Investigation of Effect of Nutritional Drink on Chemotherapy-Induced Mucosal Injury and Tumor Growth in an Established Animal Model
Abstract
:1. Introduction
2. Methods and Materials
2.1. Diets
| Clinutren® Protect (Diet A) | Placebo (Diet B) | Impact® (Diet C) | Standard Chow | |
|---|---|---|---|---|
| Energy distribution | ||||
| 25% | 25% | 22% | 23% | |
| Lipid | 35% | 25% | 25% | 12% |
| Carbohydrate | 40% | 50% | 53% | 65% |
| g/100 g powder | g/100 g powder | g/100 g powder | g/100 g powder | |
| 29.6 | 29.6 | 22.0 | 20 | |
| Casein | 12.5 | 21.75 | - | * |
| Whey | 9.25 | - | 16.9 | * |
| TGF-β | 0.009 | - | 0.0014 | * |
| Glutamine | 9.45 | - | - | * |
| Arginine | - | - | 5.10 | * |
| Alanine | - | 9.80 | - | * |
| Nitrogen | 5.25 | 4.95 | 4.38 1 | * |
| Carbohydrate | 47 | 47 | 54 | 59.4 |
| Lipid | 18.2 | 18.2 | 11 | 10.5 |
| Canola oil | 5.7 | 5.7 | - | * |
| Corn oil | 2 | 2 | 2.0 | * |
| Lecithin | 0.3 | 0.3 | - | * |
| Medium chain | 9.1 | 9.1 | 3.0 | 1.31 |
| Triglyceride | - | - | - | * |
| Fish oil | 0.5 | 0.5 | 4.6 | * |
| Minerals | 2.00 | 1.80 | 2.18 | 2.9 |
| Sodium, mg | 220 | 210 | 434 | 180 |
| Potassium, mg | 560 | 520 | 543 | 820 |
| Chloride, mg | 245 | 190 | 486 | * |
| Calcium, mg | 300 | 200 | 324 | 800 |
| Phosphorus, mg | 200 | 210 | 292 | 700 |
| Magnesium, mg | 50 | 35 | 93 | 200 |
| Manganese, μg | 500 | 500 | 81 | 104 |
| Selenium, μg | 16.1 | 16.1 | 19 | 0.4 |
| Kcal/100 g | 575 | 485 | 525 | 334 |
2.2. Study Design
2.2.1. Study 1: Safety and Tolerability of Test Diets
2.2.2. Study 2: Effects of Diet upon Tumor Growth and MTX-Induced Injury
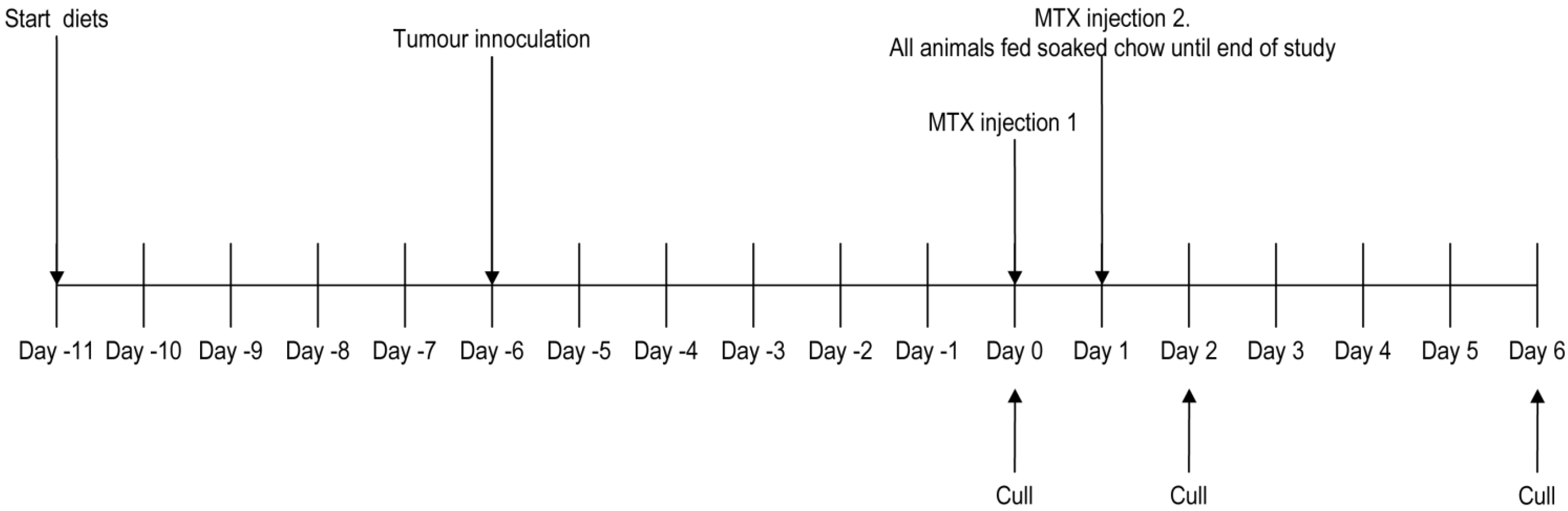
2.3. Tumor Inoculation
2.4. Chemotherapy
2.5. Necropsy
2.6. Histochemical, Biochemical and Morphometric Assessment
2.7. Statistical Analyses
3. Results and Discussion
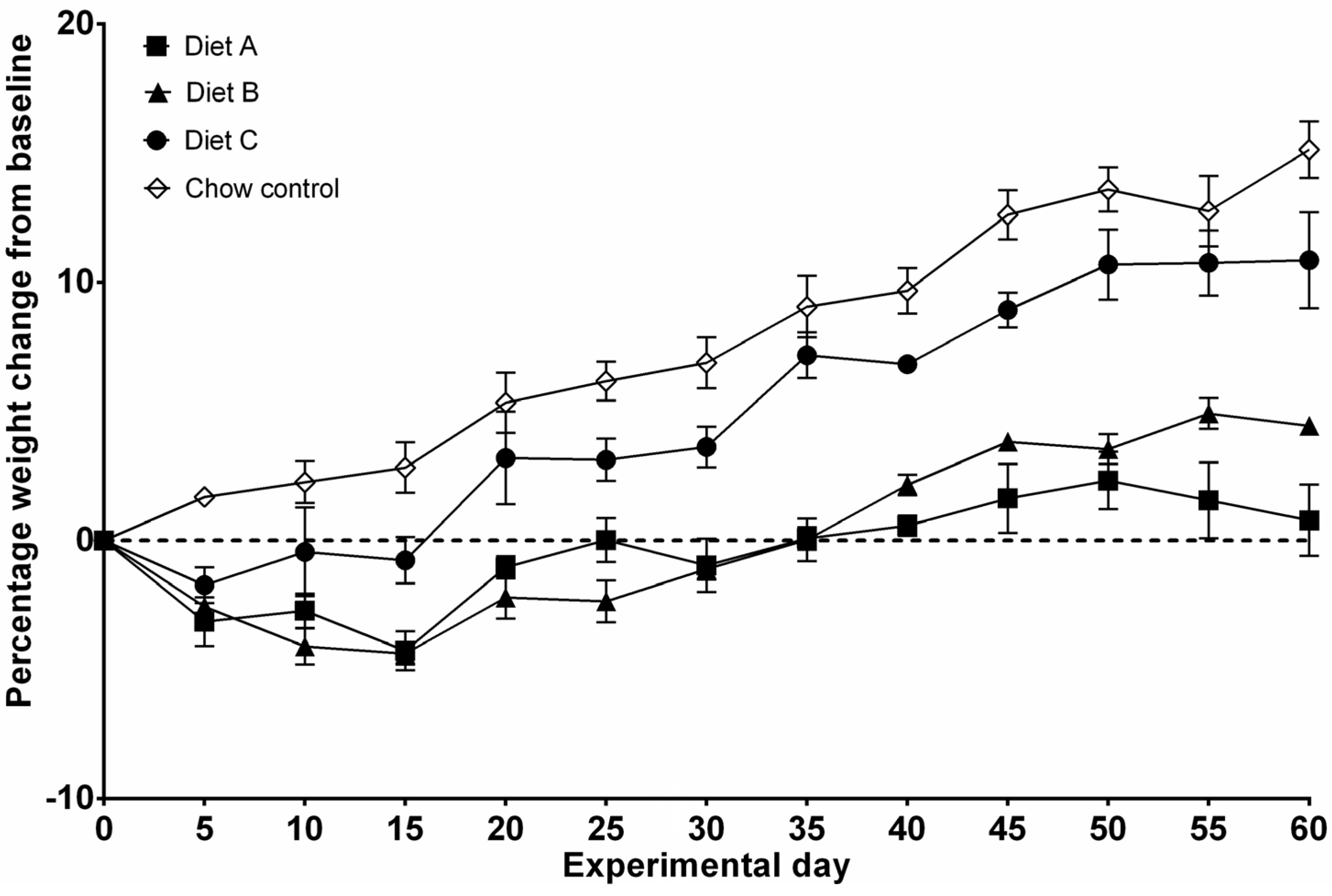
3.1. Study 1—Food Intake and Bodyweights
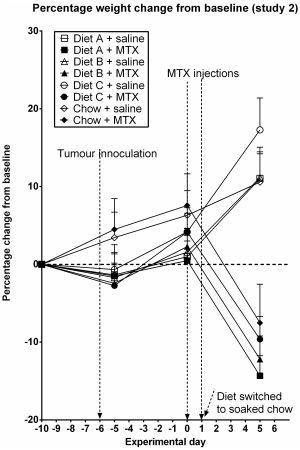
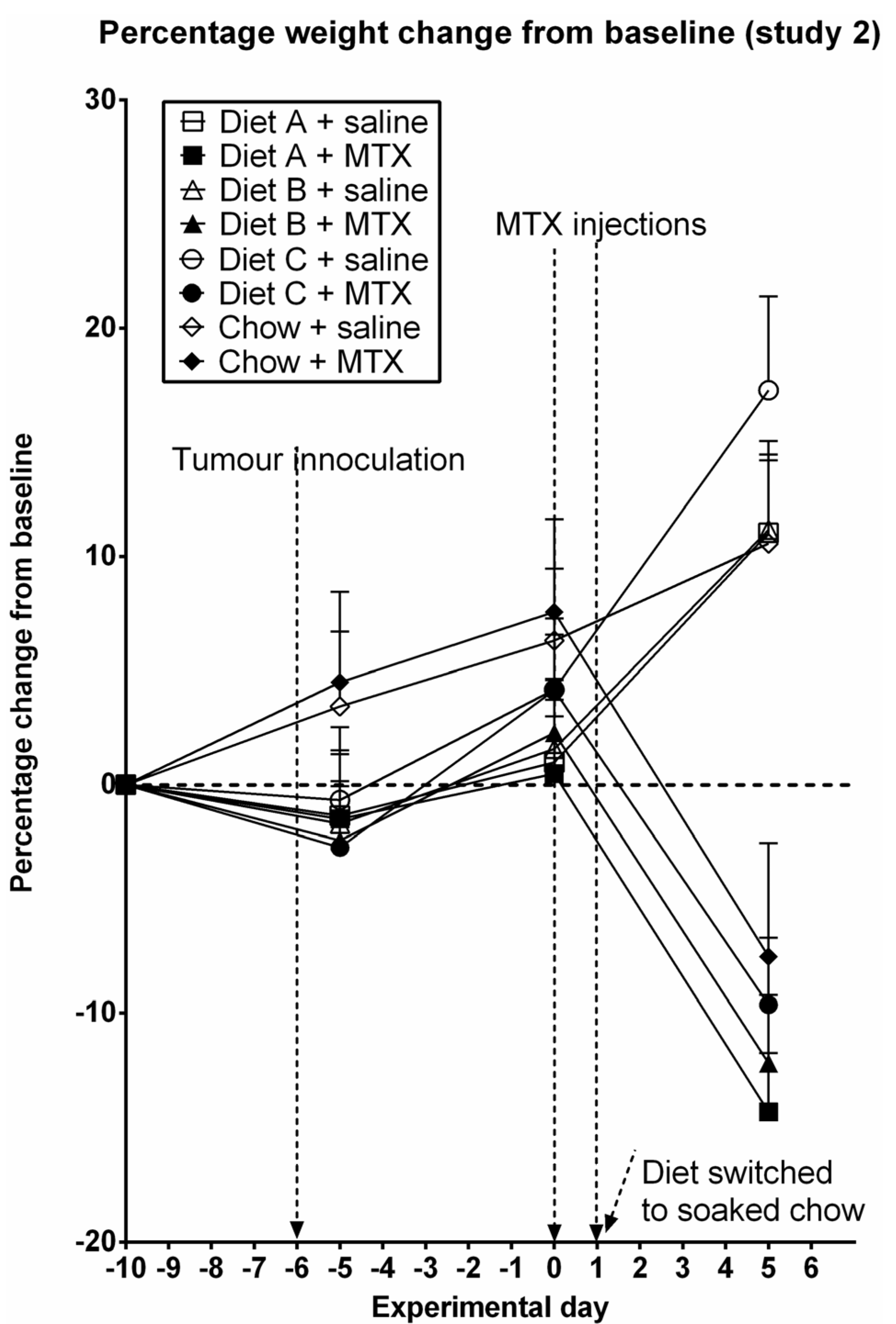
3.2. Study 2
3.3. Diarrhea
3.4. Tumor Growth

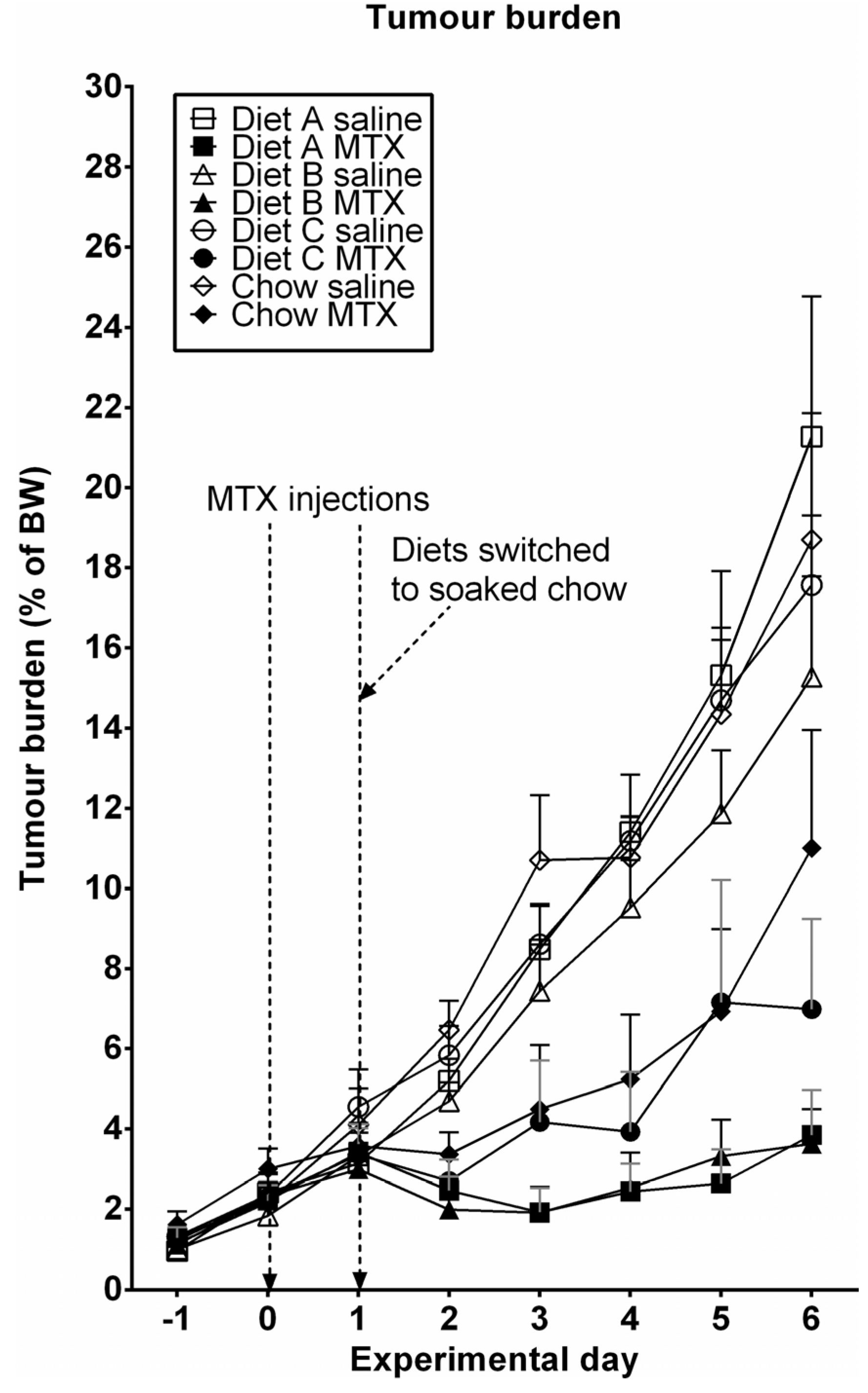
3.5. Necropsy Data
3.6. Complete Blood Examination and Blood Biochemistry
3.7. H & E and Tissue Morphometry

3.8. Markers of Cell Proliferation (Ki67) and Apoptosis (Caspase-3)
3.9. Alcian Blue/Periodic Acid-Schiff Stain for Goblet Cells
3.10. Discussion
4. Conclusions
Abbreviations
| CBE | complete blood examination |
| CP | Clinutren Protect |
| DAMA | Dark Agouti Mammary Adenocarcinoma |
| GI | gastrointestinal |
| GSH | glutathione |
| H & E | haematoxylin and eosin |
| IMVS | Institute of Medical and Veterinary Sciences |
| MBA-20 | medical biochemical analysis-20 |
| MTX | methotrexate |
| NF-κB | nuclear factor κB |
| SCFA | short chain fatty acid |
| TGF | transforming growth factor |
| TNF-α | tumor necrosis factor α |
Acknowledgments
Conflicts of Interest
References
- Al-Dasooqi, N.; Sonis, S.T.; Bowen, J.M.; Bateman, E.; Blijlevens, N.; Gibson, R.J.; Logan, R.M.; Nair, R.G.; Stringer, A.M.; Yazbeck, R.; et al. Emerging evidence on the pathobiology of mucositis. Support. Care Cancer 2013, 21, 2075–2083. [Google Scholar] [CrossRef]
- Machon, C.; Thezenas, S.; Dupuy, A.-M.; Assenat, E.; Michel, F.; Mas, E.; Senesse, P.; Cristol, J.-P. Immunonutrition before and during radiochemotherapy: Improvement of inflammatory parameters in head and neck cancer patients. Support. Care Cancer 2012, 20, 3129–3135. [Google Scholar] [CrossRef]
- Ben-Lulu, S.; Pollak, Y.; Mogilner, J.; Bejar, J.; Coran, A.G.; Sukhotnik, I. Dietary transforming growth factor-β 2 (TGF-β2) supplementation reduces methotrexate-induced intestinal mucosal injury in a rat. PLoS One 2012, 7, e45221. [Google Scholar]
- Boukhettala, N.; Ibrahim, A.; Aziz, M.; Vuichoud, J.; Saudan, K.Y.; Blum, S.; Déchelotte, P.; Breuillé, D.; Coëffier, M. A diet containing whey protein, free glutamine, and transforming growth factor-β ameliorates nutritional outcome and intestinal mucositis during repeated chemotherapeutic challenges in rats. J. Nutr. 2010, 140, 799–805. [Google Scholar] [CrossRef]
- Boukhettala, N.; Ibrahim, A.; Claeyssens, S.; Faure, M.; le Pessot, F.; Vuichoud, J.; Lavoinne, A.; Breuillé, D.; Déchelotte, P.; Coëffier, M. A diet containing whey protein, glutamine, and TGF-β modulates gut protein metabolism during chemotherapy-induced mucositis in rats. Dig. Dis. Sci. 2010, 55, 2172–2181. [Google Scholar] [CrossRef]
- Harsha, W.T.; Kalandarova, E.; McNutt, P.; Irwin, R.; Noel, J. Nutritional supplementation with transforming growth factor-β, glutamine, and short chain fatty acids minimizes methotrexate-induced injury. J. Pediatr. Gastroenterol. Nutr. 2006, 42, 53–58. [Google Scholar] [CrossRef]
- Van’t Land, B.; Meijer, H.P.; Frerichs, J.; Koetsier, M.; Jager, D.; Smeets, R.L.; M’Rabet, L.; Hoijer, M. Transforming Growth Factor-β2 protects the small intestine during methotrexate treatment in rats possibly by reducing stem cell cycling. Br. J. Cancer 2002, 87, 113–118. [Google Scholar] [CrossRef]
- Evans, M.A; Shronts, E.P. Intestinal fuels: Glutamine, short-chain fatty acids, and dietary fiber. J. Am. Diet. Assoc. 1992, 92, 1239–1246. [Google Scholar]
- Gaurav, K.; Goel, R.K.; Shukla, M.; Pandey, M. Glutamine: A novel approach to chemotherapy-induced toxicity. Indian J. Med. Paediatr. Oncol. 2012, 33, 13–20. [Google Scholar] [CrossRef]
- Logan, R.M.; Stringer, A.M.; Bowen, J.M.; Yeoh, A.S.; Gibson, R.J.; Sonis, S.T.; Keefe, D.M. The role of pro-inflammatory cytokines in cancer treatment-induced alimentary tract mucositis: Pathobiology, animal models and cytotoxic drugs. Cancer Treat. Rev. 2007, 33, 448–460. [Google Scholar] [CrossRef]
- Klimberg, V.S.; Pappas, A.A.; Nwokedi, E.; Jensen, J.C.; Broadwater, J.R.; Lang, N.P.; Westbrook, K.C. Effect of supplemental dietary glutamine on methotrexate concentrations in tumors. Arch. Surg. 1992, 127, 1317–1320. [Google Scholar] [CrossRef]
- McPherson, R.A.; Hardy, G. Clinical and nutritional benefits of cysteine-enriched protein supplements. Curr. Opin. Clin. Nutr. Metab. Care 2011, 14, 562–568. [Google Scholar] [CrossRef]
- Oz, H.S.; Chen, T.S.; Neuman, M. Nutrition intervention: A strategy against systemic inflammatory syndrome. J. Parenter. Enteral. Nutr. 2009, 33, 380–389. [Google Scholar] [CrossRef]
- Kent, K.D.; Harper, W.J.; Bomser, J.A. Effect of whey protein isolate on intracellular glutathione and oxidant-induced cell death in human prostate epithelial cells. Toxicol. In Vitro 2003, 17, 27–33. [Google Scholar] [CrossRef]
- Blijlevens, N.M.; Donnelly, J.P.; Naber, A.H.; Schattenberg, A.V.; DePauw, B.E. A randomised, double-blinded, placebo-controlled, pilot study of parenteral glutamine for allogeneic stem cell transplant patients. Support. Care Cancer 2005, 13, 790–796. [Google Scholar] [CrossRef]
- Sornsuvit, C.; Komindr, S.; Chuncharunee, S.; Wanikiat, P.; Archararit, N.; Santanirand, P. Pilot Study: Effects of parenteral glutamine dipeptide supplementation on neutrophil functions and prevention of chemotherapy-induced side-effects in acute myeloid leukaemia patients. J. Int. Med. Res. 2008, 36, 1383–1391. [Google Scholar] [CrossRef]
- Sukhotnik, I.; Mogilner, J.G.; Karry, R.; Shamian, B.; Lurie, M.; Kokhanovsky, N.; Ure, B.M.; Coran, A.G. Effect of oral glutamine on enterocyte turnover during methotrexate-induced mucositis in rats. Digestion 2009, 79, 5–13. [Google Scholar] [CrossRef]
- Rubio, I.T.; Cao, Y.; Hutchins, L.F.; Westbrook, K.C.; Klimberg, V.S. Effect of glutamine on methotrexate efficacy and toxicity. Ann. Surg. 1998, 227, 772–780. [Google Scholar] [CrossRef]
- Gibson, R.J.; Bowen, J.M.; Alvarez, E.; Finnie, J.; Keefe, D.M. Establishment of a single-dose irinotecan model of gastrointestinal mucositis. Chemotherapy 2007, 53, 360–369. [Google Scholar]
- Stringer, A.M.; Gibson, R.J.; Logan, R.M.; Bowen, J.M.; Yeoh, A.S.J.; Keefe, D.M.K. Chemotherapy-induced changes in mucin secretion and composition. J. Gastroenterol. Hepatol. 2006, 21, A266. [Google Scholar]
- Bowen, J.M.; Gibson, R.J.; Keefe, D.M.; Cummins, A.G. Cytotoxic chemotherapy upregulates pro-apoptotic Bax and Bak in the small intestine of rats and humans. Pathology 2005, 37, 56–62. [Google Scholar] [CrossRef]
- Keefe, D.M.; Brealey, J.; Goland, G.J.; Cummins, A.G. Chemotherapy for cancer causes apoptosis that precedes hypoplasia in crypts of the small intestine in humans. Gut 2000, 47, 632–637. [Google Scholar]
- Gibson, R.J.; Bowen, J.M.; Inglis, M.R.; Cummins, A.G.; Keefe, D.M. Irinotecan causes severe small intestinal damage, as well as colonic damage, in the rat with implanted breast cancer. J. Gastroenterol. Hepatol. 2003, 18, 1095–1100. [Google Scholar] [CrossRef]
- Takeda, N.; Hasegawa, S.; Morita, M.; Matsunaga, T. Pica in rats is analogous to emesis: An animal model in emesis research. Pharmacol. Biochem. Behav. 1993, 45, 817–821. [Google Scholar] [CrossRef]
- Barcelo, A.; Claustre, J.; Moro, F.; Chayvialle, J.A.; Cuber, J.C.; Plaisancie, P. Mucin secretion is modulated by luminal factors in the isolated vascularly perfused rat colon. Gut 2000, 46, 218–224. [Google Scholar] [CrossRef]
- Coyle, P.; Rofe, A.M.; Bourgeois, C.S.; Conyers, R.A. Biochemical manifestations of a rat mammary adenocarcinoma-producing cachexia: In vivo and in vitro studies. Immunol. Cell Biol. 1990, 68, 147–153. [Google Scholar] [CrossRef]
- De Jonghe, B.C.; Horn, C.C. Chemotherapy-induced pica and anorexia are reduced by common hepatic branch vagotomy in the rat. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 294, R756–R765. [Google Scholar]
- Monro, J.A. Adequate intake values for dietary fibre based on faecal bulking indexes of 66 foods. Eur. J. Clin. Nutr. 2004, 58, 32–39. [Google Scholar] [CrossRef]
- Tanabe, H.; Ito, H.; Sugiyama, K.; Kiriyama, S.; Morita, T. Dietary indigestible components exert different regional effects on luminal mucin secretion through their bulk-forming property and fermentability. Biosci. Biotechnol. Biochem. 2006, 70, 1188–1194. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bateman, E.; Bowen, J.; Stringer, A.; Mayo, B.; Plews, E.; Wignall, A.; Greenberg, N.; Schiffrin, E.; Keefe, D. Investigation of Effect of Nutritional Drink on Chemotherapy-Induced Mucosal Injury and Tumor Growth in an Established Animal Model. Nutrients 2013, 5, 3948-3963. https://doi.org/10.3390/nu5103948
Bateman E, Bowen J, Stringer A, Mayo B, Plews E, Wignall A, Greenberg N, Schiffrin E, Keefe D. Investigation of Effect of Nutritional Drink on Chemotherapy-Induced Mucosal Injury and Tumor Growth in an Established Animal Model. Nutrients. 2013; 5(10):3948-3963. https://doi.org/10.3390/nu5103948
Chicago/Turabian StyleBateman, Emma, Joanne Bowen, Andrea Stringer, Bronwen Mayo, Erin Plews, Anthony Wignall, Norman Greenberg, Eduardo Schiffrin, and Dorothy Keefe. 2013. "Investigation of Effect of Nutritional Drink on Chemotherapy-Induced Mucosal Injury and Tumor Growth in an Established Animal Model" Nutrients 5, no. 10: 3948-3963. https://doi.org/10.3390/nu5103948
APA StyleBateman, E., Bowen, J., Stringer, A., Mayo, B., Plews, E., Wignall, A., Greenberg, N., Schiffrin, E., & Keefe, D. (2013). Investigation of Effect of Nutritional Drink on Chemotherapy-Induced Mucosal Injury and Tumor Growth in an Established Animal Model. Nutrients, 5(10), 3948-3963. https://doi.org/10.3390/nu5103948