The Importance of Dose, Frequency and Duration of Vitamin D Supplementation for Plasma 25-Hydroxyvitamin D
Abstract
:1. Introduction
2. Experimental Section
3. Results
| Not taking vitamin D supplements (N = 759) | Taking any vitamin D supplements (N = 3465) | P | ||||||
|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | |||||
| 25-hydroxyvitamin D (nmol/L) | 64.92 | 28.93 | 103.31 | 47.58 | <0.001 | |||
| Age (years) | 35.78 | 11.03 | 42.48 | 11 | <0.001 | |||
| Male (%) | 68.25% | 69.99% | 0.35 | |||||
| Latitude (degrees) | 53.8 | 2.91 | 53.36 | 2.49 | <0.001 | |||
| Body weight categories (%) | <0.01 | |||||||
| Underweight | 1.19% | 0.52% | ||||||
| Normal weight | 28.59% | 24.01% | ||||||
| Overweight | 36.50% | 37.09% | ||||||
| Obesity | 33.73% | 38.38% | ||||||
| General Health (first visits) (%) | <0.01 | |||||||
| Excellent | 4.87% | 6.58% | ||||||
| Very good | 23.98% | 0.2771 | ||||||
| Good | 41.90% | 42.60% | ||||||
| Fair | 17.13% | 14.31% | ||||||
| Needs improvement | 12.12% | 8.80% | ||||||
| April to October (%) | 49.28% | 48.89% | 0.85 | |||||
| Health behaviors | ||||||||
| Physical activity level (%) | <0.01 | |||||||
| Low | 37.88% | 33.58% | ||||||
| Moderate | 43.29% | 51.87% | ||||||
| High | 18.82% | 14.56% | ||||||
| Smoking (%) | 16.16% | 15.48% | 0.64 | |||||
| Food servings | ||||||||
| Milk (per day) (%) | 0.49 | |||||||
| 0 | 6.18% | 6.82% | ||||||
| 1 to 2 | 64.61% | 66.69% | ||||||
| 3 or more | 29.22% | 26.49% | ||||||
| Fish (per week) (%) | <0.001 | |||||||
| 0 | 28.25% | 19.17% | ||||||
| 1 to 2 | 50.28% | 59.11% | ||||||
| 3 or more | 21.47% | 21.72% | ||||||
| Margarine consumption frequency (%) | 0.11 | |||||||
| Never | 24.04% | 27.99% | ||||||
| Rarely | 34.13% | 32.16% | ||||||
| Often | 34.38% | 30.28% | ||||||
| Always | 7.45% | 9.57% | ||||||
| Not taking vitamin D supplementation | Taking any supplements | P | ||||||
|---|---|---|---|---|---|---|---|---|
| No. of obs. | Mean | SD | No. of obs. | Mean | SD | |||
| 759 | 64.76 | 28.86 | 3465 | 103.31 | 47.58 | <0.001 * | ||
| Dose (IU) | ||||||||
| 1000 to 2000 | 1433 | 82.99 | 34.77 | <0.001 ** | ||||
| 2000 to 3000 | 524 | 98.89 | 36.53 | |||||
| 3000 to 4000 | 432 | 110.56 | 39.93 | |||||
| 5000 or more | 1076 | 129.63 | 55.88 | |||||
| Frequency, times/week | ||||||||
| 1–2/week | 738 | 75.15 | 31.86 | <0.001 ** | ||||
| 3–4/week | 762 | 89.28 | 37.94 | |||||
| 5–6/week | 935 | 108.27 | 41.76 | |||||
| 7/week | 1030 | 129.37 | 53.2 | |||||
| Duration (months) | ||||||||
| 1 | 629 | 83.35 | 41.08 | <0.001 ** | ||||
| 2 | 258 | 85.72 | 36.99 | |||||
| 3 | 281 | 90.84 | 40.63 | |||||
| 4 | 225 | 96.8 | 42.23 | |||||
| 5 or more | 2072 | 113.96 | 48.89 | |||||
| Models | Univariable analysis | Multivariable analysis | ||||
|---|---|---|---|---|---|---|
| β | (95% CI) | β | (95% CI) | |||
| Vitamin D supplementation | ||||||
| Taking minimum supplementation (compared to not taking any supplements) | 36.84 *** | (33.39 to | 40.29) | 3.08 | (−1.10 to | 7.26) |
| Dose (IU) | ||||||
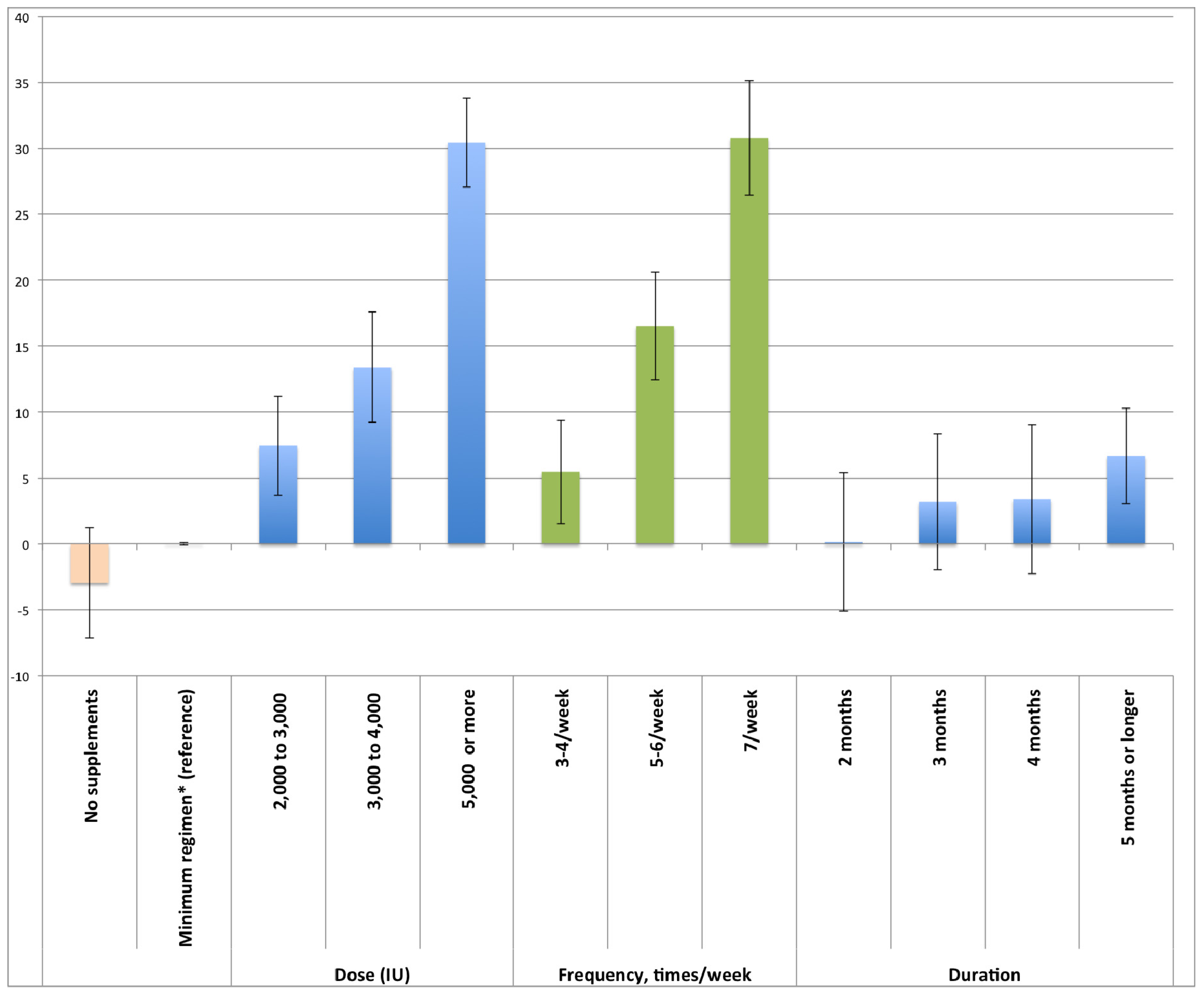
| 1000 to 2000 | (minimum regimen as reference) | (minimum regimen as reference) | ||||
| 2000 to 3000 | 14.27 *** | (10.47 to | 18.08) | 7.49 *** | (3.73 to | 11.24) |
| 3000 to 4000 | 26.56 *** | (22.44 to | 30.68) | 13.19 *** | (9.02 to | 17.36) |
| 5000 or more | 47.33 *** | (44.25 to | 50.41) | 30.22 *** | (26.86 to | 33.59) |
| Dose frequency | ||||||
| 1–2/week | (minimum regimen as reference) | (minimum regimen as reference) | ||||
| 3–4/week | 14.09 *** | (10.07 to | 18.10) | 5.44 ** | (1.52 to | 9.35) |
| 5–6/week | 33.05 *** | (29.13 to | 36.97) | 16.52 *** | (12.41 to | 20.64) |
| 7/week | 53.02 *** | (49.09 to | 56.95) | 30.69 *** | (26.35 to | 35.03) |
| Duration (months) | ||||||
| 1 | (minimum regimen as reference) | (minimum regimen as reference) | ||||
| 2 | 2.87 | (−3.11 to | 8.85) | 0.18 | (−5.07 to | 5.44) |
| 3 | 6.61 * | (0.82 to | 12.39) | 3.22 | (−1.94 to | 8.38) |
| 4 | 11.10 *** | (4.86 to | 17.35) | 3.31 | (−2.34 to | 8.97) |
| 5 or more | 28.56 *** | (24.82 to | 32.30) | 6.68 *** | (3.05 to | 10.31) |
| Age (per 10 years) | 7.65 *** | (6.35 to | 8.96) | 1.46 * | (0.28 to | 2.63) |
| Male (female as reference) | −6.79 *** | (−10.19 to | −3.38) | −1.23 | (−4.10 to | 1.64) |
| Body weight status | ||||||
| Underweight | −10.27 | (−28.06 to | 7.52) | −3.45 | (−17.87 to | 10.96) |
| Normal | (reference) | (reference) | ||||
| Overweight | −6.56 ** | (−10.36 to | −2.76) | −7.13 *** | (−10.33 to | −3.93) |
| Obesity | −14.85 *** | (−18.71 to | −11.00) | −14.59 *** | (−17.97 to | −11.21) |
| General health (first visit) | ||||||
| Excellent | (reference) | (reference) | ||||
| Very good | −11.01 ** | (−17.89 to | −4.14) | −4.29 | (−9.94 to | 1.36) |
| Good | −19.08 *** | (−25.70 to | −12.46) | −7.98 ** | (−13.52 to | −2.44) |
| Fair | −27.34 *** | (−34.73 to | −19.96) | −12.92 *** | (−19.15 to | −6.69) |
| Needs improvement | −30.20 *** | (−38.11 to | −22.29) | −11.24 ** | (−17.97 to | −4.51) |
| Latitude (per 10°) | −17.50 *** | (−23.37 to | −11.63) | −5.39 * | (−10.17 to | −0.61) |
| Summer months (April to October) | 1.18 | (−1.50 to | 3.86) | 2.91 ** | (0.74 to | 5.09) |
| Physical activity levels | ||||||
| Low | (reference) | (reference) | ||||
| Moderate | 6.05 ** | (2.58 to | 9.52) | 1.96 * | (−1.01 to | 4.92) |
| High | 9.99 *** | (5.06 to | 14.93) | 4.36 | (0.04 to | 8.68) |
| Smoking | ||||||
| Non-smoker | (reference) | (reference) | ||||
| Smoker | −0.91 | (−4.79 to | 2.97) | −5.52 ** | (−8.88 to | −2.17) |
| Milk servings/day | ||||||
| 0 | (reference) | (reference) | ||||
| 1 to 2 | −5.14 | (−11.69 to | 1.41) | −3.92 | (−9.46 to | 1.62) |
| 3 or more | −2.56 | (−9.58 to | 4.46) | −1.76 | (−7.72 to | 4.19) |
| Fish servings/week | ||||||
| 0 | (reference) | (reference) | ||||
| 1 to 2 | 0.33 * | (9.45 to | 2.10) | −0.51 | (−4.31 to | 3.30) |
| 3 or more | 2.52 ** | (13.51 to | 2.86) | 2.59 | (−2.00 to | 7.18) |
| Margarine consumption frequency | ||||||
| Never | (reference) | (reference) | ||||
| Rarely | −6.12 ** | (−10.33 to | −1.91) | −2.22 | (−5.78 to | 1.34) |
| Often | −12.89 *** | (−17.19 to | −8.59) | −4.75 * | (−8.43 to | −1.08) |
| Always | −7.60 * | (−13.74 to | −1.47) | −2.74 | (−7.96 to | 2.47) |
| Constant | (not shown) | 114.46 *** | (87.25 to | 141.68) | ||
| No. of visits | 4224 | 4224 | ||||
| No. of individuals | 2714 | 2714 | ||||
4. Discussion
5. Conclusions
Acknowledgements
Conflicts of Interest
References
- Ceglia, L.; Simpson, R.U. Vitamin D and Skeletal Muscle Function. In Vitamin D; Feldman, D., Pike, J.W., Adams, J.S., Eds.; Elsevier Science: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Institute of Medicine Committee to Review Dietary Reference Intakes for Vitamin D and Calcium Institute of Medicine. In Dietary Reference Intakes for Calcium and Vitamin D; The National Academies Press: Washington, DC, USA, 2011.
- Chung, M.; Balk, E.; Brendel, M.; Ip, S.; Lau, J.; Lee, J.; Lichtenstein, A.; Patel, K.; Raman, G.; Tatsioni, A.; et al. Vitamin D and Calcium: Systematic Review of Health Outcomes; Evidence Report/Technology Assessment No. 183; (Prepared by Tufts Evidence-Based Practice Center under Contract No. 290-2007-10055-i); Agency for Healthcare Research and Quality: Rockville, MD, USA, 2009. [Google Scholar]
- Holick, M.F. Vitamin D: Physiology, Molecular Biology, and Clinical Applications; Humana Press: New York, NY, USA, 2010. [Google Scholar]
- Heaney, R.P. Serum 25-hydroxyvitamin d is a reliable indicator of vitamin D status. Am. J. Clin. Nutr. 2011, 94, 619–620. [Google Scholar] [CrossRef]
- Lappe, J.M.; Davies, K.M.; Travers-Gustafson, D.; Heaney, R.P. Vitamin D status in a rural postmenopausal female population. J. Am. Coll. Nutr. 2006, 25, 395–402. [Google Scholar] [CrossRef]
- Viljakainen, H.T.; Palssa, A.; Karkkainen, M.; Jakobsen, J.; Lamberg-Allardt, C. How much vitamin D3 do the elderly need? J. Am. Coll. Nutr. 2006, 25, 429–435. [Google Scholar] [CrossRef]
- Heaney, R.P. Vitamin d in health and disease. Clin. J. Am. Soc. Nephrol. 2008, 3, 1535–1541. [Google Scholar] [CrossRef]
- Heaney, R.P. The vitamin D requirement in health and disease. J. Steroid Biochem. Mol. Biol. 2005, 97, 13–19. [Google Scholar] [CrossRef]
- Vieth, R. Vitamin D supplementation, 25-hydroxyvitamin d concentrations, and safety. Am. J. Clin. Nutr. 1999, 69, 842–856. [Google Scholar]
- Vieth, R. The Pharmacology of Vitamin D, Including Fortification Strategies. In Vitamin D; Feldman, D., Glorieux, F., Pike, J., Eds.; Elsevier: New York, NY, USA, 2005; pp. 995–1015. [Google Scholar]
- Holick, M.F. Vitamin D. In Modern Nutrition in Health and Disease; Shils, M.E., Shike, M.E.S.M., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2006; Volume 2, pp. 376–393. [Google Scholar]
- Hathcock, J.N.; Shao, A.; Vieth, R.; Heaney, R. Risk assessment for vitamin D. Am. J. Clin. Nutr. 2007, 85, 6–18. [Google Scholar]
- Plum, L.A.; DeLuca, H.F. Vitamin d, disease and therapeutic opportunities. Nat. Rev. Drug Discov. 2010, 9, 941–955. [Google Scholar] [CrossRef]
- Saliba, W.; Barnett, O.; Stein, N.; Kershenbaum, A.; Rennert, G. The longitudinal variability of serum 25(OH)D levels. Eur. J. Intern. Med. 2012, 23, e106–e111. [Google Scholar] [CrossRef]
- Heaney, R.P.; French, C.B.; Nguyen, S.; Baggerly, L.L.; Brunel, L.; Veugelers, P.J. A novel approach localizes the association of vitamin D status with insulin resistance to one region of the 25-hydroxyvitamin d continuum. Adv. Nutr. 2013, 4, 303–310. [Google Scholar] [CrossRef]
- Calgary Laboratry Services. Vitamin D, 25 Hydroxy. Available online: http://www.calgarylabservices.com/lab-services-guide/lab-tests/AlphabeticalListing/V/Vitamin-D-25-Hydroxy.htm (accessed on 9 December 2012).
- Craig, C.L.; Marshall, A.L.; Sjostrom, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-Country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef]
- Mello, M.M.; Stearns, S.C.; Norton, E.C.; Ricketts, T.C., III. Understanding biased selection in Medicare HMOs. Health Serv. Res. 2003, 38, 961–992. [Google Scholar]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Jackson, R.D.; LaCroix, A.Z.; Gass, M.; Wallace, R.B.; Robbins, J.; Lewis, C.E.; Bassford, T.; Beresford, S.A.A.; Black, H.R.; Blanchette, P.; et al. Calcium plus vitamin D supplementation and the risk of fractures. N. Engl. J. Med. 2006, 354, 669–683. [Google Scholar] [CrossRef]
- Wactawski-Wende, J.; Kotchen, J.M.; Anderson, G.L.; Assaf, A.R.; Brunner, R.L.; O’Sullivan, M.J.; Margolis, K.L.; Ockene, J.K.; Phillips, L.; Pottern, L.; et al. Calcium plus vitamin D supplementation and the risk of colorectal cancer. N. Engl. J. Med. 2006, 354, 684–696. [Google Scholar] [CrossRef]
- Stroud, M.L.; Stilgoe, S.; Stott, V.E.; Alhabian, O.; Salman, K. Vitamin D—A review. Aust. Fam. Phys. 2008, 37, 1002–1005. [Google Scholar]
- Holick, M.F. Vitamin D deficiency: What a pain it is. Mayo Clin. Proc. 2003, 78, 1457–1459. [Google Scholar] [CrossRef]
- Lips, P. Vitamin D deficiency and secondary hyperparathyroidism in the elderly: Consequences for bone loss and fractures and therapeutic implications. Endocr. Rev. 2001, 22, 477–501. [Google Scholar] [CrossRef]
- Drincic, A.T.; Armas, L.A.; van Diest, E.E.; Heaney, R.P. Volumetric dilution, rather than sequestration best explains the low vitamin D status of obesity. Obesity (Silver Spring) 2012, 20, 1444–1448. [Google Scholar] [CrossRef]
- Heaney, R.P.; Holick, M.F. Why the iom recommendations for vitamin D are deficient. J. Bone Miner. Res. 2011, 26, 455–457. [Google Scholar] [CrossRef]
- Pittas, A.G.; Chung, M.; Trikalinos, T.; Mitri, J.; Brendel, M.; Patel, K.; Lichtenstein, A.H.; Lau, J.; Balk, E.M. Systematic review: Vitamin D and cardiometabolic outcomes. Ann. Intern. Med. 2010, 152, 307–314. [Google Scholar]
- Scragg, R. Vitamin D and Cardiovascular Disease: A Review of the Epidemiological and Clinical Evidence. In Vitamins in the Prevention of Human Diseases; Herrmann, W., Obeid, R., Eds.; Walter de Gruyter GmbH & Company KG: Berlin, Germany, 2011. [Google Scholar]
- Heaney, R.P. Vitamin D—Baseline status and effective dose. N. Engl. J. Med. 2012, 367, 77–78. [Google Scholar] [CrossRef]
- Gallagher, J.C.; Sai, A.; Templin, T., II; Smith, L. Dose response to vitamin D supplementation in postmenopausal women: A randomized trial. Ann. Intern. Med. 2012, 156, 425–437. [Google Scholar] [CrossRef]
- Lips, P.; Wiersinga, A.; van Ginkel, F.C.; Jongen, M.J.; Netelenbos, J.C.; Hackeng, W.H.; Delmas, P.D.; van der Vijgh, W.J. The effect of vitamin D supplementation on vitamin d status and parathyroid function in elderly subjects. J. Clin. Endocrinol. Metab. 1988, 67, 644–650. [Google Scholar] [CrossRef]
- Ginde, A.A.; Sullivan, A.F.; Mansbach, J.M.; Camargo, C.A., Jr. Vitamin D insufficiency in pregnant and nonpregnant women of childbearing age in the united states. Am. J. Obstet. Gynecol. 2010, 202, 431–438. [Google Scholar]
- Ginde, A.A.; Liu, M.C.; Camargo, C.A., Jr. Demographic differences and trends of vitamin D insufficiency in the US population, 1988–2004. Arch. Intern. Med. 2009, 169, 626–632. [Google Scholar] [CrossRef]
- Langlois, K.; Greene-Finestone, L.; Little, J.; Hidiroglou, N.; Whiting, S. Vitamin D status of canadians as measured in the 2007 to 2009 Canadian Health Measures Survey. Health Reports 2010, 21, 47–55. [Google Scholar]
Supplementary Files
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chao, Y.-S.; Brunel, L.; Faris, P.; Veugelers, P.J. The Importance of Dose, Frequency and Duration of Vitamin D Supplementation for Plasma 25-Hydroxyvitamin D. Nutrients 2013, 5, 4067-4078. https://doi.org/10.3390/nu5104067
Chao Y-S, Brunel L, Faris P, Veugelers PJ. The Importance of Dose, Frequency and Duration of Vitamin D Supplementation for Plasma 25-Hydroxyvitamin D. Nutrients. 2013; 5(10):4067-4078. https://doi.org/10.3390/nu5104067
Chicago/Turabian StyleChao, Yi-Sheng, Ludovic Brunel, Peter Faris, and Paul J. Veugelers. 2013. "The Importance of Dose, Frequency and Duration of Vitamin D Supplementation for Plasma 25-Hydroxyvitamin D" Nutrients 5, no. 10: 4067-4078. https://doi.org/10.3390/nu5104067
APA StyleChao, Y.-S., Brunel, L., Faris, P., & Veugelers, P. J. (2013). The Importance of Dose, Frequency and Duration of Vitamin D Supplementation for Plasma 25-Hydroxyvitamin D. Nutrients, 5(10), 4067-4078. https://doi.org/10.3390/nu5104067




