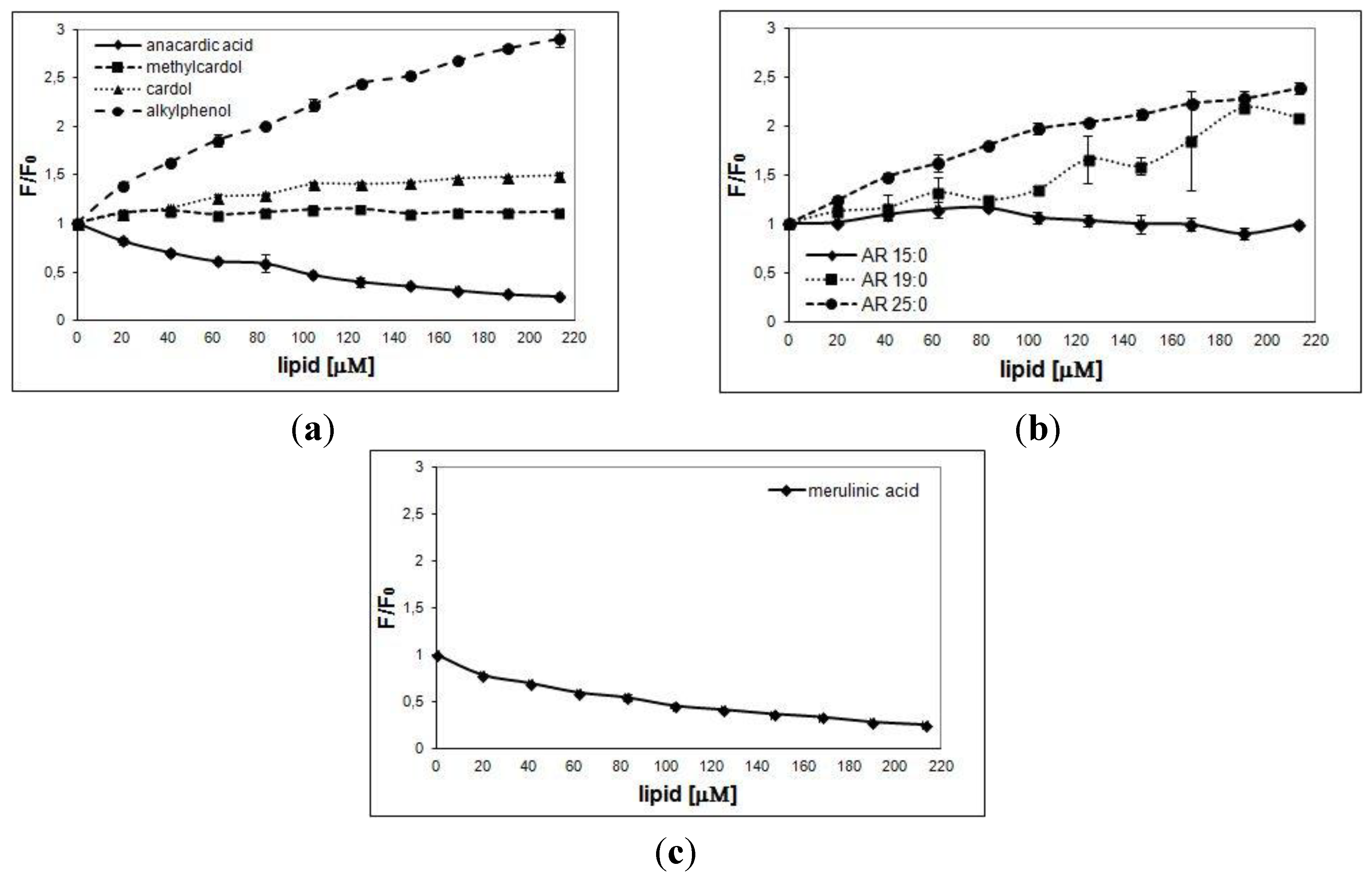
3. Results

| Studied Compound | IC50 ± SD [μM] |
|---|---|
| Cardol | 3.5 ± 0.2 |
| Methylcardol | 5 ± 0.41 |
| Alkylphenol | 4 ± 0.24 |
| Anacardic acid | 3 ± 0.24 |
| AR 15:0 | >94 |
| AR 19:0 | 77 ± 1.25 |
| AR 21:0 | 44 ± 0.47 |
| AR 25:0 | 44 ± 4.24 |
| Merulinic acid | >94 |
4. Discussion
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Kozubek, A.; Tyman, J.H.P. Resorcinolic lipids, the natural non-isoprenoic amphiphiles and their biological activity. Chem. Rev. 1999, 99, 1–26. [Google Scholar] [CrossRef]
- Stasiuk, M.; Kozubek, A. Biological activity of phenolic lipids. Cell. Mol. Life Sci. 2010, 67, 841–860. [Google Scholar] [CrossRef]
- Logrado, L.P.L.; dos Santos, M.L.; Silveira, D.; Romeiro, L.A.S.; Moraes, M.O.; Cavalcanti, B.C.; Costa-Lotufo, L.V.; do Ó Pessoa, C.; dos Santos, M.L. Synthesis and biological evaluation of new salicylate macrolactones from anacardic acids. J. Braz. Chem. Soc. 2005, 16, 1217–1225. [Google Scholar] [CrossRef]
- Ross, A.B.; Shepherd, M.J.; Schupphaus, M.; Sinclair, V.; Alfaro, B.; Kamal Eldin, A.; Aman, P. Alkylresorcinols in cereals and cereal products. J. Agric. Food Chem. 2003, 51, 4111–4118. [Google Scholar] [CrossRef]
- Ross, A.B.; Shepherd, M.J.; Bach Knudsen, K.E.; Glitso, L.V.; Bowey, E.; Phillips, J.; Rowland, I.; Guo, Z.-X.; Massy, D.J.; Aman, P.; et al. Absorption of dietary alkylresorcinols in ileal cannulated pigs and rats. Br. J. Nutr. 2003, 90, 787–794. [Google Scholar] [CrossRef]
- Linko, A.M.; Parikka, K.; Wahala, K.; Adlercreutz, H. Gas chromatographic-mass spectrometric method for the determination of alkylresorcinols in human plasma. Anal. Biochem. 2002, 308, 307–313. [Google Scholar] [CrossRef]
- Linko, A.M.; Juntunen, K.S.; Mykkänen, H.M.; Adlercreutz, H. Whole-grain rye bread consumption by women correlates with plasma alkylresorcinols and increases their concentration compared with low-fiber wheat bread. J. Nutr. 2005, 135, 580–583. [Google Scholar]
- Ross, A.B.; Kamal-Eldin, A.; Lundin, E.A.; Zhang, J.X.; Hallmans, G.; Aman, P. Cereal alkylresorcinols are absorbed by humans. J. Nutr. 2003, 133, 2222–2224. [Google Scholar]
- Linko, A.M.; Aldercreutz, H. Whole-grain rye and wheat alkylresorcinols are incorporated into human erythrocyte membranes. Br. J. Nutr. 2005, 93, 11–13. [Google Scholar] [CrossRef]
- Linko, A.M.; Landberg, R.; Tikkanen, M.J.; Adlercreutz, H.; Penalvo, J.L. Alkylresircinols from whole-grain wheat and rye are transported in human plasma lipoproteins. J. Nutr. 2007, 137, 1137–1142. [Google Scholar]
- Stasiuk, M.; Jaromin, A.; Kozubek, A. The effect of merulinic acid on biomembranes. Biochim. Biophys. Acta 2004, 1667, 215–221. [Google Scholar]
- Wessler, I.; Kilbinger, H.; Bittinger, F.; Kirkpatrick, C.J. The biological role of non-neuronal acetylcholine in plants and humans. Jpn. J. Pharmacol. 2001, 85, 2–10. [Google Scholar] [CrossRef]
- Paleari, L.; Grozio, A.; Cesario, A.; Russo, P. The cholinergic system and cancer. Semin. Cancer Biol. 2008, 18, 211–217. [Google Scholar] [CrossRef]
- Silman, J.; Sussman, L. Acetylcholinesterase: “Classical” and “nonclassical” functions and pharmacology. Curr. Opin. Pharmacol. 2005, 5, 293–302. [Google Scholar] [CrossRef]
- Battisti, V.; Schetinger, M.R.C.; Maders, L.D.K.; Santos, K.F.; Bagatini, M.D.; Correa, M.C.; Spanevello, R.M.; do Carmo Araújo, M.; Morsch, V.M. Changes in acetylcholinesterase (AchE) activity in lymphocytes and whole blood in acute lymphoblastic leukemia patients. Clin. Chim. Acta 2009, 402, 114–118. [Google Scholar] [CrossRef]
- Razani-Boroujerdi, S.; Behl, M.; Hahn, F.; Pena, J.C.; Hutt, J.; Sopori, M.L. Role of muscarinic receptors in the regulation of immune and inflammatory responses. J. Neuroimmunol. 2008, 194, 83–88. [Google Scholar]
- Benzi, G.; Moretti, A. Is there a rationale for the use of acetylcholinesterase inhibitors in the therapy of Alzheimer’s disease? Eur. J. Pharmacol. 1998, 346, 1–13. [Google Scholar] [CrossRef]
- Mega, M.S. The cholinergic deficit in Alzheimer’s disease: Impact on cognition, behavior and function. Int. J. Neuropsychopharmacol. 2000, 3, 3–12. [Google Scholar]
- Valle, A.M.; Radic, Z.; Rana, B.K.; Mahboubi, V.; Wessel, J.; Shih, P.B.; Rao, F.; Connor, D.T.O.; Taylor, P. Naturally occurring variations in the human cholinesterase genes: Heritability and association with cardiovascular and metabolic traits. J. Pharmacol. Exp. Ther. 2011, 338, 125–133. [Google Scholar] [CrossRef]
- Sugimoto, H.; Yamanishi, Y.; Iimura, Y.; Kawakami, Y. Donepezil hydrochloride (E2020) and other acetylcholinesterase inhibitors. Curr. Med. Chem. 2000, 7, 303–339. [Google Scholar] [CrossRef]
- Stasiuk, M.; Kozubek, A. Membrane perturbing properties of natural phenolic and resorcinolic lipids. FEBS Lett. 2008, 582, 3607–3613. [Google Scholar] [CrossRef]
- Stasiuk, M.; Kleta, M.; Kozubek, A. Dual effect of free and liposomal forms of phenolic lipids on the activity of GPI-anchor-deprived acetylcholinesterase from erythrocytes. Food Chem. 2011, 125, 508–512. [Google Scholar] [CrossRef]
- Przeworska, E.; Gubernator, J.; Kozubek, A. Formation of liposomes by resorcinolic lipids, single chain phenolic amphiphiles from Anacardium occidentale L. Biochim. Biophys. Acta 2001, 1513, 75–81. [Google Scholar] [CrossRef]
- Kozubek, A. Isolation of 5-n-alkyl-, 5-n-alkenyl- and 5-n-alkadienyl-homologs of alk(en)ylresorcinols from rye grains. Acta Aliment. Pol. 1985, 9, 185–198. [Google Scholar]
- Giannetti, B.M.; Steglich, W.; Quack, W.; Anke, T.; Oberwinkler, F. Antibiotika aus basidiomyceten: VI. Merulinsauren A, B und C, neue antibiotika aus Merulius tremellosus Fr. und Phlebia radiata Fr. Z. für Naturforschung 1978, 33, 807–816. [Google Scholar]
- Ellman, G.L.; Courtney, D.; Andres, D.; Featherstone, R.M. A new rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Zhao, H.; Sood, R.; Jutila, A.; Bose, S.; Fimland, G.; Nissen-Meyer, J.; Kinnunen, P.K.J. Interaction of the antimicrobial peptide pheromone Plantaricin A with model membranes: Implications for a novel mechanism of action. Biochim. Biophys. Acta 2006, 1758, 1461–1474. [Google Scholar]
- Liu, S.; Shibata, A.; Ueno, S.; Xu, F.; Baba, Y.; Jiang, D.; Li, Y. Investigation of interaction of Leu-enkephalin with lipid membranes. Colloids Surf. B 2006, 48, 148–158. [Google Scholar] [CrossRef]
- Loura, L.M.S.; Almeida, R.F.M.; Coutinho, A.; Prieto, M. Interaction of peptides with binary phospholipid membranes: Application of fluorescence methodologies. Chem. Phys. Lipids 2003, 122, 77–96. [Google Scholar] [CrossRef]
- Terlecki, G.; Czapinska, E.; Rogozik, K.; Lisowski, M.; Gutowicz, J. Investigation of the interaction of pig muscle lactate dehydrogenase with acidic phospholipids at low pH. Biochim. Biophys. Acta 2006, 1758, 133–144. [Google Scholar]
- Sussman, J.L.; Harel, M.; Frolow, F.; Oefner, C.; Goldman, A.; Toker, L.; Silman, I. Atomic structure of acetylcholinesterase from Torpedo californica: A prototypic acetylcholine-binding protein. Science 1991, 253, 872–879. [Google Scholar] [CrossRef]
- Ordentlich, A.; Barak, D.; Kronman, C.; Flashner, Y.; Leitner, M.; Segall, Y.; Ariel, N.; Cohen, S.; Velan, B.; Shafferman, A. Dissection of the human acetylcholinesterase active center determinants of substrate specificity. Identification of residues constituting the anionic site, the hydrophobic site, and the acyl pocket. J. Biol. Chem. 1993, 268, 17083–17095. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Stasiuk, M.; Janiszewska, A.; Kozubek, A. Phenolic Lipids Affect the Activity and Conformation of Acetylcholinesterase from Electrophorus electricus (Electric eel). Nutrients 2014, 6, 1823-1831. https://doi.org/10.3390/nu6051823
Stasiuk M, Janiszewska A, Kozubek A. Phenolic Lipids Affect the Activity and Conformation of Acetylcholinesterase from Electrophorus electricus (Electric eel). Nutrients. 2014; 6(5):1823-1831. https://doi.org/10.3390/nu6051823
Chicago/Turabian StyleStasiuk, Maria, Alicja Janiszewska, and Arkadiusz Kozubek. 2014. "Phenolic Lipids Affect the Activity and Conformation of Acetylcholinesterase from Electrophorus electricus (Electric eel)" Nutrients 6, no. 5: 1823-1831. https://doi.org/10.3390/nu6051823
APA StyleStasiuk, M., Janiszewska, A., & Kozubek, A. (2014). Phenolic Lipids Affect the Activity and Conformation of Acetylcholinesterase from Electrophorus electricus (Electric eel). Nutrients, 6(5), 1823-1831. https://doi.org/10.3390/nu6051823




