Bone Mass and Mineral Metabolism Alterations in Adult Celiac Disease: Pathophysiology and Clinical Approach
Abstract
:1. Introduction
2. Bone Damage and Mineral Metabolism Derangement in Celiac Disease
2.1. Effect of GFD
3. Fracture Risk
4. Clinical Management
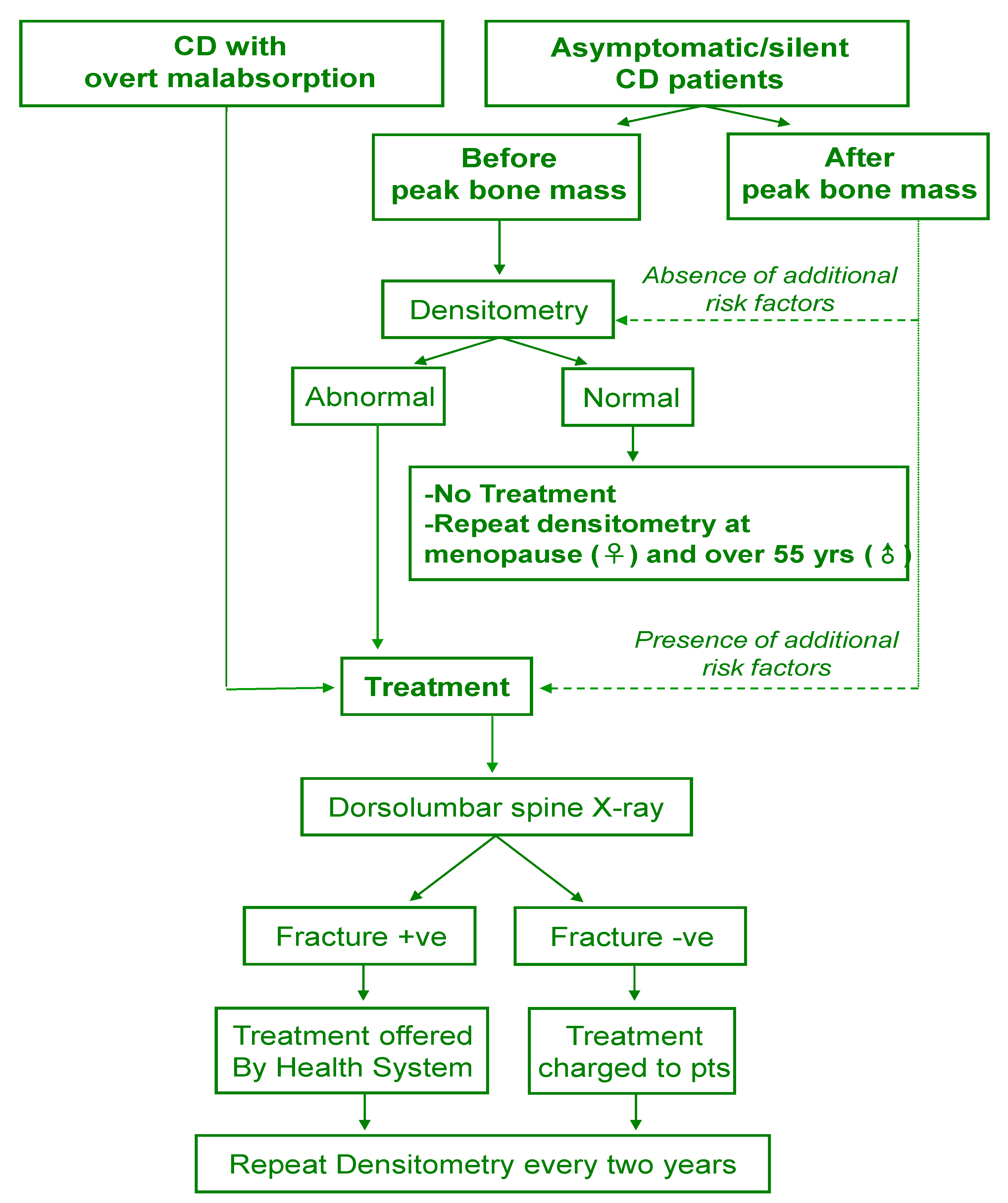
5. Conclusions
Conflicts of Interest
References
- Consensus development conference: Prophylaxis and treatment of osteoporosis. Osteoporos. Int. 1991, 1, 114–117. [CrossRef]
- WHO Scientific Group on the Assessment of Osteoporosis at Primary Health Care Levels. In Summary Meeting Report 2004; World Health Organization: Brussels, Belgium, 2007; pp. 1–17.
- Corazza, G.R.; Di Sario, A.; Cecchetti, A.; Jorizzo, R.A.; Di Stefano, M.; Minguzzi, L.; Brusco, G.; Bernardi, M.; Gasbarrini, G. Influence of pattern of clinical presentation and gluten-free diet on bone mass and metabolism in adult coeliac disease. Bone 1996, 18, 525–530. [Google Scholar] [CrossRef]
- Mazure, R.; Vazquez, H.; Gonzalez, D.; Mautalen, C.; Pedreira, S.; Boerr, L.; Bai, J.C. Bone mineral affectation in asymptomatic adult patients with celiac disease. Am. J. Gastroenterol. 1994, 89, 2130–2134. [Google Scholar]
- Corazza, G.R.; di Sario, A.; Cecchetti, A.; Tarozzi, C.; Corrao, G.; Bernardi, M.; Gasbarrini, G. Bone mass and metabolism in patients with celiac disease. Gastroenterology 1995, 109, 122–128. [Google Scholar] [CrossRef]
- McFarlane, X.A.; Bhalla, A.K.; Robertson, D.A.F. Effect of gluten free diet on osteopenia in adults with newly diagnosed celiac disease. Gut 1996, 39, 180–184. [Google Scholar] [CrossRef]
- Keaveny, A.P.; Freaney, R.; McKenna, M.J.; Masterson, J.; O’Donoghue, D.P. Bone remodeling indices and secondary hyperparathyroidism in celiac disease. Am. J. Gastroenterol. 1996, 91, 1226–1231. [Google Scholar]
- Valdimarsson, T.; Löfman, O.; Toss, G.; Ström, M. Reversal of osteopenia with diet in adult coeliac disease. Gut 1996, 38, 322–327. [Google Scholar] [CrossRef]
- Mustalahti, K.; Collin, P.; Sievänen, H.; Salmi, J.; Mäki, M. Osteopenia in patients with clinically silent coeliac disease warrants screening. Lancet 1999, 354, 744–745. [Google Scholar] [CrossRef]
- Duerksen, D.R.; Leslie, W.D. Positive celiac disease serology and reduced bone mineral density in adult women. Can. J. Gastroenterol. 2010, 24, 103–107. [Google Scholar]
- Stenson, W.F.; Newberry, R.; Lorenz, R.; Baldus, C.; Civitelli, R. Increased prevalence of celiac disease and need for routine screening among patients with osteoporosis. Arch. Intern. Med. 2005, 165, 393–399. [Google Scholar] [CrossRef]
- Armagan, O.; Uz, T.; Tascioglu, F.; Colak, O.; Oner, C.; Akgun, Y. Serological screening for celiac disease in premenopausal women with idiopathic osteoporosis. Clin. Rheumatol. 2005, 24, 239–243. [Google Scholar] [CrossRef]
- Drummond, F.J.; Annis, P.; O’Sullivan, K.; Wynne, F.; Daly, M.; Shanahan, F.; Quane, K.A.; Molloy, M.G. Screening for asymptomatic celiac disease among patients referred for bone densitometry measurement. Bone 2003, 33, 970–974. [Google Scholar] [CrossRef]
- Mather, K.J.; Meddings, J.B.; Beck, P.L.; Scott, R.B.; Hanley, D.A. Prevalence of IgA-antiendomysial antibody in asymptomatic low bone mineral density. Am. J. Gastroenterol. 2001, 96, 120–125. [Google Scholar]
- Karakan, T.; Ozyemisci-Taskiran, O.; Gunendi, Z.; Atalay, F.; Tuncer, C. Prevalence of IgA-antiendomysial antibody in a patient cohort with idiopathic low bone mineral density. World J. Gastroenterol. 2007, 21, 2978–2982. [Google Scholar]
- Molteni, N.; Bardella, M.T.; Vezzoli, G.; Pozzoli, E.; Bianchi, P. Intestinal calcium absorption as shown by stable strontium test in celiac disease before and after gluten-free diet. Am. J. Gastroenterol. 1995, 90, 2025–2028. [Google Scholar]
- Pazianas, M.; Butcher, G.P.; Subhani, J.M.; Finch, P.J.; Ang, L.; Collins, C.; Heaney, R.P.; Zaidi, M.; Maxwell, J.D. Calcium absorption and bone mineral density in celiacs after long term treatment with gluten-free diet and adequate calcium intake. Osteoporos. Int. 2005, 16, 56–63. [Google Scholar] [CrossRef]
- Kinsey, L.; Burden, S.T.; Bannermann, E. A dietary survey to determine if patients with celiac disease are meeting current healthy eating guidelines and how their diet compares to that of the british general population. Eur. J. Clin. Nutr. 2008, 62, 1333–1342. [Google Scholar] [CrossRef]
- Plotkin, G.R.; Isselbacher, K.J. Secondary disaccharidase deficiency in adult celiac disease (nontropical sprue) and other malabsorption states. N. Engl. J. Med. 1964, 271, 1033–1037. [Google Scholar] [CrossRef]
- Valdimarsson, T.; Toss, G.; Löfman, O.; Ström, M. Three years’ follow-up of bone density in adult coeliac disease: significance of secondary hyperparathyroidism. Scand. J. Gastroenterol. 2000, 35, 274–280. [Google Scholar] [CrossRef]
- Corazza, G.R.; di Stefano, M.; Maurino, E.; Bai, J.C. Bones in celiac disease: diagnosis and treatment. Best Pract. Res. Clin. Gastroenterol. 2005, 19, 453–465. [Google Scholar] [CrossRef]
- Staun, M.; Jarnum, S. Measurement of the 10,000-molecular weight calcium-binding protein in small-intestinal biopsy specimens from patients with malabsorption syndromes. Scand. J. Gastroenterol. 1988, 23, 827–832. [Google Scholar] [CrossRef]
- Colston, K.W.; Mackay, A.G.; Finlayson, C.; Wu, J.C.; Maxwell, J.D. Localisation of vitamin D receptor in normal human duodenum and in patients with coeliac disease. Gut 1994, 35, 1219–1225. [Google Scholar] [CrossRef]
- Valdimarsson, T.; Arnqvist, H.J.; Toss, G.; Järnerot, G.; Nyström, F.; Ström, M. Low circulating insulin-like growth factor i in coeliac disease and its relation to bone mineral density. Scand. J. Gastroenterol. 1999, 34, 904–908. [Google Scholar] [CrossRef]
- Jameson, S. Coeliac disease, insulin-like growth factor, bone mineral density, and zinc. Scand. J. Gastroenterol. 2000, 35, 894–896. [Google Scholar] [CrossRef]
- Kontakou, M.; Przemioslo, R.T.; Sturgess, R.P.; Limb, A.G.; Ciclitira, P.J. Expression of tumour necrosis factor-alpha, interleukin-6, and interleukin-2 mRNA in the jejunum of patients with coeliac disease. Scand. J. Gastroenterol. 1995, 30, 456–463. [Google Scholar] [CrossRef]
- Fornari, M.C.; Pedreira, S.; Niveloni, S.; González, D.; Diez, R.A.; Vázquez, H.; Mazure, R.; Sugai, E.; Smecuol, E.; Boerr, L.; et al. Pre- and post-treatment serum levels of cytokines IL-1beta, IL-6, and IL-1 receptor antagonist in celiac disease. Are they related to the associated osteopenia? Am. J. Gastroenterol. 1998, 93, 413–418. [Google Scholar]
- Wei, S.; Kitaura, H.; Zhou, P.; Ross, F.P.; Teitelbaum, S.L. IL-1 mediates TNF-induced osteoclastogenesis. J. Clin. Investig. 2005, 115, 282–290. [Google Scholar]
- Raisz, L.G. Local and systemic factors in the pathogenesis of osteoporosis. N. Engl. J. Med. 1988, 318, 818–828. [Google Scholar] [CrossRef]
- Di Stefano, M.; Sciarra, G.; Jorizzo, R.A.; Grillo, R.L.; Cecchetti, L.; Speziale, D.; Pichini, L.; Gasbarrini, G.; Corazza, G.R. Local and gonadal factors in the pathogenesis of coeliac bone loss. Ital. J. Gastroenterol. 1997, 29, 31. [Google Scholar]
- Taranta, A.; Fortunati, D.; Longo, M.; Rucci, N.; Iacomino, E.; Aliberti, F.; Facciuto, E.; Migliaccio, S.; Bardella, M.T.; Dubini, A. Imbalance of Osteoclastogenesis-regulating Factors in Patients with Celiac Disease. J. Bone Miner. Res. 2004, 19, 1112–1121. [Google Scholar] [CrossRef]
- Horwood, N.J.; Elliott, J.; Martin, T.J.; Gillespie, M.T. IL-12 alone and in synergy with IL-18 inhibits osteoclast formation in vitro. J. Immunol. 2001, 166, 4915–4921. [Google Scholar]
- Yamada, N.; Niwa, S.; Tsujimura, T.; Iwasaki, T.; Sugihara, A.; Futani, H.; Hayashi, S.; Okamura, H.; Akedo, H.; Terada, N. Interleukin-18 and interleukin-12 synergistically inhibit osteoclastic bone-resorbing activity. Bone 2002, 6, 901–908. [Google Scholar]
- Khosla, S. Minireview: The OPG/RANKL/RANK system. Endocrinology 2001, 142, 5050–5055. [Google Scholar] [CrossRef]
- Fiore, C.E.; Pennisi, P.; Ferro, G.; Ximenes, B.; Privitelli, L.; Mangiafico, R.A.; Santoro, F.; Parisi, N.; Lombardo, T. Altered osteoprotegerin/RANKL ratio and low bone mineral density in celiac patients on long-term treatment with gluten-free diet. Horm. Metab. Res. 2006, 38, 417–422. [Google Scholar] [CrossRef]
- Riches, P.L.; McRorie, E.; Fraser, W.D.; Determann, C.; van’t Hof, R.; Ralston, S.H. Osteoporosis associated with neutralizing autoantibodies against osteoprotegerin. N. Engl. J. Med. 2009, 361, 1459–1465. [Google Scholar] [CrossRef]
- Larussa, T.; Suraci, E.; Nazionale, I.; Leone, I.; Montalcini, T.; Abenavoli, L.; Imeneo, M.; Pujia, A.; Luzza, F. No evidence of circulating autoantibodies against osteoprotegerin in patients with celiac disease. World J. Gastroenterol. 2012, 18, 1622–1627. [Google Scholar] [CrossRef]
- Ozgör, B.; Selimoğlu, M.A. Coeliac disease and reproductive disorders. Scand. J. Gastroenterol. 2010, 45, 395–402. [Google Scholar] [CrossRef]
- Green, J.R.; Goble, H.L.; Edwards, C.R.; Dawson, A.M. Reversible insensitivity to androgens in men with untreated gluten enteropathy. Lancet 1977, 1, 280–282. [Google Scholar]
- Farthing, M.J.; Rees, L.H.; Dawson, A.M. Male gonadal function in coeliac disease: III. Pituitary regulation. Clin. Endocrinol. 1983, 19, 661–671. [Google Scholar] [CrossRef]
- Stazi, A.V.; Trecca, A.; Trinti, B. Osteoporosis in celiac disease and in endocrine and reproductive disorders. World J. Gastroenterol. 2008, 14, 498–505. [Google Scholar] [CrossRef]
- Di Sabatino, A.; Corazza, G.R. Coeliac disease. Lancet 2009, 373, 1480–1493. [Google Scholar] [CrossRef]
- Mosekilde, L.; Eriksen, E.F.; Charles, P. Effects of thyroid hormones on bone and mineral metabolism. Endocrinol. Metab. Clin. 1990, 19, 35–63. [Google Scholar]
- Hofbauer, L.C.; Brueck, C.C.; Singh, S.K.; Dobnig, H. Osteoporosis in patients with diabetes mellitus. J. Bone Miner. Res. 2007, 22, 1317–1328. [Google Scholar] [CrossRef]
- Mora, S.; Barera, G.; Beccio, S.; Menni, L.; Proverbio, M.C.; Bianchi, C.; Chiumello, G. A prospective, longitudinal study of the long-term effect of treatment on bone density in children with celiac disease. J. Pediatr. 2001, 139, 516–521. [Google Scholar] [CrossRef]
- Mora, S.; Barera, G.; Ricotti, A.; Weber, G.; Bianchi, C.; Chiumello, G. Reversal of low bone density with a gluten-free diet in children and adolescents with celiac disease. Am. J. Clin. Nutr. 1998, 67, 477–481. [Google Scholar]
- Bodé, S.; Hassager, C.; Gudmand-Høyer, E.; Christiansen, C. Body composition and calcium metabolism in adult treated coeliac disease. Gut 1991, 32, 1342–1345. [Google Scholar] [CrossRef]
- Valdimarsson, T.; Toss, G.; Ross, I.; Löfman, O.; Ström, M. Bone mineral density in coeliac disease. Scand. J. Gastroenterol. 1994, 29, 457–461. [Google Scholar] [CrossRef]
- Walters, J.R.; Banks, L.M.; Butcher, G.P.; Fowler, C.R. Detection of low bone mineral density by dual energy x-ray absorptiometry in unsuspected suboptimally treated coeliac disease. Gut 1995, 37, 220–224. [Google Scholar] [CrossRef]
- McFarlane, X.A.; Bhalla, A.K.; Reeves, D.E.; Morgan, L.M.; Robertson, D.A. Osteoporosis in treated adult coeliac disease. Gut 1995, 36, 710–714. [Google Scholar] [CrossRef]
- Ferretti, J.; Mazure, R.; Tanoue, P.; Marino, A.; Cointry, G.; Vazquez, H.; Niveloni, S.; Pedreira, S.; Mauriño, E.; Zanchetta, J.; et al. Analysis of the structure and strength of bones in celiac disease patients. Am. J. Gastroenterol. 2003, 98, 382–390. [Google Scholar]
- González, D.; Mazure, R.; Mautalen, C.; Vazquez, H.; Bai, J. Body composition and bone mineral density in untreated and treated patients with celiac disease. Bone 1995, 16, 231–234. [Google Scholar] [CrossRef]
- Kemppainen, T.; Kröger, H.; Janatuinen, E.; Arnala, I.; Lamberg-Allardt, C.; Kärkkäinen, M.; Kosma, V.M.; Julkunen, R.; Jurvelin, J.; Alhava, E.; et al. Bone recovery after a gluten-free diet: A 5-year follow-up study. Bone 1999, 25, 355–360. [Google Scholar] [CrossRef]
- Corazza, G.R.; di Stefano, M.; Jorizzo, R.A.; Cecchetti, L.; Minguzzi, L.; Gasbarrini, G. Propeptide of Type I procollagen is predictive of posttreatment bone mass gain in adult celiac disease. Gastroenterology 1997, 113, 67–71. [Google Scholar] [CrossRef]
- Bai, J.C.; Gonzalez, D.; Mautalen, C.; Mazure, R.; Pedreira, S.; Vazquez, H.; Smecuol, E.; Siccardi, A.; Cataldi, M.; Niveloni, S.; et al. Long-term effect of gluten restriction on bone mineral density of patients with coeliac disease. Aliment. Pharm. Ther. 1997, 11, 157–164. [Google Scholar]
- Pistorius, L.R.; Sweidan, W.H.; Purdie, D.W.; Steel, S.A.; Howey, S.; Bennett, J.R.; Sutton, D.R. Coeliac disease and bone mineral density in adult female patients. Gut 1995, 37, 639–642. [Google Scholar] [CrossRef]
- Kemppainen, T.; Kröger, H.; Janatuinen, E.; Arnala, I.; Kosma, V.M.; Pikkarainen, P.; Julkunen, R.; Jurvelin, J.; Alhava, E.; Uusitupa, M. Osteoporosis in adult patients with celiac disease. Bone 1999, 24, 249–255. [Google Scholar] [CrossRef]
- Selby, P.L.; Davies, M.; Adams, J.E.; Mawer, E.B. Bone loss in celiac disease is related to secondary hyperparathyroidism. J.Bone Miner. Res. 1999, 14, 652–657. [Google Scholar] [CrossRef]
- Lemieux, B.; Boivin, M.; Brossard, J.H.; Lepage, R.; Picard, D.; Rousseau, L.; D’amour, P. Normal parathyroid function with decreased bone mineral density in treated celiac disease. Can.J.Gastroenterol. 2001, 15, 302–307. [Google Scholar]
- Christensen, M.S.; Nielsen, H.E.; Torring, S. Hypercalcemia and parathyroid function after renal transplantation. Acta Med. Scand. 1977, 201, 35–39. [Google Scholar] [CrossRef]
- Liberman, U.A.; Weiss, S.R.; Bröll, J.; Minne, H.W.; Quan, H.; Bell, N.H.; Rodriguez-Portales, J.; Downs, R.W., Jr.; Dequeker, J.; Favus, M. Effect of oral alendronate on bone mineral density and the incidence of fractures in postmenopausal osteoporosis. N. Engl. J. Med. 1995, 333, 1437–1443. [Google Scholar] [CrossRef]
- Di Stefano, M.; Mengoli, C.; Tomarchio, O.; Bergonzi, M.; Pagani, E.; de Amici, M.; Ilardo, D.; Vattiato, C.; Biagi, F.; Zanaboni, A.; et al. Increased Osteoprotegerin and Decreased COOH-Terminal Propeptide of Type I Collagen Select Celiac Disease Patients on Long Term Gluten Free Diet with Persisting Low Bone Mass. In Proceedings of UEGW (21st United European Gastroenterology Week), Belin, Germany, 12–16 October 2013.
- Sánchez, M.I.; Mohaidle, A.; Baistrocchi, A.; Matoso, D.; Vázquez, H.; González, A.; Mazure, R.; Maffei, E.; Ferrari, G.; Smecuol, E.; et al. Risk of fracture in celiac disease: Gender, dietary compliance, or both? World J. Gastroenterol. 2011, 17, 3035–3042. [Google Scholar] [CrossRef]
- Bianchi, M.L.; Bardella, M.T. Bone in celiac disease. Osteoporos. Int. 2008, 19, 1705–1716. [Google Scholar] [CrossRef]
- West, J.; Logan, R.F.; Card, T.R.; Smith, C.; Hubbard, R. Fracture risk in people with celiac disease: A population-based cohort study. Gastroenterology 2003, 125, 429–436. [Google Scholar] [CrossRef]
- Jafri, M.R.; Nordstrom, C.W.; Murray, J.A.; van Dyke, C.T.; Dierkhising, R.A.; Zinsmeister, A.R.; Melton, L.J., III. Long-term fracture risk in patients with celiac disease: A population-based study in Olmsted County, Minnesota. Dig. Dis. Sci. 2008, 53, 964–971. [Google Scholar] [CrossRef]
- Ludvigsson, J.F.; Michaelsson, K.; Ekbom, A.; Montgomery, S.M. Coeliac disease and the risk of fractures—A general population-based cohort study. Aliment. Pharm. Ther. 2007, 25, 273–285. [Google Scholar]
- Vasquez, H.; Mazure, R.; Gonzalez, D.; Flores, D.; Pedreira, S.; Niveloni, S.; Smecuol, E.; Mauriño, E.; Bai, J.C. Risk of fractures in celiac disease patients: A cross-sectional, case-control study. Am. J. Gastroenterol. 2000, 95, 183–189. [Google Scholar] [CrossRef]
- Fickling, W.E.; McFarlane, X.A.; Bhalla, A.K.; Robertson, D.A. The clinical impact of metabolic bone disease in coeliac disease. Postgrad. Med. J. 2001, 77, 33–36. [Google Scholar] [CrossRef]
- Davie, M.W.; Gaywood, I.; George, E.; Jones, P.W.; Masud, T.; Price, T.; Summers, G.D. Excess non-spine fractures in women over 50 years with celiac disease: A cross-sectional, questionnaire-based study. Osteoporos. Int. 2005, 16, 1150–1155. [Google Scholar] [CrossRef]
- Moreno, M.L.; Vazquez, H.; Mazure, R.; Smecuol, E.; Niveloni, S.; Pedreira, S.; Sugai, E.; Mauriño, E.; Gomez, J.C.; Bai, J.C. Stratification of bone fracture risk in patients with celiac disease. Clin. Gastroenterol. Hepatol. 2004, 2, 127–134. [Google Scholar] [CrossRef]
- Olmos, M.; Antelo, M.; Vazquez, H.; Smecuol, E.; Mauriño, E.; Bai, J.C. Systematic review and meta-analysis of observational studies on the prevalence of fractures in coeliac disease. Dig. Liver Dis. 2008, 40, 46–53. [Google Scholar] [CrossRef]
- Vestergaard, P.; Mosekilde, L. Fracture risk in patients with celiac disease, crohn’s disease, and ulcerative colitis: A nationwide follow-up study of 16,416 patients in denmark. Am. J. Epidemiol. 2002, 156, 1–10. [Google Scholar] [CrossRef]
- Glüer, C.C. Quantitative ultrasound techniques for the assessment of osteoporosis: Expert agreement on current status. The international quantitative ultrasound consensus group. J. Bone Miner. Res. 1997, 12, 1280–1288. [Google Scholar] [CrossRef]
- Njeh, C.F.; Boivin, C.M.; Langton, C.M. The role of ultrasound in the assessment of oeteoporosis: A review. Osteoporos.Int. 1997, 7, 7–22. [Google Scholar] [CrossRef]
- Scott, E.M.; Gaywood, I.; Scott, B.B. Guidelines for osteoporosis in coeliac disease and inflammatory bowel disease. Gut 2000, 46, I1–I8. [Google Scholar] [CrossRef]
- Thomason, K.; West, J.; Logan, R.F.; Coupland, C.; Holmes, G.K. Fracture experience of patients with coeliac disease: A population based survey. Gut 2003, 52, 518–522. [Google Scholar] [CrossRef]
- Compston, J. Is fracture risk increased in patients with coeliac disease? Gut 2003, 52, 459–460. [Google Scholar] [CrossRef]
- Lewis, N.R.; Scott, B.B. Should patients with coeliac disease have their bone mineral density measured? Eur. J. Gastroenterol. Hepatol. 2005, 17, 1065–1070. [Google Scholar] [CrossRef]
- American Gastroenterological Association. American gastroenterological association medical position statement: Guidelines on osteoporosis in gastrointestinal diseases. Gastroenterology 2003, 124, 791–794. [CrossRef]
- Fouda, M.A.; Khan, A.A.; Sultan, M.S.; Rios, L.P.; McAssey, K.; Armstrong, D. Evaluation and management of skeletal health in celiac disease: Position statement. Can. J. Gastroenterol. 2012, 26, 819–829. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Di Stefano, M.; Mengoli, C.; Bergonzi, M.; Corazza, G.R. Bone Mass and Mineral Metabolism Alterations in Adult Celiac Disease: Pathophysiology and Clinical Approach. Nutrients 2013, 5, 4786-4799. https://doi.org/10.3390/nu5114786
Di Stefano M, Mengoli C, Bergonzi M, Corazza GR. Bone Mass and Mineral Metabolism Alterations in Adult Celiac Disease: Pathophysiology and Clinical Approach. Nutrients. 2013; 5(11):4786-4799. https://doi.org/10.3390/nu5114786
Chicago/Turabian StyleDi Stefano, Michele, Caterina Mengoli, Manuela Bergonzi, and Gino Roberto Corazza. 2013. "Bone Mass and Mineral Metabolism Alterations in Adult Celiac Disease: Pathophysiology and Clinical Approach" Nutrients 5, no. 11: 4786-4799. https://doi.org/10.3390/nu5114786
APA StyleDi Stefano, M., Mengoli, C., Bergonzi, M., & Corazza, G. R. (2013). Bone Mass and Mineral Metabolism Alterations in Adult Celiac Disease: Pathophysiology and Clinical Approach. Nutrients, 5(11), 4786-4799. https://doi.org/10.3390/nu5114786




