Gastric Lipase Secretion in Children with Gastritis
Abstract
:1. Introduction
2. Experimental Section
2.1. Groups of Patients
2.2. Gastric Juice Collection Method
2.3. Laboratory Tests
2.3.1. Measurements of pH
2.3.2. Measurements of Human Gastric Lipase Activity in Gastric Juice
2.3.3. Measurements of Peptide Concentrations in Blood
2.4. Statistical Analysis
3. Results
3.1. Anthropometric Measurements
3.2. pH
3.3. HGL Activity

3.4. Peptide Concentrations
3.5. Correlations
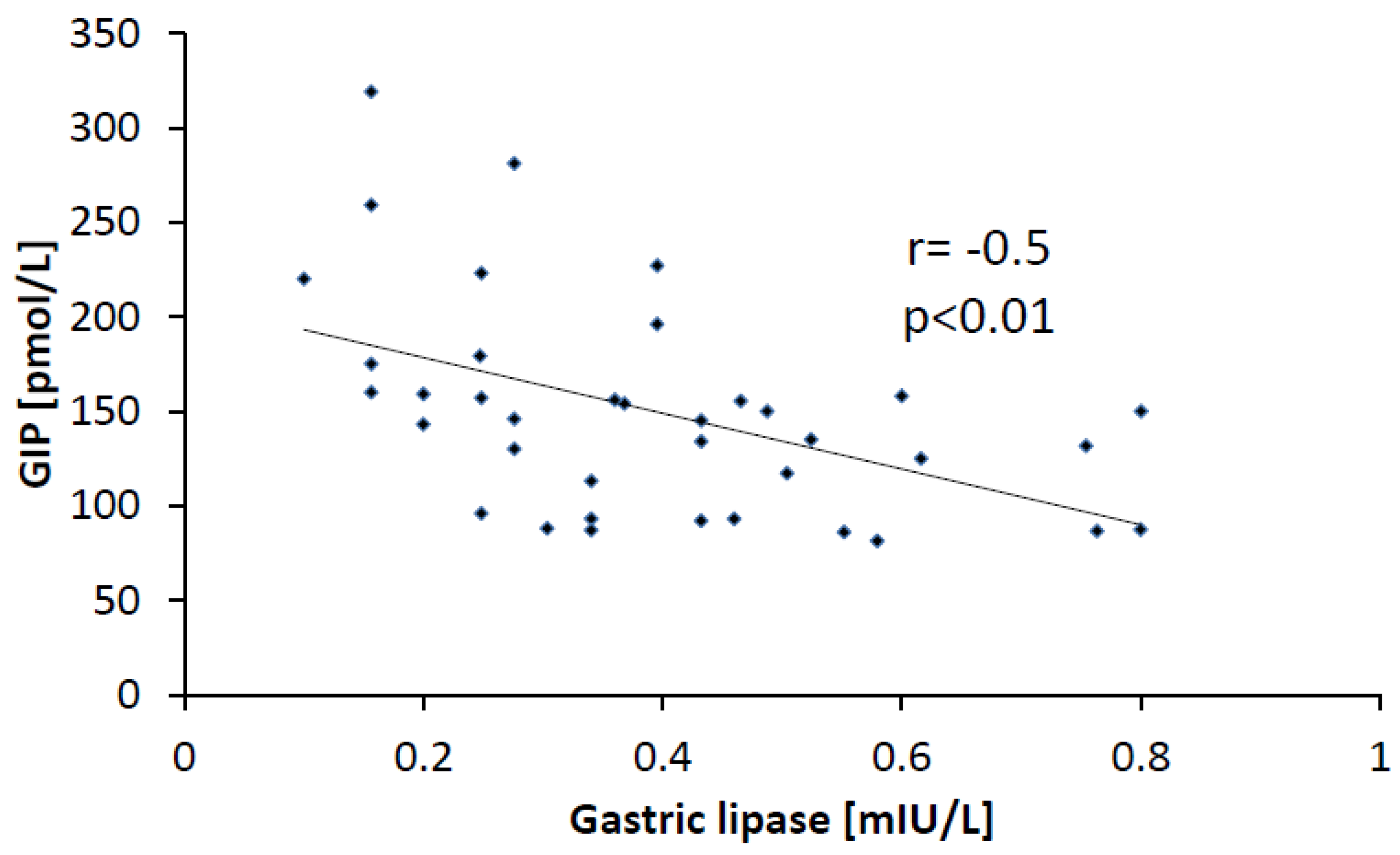
4. Discussion
5. Conclusions
- The activity of HGL is reduced in patients with non-H. pylori gastritis.
- Regulation of HGL activity by GLP-1 and CCK is abnormal in patients with gastritis.
- GIP is a potent regulator of HGL activity.
Acknowledgments
Conflict of Interest
References
- DeNigris, S.J.; Hamosh, M.; Kasbekar, D.K.; Lee, T.C.; Hamosh, P. Lingual and gastric lipases: Species differences in the origin of prepancreatic digestive lipases and in the localization of gastric lipase. Biochim. Biophys. Acta 1988, 959, 38–45. [Google Scholar] [CrossRef]
- Carrière, F.; Raphel, V.; Moreau, H.; Bernadac, A.; Devaux, M.A.; Grimaud, R.; Barrowman, J.A.; Bénicourt, C.; Junien, J.L.; Laugier, R.; et al. Dog gastric lipase: Stimulation of its secretion in vivo and cytolocalization in mucous pit cells. Gastroenterology 1992, 102, 1535–1545. [Google Scholar]
- Hamosh, M.; Henderson, T.R.; Hamosh, P. Gastric lipase and pepsin activities in the developing ferret: Nonparallel development of the two gastric digestive enzymes. J. Pediatr. Gastroenterol. Nutr. 1998, 26, 162–166. [Google Scholar] [CrossRef]
- Moreau, H.; Bernadac, A.; Gargouri, Y.; Benkouka, F.; Laugier, R.; Verger, R. Immunocytolocalization of human gastric lipase in chief cells of the fundic mucosa. Histochemistry 1989, 91, 419–423. [Google Scholar] [CrossRef]
- Lindquist, S.; Hernell, O. Lipid digestion and absorption in early life: An update. Opin. Clin. Nutr. Metab. Care 2010, 13, 314–320. [Google Scholar] [CrossRef]
- Gargouri, Y.; Pieroni, G.; Rivière, C.; Lowe, P.A.; Saunière, J.F.; Sarda, L. Importance of human gastric lipase for intestinal lipolysis: An in vitro study. Biochim. Biophys. Acta 1986, 879, 419–423. [Google Scholar] [CrossRef]
- Carrière, F.; Grandval, P.; Gregory, P.C.; Renou, C.; Henniges, F.; Sander-Struckmeier, S.; Laugier, R. Does the pancreas really produce much more lipase than required for fat digestion? Pancreas 2006, 6, 206–215. [Google Scholar] [CrossRef]
- Armand, M.; Hamosh, M.; Philpott, J.R.; Resnik, A.K.; Rosenstein, B.J.; Hamosh, A.; Perman, J.A.; Hamosh, P. Gastric function in children with cystic fibrosis: Effect of diet on gastric lipase levels and fat digestion. Pediatr. Res. 2004, 55, 457–465. [Google Scholar] [CrossRef]
- Bansi, D.; Price, A.; Russell, C.; Sarner, M. Fibrosing colonopathy in an adult owing to over use of pancreatic enzyme supplements. Gut 2000, 46, 283–285. [Google Scholar] [CrossRef]
- FitzSimmons, S.C.; Burkhart, G.A.; Borowitz, D.; Grand, R.J.; Hammerstrom, T.; Durie, P.R.; Lloyd-Still, J.D.; Lowenfels, A.B. Highdose pancreatic-enzyme supplements and fibrosing colonopathy in children with cystic fibrosis. N. Engl. J. Med. 1997, 336, 1283–1289. [Google Scholar] [CrossRef]
- Romo Vaquero, M.; Yáñez-Gascón, M.J.; García Villalba, R.; Larrosa, M.; Fromentin, E.; Ibarra, A.; Roller, M.; Tomás-Barberán, F.; Espín de Gea, J.C.; García-Conesa, M.T. Inhibition of gastric lipase as a mechanism for body weight and plasma lipids reduction in zucker rats fed a rosemary extract rich in carnosic acid. PLoS One 2012, 7, e39773. [Google Scholar] [CrossRef]
- Wojdemann, M.; Riber, C.; Bisgaard, T.; Sternby, B.; Larsen, S.; Rehfeld, J.F.; Holst, J.J.; Olsen, O. Inhibition of human gastric lipase by intraduodenal fat involves glucagon-like peptide-1 and cholecystokinin. Regul. Pept. 1999, 80, 101–106. [Google Scholar] [CrossRef]
- Sarles, J.; Moreau, H.; Verger, R. Human gastric lipase: Ontogeny and variations in children. Acta Paediatr. 1992, 81, 511–513. [Google Scholar] [CrossRef]
- Gargouri, Y.; Pieroni, G.; Riviere, C.; Sauniere, J.F.; Lowe, P.A.; Sarda, L.; Verger, R. Kinetic assay of human gastric lipase on short-chain and long-chain triacylglycerol emulsions. Gastroenterology 1986, 91, 919–925. [Google Scholar]
- Levy, E.; Goldstein, R.; Freier, S.; Shafrir, E. Gastric lipase in the newborn rat. Pediatr. Res. 1982, 16, 69–74. [Google Scholar]
- Hamosh, M.; Scanlon, J.W.; Ganot, D.; Likel, M.; Scanlon, K.B.; Hamosh, P. Fat digestion in the newborn. Characterization of lipase in gastric aspirates of premature and term infants. J. Clin. Investig. 1981, 67, 838–846. [Google Scholar] [CrossRef]
- Nègre, A.; Salvayre, R.; Dousset, N.; Rogalle, P.; Dang, Q.Q.; Douste-Blazy, L. Hydrolysis of fluorescent pyrenetriacylglycerols by lipases from human stomach and gastric juice. Biochim. Biophys. Acta 1988, 963, 340–348. [Google Scholar] [CrossRef]
- Olsen, O.; Wojdemann, M.; Berner, B.; Christiansen, G.; Sternby, B. Secretin and gastric lipase secretion. Digestion 1998, 59, 655–659. [Google Scholar] [CrossRef]
- Wojdemann, M.; Wettergren, A.; Sternby, B.; Holst, J.J.; Larsen, S; Rehfeld, J.F.; Olsen, O. Inhibition of human gastric lipase secretion by glucagon-like peptide-1. Dig. Dis. Sci. 1998, 43, 799–805. [Google Scholar] [CrossRef]
- Borovicka, J.; Schwizer, W.; Mettraux, C.; Kreiss, C.; Remy, B.; Asal, K.; Jansen, J.B.; Douchet, I.; Verger, R.; Fried, M. Regulation of gastric and pancreatic lipase secretion by CCK and cholinergic mechanisms in humans. Am. J. Physiol. 1997, 273, G374–G380. [Google Scholar]
- Kim, S.J.; Nian, C.; McIntosh, C.H. GIP increases human adipocyte LPL expression through CREB and TORC2-mediated trans-activation of the LPL gene. J. Lipid Res. 2010, 51, 3145–3157. [Google Scholar] [CrossRef]
- Duan, R.D.; Erlanson-Albertsson, C. Gastric inhibitory polypeptide stimulates pancreatic lipase and colipase synthesis in rats. Am. J. Physiol. 1992, 262, G779–G784. [Google Scholar]
- Pedersen, P.B.; Vilmann, P.; Bar-Shalom, D.; Müllertz, A.; Baldursdottir, S. Characterization of fasted human gastric fluid for relevant rheological parameters and gastric lipase activities. Eur. J. Pharm. Biopharm. 2013, in press. [Google Scholar]
- Carriere, F.; Barrowman, J.A.; Verger, R.; Laugier, R. Secretion and contribution to lipolysis of gastric and pancreatic lipases during a test meal in humans. Gastroenterology 1993, 105, 876–888. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tomasik, P.J.; Wędrychowicz, A.; Rogatko, I.; Zając, A.; Fyderek, K.; Sztefko, K. Gastric Lipase Secretion in Children with Gastritis. Nutrients 2013, 5, 2924-2932. https://doi.org/10.3390/nu5082924
Tomasik PJ, Wędrychowicz A, Rogatko I, Zając A, Fyderek K, Sztefko K. Gastric Lipase Secretion in Children with Gastritis. Nutrients. 2013; 5(8):2924-2932. https://doi.org/10.3390/nu5082924
Chicago/Turabian StyleTomasik, Przemyslaw J., Andrzej Wędrychowicz, Iwona Rogatko, Andrzej Zając, Krzysztof Fyderek, and Krystyna Sztefko. 2013. "Gastric Lipase Secretion in Children with Gastritis" Nutrients 5, no. 8: 2924-2932. https://doi.org/10.3390/nu5082924



