Naringin Improves Diet-Induced Cardiovascular Dysfunction and Obesity in High Carbohydrate, High Fat Diet-Fed Rats
Abstract
:1. Introduction
2. Experimental Section
2.1. Rats and Diets
2.2. Mitochondrial Preparations
2.3. Histology
2.4. Statistical Analysis
3. Results
3.1. Metabolic Parameters
| Parameters | C | CN | H | HN | p value | ||
|---|---|---|---|---|---|---|---|
| Diet | Treatment | Interaction | |||||
| Initial body weight, g | 330 ± 3 | 327 ± 2 | 327 ± 3 | 327 ± 2 | 0.56 | 0.56 | 0.56 |
| Final body weight, g | 451 ± 13 b | 472 ± 12 ab | 509 ± 6 a | 503 ± 14 a | 0.0006 | 0.53 | 0.26 |
| % of weight gain at week 16 | 8.3 ± 1.1 b | 7.6 ± 1.5 b | 13.6 ± 1.6 a | 13.4 ± 1.5 a | 0.0004 | 0.81 | 0.92 |
| Food intake at week 16, g/day | 33.4 ± 0.7 a | 32.2 ± 0.4 a | 28.0 ± 1.1 b | 28.1 ± 0.8 b | <0.0001 | 0.49 | 0.42 |
| Water intake at week 16, mL/day | 31.9 ± 3.4 a | 34.3 ± 0.8 a | 22.8 ± 1.4 b | 28.7 ± 0.6 a | 0.0005 | 0.37 | 0.37 |
| Energy intake at week 16, kJ/day | 407 ± 9 a | 396 ± 5 a | 590 ± 20 b | 603 ± 11 b | <0.0001 | 0.94 | 0.35 |
| Kidney weight, mg/mm | 47.0 ± 1.5 c | 57.8 ± 1.6 a | 55.5 ± 2.7 ab | 55.1 ± 1.2 ab | 0.1 | 0.0075 | 0.0032 |
| Spleen, mg/mm | 17.1 ± 0.7 | 19.1 ± 0.9 | 20.8 ± 2.3 | 19.4 ± 0.6 | 0.14 | 0.82 | 0.21 |
| Liver wet weight, mg/mm | 243.5 ± 5.4 a | 297.9 ± 11.5 c | 339.0 ± 14.2 b | 289.7 ± 8.5 c | 0.0002 | 0.81 | <0.0001 |
| Abdominal fat pads, mg/mm | 449.2 ± 37.3 b | 382.0 ± 46.1 b | 730.3 ± 34.3 a | 695.8 ± 70.0 a | <0.0001 | 0.31 | 0.74 |
| Retroperitoneal fat, mg/mm | 266.0 ± 21.9 b | 188.2 ± 21.3 c | 422.9 ± 18.7 a | 350.8 ± 37.0 a | <0.0001 | 0.0072 | 0.92 |
| Epididymal fat, mg/mm | 97.4 ± 13.4 b | 97.5 ± 11.3 b | 173.4 ± 15.7 a | 167.7 ± 15.2 a | <0.0001 | 0.84 | 0.84 |
| Omental fat, mg/mm | 85.8 ± 6.7 b | 96.2 ± 15.2 b | 134.0 ± 12.8 b | 177.3 ± 21.2 a | 0.0002 | 0.08 | 0.28 |
| Abdominal circumference at 16 weeks, cm | 21.7 ± 0.3 bc | 21.2 ± 0.3 c | 23.8 ± 0.15 a | 22.3 ± 0.2 b | <0.0001 | 0.0004 | 0.05 |
| Pancreas wet weight, mg/mm | 45.3 ± 1.9 b | 47.0 ± 3.9 b | 60.8 ± 2.9 a | 50.0 ± 0.4 b | 0.0014 | 0.09 | 0.0239 |
| Oral glucose tolerance test (area under curve) | |||||||
| Area under curve (AUC) at 0 week | 683 ± 21 | 679 ± 19 | 671 ± 18 | 684 ± 20 | 0.85 | 0.84 | 0.68 |
| AUC at 8 weeks | 682 ± 12 a | 700 ± 12 a | 802 ± 19 b | 781 ± 16 b | <0.0001 | 0.91 | 0.21 |
| AUC at 16 weeks | 707 ± 8 b | 687 ± 21 b | 854 ± 18 a | 709 ± 18 b | <0.0001 | <0.0001 | 0.0011 |
| Parameters | C | CN | H | HN | p values | ||
|---|---|---|---|---|---|---|---|
| Diet | Treatment | Interaction | |||||
| ALT, U/L | 28.4 ± 4.1 b | 33.8 ± 1.0 ab | 41.9 ± 2.1 a | 37.5 ± 2.3 ab | 0.0028 | 0.85 | 0.07 |
| AST, U/L | 68.6 ± 3.5 b | 67.8 ± 4.1 b | 84.4 ± 5.9 a | 72.8 ± 2.7 ab | 0.0201 | 0.15 | 0.21 |
| Triglycerides, mmol/L | 0.6 ± 0.1 | 1.2 ± 0.2 | 1.4 ± 0.4 | 1.6 ± 0.3 | 0.0369 | 0.16 | 0.47 |
| Total cholesterol, mmol/L | 1.6 ± 0.1 b | 1.7 ± 0.1 b | 2.2 ± 0.1 a | 1.6 ± 0.1 b | 0.0186 | 0.0186 | 0.0016 |
| NEFA, mmol/L | 1.4 ± 0.2 b | 0.7 ± 0.1 b | 3.8 ± 0.7 a | 0.8 ± 0.1 b | 0.0022 | <0.0001 | 0.0044 |
| Uric acid, mmol/L | 46.7 ± 1.8 a | 29.6 ± 2.1 b | 49.0 ± 2.1 a | 43.9 ± 5.9 a | 0.022 | 0.0031 | 0.09 |
| Insulin, μmol/L | 1.9 ± 0.2 b | 2.2 ± 0.5 b | 4.2 ± 0.8 a | 1.7 ± 0.2 b | 0.08 | 0.0337 | 0.0083 |
| Plasma TBARS, µmol/L | 12.4 ± 1.3 b | 14.4 ± 0.9 b | 37.4 ± 6.1 a | 17.1 ± 1.8 b | 0.0097 | 0.0004 | 0.0025 |
3.2. Cardiovascular Structure and Function
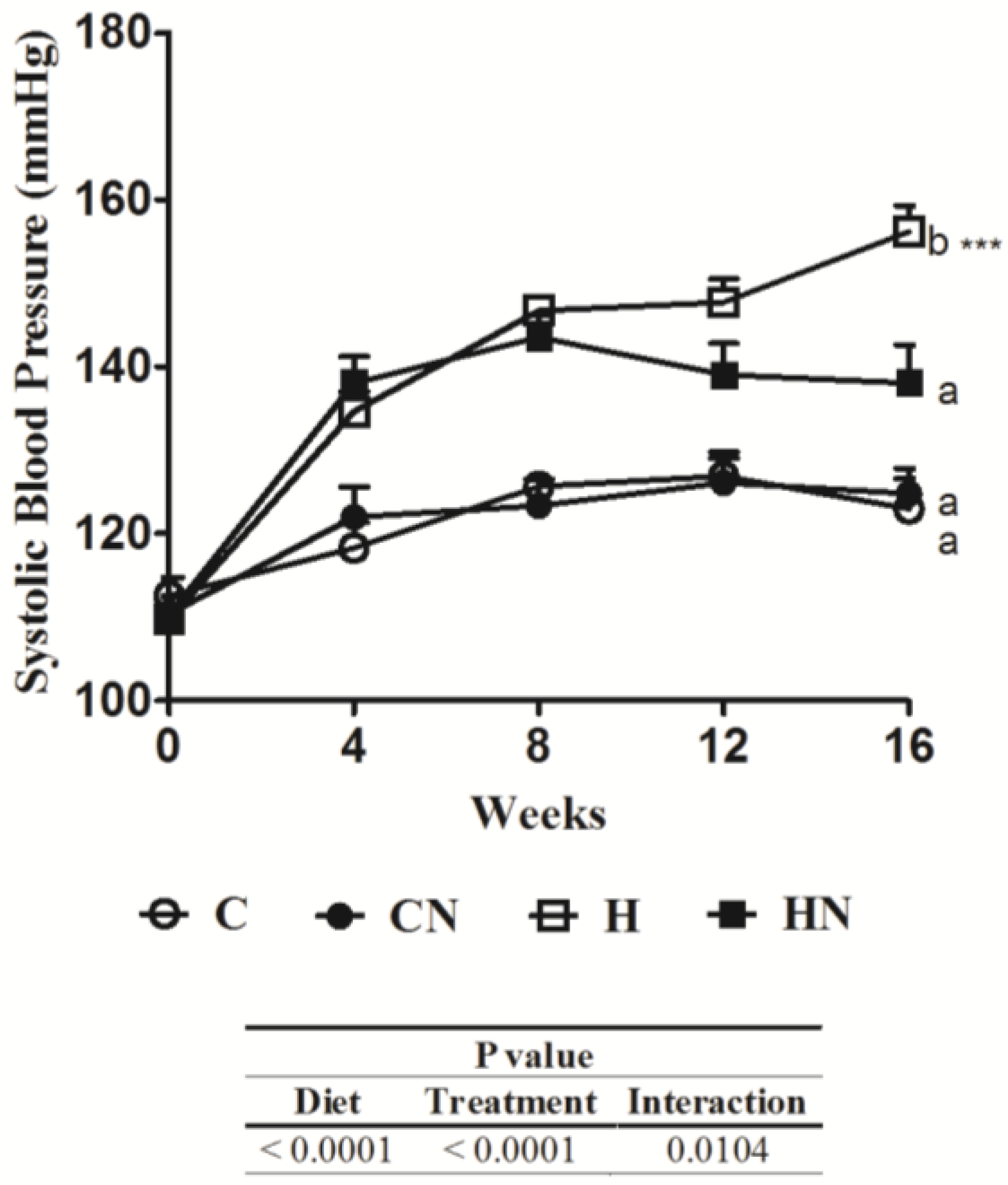
| Parameters | C | CN | H | HN | p value | ||
|---|---|---|---|---|---|---|---|
| Diet | Treatment | Interaction | |||||
| LVIDd, mm | 7.14 ± 0.16 b | 7.59 ± 0.18 ab | 8.35 ± 0.26 a | 7.62 ± 0.21 ab | <0.0001 | <0.0001 | <0.0001 |
| LVPWd, mm | 1.84 ± 0.03 b | 1.94 ± 0.12 b | 2.22 ± 0.05 a | 1.92 ± 0.05 b | 0.0215 | 0.18 | 0.0116 |
| Fractional shortening, % | 48.2 ± 1.2 a | 56.4 ± 4.9 a | 35.7 ± 1.2 b | 48.3 ± 2.8 a | 0.0021 | 0.002 | 0.47 |
| Ejection fraction, % | 90.7 ± 1.2 b | 89.4 ± 1.5 b | 96.5 ± 0.3 a | 92.5 ± 1.3 b | 0.001 | 0.0312 | 0.26 |
| Relative wall thickness | 0.49 ± 0.01 a | 0.57 ± 0.06 b | 0.49 ± 0.01 a | 0.50 ± 0.02 a | 0.08 | 0.0296 | 0.08 |
| Ascending aortic flow, m/s | 0.86 ± 0.02 | 0.98 ± 0.13 | 1.08 ± 0.05 | 0.82 ± 0.08 | 0.7 | 0.37 | 0.0203 |
| Descending aortic flow, m/s | 0.79 ± 0.6 | 0.73 ± 0.8 | 0.96 ± 0.7 | 0.77 ± 0.7 | 0.15 | 0.09 | 0.36 |
| E:A ratio | 1.9 ± 0.2 | - * | 1.4 ± 0.2 | 1.7 ± 0.4 | - | - | - |
| Ejection time, ms | 87.3 ± 1.5 a | 69.3 ± 8.6 b | 77.0 ± 2.8 ab | 81.8 ± 2.7 ab | 0.81 | 0.16 | 0.0187 |
| Deceleration time, ms | 58.5 ± 1.9 a | 37.9 ± 3.6 b | 45.4 ± 2.9 b | 47.0 ± 3.8 b | 0.06 | 0.0472 | 0.0151 |
| Estimated LV mass, g | 0.84 ± 0.04 b | 1.11 ± 0.09 ab | 1.25 ± 0.09 a | 1.01 ± 0.05 ab | 0.0411 | 0.84 | 0.0017 |
| Heart wet weight, mg/mm | 26.5 ± 0.9 b | 25.4 ± 0.6 b | 30.1 ± 1.4 a | 25.6 ± 0.5 b | 0.05 | 0.0055 | 0.08 |
| LV wet weight, mg/mm | 20.5 ± 0.4 b | 18.8 ± 0.5 b | 22.7 ± 0.8 a | 19.8 ± 0.4 b | 0.007 | 0.0002 | 0.29 |
| RV wet weight, mg/mm | 4.7 ± 0.2 | 4.4 ± 0.2 | 4.6 ± 0.2 | 4.3 ± 0.2 | 0.62 | 0.14 | 1 |
| LV stiffness constant, κ | 22.9 ± 0.4 b | 24.6 ± 0.9 b | 29.3 ± 0.5 a | 25.1 ± 1.0 b | <0.0001 | 0.11 | 0.0005 |
| LV interstitial collagen, % | 5.8 ± 0.5 b | 4.8 ± 0.9 b | 18.4 ± 0.8 a | 13.5 ± 0.5 b | 0.0212 | 0.0025 | <0.0001 |

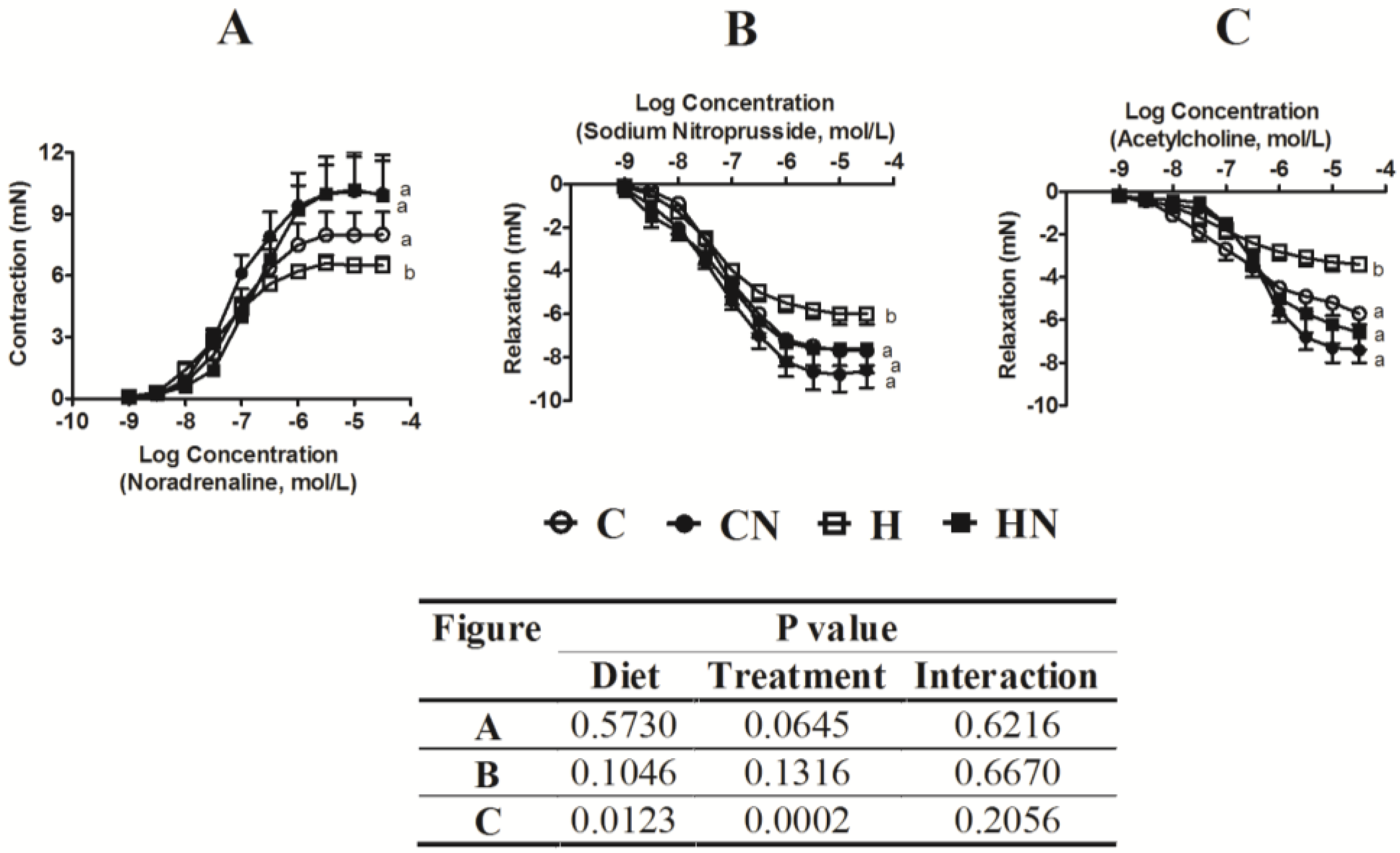
3.3. Liver Structure and Function
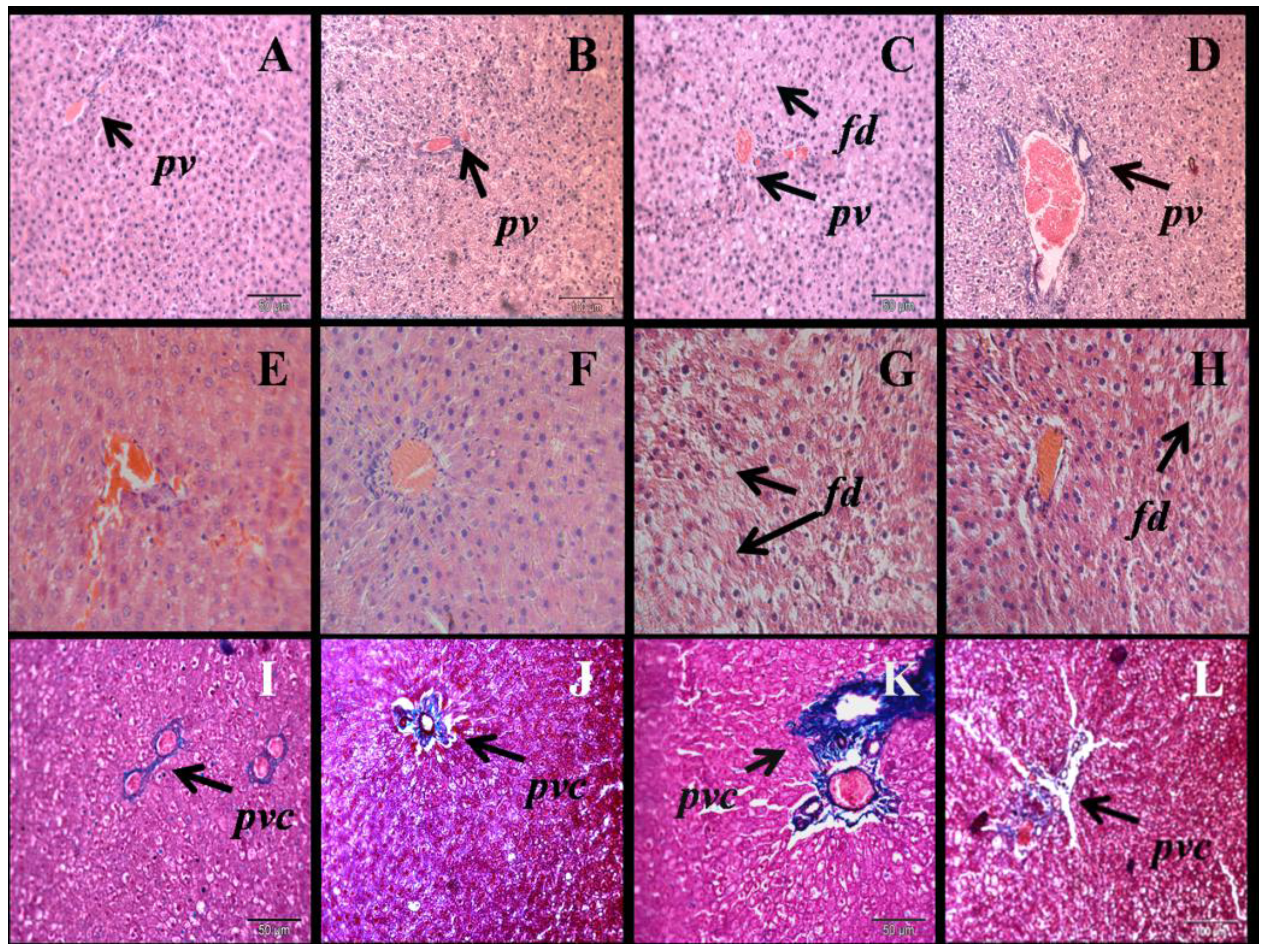
| Respiratory parameter | C | CN | H | HN | p Value | ||
|---|---|---|---|---|---|---|---|
| Diet | Treatment | Interaction | |||||
| Succinate + Rotenone-dependent | |||||||
| State 3, nmol/min/mg protein | 121.8 ± 15.6 a | 120.6 ± 17.3 a | 55.7 ± 6.6 b | 111.4 ± 2.8 a | 0.0069 | 0.0394 | 0.0324 |
| State 4, nmol/min/mg protein | 19.2 ± 3.2 | 22.8 ± 2.0 | 17.4 ± 1.2 | 26.7 ± 3.4 | 0.71 | 0.0343 | 0.32 |
| RCR | 6.8 ± 1.0 a | 5.6 ± 1.1 ab | 3.2 ± 0.3 b | 4.5 ± 0.6 ab | 0.0108 | 0.95 | 0.14 |
4. Discussion
5. Conclusions
Acknowledgments
Conflict of Interest
References
- Hossain, P.; Kawar, B.; El Nahas, M. Obesity and diabetes in the developing world—a growing challenge. N. Eng. J. Med. 2007, 356, 213–215. [Google Scholar] [CrossRef]
- Phillips, L.; Prins, J. The link between abdominal obesity and the metabolic syndrome. Curr. Hypertens. Rep. 2008, 10, 156–164. [Google Scholar] [CrossRef]
- Tripoli, E.; Guardia, M.L.; Giammanco, S.; Majo, D.D.; Giammanco, M. Citrus flavonoids: Molecular structure, biological activity and nutritional properties: A review. Food Chem. 2007, 104, 466–479. [Google Scholar]
- Jeon, S.M.; Park, Y.B.; Choi, M.S. Antihypercholesterolemic property of naringin alters plasma and tissue lipids, cholesterol-regulating enzymes, fecal sterol and tissue morphology in rabbits. Clin. Nutr. 2004, 23, 1025–1034. [Google Scholar] [CrossRef]
- Rajadurai, M.; Prince, P.S.M. Preventive effect of naringin on lipids, lipoproteins and lipid metabolic enzymes in isoproterenol-induced myocardial infarction in Wistar rats. J. Biochem. Mol. Toxicol. 2006, 20, 191–197. [Google Scholar] [CrossRef]
- Rajadurai, M.; Prince, P.S.M. Preventive effect of naringin on lipid peroxides and antioxidants in isoproterenol-induced cardiotoxicity in Wistar rats: Biochemical and histopathological evidences. Toxicology 2006, 228, 259–268. [Google Scholar] [CrossRef]
- Kannappan, S.; Anuradha, C.V. Naringenin enhances insulin-stimulated tyrosine phosphorylation and improves the cellular actions of insulin in a dietary model of metabolic syndrome. Eur. J. Nutr. 2010, 49, 101–109. [Google Scholar] [CrossRef]
- Sharma, A.K.; Bharti, S.; Ojha, S.; Bhatia, J.; Kumar, N.; Ray, R.; Kumari, S.; Arya, D.S. Up-regulation of PPARγ, heat shock protein-27 and -72 by naringin attenuates insulin resistance, β-cell dysfunction, hepatic steatosis and kidney damage in a rat model of type 2 diabetes. Br. J. Nutr. 2011, 106, 1713–1723. [Google Scholar] [CrossRef]
- Pu, P.; Gao, D.-M.; Mohamed, S.; Chen, J.; Zhang, J.; Zhou, X.-Y.; Zhou, N.-J.; Xie, J.; Jiang, H. Naringin ameliorates metabolic syndrome by activating AMP-activated protein kinase in mice fed a high-fat diet. Arch. Biochem. Biophys. 2012, 518, 61–70. [Google Scholar]
- Jung, U.J.; Lee, M.K.; Park, Y.B.; Kang, M.A.; Choi, M.S. Effect of citrus flavonoids on lipid metabolism and glucose-regulating enzyme mRNA levels in type-2 diabetic mice. Int. J. Biochem. Cell. Biol. 2006, 38, 1134–1145. [Google Scholar] [CrossRef]
- Panchal, S.K.; Poudyal, H.; Iyer, A.; Nazer, R.; Alam, A.; Diwan, V.; Kauter, K.; Sernia, C.; Campbell, F.; Ward, L.; et al. High-carbohydrate high-fat diet-induced metabolic syndrome and cardiovascular remodeling in rats. J. Cardiovasc. Pharmacol. 2011, 57, 611–624. [Google Scholar] [CrossRef]
- Panchal, S.K.; Poudyal, H.; Arumugam, T.V.; Brown, L. Rutin attenuates metabolic changes, nonalcoholic steatohepatitis, and cardiovascular remodeling in high-carbohydrate, high-fat diet-fed rats. J. Nutr. 2011, 141, 1062–1069. [Google Scholar] [CrossRef]
- Panchal, S.K.; Poudyal, H.; Brown, L. Quercetin ameliorates cardiovascular, hepatic, and metabolic changes in diet-induced metabolic syndrome in rats. J. Nutr. 2012, 142, 1026–1032. [Google Scholar] [CrossRef]
- Panchal, S.K.; Poudyal, H.; Waanders, J.; Brown, L. Coffee extract attenuates changes in cardiovascular and hepatic structure and function without decreasing obesity in high-carbohydrate, high-fat diet-fed male rats. J. Nutr. 2012, 142, 690–697. [Google Scholar] [CrossRef]
- Niehius, W.G.; Samuelsson, B. Formation of malondialdehyde from phospholipids arachidonate during microsomal lipidperoxidation. Eur. J. Biochem. 1968, 6, 126–130. [Google Scholar] [CrossRef]
- Frezza, C.; Cipolat, S.; Scorrano, L. Organelle isolation: Functional mitochondria from mouse liver, muscle and cultured filroblasts. Nat. Protoc. 2007, 2, 287–295. [Google Scholar] [CrossRef]
- Chanet, A.; Milenkovic, D.; Manach, C.; Mazur, A.; Morand, C. Citrus flavanones: What is their role in cardiovascular protection? J. Agric. Food Chem. 2012, 60, 8809–8822. [Google Scholar]
- Ikemura, M.; Sasaki, Y.; Giddings, J.C.; Yamamoto, J. Preventive effects of hesperidin, glucosyl gesperidin and naringin on hypertension and cerebral thrombosis in stroke-prone spontaneously hypertensive rats. Phytother. Res. 2012, 26, 1272–1277. [Google Scholar] [CrossRef]
- Rajadurai, M.; Prince, P.S. Naringin ameliorates mitochondrial lipid peroxides, antioxidants and lipids in isoproterenol-induced myocardial infarction in Wistar rats. Phytother. Res. 2009, 23, 358–362. [Google Scholar] [CrossRef]
- Johnson, A.R.; Milner, J.J.; Makowski, L. The inflammation highway: Metabolism accelerates inflammatory traffic in obesity. Immunol. Rev. 2012, 249, 218–238. [Google Scholar] [CrossRef]
- Jain, M.; Parmar, H.S. Evaluation of antioxidative and anti-inflammatory potential of hesperidin and naringin on the rat air pouch model of inflammation. Inflamm. Res. 2011, 60, 483–491. [Google Scholar] [CrossRef]
- Mahmoud, A.M.; Ashour, M.B.; Abel-Moneim, A.; Ahmed, O.M. Hesperidin and naringin attenuate hyperglycemia-mediated oxidative stress andproinflammatory cytokine production in high fat fed/streptozotocin-inducedtype 2 diabetic rats. J. Diabetes Complicat. 2012, 26, 483–490. [Google Scholar] [CrossRef]
- Morikawa, K.; Nonaka, M.; Mochizuki, H.; Handa, K.; Hanada, H.; Hirota, K. Naringenin and hesperetin induce growth arrest, apoptosis, and cytoplasmic fat deposit in human preadipocytes. J. Agric. Food Chem. 2008, 26, 11030–11037. [Google Scholar]
- Jung, U.J.; Lee, M.K.; Jeong, K.S.; Choi, M.S. The hypoglycemic effects of hesperidin and naringin are partly mediated by hepatic glucose-regulating enzymes in C57BL/KsJ-db/db mice. J. Nutr. 2004, 134, 2499–2503. [Google Scholar]
- Parmer, H.S.; Jain, P.; Chauhan, D.S.; Bhinchar, M.K.; Munjal, V.; Yusuf, M.; Choube, K.; Tawani, A.; Tiwari, V.; Manivannan, E.; Kumar, A. DPP-IV inhibitory potential of naringin: An in silico, in vitro and in vivo study. Diabetes Res. Clin. Pract. 2012, 97, 105–111. [Google Scholar] [CrossRef]
- Kim, S.-Y.; Kim, H.-J.; Lee, M.-K.; Jeon, S.-M.; Do, G.-M.; Kwon, E.-Y.; Cho, Y.Y.; Kim, D.J.; Jeong, K.S.; Park, Y.B.; et al. Naringin time-dependently lowers hepatic cholesterol biosynthesis and plasma cholesterol in rats fed high-fat and high-cholesterol diet. J. Med. Food 2006, 9, 582–586. [Google Scholar] [CrossRef]
- Demonty, I.; Lin, Y.; Zebregs, Y.E.; Vermeer, M.A.; van der Knaap, H.C.; Jaekel, M.; Trautwein, E.A. The citrus flavonoids hesperidin and naringin do not affect serum cholesterol in moderately hypercholesterolemic men and women. J. Nutr. 2010, 140, 1615–1620. [Google Scholar] [CrossRef]
- Ali, M.M.; El Kader, M.A. The influence of naringin on the oxidative state of rats with streptozotocin-induced acute hyperglycaemia. Z. Naturforsch. C 2004, 59, 726–733. [Google Scholar]
- Mantena, S.K.; Vaughn, D.P.; Andringa, K.K.; Eccleston, H.B.; King, A.L.; Abrams, G.A.; Doeller, J.E.; Kraus, D.W.; Darley-Usmar, V.M.; Bailey, S.M. High fat diet induces dysregulation of hepatic oxygen gradients and mitochondrial function in vivo. Biochem. J. 2009, 417, 183–193. [Google Scholar] [CrossRef]
- Crespy, V.; Morand, C.; Besson, C.; Cotelle, N.; Vezin, H.; Demigne, C.; Remesy, C. The splanchnic metabolism of flavonoids highly differed according to the nature of the compound. Am. J. Physiol. Gastrointest. Liver Physiol. 2003, 284, G980–G988. [Google Scholar]
- Bokkenheuser, V.D.; Shackleton, C.H.; Winter, J. Hydrolysis of dietary flavonoid glycosides by strains of intestinal Bacteroides from humans. Biochem. J. 1987, 248, 953–956. [Google Scholar]
- Erlund, I.; Meririnne, E.; Alfthan, G.; Aro, A. Plasma kinetics and urinary excretion of the flavanones naringenin and hesperetin in humans after ingestion of orange juice and grapefruit juice. J. Nutr. 2001, 131, 235–241. [Google Scholar]
- Kanaze, F.I.; Bounartzi, M.I.; Georgarakis, M.; Niopas, I. Pharmacokinetics of the citrus flavonone aglycones hesperetin and naringenin after single oral administration in human subjects. Eur. J. Clin. Nutr. 2007, 61, 472–477. [Google Scholar]
- Vallejo, F.; Larrosa, M.; Escudero, E.; Zafrilla, M.P.; Cerdá, B.; Boza, J.; García-Conesa, M.T.; Espín, J.C.; Tomás-Barberán, F.A. Concentration and solubility of flavanones in orange beverages affect their bioavailability in humans. J. Agric. Food Chem. 2010, 58, 6516–6524. [Google Scholar]
- Vajro, P.; Paolella, G.; Fasano, A. Microbiota and gut-liver axis: A mini-review on their influence on obesity and obesity related liver disease. J. Pediatr. Gastroenterol. Nutr. 2013. [Google Scholar] [CrossRef]
- Bolca, S.; van de Wiele, T.; Possemiers, S. Gut metabotypes govern health effects of dietary polyphenols. Curr. Opin. Biotechnol. 2012. [Google Scholar] [CrossRef]
- Duda-Chodak, A. The inhibitory effect of polyphenols on human gut microbiota. J. Physiol. Pharmacol. 2012, 63, 497–503. [Google Scholar]
- Tuohy, K.M.; Conferno, L.; Gasperotti, M.; Viola, R. Up-regulating the human intestinal microbiome using whole plant foods, polyphenols, and/or fiber. J. Agric. Food Chem. 2012, 60, 8776–8782. [Google Scholar] [CrossRef]
- Bachmann, K.; Pardoe, D.; White, D. Scaling basic toxicokinetic parameters from rat to man. Environ. Health Perspect. 1996, 104, 400–407. [Google Scholar] [CrossRef]
- Reagan-Shaw, S.; Nihal, M.; Ahmad, N. Dose translation from animal to human studies revisited. FASEB J. 2008, 22, 659–661. [Google Scholar] [CrossRef]
- Scalbert, A.; Williamson, G. Dietary intake and bioavailability of polyphenols. J. Nutr. 2000, 130, 2073S–2085S. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Alam, M.A.; Kauter, K.; Brown, L. Naringin Improves Diet-Induced Cardiovascular Dysfunction and Obesity in High Carbohydrate, High Fat Diet-Fed Rats. Nutrients 2013, 5, 637-650. https://doi.org/10.3390/nu5030637
Alam MA, Kauter K, Brown L. Naringin Improves Diet-Induced Cardiovascular Dysfunction and Obesity in High Carbohydrate, High Fat Diet-Fed Rats. Nutrients. 2013; 5(3):637-650. https://doi.org/10.3390/nu5030637
Chicago/Turabian StyleAlam, Md. Ashraful, Kathleen Kauter, and Lindsay Brown. 2013. "Naringin Improves Diet-Induced Cardiovascular Dysfunction and Obesity in High Carbohydrate, High Fat Diet-Fed Rats" Nutrients 5, no. 3: 637-650. https://doi.org/10.3390/nu5030637
APA StyleAlam, M. A., Kauter, K., & Brown, L. (2013). Naringin Improves Diet-Induced Cardiovascular Dysfunction and Obesity in High Carbohydrate, High Fat Diet-Fed Rats. Nutrients, 5(3), 637-650. https://doi.org/10.3390/nu5030637






