Iron Transport through Ferroportin Is Induced by Intracellular Ascorbate and Involves IRP2 and HIF2α
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Antibodies
2.3. Cell Line
2.4. Cell Culture and Iron-Uptake Experiments
2.5. Harvesting of Caco-2 Cells for Protein Analysis
2.6. Sandwich ELISA for Estimation of Ferroportin, IRP2, and HIF2α Protein Expression
2.7. SDS-PAGE and Western Blot for Detection of Ferroportin Levels
2.8. 55Fe Transmembrane Transport
2.9. RNA Purification and cDNA Synthesis
2.10. Real Time PCR Measurements of Ferroportin mRNA Expression
2.11. Statistics
3. Results
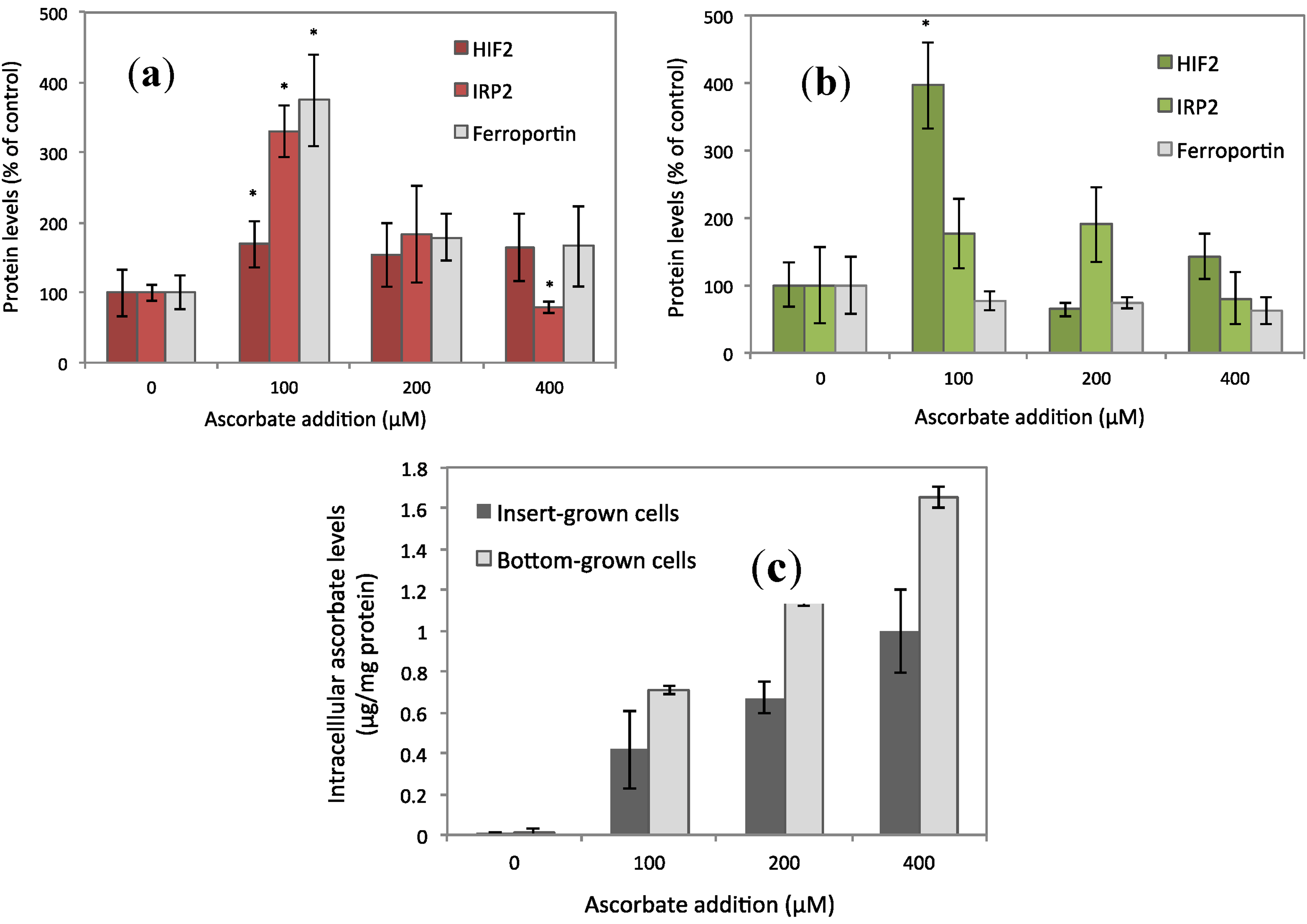
3.1. Ferroportin, IRP2, and HIF2α Levels Peaked at 100 μM of Ascorbate
3.2. Increased Ferroportin Levels, Increased Transmembrane Fe Transport
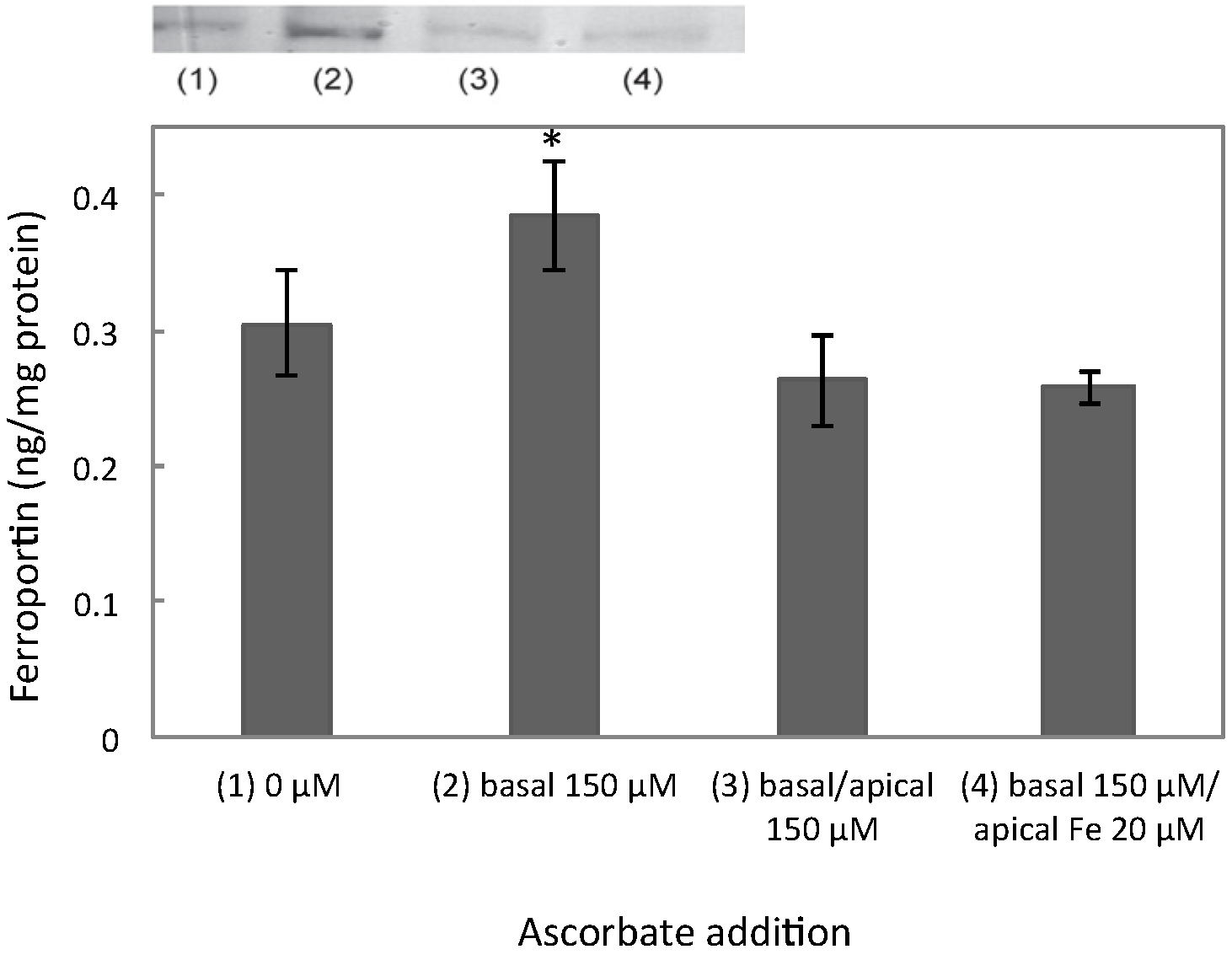

3.3. Ferroportin mRNA Levels Were Unchanged at 150 μM Ascorbate
4. Discussion
4.1. The Effects of Ascorbate—Just a Change in Redox Balance of the Cell?
4.2. Insights from Comparison of Permeable and Impermeable Basal Support for Cell Cultures
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Hutton, J.J., Jr.; Tappel1, A.L.; Udenfriend, S. Cofactor and substrate requirements of collagen prolin hydroxylase. Arch. Biochem. Phys. 1967, 118, 231–240. [Google Scholar] [CrossRef]
- Levine, M.; Morita, K.; Heldman, E.; Pollard, H.B. Ascorbic acid regulation of norepinephrine biosynthesis in isolated chromaffin granules from bovine adrenal medulla. J. Biol. Chem. 1985, 260, 15598–15603. [Google Scholar]
- Hallberg, L.; Brune, M.; Rossander, L. Effect of ascorbic acid on iron absorption from different types of meals. Studies with ascorbic-acid-rich foods and synthetic ascorbic acid given in different amounts with different meals. Hum. Nutr. Appl. Nutr. 1980, 40, 97–113. [Google Scholar]
- Yun, S.; Habicht, J.-P.; Miller, D.D.; Glahn, R.P. An in vitro digestion/Caco-2 cell culture system accurately predicts the effects of ascorbic acid and polyphenolic compounds on iron bioavailability in humans. J. Nutr. 2004, 134, 2717–2721. [Google Scholar]
- Thumser, A.E.; Rashed, A.A.; Sharp, P.A.; Lodge, J.K. Ascorbate enhances iron uptake into intestinal cells through formation of a FeCl3–ascorbate complex. Food Chem. 2010, 123, 281–285. [Google Scholar] [CrossRef] [Green Version]
- Plug, C.M.; Dekker, D.; Bult, A. Complex stability of ferrous ascorbate in aqueous solution and its significance in iron absorption. Pharm. World Sci. 1984, 6, 245–248. [Google Scholar]
- Scheers, N.; Andlid, T.; Alminger, M.; Sandberg, A.-S. Determination of Fe2+ and Fe3+ in aqueous solutions containing food chelators by differential pulse anodic stripping voltammetry. Electroanalysis 2010, 22, 1090–1096. [Google Scholar] [CrossRef]
- Scheers, N.; Sandberg, A.-S. Iron regulates the uptake of ascorbic acid and the expression of sodium-dependent vitamin C transporter 1 (SVCT1) in human intestinal Caco-2 cells. Br. J. Nutr. 2011, 105, 1734–1740. [Google Scholar] [CrossRef]
- Scheers, N.; Sandberg, A.-S. Ascorbic acid uptake affects ferritin, Dcytb and Nramp2 expression in Caco-2 cells. Eur. J. Nutr. 2008, 47, 401–408. [Google Scholar] [CrossRef]
- Cook, J.D.; Reddy, M.B. Effect of ascorbic acid intake on nonheme-iron absorption from a complete diet. Am. J. Clin. Nutr. 2001, 73, 93–98. [Google Scholar]
- Scheers, N. Regulatory effects of Cu, Zn, and Ca on Fe absorption: The intricate play between nutrient transporters. Nutrients 2013, 5, 957–970. [Google Scholar] [CrossRef]
- McKie, A.T.; Marciani, P.; Rolfs, A.; Brennan, K.; Wehr, K.; Barrow, D.; Miret, S.; Bomford, A.; Peters, T.J.; Farzaneh, F.; et al. A novel duodenal iron-regulated transporter, IREG1, implicated in the basolateral transfer of iron to the circulation. Mol. Cell 2000, 5, 299–309. [Google Scholar] [CrossRef]
- Abboud, S.; Haile, D.J. A novel mammalian iron-regulated protein involved in intracellular iron metabolism. J. Biol. Chem. 2000, 275, 19906–19912. [Google Scholar] [CrossRef]
- Walden, W.E.; Selezneva, A.I.; Dupuy, J.; Volbeda, A.; Fontecilla-Camps, J.C.; Theil, E.C.; Volz, K. Structure of dual function iron regulatory protein 1 complexed with ferritin IRE-RNA. Science 2006, 314, 1903–1908. [Google Scholar] [CrossRef]
- Vashisht, A.A.; Zumbrennen, K.B.; Huang, X.; Powers, D.N.; Durazo, A.; Sun, D.; Bhaskaran, N.; Persson, A.; Uhlen, M.; Sangfelt, O.; et al. Control of iron homeostasis by an iron-regulated ubiquitin ligase. Science 2009, 326, 718–721. [Google Scholar] [CrossRef]
- Salahudeen, A.A.; Thompson, J.W.; Ruiz, J.C.; Ma, H.-W.; Kinch, L.N.; Li, Q.; Grishin, N.V.; Bruick, R.K. An E3 ligase possessing an iron-responsive hemerythrin domain is a regulator of iron homeostasis. Science 2009, 326, 722–726. [Google Scholar] [CrossRef]
- Scheers, N.M.; Hoffmann, K.B.; Sandberg, A.-S. The iron transporter ferroportin is regulated by ascorbate. FASEB J. 2012, 26, 641.20. [Google Scholar]
- Margolis, S.; Paule, R.C.; Ziegler, R.G. Ascorbic and dehydroascorbic acids measured in plasma preserved with dithiothreitol or metaphosphoric acid. Clin. Chem. 1990, 36, 1750–1755. [Google Scholar]
- Aguirre, P.; Mena, N.; Tapia, V.; Arredondo, M.; Núñez, M.T. Iron homeostasis in neuronal cells: A role for IREG1. BMC Neurosci. 2005, 6, 3. [Google Scholar] [CrossRef] [Green Version]
- Martini, L.A.; Tchack, L.; Wood, R.J. Iron treatment downregulates DMT1 and IREG1 mRNA expression in Caco-2 Cells. J. Nutr. 2002, 132, 693–696. [Google Scholar]
- Latunde-Dada, G.O.; van der Westhuizen, J.; Vulpe, C.D.; Anderson, G.J.; Simpson, R.J.; McKie, A.T. Molecular and functional roles of duodenal cytochrome B (Dcytb) in iron metabolism. Blood Cells Mol. Dis. 2002, 29, 356–360. [Google Scholar] [CrossRef]
- Lane, D.J.; Robinson, S.R.; Czerwinska, H.; Bishop, G.M.; Lawen, A. Two routes of iron accumulation in astrocytes: Ascorbate-dependent ferrous iron uptake via the divalent metal transporter (DMT1) plus an independent route for ferric iron. Biochem. J. 2010, 432, 123–132. [Google Scholar] [CrossRef]
- Zumbrennen, K.B.; Wallander, M.L.; Romney, S.J.; Leibold, E.A. Cysteine oxidation regulates the RNA-binding activity of iron regulatory protein 2. Mol. Cell. Biol. 2009, 29, 2219–2229. [Google Scholar] [CrossRef]
- Lane, D., Jr.; Chikhani, S.; Richardson, V.; Richardson, D.R. Transferrin iron uptake is stimulated by ascorbate via an intracellular reductive mechanism. Biochim. Biophys. Acta 2013, 1833, 1527–1541. [Google Scholar]
- Taylor, M.; Qu, A.; Anderson, E.R.; Matsubara, T.; Martin, A.; Gonzalez, F.J.; Shah, Y.M. Hypoxia-inducible factor-2α mediates the adaptive increase of intestinal ferroportin during iron deficiency in mice. Gastroenterology 2011, 140, 2044–2055. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Scheers, N.; Sandberg, A.-S. Iron Transport through Ferroportin Is Induced by Intracellular Ascorbate and Involves IRP2 and HIF2α. Nutrients 2014, 6, 249-260. https://doi.org/10.3390/nu6010249
Scheers N, Sandberg A-S. Iron Transport through Ferroportin Is Induced by Intracellular Ascorbate and Involves IRP2 and HIF2α. Nutrients. 2014; 6(1):249-260. https://doi.org/10.3390/nu6010249
Chicago/Turabian StyleScheers, Nathalie, and Ann-Sofie Sandberg. 2014. "Iron Transport through Ferroportin Is Induced by Intracellular Ascorbate and Involves IRP2 and HIF2α" Nutrients 6, no. 1: 249-260. https://doi.org/10.3390/nu6010249




