Linseed Dietary Fibers Reduce Apparent Digestibility of Energy and Fat and Weight Gain in Growing Rats
Abstract
:1. Introduction
2. Experimental Section
2.1. Animals and Study Design
2.2. Experimental Diets
2.3. Feed Analyses of Major Nutritional Compounds
| Diet | C | CEL | 5-WL | 5-GL | 5-LDF | 10-LDF |
|---|---|---|---|---|---|---|
| Ingredient, % (calculated) | ||||||
| Maize starch | 63.7 | 58.7 | 52.7 | 52.7 | 48.9 | 39.0 |
| Linseed oil | 10.00 | 10.00 | 6.45 | 6.45 | 9.20 | 8.40 |
| Animal fat (pig) | 10.00 | 10.00 | 6.45 | 6.45 | 9.20 | 8.40 |
| Casein + 1% methionine | 11.80 | 11.80 | 7.00 | 7.00 | 9.70 | 7.65 |
| Vitamin mix | 0.81 | 0.81 | 0.81 | 0.81 | 0.81 | 0.81 |
| Mineral mix | 3.70 | 3.70 | 3.70 | 3.70 | 3.70 | 3.70 |
| Linseed | 0.00 | 0.00 | 17.9 | 17.9 | 0.00 | 0.00 |
| Linseed mucilage | 0.00 | 0.00 | 0.00 | 0.00 | 13.5 | 27.0 |
| Cellulose | 0.00 | 5.00 | 5.00 | 5.00 | 5.00 | 5.00 |
| Chemical composition (measured) | ||||||
| DM, g/kg | 973 | 989 | 973 | 975 | 979 | 976 |
| Ash, g/kg DM | 30.6 | 33.0 | 34.7 | 36.7 | 39.0 | 50.9 |
| Protein, g/kg DM | 111 | 110 | 110 | 109 | 107 | 111 |
| Fat, g/kg DM | 209 | 205 | 208 | 205 | 212 | 202 |
| Gross E, kcal/kg DM | 5280 | 5088 | 5232 | 5184 | 5179 | 5139 |
| NSP, g/kg DM | 14 | 68 | 76 | 76 | 99 | 158 |
| Lignin, g/kg DM | 0 | 3 | 16 | 16 | 9 | 31 |
| DF g/kg DM | 14 | 71 | 92 | 92 | 108 | 189 |
2.4. Analyses of Faecal and Tissue Samples
2.5. Calculations and Statistical Analyses
3. Results
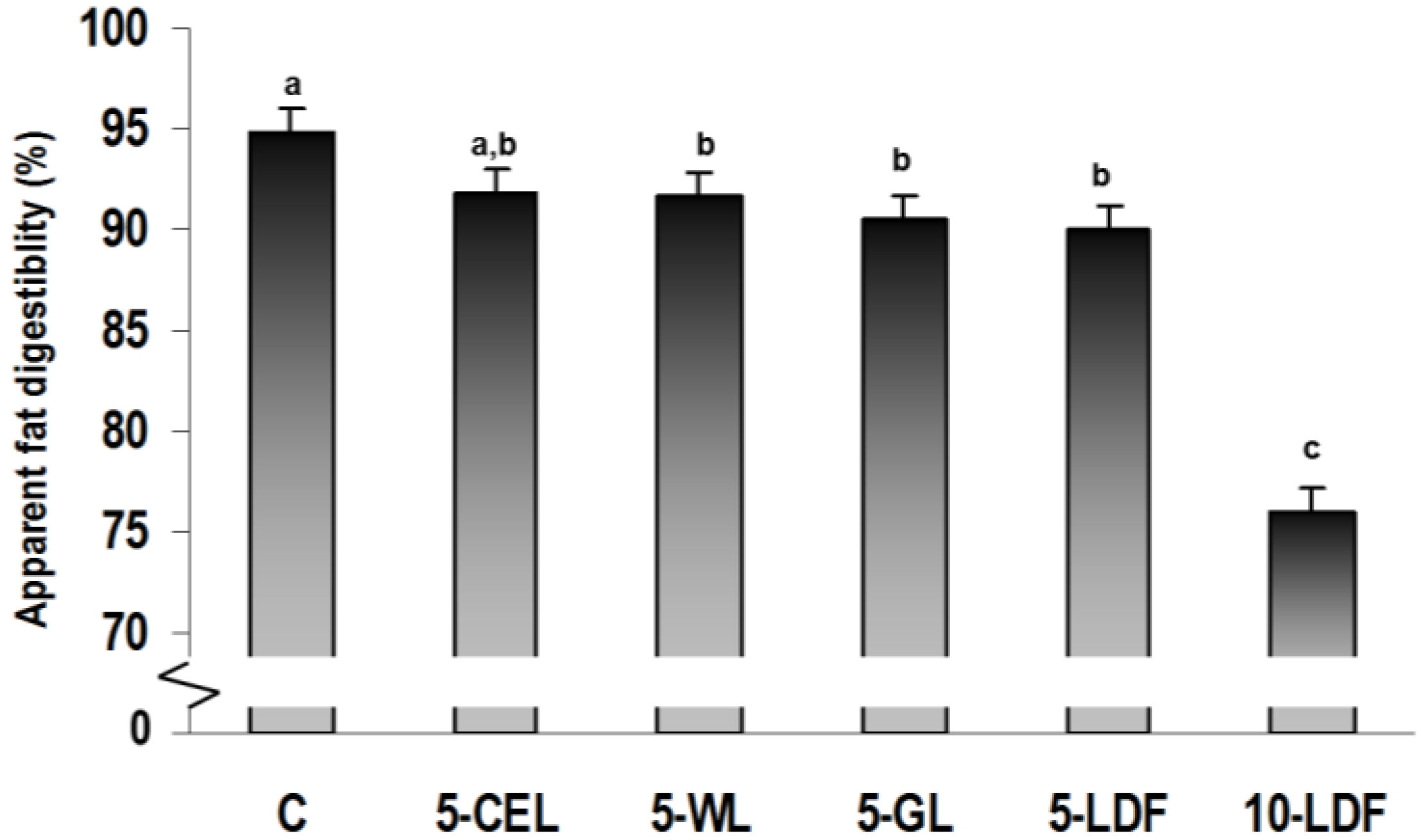
| Outcome parameter | C | 5-CEL | 5-WL | 5-GL | 5-LDF | 10-LDF * | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SE | Mean | SE | Mean | SE | Mean | SE | Mean | SE | Mean | SE | |
| Nutrient digestibility | ||||||||||||
| Feed intake (g/day) | 14.9 c | 0.4 | 17.8 a | 0.3 | 15.7 bc | 0.3 | 16.1 bc | 0.3 | 15.4 bc | 0.4 | 16.5 ab | 0.4 |
| Energy intake (kcal/day) | 74.6 b | 1.9 | 90.4 a | 1.9 | 80.0 b | 1.9 | 81.5 b | 1.9 | 78.4 b | 1.9 | 82.6 b | 2.1 |
| Fat intake (g/day) | 3.10 b | 0.07 | 3.51 a | 0.07 | 3.24 b | 0.07 | 3.30 b | 0.07 | 3.27 b | 0.07 | 3.37 b | 0.08 |
| Faecal volume (g/day) | 0.92 e | 0.23 | 2.34 d | 0.23 | 3.29 cd | 0.24 | 3.33 c | 0.23 | 4.59 b | 0.23 | 7.71 a | 0.27 |
| Energy excretion (kcal/day) | 2.98 c | 0.55 | 7.85 b | 0.55 | 8.67 ab | 0.57 | 9.42 ab | 0.55 | 10.27 a | 0.56 | 19.08 a | 0.64 |
| Fat excretion (mg/day) | 154 c | 33 | 287 bc | 33 | 294 b | 33 | 349 b | 33 | 306 b | 33 | 829 a | 38 |
| GIT mass and content | ||||||||||||
| Small intestine weight (g) | 5.78 c | 0.24 | 5.11 c | 0.17 | 5.74 c | 0.24 | 6.23 b | 0.24 | 6.91 b | 0.23 | 9.11 a | 0.30 |
| Small intestine content (g) | 1.34 d | 0.25 | 2.66 c | 0.17 | 2.92 bc | 0.25 | 2.54 c | 0.25 | 3.66 b | 0.24 | 7.57 a | 0.31 |
| Caecum weight (g) | 0.80 b | 0.09 | 0.89 b | 0.07 | 0.78 b | 0.09 | 0.78 b | 0.09 | 1.17 b | 0.09 | 2.07 a | 0.12 |
| Caecum content (g) | 1.68 d | 0.46 | 2.79 cd | 0.32 | 3.45 cd | 0.46 | 3.69 bc | 0.46 | 5.47 b | 0.45 | 10.82 a | 0.57 |
| Large intestine weight (g) | 1.00 c | 0.09 | 1.08 c | 0.06 | 1.28 bc | 0.09 | 1.30 bc | 0.09 | 1.39 b | 0.08 | 1.76 a | 0.11 |
| Large intestine content (g) | 1.60 b | 0.33 | 1.73 b | 0.23 | 2.06 b | 0.33 | 2.21 b | 0.34 | 2.65 b | 0.33 | 4.88 a | 0.41 |
| Body composition | ||||||||||||
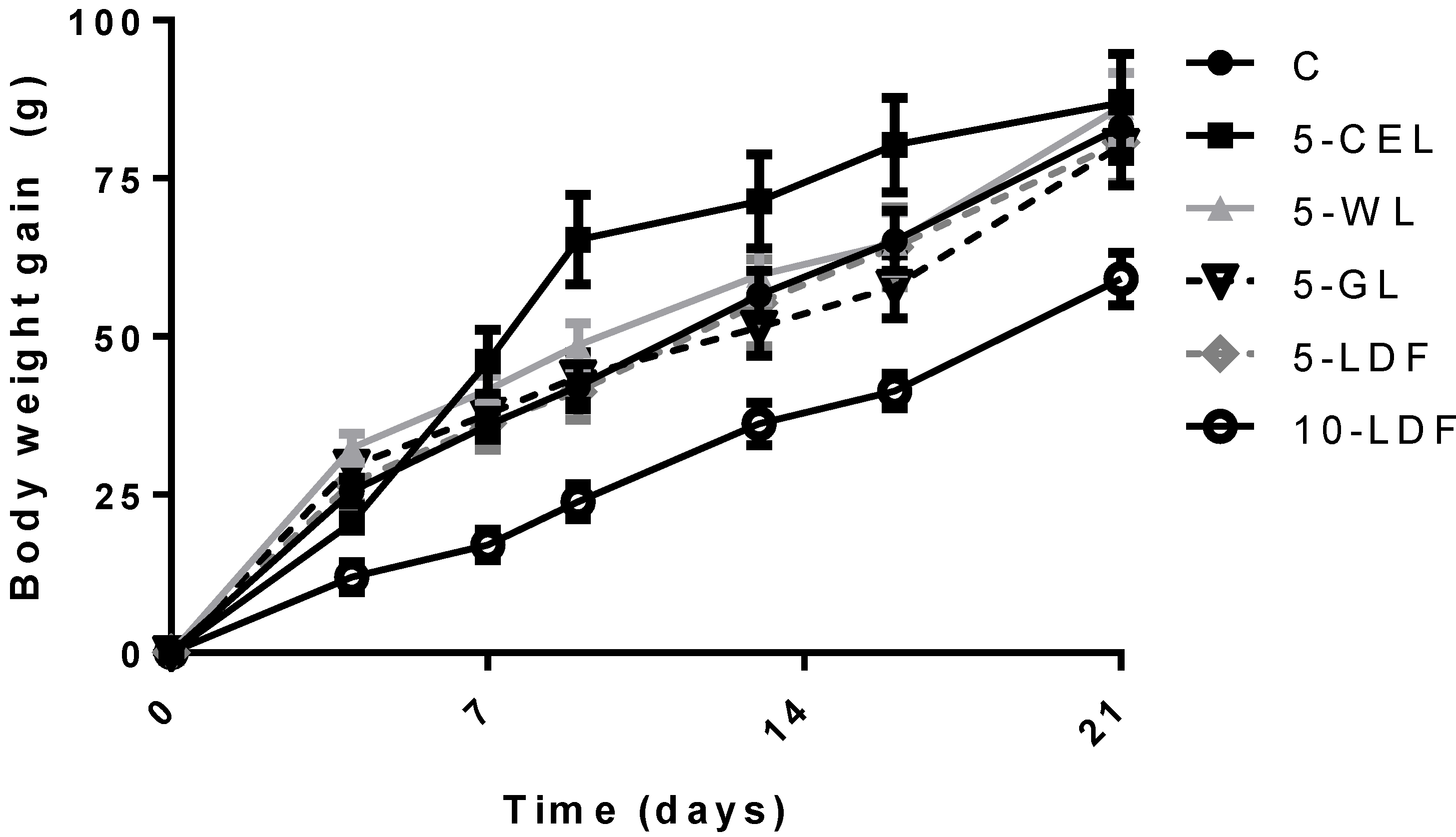
| Final body weight (g) | 282 a | 5.9 | 281 a | 5.9 | 285 a | 5.9 | 278 ab | 5.9 | 279 ab | 5.9 | 258 b | 6.2 |
| Empty body weight (g) | 275 a | 5.9 | 266 ab | 5.9 | 269 ab | 5.9 | 270 ab | 5.9 | 253 b | 5.9 | 222 c | 6.6 |
| Abdominal fat (g) † | 14.1 b | 1.3 | 22.8 a | 0.9 | 14.5 b | 1.3 | 14.0 b | 1.3 | 11.9 b | 1.2 | 10.9 b | 1.6 |
| Protein (%) †‡ | 21.3 | 0.33 | 20.8 | 0.66 | 21.4 | 0.33 | 21.5 | 0.28 | 21.8 | 0.31 | 20.7 | 0.66 |
| Fat (%) †‡ | 13.1 ab | 0.99 | 16.5 a | 1.98 | 12.2 ab | 0.97 | 12.6 ab | 1.0 | 11.7 ab | 0.82 | 10.4 b | 0.82 |
| Heart (mg) † | 949 | 29 | 1028 | 20 | 984 | 29 | 992 | 29 | 974 | 29 | 906 | 36 |
| Liver (g) † | 10.56 a | 0.17 | 9.34 c | 0.11 | 10.11 ab | 0.16 | 9.99 ab | 0.17 | 9.58 bc | 0.15 | 9.73 bc | 0.20 |
4. Discussion
5. Conclusions
Acknowledgements
Conflict of Interest
References
- Bazzano, L.A.; Song, Y.; Bubes, V.; Good, C.K.; Manson, J.E.; Liu, S. Dietary intake of whole and refined grain breakfast cereals and weight gain in men. Obes. Res. 2005, 13, 1952–1960. [Google Scholar] [CrossRef]
- Koh-Banerjee, P.; Franz, M.; Sampson, L.; Liu, S.; Jacobs, D.R., Jr.; Spiegelman, D.; Willett, W.; Rimm, E. Changes in whole-grain, bran, and cereal fiber consumption in relation to 8-y weight gain among men. Am. J. Clin. Nutr. 2004, 80, 1237–1245. [Google Scholar]
- Liu, S.; Willett, W.C.; Manson, J.E.; Hu, F.B.; Rosner, B.; Colditz, G. Relation between changes in intakes of dietary fiber and grain products and changes in weight and development of obesity among middle-aged women. Am. J. Clin. Nutr. 2003, 78, 920–927. [Google Scholar]
- Wanders, A.J.; van den Borne, J.J.; de Graaf, C.; Hulshof, T.; Jonathan, M.C.; Kristensen, M.; Mars, M.; Schols, H.A.; Feskens, E.J. Effects of dietary fibre on subjective appetite, energy intake and body weight: A systematic review of randomized controlled trials. Obes. Rev. 2011, 12, 724–739. [Google Scholar]
- Pittler, M.H.; Ernst, E. Guar gum for body weight reduction: Meta-analysis of randomized trials. Am. J. Med. 2001, 110, 724–730. [Google Scholar] [CrossRef]
- Jull, A.B.; Ni, M.C.; Bennett, D.A.; Dunshea-Mooij, C.A.; Rodgers, A. Chitosan for overweight or obesity. Cochrane Database. Syst. Rev. 2008. [Google Scholar] [CrossRef]
- Pasquier, B.; Armand, M.; Castelain, C.; Guillon, F.; Borel, P.; Lafont, H.; Lairon, D. Emulsification and lipolysis of triacylglycerols are altered by viscous soluble dietary fibres in acidic gastric medium in vitro. Biochem. J. 1996, 314, 269–275. [Google Scholar]
- Isken, F.; Klaus, S.; Osterhoff, M.; Pfeiffer, A.F.H.; Weickert, M.O. Effects of long-term soluble vs. insoluble dietary fiber intake on high-fat diet-induced obesity in C57BL/6J mice. J. Nutr. Biochem. 2010, 21, 278–284. [Google Scholar] [CrossRef]
- Warrand, J.; Michaud, P.; Picton, L.; Muller, G.; Courtois, B.; Ralainirina, R.; Courtois, J. Structural investigations of the neutral polysaccharide of Linum usitatissimum L. seeds mucilage. Int. J. Biol. Macromol. 2005, 35, 121–125. [Google Scholar] [CrossRef]
- Cui, W.; Mazza, G.; Biliaderis, C.G. Chemical structure, molecular size distributions, and rheological properties of flaxseed gum. J. Agric. Food Chem. 1994, 42, 1891–1895. [Google Scholar] [CrossRef]
- Alzueta, C.; Rodriguez, M.L.; Cutuli, M.T.; Rebole, A.; Ortiz, L.T.; Centeno, C.; Trevino, J. Effect of whole and demucilaged linseed in broiler chicken diets on digesta viscosity, nutrient utilisation and intestinal microflora. Br. Poult. Sci. 2003, 44, 67–74. [Google Scholar] [CrossRef]
- Berggren, A.M.; Björck, I.M.E.; Nyman, E.M.G.L.; Eggum, B.O. Short-chain fatty acid content and pH in caecum of rats given various sources of carbohydrates. J. Sci. Food Agric. 1993, 63, 397–406. [Google Scholar] [CrossRef]
- Kristensen, M.; Damgaard, T.M.; Sorensen, A.D.; Raben, A.; Lindelov, T.S.; Thomsen, A.D.; Bjergegaard, C.; Sorensen, H.; Astrup, A.; Tetens, I. Whole flaxseeds but not sunflower seeds in rye bread reduce apparent digestibility of fat in healthy volunteers. Eur. J. Clin. Nutr. 2008, 62, 961–967. [Google Scholar] [CrossRef]
- Kristensen, M.; Savorani, F.; Christensen, S.; Engelsen, S.B.; Bügel, S.; Toubro, S.; Tetens, I.; Astrup, A. Flaxseed dietary fibers suppress postprandial lipemia and appetite sensation in young men. Nutr. Metab. Cardiovasc. Dis. 2011, 26, 136–143. [Google Scholar]
- Kristensen, M.; Jensen, M.G.; Aarestrup, J.; Petersen, K.E.; Sondergaard, L.; Mikkelsen, M.S.; Astrup, A. Flaxseed dietary fibers lower cholesterol and increase fecal fat excretion, but magnitude of effect depend on food type. Nutr. Metab. (Lond.) 2012, 9, 8. [Google Scholar] [CrossRef]
- Prosky, L.; Asp, N.G.; Furda, I.; DeVries, J.W.; Schweizer, T.F.; Harland, B.F. Determination of total dietary fiber in foods and food products: Collaborative study. J. Assoc. Off. Anal. Chem. 1985, 68, 677–679. [Google Scholar]
- Saxholt, E.; Christensen, A.T.; Møller, A.; Hartkopp, H.B.; Hess, Y.; Hels, O. Danish Food Composition Databank, Revision 7; Department of Nutrition, National Food Institute, Technical University of Denmark: Søborg, Dermark, 2008. [Google Scholar]
- National Research Council, Nutrient Requirements of Laboratory Animals, 4th ed.; National Academy Press: Washington, DC, USA, 1995.
- AOAC, Official Methods of Analysis, 15th ed.; Association of Official Analytical Chemists Inc.: Arlington, VA, USA, 1990.
- Hansen, B. Determination of nitrogen as elementary N, an alternative to Kjeldahl. Acta Agric. Scand. 1989, 39, 113–118. [Google Scholar] [CrossRef]
- Bach Knudsen, K.E. Carbohydrate and lignin contents of plant materials used in animal feeding. Anim. Feed Sci. Technol. 2009, 67, 319–338. [Google Scholar] [CrossRef]
- Lauridsen, C.; Christensen, T.B.; Halekoh, U.; Jensen, S.K. Alternative fat sources to animal fat for pigs. Lipid Technol. 2007, 19, 156–159. [Google Scholar] [CrossRef]
- Ibrugger, S.; Kristensen, M.; Mikkelsen, M.S.; Astrup, A. Flaxseed dietary fiber supplements for suppression of appetite and food intake. Appetite 2012, 58, 490–495. [Google Scholar] [CrossRef]
- Artiss, J.D.; Brogan, K.; Brucal, M.; Moghaddam, M.; Jen, K.-L.C. The effects of a new soluble dietary fiber on weight gain and selected blood parameters in rats. Metabolism 2006, 55, 195–202. [Google Scholar] [CrossRef]
- Woo, M.N.; Bok, S.H.; Lee, M.K.; Kim, H.J.; Jeon, S.M.; Do, G.M.; Shin, S.K.; Ha, T.Y.; Choi, M.S. Anti-obesity and hypolipidemic effects of a proprietary herb and fiber combination (S&S PWH) in rats fed high-fat diets. J. Med. Food 2008, 11, 169–178. [Google Scholar] [CrossRef]
- Gonzalez-Esquerra, R.; Leeson, S. Studies on the metabolizable energy content of ground full-fat flaxseed fed in mash, pellet, and crumbled diets assayed with birds of different ages. Poult. Sci. 2000, 79, 1603–1607. [Google Scholar]
- Petit, H.V. Digestion, milk production, milk composition, and blood composition of dairy cows fed whole flaxseed. J. Dairy Sci. 2002, 85, 1482–1490. [Google Scholar] [CrossRef]
- Dikeman, C.L.; Murphy, M.R.; Fahey, G.C., Jr. Dietary fibers affect viscosity of solutions and simulated human gastric and small intestinal digesta. J. Nutr. 2006, 136, 913–919. [Google Scholar]
- Zhao, X.; Jorgensen, H.; Eggum, B.O. The influence of dietary fibre on body composition, visceral organ weight, digestibility and energy balance in rats housed in different thermal environments. Br. J. Nutr. 1995, 73, 687–699. [Google Scholar] [CrossRef]
- Sklan, D.; Smirnov, A.; Plavnik, I. The effect of dietary fibre on the small intestines and apparent digestion in the turkey. Br. Poult. Sci. 2003, 44, 735–740. [Google Scholar] [CrossRef]
- Dvir, I.; Chayoth, R.; Sod-Moriah, U.; Shany, S.; Nyska, A.; Stark, A.H.; Madar, Z.; Arad, S.M. Soluble polysaccharide and biomass of red microalga Porphyridium sp. alter intestinal morphology and reduce serum cholesterol in rats. Br. J. Nutr. 2000, 84, 469–476. [Google Scholar]
- Jensen, B.B.; Jorgensen, H. Effect of dietary fiber on microbial activity and microbial gas production in various regions of the gastrointestinal tract of pigs. Appl. Environ. Microbiol. 1994, 60, 1897–1904. [Google Scholar]
- Bach Knudsen, K.E.; Wisker, E.; Daniel, M.; Feldheim, W.; Eggum, B.O. Digestibility of energy, protein, fat and non-starch polysaccharides in mixed diets: Comparative studies between man and the rat. Br. J. Nutr. 1994, 71, 471–487. [Google Scholar] [CrossRef]
- Sembries, S.; Dongowski, G.; Mehrlander, K.; Will, F.; Dietrich, H. Dietary fiber-rich colloids from apple pomace extraction juices do not affect food intake and blood serum lipid levels, but enhance fecal excretion of steroids in rats. J. Nutr. Biochem. 2004, 15, 296–302. [Google Scholar] [CrossRef]
- Wisker, E.; Bach Knudsen, K.E.; Daniel, M.; Feldheim, W.; Eggum, B.O. Digestibilities of energy, protein, fat and nonstarch polysaccharides in a low fiber diet and diets containing coarse or fine whole meal rye are comparable in rats and humans. J. Nutr. 1996, 126, 481–488. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kristensen, M.; Knudsen, K.E.B.; Jørgensen, H.; Oomah, D.; Bügel, S.; Toubro, S.; Tetens, I.; Astrup, A. Linseed Dietary Fibers Reduce Apparent Digestibility of Energy and Fat and Weight Gain in Growing Rats. Nutrients 2013, 5, 3287-3298. https://doi.org/10.3390/nu5083287
Kristensen M, Knudsen KEB, Jørgensen H, Oomah D, Bügel S, Toubro S, Tetens I, Astrup A. Linseed Dietary Fibers Reduce Apparent Digestibility of Energy and Fat and Weight Gain in Growing Rats. Nutrients. 2013; 5(8):3287-3298. https://doi.org/10.3390/nu5083287
Chicago/Turabian StyleKristensen, Mette, Knud Erik Bach Knudsen, Henry Jørgensen, David Oomah, Susanne Bügel, Søren Toubro, Inge Tetens, and Arne Astrup. 2013. "Linseed Dietary Fibers Reduce Apparent Digestibility of Energy and Fat and Weight Gain in Growing Rats" Nutrients 5, no. 8: 3287-3298. https://doi.org/10.3390/nu5083287
APA StyleKristensen, M., Knudsen, K. E. B., Jørgensen, H., Oomah, D., Bügel, S., Toubro, S., Tetens, I., & Astrup, A. (2013). Linseed Dietary Fibers Reduce Apparent Digestibility of Energy and Fat and Weight Gain in Growing Rats. Nutrients, 5(8), 3287-3298. https://doi.org/10.3390/nu5083287




