Citrus medica “Otroj”: Attenuates Oxidative Stress and Cardiac Dysrhythmia in Isoproterenol-Induced Cardiomyopathy in Rats
Abstract
:1. Introduction
2. Experimental Section
2.1. Animals
2.2. Plant Material and Preparation of Extract
2.3. Acute Toxicity Test
2.4. Induction of Myocardial Disturbance
2.5. Preparation of Cardiac Tissue Homogenate
2.6. Estimation of Marker Enzymes
2.7. Estimation of Lipid Profile
2.8. Lipid Peroxidation (LPO) Determination
2.9. Estimation of Non-Protein Sulfhydryl Groups (NP-SH)
2.10. Determination of Total Protein (TP)
2.11. Histopathological Studies
2.12. Studies of the in Vitro Antioxidant Activity
2.12.1. Scavenging Activity of DPPH Radical
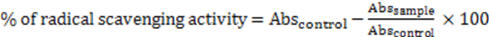
2.12.2. β-Carotene-Linoleic Acid Assay
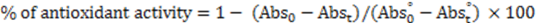
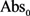 and
and 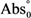 are the absorbance values measured at zero time of incubation for sample extract and control, respectively.
are the absorbance values measured at zero time of incubation for sample extract and control, respectively. and
and  are the absorbance values for sample extract and control, respectively, at t = 120 min.
are the absorbance values for sample extract and control, respectively, at t = 120 min.2.13. Total Phenolic Content
2.14. Total Flavonoid Content
2.15. Statistical Analysis
3. Results
3.1. Acute Toxicity Test
3.2. Effect of Ethanolic Extract of Otroj (EEOT) on Marker Enzymes
3.3. Effect of EEOT on Lipid Profile
| Treatment Group (n = 6) | AST (U/L) | ALT (U/L) | LDH (U/L) | CK (U/L) |
|---|---|---|---|---|
| Normal control | 72.15 ± 2.34 | 29.28 ± 2.17 | 84.11 ± 2.62 | 139.50 ± 4.26 |
| ISO (85 mg/kg) | 166.66 ± 10.61 ***, a | 92.13 ± 4.88 ***, a | 131.77 ± 4.01 ***, a | 198.50 ± 6.14 ***, a |
| EEOT (250 mg/kg) + ISO | 161.16 ± 5.85 b | 84.25 ± 5.39 b | 118.45 ± 2.86 *, b | 171.33 ± 3.58 **, b |
| EEOT (500 mg/kg) + ISO | 137.00 ± 3.44 * | 69.11 ± 3.08 **, b | 107.22 ± 3.38 ***, b | 151.50 ± 3.38 ***, b |
| Treatment Group (n = 6) | Cholesterol (mg/dL) | Triglycerides (mg/dL) | HDL-C (mg/dL) | LDL-C (mg/dL) | VLDL-C (mg/dL) |
|---|---|---|---|---|---|
| Normal control | 108.84 ± 5.83 | 58.66 ± 4.66 | 51.14 ± 2.88 | 45.96 ± 5.08 | 11.73 ± 0.93 |
| ISO (85 mg/kg) | 225.17 ± 9.74 *** | 121.51 ± 5.46 ***,a | 29.68 ± 2.17 ***,a | 171.18 ± 9.14 ***,a | 24.30 ± 1.09 ***,a |
| EEOT (250 mg/kg) + ISO | 189.11 ± 6.72 * | 110.54 ± 6.15 b | 42.01 ± 3.65 *,b | 124.99 ± 9.65 **,b | 22.10 ± 1.23 b |
| EEOT (500 mg/kg) + ISO | 157.82 ± 6.38 *** | 69.19 ± 3.61 ***,b | 40.18 ± 2.41 **,b | 103.79 ± 6.21 ***,b | 13.83 ± 0.72 ***,b |
3.4. Effect of EEOT on MDA, NP-SH, and TP
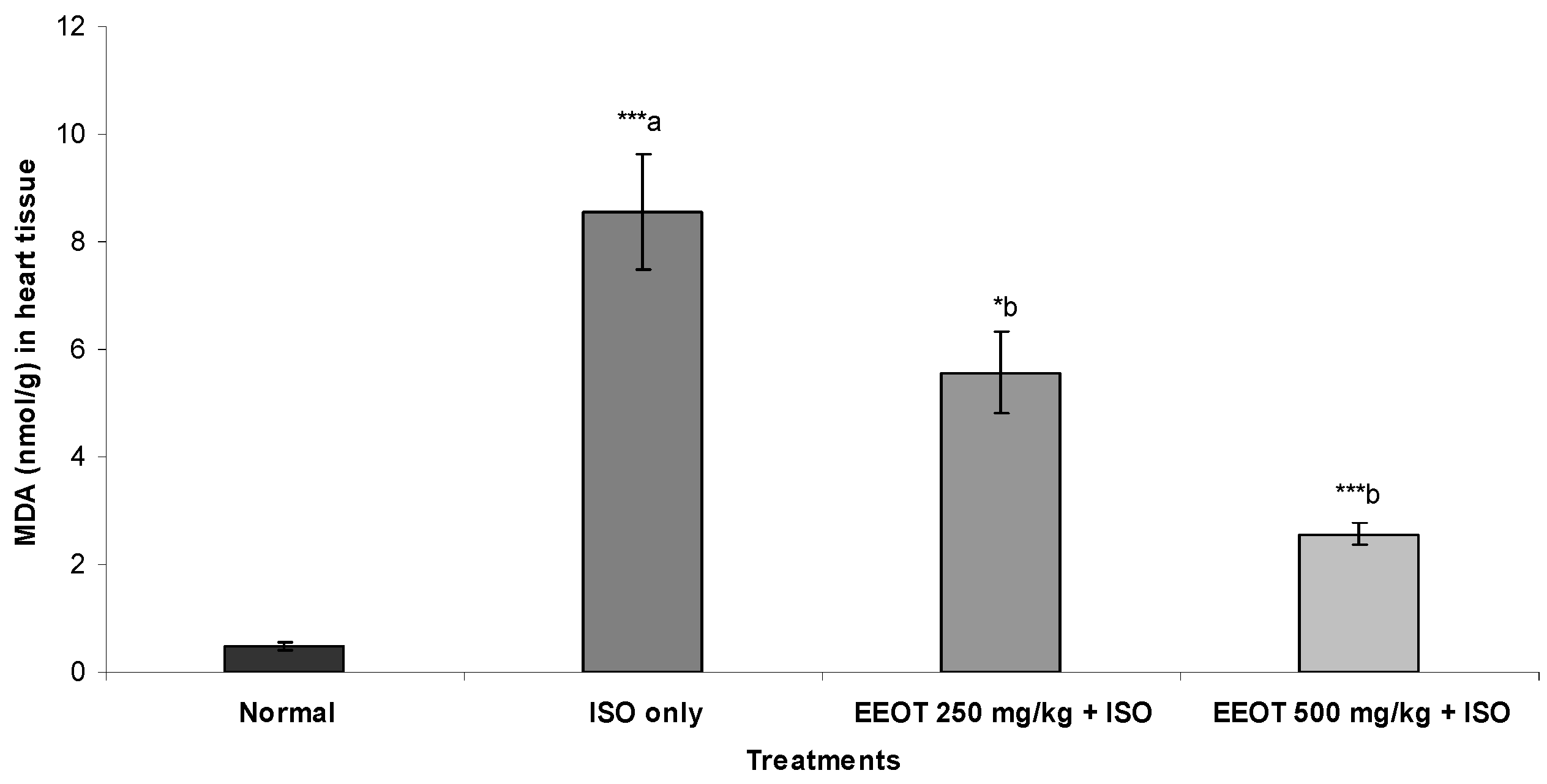


3.5. Effect of EEOT on Histopathological Evaluation
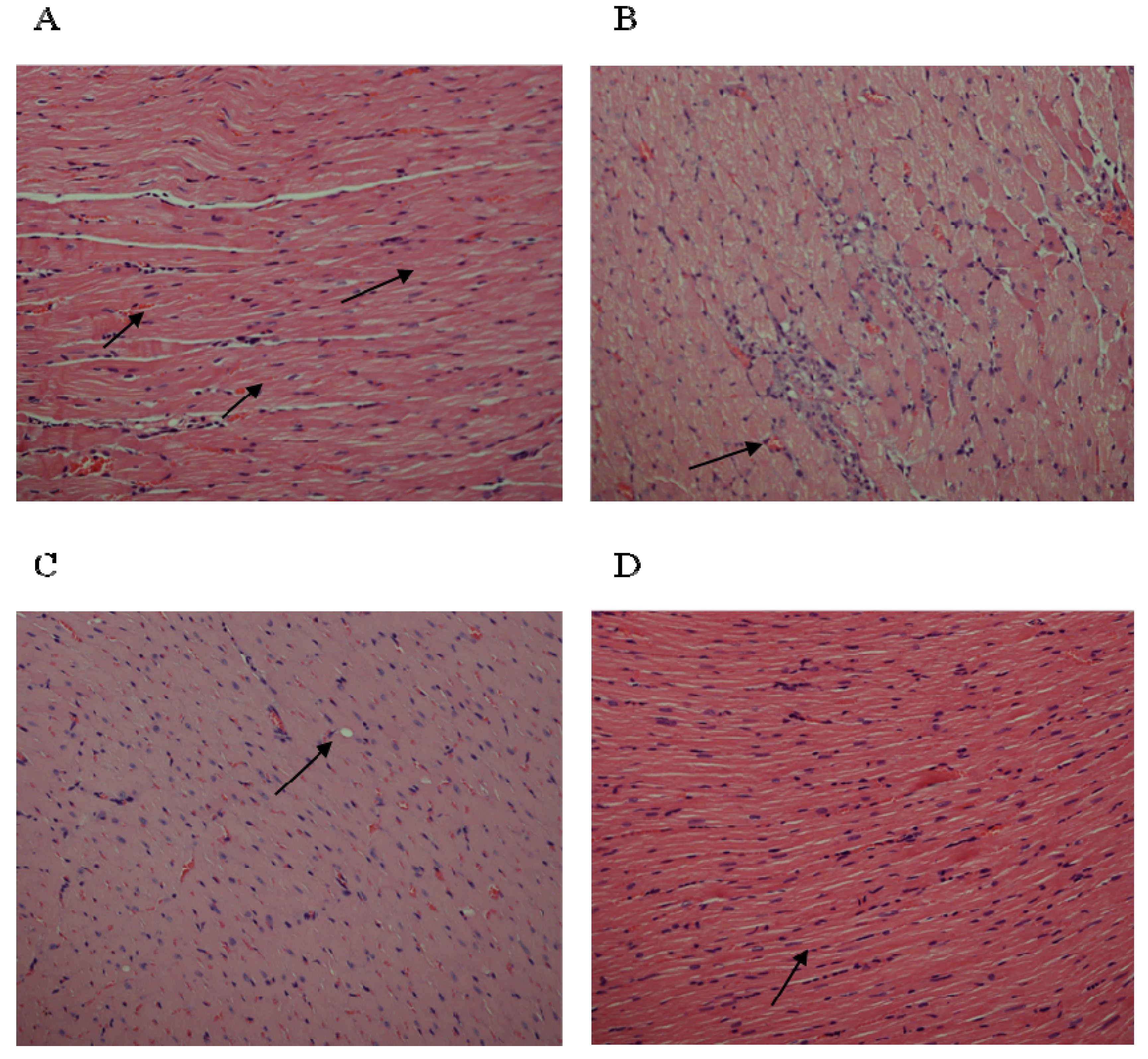
3.6. Effect of EEOT on Cardiac Rhythm
| Treatment Group (n = 6) | % Increase in Heart Rate | % Effectiveness in Suppressing Isoproterenol-Induced Tachycardia |
|---|---|---|
| Isoproterenol (85 mg/kg) s.c. | 53.35 ± 3.1 | - |
| EEOT (250 mg/kg) + ISO | 21.4 ± 2.1 | 60 ± 5.9 * |
| EEOT (500 mg/kg) + ISO | 17.8 ± 2.5 | 66.7 ± 6.1 * |
3.7. Antioxidant Activity and Phenolic and Flavonoidal Contents
| Plant Species | Radical Scavenging Activity in (%) | Total Antioxidant Activity in (%) | TPC (mg GAE/100 g) | TFC (mg QE/100 g) | ||||
|---|---|---|---|---|---|---|---|---|
| 10 | 50 | 100 | 500 | 1000 | 1000 (μg/mL) | |||
| EEOT | 13.1 | 39.0 | 72.9 | 87.0 | 93.5 | 92.8 ± 6.91 | 192.4 ± 2.52 | 74.1 ± 3.12 |
| Ascorbic acid | 19.5 | 71.2 | 85.5 | 92.7 | 94.1 | - | ||
| Rutin | 93.1 ± 7.22 | |||||||
4. Discussion
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- MacKay, J.; Mensah, G. The Atlas of Heart Disease and Stroke; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Murugesan, M.; Revathi, R.; Manju, V. Cadrioprotective effect of fenugreek on isoproterenol induced myocardial infarction in rats. Indian J. Pharmacol. 2011, 43, 516–519. [Google Scholar] [CrossRef]
- Aroow, W.S. Epidemiology, pathophysiology, prognosis, and treatment of systolic and diastolic heart failure. Cardiol. Rev. 2006, 14, 108–124. [Google Scholar] [CrossRef]
- Wexler, B.C. Myocardial infarction in young vs old male rats: Pathophysiologic changes. Am. Heart J. 1978, 96, 70–80. [Google Scholar] [CrossRef]
- Sood, S.; Bansal, S.; Muthuraman, A.; Gill, N.S.; Bali, M. Therapeutic potential of Citrus medica L. peel extract in carrageenan induced inflammatory pain in rat. Res. J. Med. Plant 2009, 3, 123–133. [Google Scholar] [CrossRef]
- Sood, S.; Muthuraman, A.; Arora, B.; Bansal, S.; Bali, M.; Sharma, P.D. Potential effect of Citrus decumana extract on stress induced peptic ulcer in rat. Lat. Am. J. Pharm. 2009, 40, 945–948. [Google Scholar]
- Hartwell, J.L. Plants Used Against Cancer, a Survey; Quarterman Publications, Inc.: Lawrence, MA, USA, 1982; p. 154. [Google Scholar]
- OECD, Guidelines 423, Acute Oral Toxicity: Acute Toxic Class Method. In OECD Guidelines for Testing Chemicals; OECD: Paris, France, 2001.
- Panda, V.S.; Naik, S.R. Cardioprotective activity of Ginkgo biloba phytosomes in isoproterenol-induced myocardial necrosis in rats: A biochemical and histoarchitectural evaluation. Exp. Toxicol. Pathol. 2008, 60, 397–404. [Google Scholar] [CrossRef]
- Bakheet, D.M.; ElTahir, K.E.H.; Al-Sayed, M.I.; El-Obeid, H.A.; Al-Rashood, K.A. Studies on the cardiovascular effect of N-ethyl- and N-benzyl-1,2-diphenyl ethanolamines in the rat: Elucidation of the mechanisms of action. Gen. Pharmacol. 1999, 33, 17–22. [Google Scholar] [CrossRef]
- Reitman, S.A.; Frankel, A. A colorimetric method for the determination of serum glutamic oxaloaceric acid and glutamic pyruvic transaminases. Am. J. Clin. Pathol. 1957, 28, 56–63. [Google Scholar]
- Bergmeyer, H.U. Method of Enzymatic Analysis; Academic Press: London, UK, 1974. [Google Scholar]
- Okinada, S.; Kumagai, H.; Ebashi, S.; Sugita, H.; Momoi, H.; Toyokura, Y. Serum creatinine phosphokinase. Activity in progressive muscular dystrophy and neuromuscular disease. Arch. Nurol. 1961, 4, 520–525. [Google Scholar] [CrossRef]
- Demacher, P.N.M.; Hijamaus, A.G.M. A study of the use of polyethylene glycol in estimating cholesterol. Clin. Chem. 1980, 26, 1775–1778. [Google Scholar]
- Foster, L.B.; Dunn, R.T. Stable reagents for the determination of serum triglycerides by a colorimetric Hantzch condensation method. Clin. Chem. 1973, 19, 338–340. [Google Scholar]
- Burstein, M.; Scholnick, H.R. Lipoprotein-polyanion-metal interaction. Adv. Lipid Res. 1973, 11, 67–108. [Google Scholar]
- Utley, H.C.; Bernheim, F.; Hochslein, P. Effect of sulfhydryl reagent on peroxidation in microsome. Arch. Biochem. Biophys. 1967, 260, 521–531. [Google Scholar]
- Sedlak, J.; Lindsay, R.H. Estimation of total, protein bound and non-protein SH groups in tissue with Ellman’s reagent. Anal. Biochem. 1968, 25, 192–205. [Google Scholar] [CrossRef]
- Culling, C.F.A. Handbook of Histopathological and Histochemical Techniques, 3rd ed.; Butterworth and Co.: London, UK, 1974; pp. 73–159. [Google Scholar]
- Brand, W.W.; Cuvelier, H.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. Food Sci. Technol. 1995, 82, 25–30. [Google Scholar]
- Mothana, R.A. Anti-inflammatory, antinociceptive and antioxidant activities of the endemic Soqotraen Boswellia elongata Balf. f. and Jatropha unicostata Balf. f. in different experimental models. Food Chem. Toxicol. 2011, 49, 2594–2599. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidant by means of Folin-Ciocalteu reagent. Method Enzymol. 1999, 299, 152–178. [Google Scholar] [CrossRef]
- Djeridane, A.; Yousfi, M.; Nadjemi, B.; Boutassouna, D.; Stocker, P.; Vidal, N. Antioxidant activity of some Algerian medicinal plants extracts containing phenolic compounds. Food Chem. 2006, 97, 654–660. [Google Scholar] [CrossRef]
- Selvaraj, P.; Pugalendi, K.V. Efficacy of hesperidin on plasma, heart and liver tissue lipids in rats subjected to isoproterenol-induced cardiotoxicity. Exp. Toxicol. Pathol. 2012, 64, 449–452. [Google Scholar] [CrossRef]
- Dhalla, N.S.; Yates, J.C.; Lee, S.L.; Singh, A. Functional and subcellular changes in the isolated rat heart perused with oxidised isoproterenol. J. Mol. Cell. Cardiol. 1978, 10, 31–41. [Google Scholar]
- Zhou, B.; Wu, L.J.; Li, L.H.; Tashiro, S.; Onodera, S.; Uchiumi, F.; Ikejima, T. Silibinin protects against isoproterenol-induced rat cardiac myocyte injury through mitochondrial pathway after up-regulation of SIRTl. J. Pharmacol. Sci. 2006, 102, 387–395. [Google Scholar] [CrossRef]
- Yeager, J.C.; Whitehurst, M.E. Verapamil prevents isoproterenol induced cardiac failure in the rat. Life Sci. 1982, 30, 299–306. [Google Scholar] [CrossRef]
- Bloom, S.; Davis, D.L. Calcium as mediator of isoproterenol-induced myocardial necrosis. Am. J. Pathol. 1972, 69, 459–470. [Google Scholar]
- Singal, P.K.; Kapur, N.; Dhillon, K.S.; Beamish, R.E.; Dhalla, N.S. Role of free radicals in catecholamine-induced cardiomyopathy. Can. J. Physiol. Pharmacol. 1982, 60, 1390–1397. [Google Scholar]
- Senthil Kumar, H.; Anandan, R.; Santhosh Kumar, M. Cardioprotective effect of Picorrhiza kurrooa against isoproterenol induced myocardial stress in rats. Fitotherapia 2001, 72, 402–405. [Google Scholar] [CrossRef]
- Gürgün, C.; Ildizlt, M.; Yavuzgil, O.; Sin, A.; Apaydin, A.; Cinar, C.; Kúltúrsay, H. The effects of short term statin treatment on left ventricular function and inflammatory markers in patients with chronic heart failure. Int. J. Cardiol. 2008, 123, 102–107. [Google Scholar] [CrossRef]
- Karthikeyan, K.; Sarala, B.R.; Devaraj, N.S. Efficacy of grape seed proanthocyanidins on serum and heart tissue lipids in rats subjected to isoproterenol-induced myocardial injury. Vasc. Pharmacol. 2007, 47, 295–301. [Google Scholar]
- Gokkusu, C.; Mostafazadeh, T. Changes of oxidative stress in various tissues by long-term administration of vitamin E in hypercholesterolemic rats. Clin. Chim. Acta 2003, 328, 155–161. [Google Scholar] [CrossRef]
- Nair, P.S.; Devi, C.S.S. Efficacy of mangiferin on serum and heart tissue lipids in rats subjected to isoproterenol induced cardiotoxicity. Toxicology 2006, 228, 135–139. [Google Scholar] [CrossRef]
- Prince, P.S.M.; Suman, S.; Devika, P.T.; Vaithianathan, M. Cardioprotective effect of ‘Marutham’ a polyherbal formulation on isoproterenol induced myocardial infarction in Wistar rats. Fitoterapia 2008, 79, 433–438. [Google Scholar] [CrossRef]
- Kota, S.K.; Jammula, S.; Kota, S.K.; Venkata, S.; Krisbna, S.; Meher, L.K.; Rao, E.S.; Modi, K.D. Nutraceuticals in dyslipidemia management. J. Med. Nutr. Nutraceut. 2013, 2, 26–40. [Google Scholar] [CrossRef]
- Prabhu, S.; Jainu, M.; Sabitha, K.E.; Devi, C.S. Cardioprotective effect of mangiferin on isoproterenol induced myocardial infarction in rats. Indian J. Exp. Biol. 2006, 44, 209–215. [Google Scholar]
- Brindha, E.; Pandian, M.R.; Indira, V. Phytic acid protects rat myocardium during isoproterenol-induced myocardial infarction: A biochemical and histological study. Int. J. Res. Pharm. Biomed. Sci. 2012, 3, 1779–1786. [Google Scholar]
- Halliwell, B.; Aeschbach, R.; Loliger, J.; Aruoma, O.I. The characterization of antioxidants. Food Chem. Toxicol. 1995, 33, 601–617. [Google Scholar] [CrossRef]
- Fahmy, S.R.; Hamdi, S.A.H. Antioxidant effect of the Egyptian freshwater Procambarus clarkii extract in rat liver and erythrocytes. Afr. J. Pharm. Pharmacol. 2011, 5, 776–785. [Google Scholar]
- Punithavathi, V.R.; Prince, P.S.M. Combined effects of quercetin and α-tocopherol on lipids and glycoprotein components in isoproterenol induced myocardial infarcted Wistar rats. Chem. Biol. Afr. J. Pharm. Pharmacol. 2009, 181, 322–327. [Google Scholar]
- Al-Said, M.S.; Mothana, R.A.; Al-Yahya, M.A.; Al-Blowi, A.S.; Al-Sohaibani, M.; Ahmed, A.F.; Rafatullah, S. Edible oils for liver protection: Hepatoprotective potentiality of Moringa oleifera seed oil against chemical-induced hepatitis in rats. J. Food Sci. 2012, 77, T124–T130. [Google Scholar] [CrossRef]
- Pushpakiran, G.; Mahalakshmi, K.; Anuradha, C.V. Protective effects of taurine on glutathione and glutathione-dependent enzymes in ethanol-fed rats. Pharmazie 2004, 59, 869–872. [Google Scholar]
- Borkowski, T.; Szymusiak, H.; Gliszczynska-Rwiglo, A.; Rietjens, I.; Tyrakowska, B. Radical scavenging capacity of wine anthocyanins is strongly pH-dependent. J. Agric. Food Chem. 2005, 53, 5526–5534. [Google Scholar]
- Fukumoto, L.R.; Mazza, G. Assessing antioxidant and prooxidant activities of phenolic compounds. J. Agric. Food Chem. 2000, 48, 3597–3604. [Google Scholar] [CrossRef]
- Ziberna, L.; Vanzo, A.; Passamonti, S.; Lunder, M.; Tramer, F.; Moze, S.; Drevensek, G. Acute cardioprotective and cardiotoxic effects of bilberry anthocyanins in ischemia-reperfusion injury: Beyond concentration-dependent antioxidant activity. Cardiovasc. Toxicol. 2010, 10, 283–294. [Google Scholar] [CrossRef]
- Scarabelli, T.M.; Mariotto, S.; Abdel-Azeim, S.; Shoji, K.; Darra, E.; Stephanou, A.; Chen-Scarabelli, C; Marechal, J.D.; Knight, R.; Ciampa, A.; et al. Targeting STAT1 by myricetin and delphinidin provides efficient protection of the heart from ischemia/reperfusion-induced injury. FEBS Lett. 2009, 583, 531–541. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Al-Yahya, M.A.; Mothana, R.A.; Al-Said, M.S.; El-Tahir, K.E.; Al-Sohaibani, M.; Rafatullah, S. Citrus medica “Otroj”: Attenuates Oxidative Stress and Cardiac Dysrhythmia in Isoproterenol-Induced Cardiomyopathy in Rats. Nutrients 2013, 5, 4269-4283. https://doi.org/10.3390/nu5114269
Al-Yahya MA, Mothana RA, Al-Said MS, El-Tahir KE, Al-Sohaibani M, Rafatullah S. Citrus medica “Otroj”: Attenuates Oxidative Stress and Cardiac Dysrhythmia in Isoproterenol-Induced Cardiomyopathy in Rats. Nutrients. 2013; 5(11):4269-4283. https://doi.org/10.3390/nu5114269
Chicago/Turabian StyleAl-Yahya, Mohammed A., Ramzi A. Mothana, Mansour S. Al-Said, Kamal Elddin El-Tahir, Mohammed Al-Sohaibani, and Syed Rafatullah. 2013. "Citrus medica “Otroj”: Attenuates Oxidative Stress and Cardiac Dysrhythmia in Isoproterenol-Induced Cardiomyopathy in Rats" Nutrients 5, no. 11: 4269-4283. https://doi.org/10.3390/nu5114269
APA StyleAl-Yahya, M. A., Mothana, R. A., Al-Said, M. S., El-Tahir, K. E., Al-Sohaibani, M., & Rafatullah, S. (2013). Citrus medica “Otroj”: Attenuates Oxidative Stress and Cardiac Dysrhythmia in Isoproterenol-Induced Cardiomyopathy in Rats. Nutrients, 5(11), 4269-4283. https://doi.org/10.3390/nu5114269




