Grape Consumption Increases Anti-Inflammatory Markers and Upregulates Peripheral Nitric Oxide Synthase in the Absence of Dyslipidemias in Men with Metabolic Syndrome
Abstract
:1. Introduction
2. Experimental Section
2.1. Study Protocol and Population
2.2. Classification of Participants
2.3. Antropometrics, Blood Pressure and Blood Collection
2.4. Plasma Inflammatory Cytokines and Adiponectin
2.5. Plasma LDL Oxidation and Urinary 8-Isoprostanes
2.6. Peripheral Blood Mononuclear Cells (PBMC) Isolation
2.7. RNA Extraction and Real-Time Polymerase Chain Reaction (PCR)
2.8. Statistical Analysis
3. Results
3.1. MetS Criteria & Study Population
| Parameter | Dyslipidemia ( n = 11) | Non-Dyslipidemia ( n = 13) | p Value |
|---|---|---|---|
| Age | 48.1 ± 11.3 | 53.9 ± 7.4 | NS |
| Waist Circumference (cm) | 107.4 ± 10.5 | 109.2 ± 16.1 | NS |
| Plasma Triglycerides (mg/dL) | 222 ± 83 a | 146 ± 70 b | p < 0.025 |
| Plasma HDL-C (mg/dL) | 30 ± 5 a | 43 ± 6 b | p < 0.001 |
| Plasma Glucose (mg/dL) | 99 ± 7 a | 107 ± 9 b | p < 0.025 |
| Systolic Blood Pressure (mm Hg) | 126 ± 13 | 131 ± 11 | NS |
| Diastolic Blood Pressure (mm Hg) | 83 ± 7 | 85 ± 7 | NS |
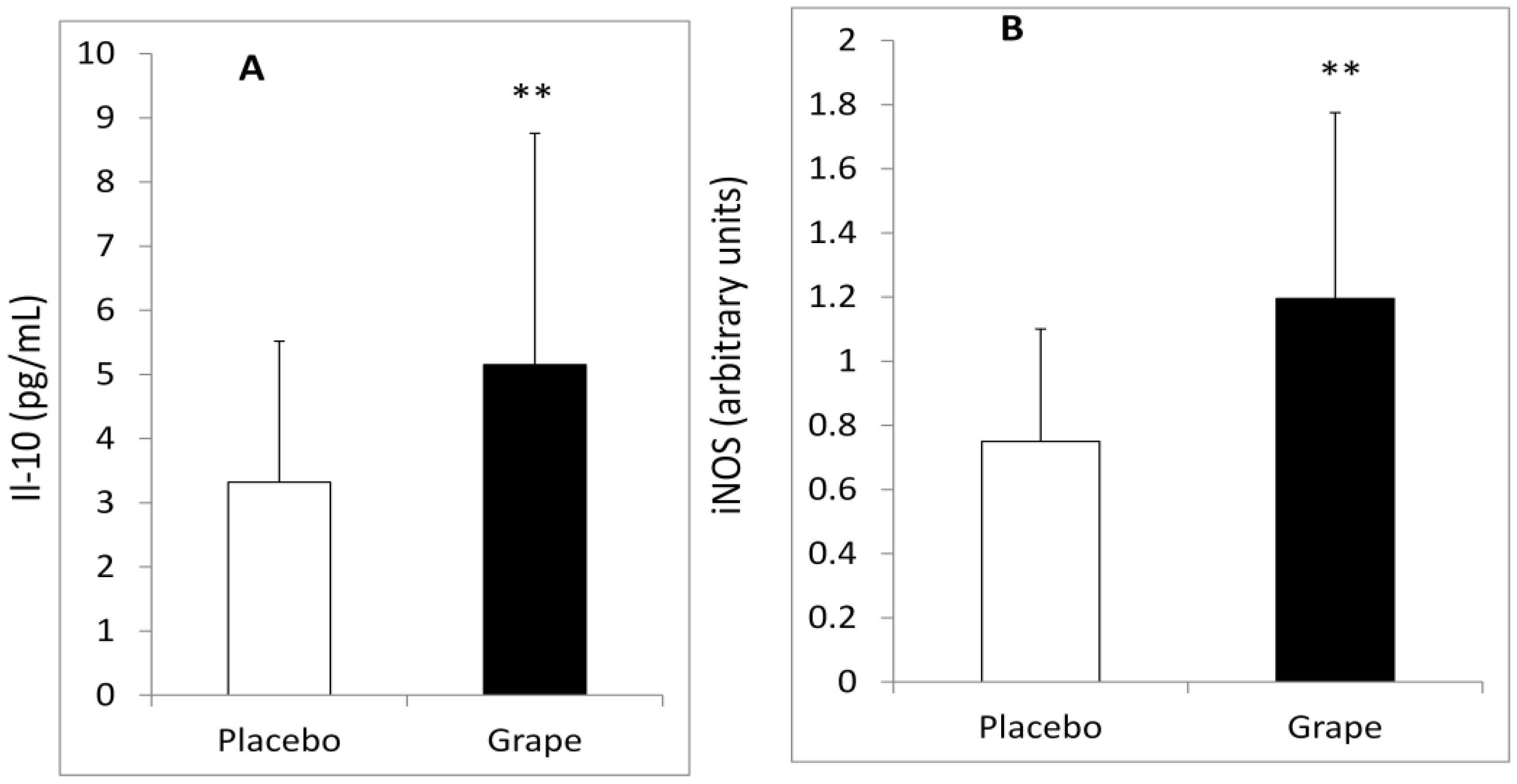
3.2. Plasma Inflammatory Markers and Adiponectin
| Change between GRAPE and Placebo | Dyslipidemia ( n = 11) | Non-Dyslipidemia ( n = 13) | p Value |
|---|---|---|---|
| Adiponectin (μg/mL) | −1.749 ± 3.341 a | 1.137 ± 3.930 b | p < 0.05 |
| IL-10 (pg/mL) | −2.830 ± 3.988 a | 1.838 ± 2.738 b | p < 0.005 |
| IL-6 (pg/mL) | 0.264 ± 1.085 | −0.175 ± 1.171 | NS |
| IL-8 (pg/mL) | −0.061 ± 0.534 | 0.115 ± 0.383 | NS |
| TNF-α (pg/mL) | 0.057 ± 0.782 | 0.175 ± 0.647 | NS |
3.3. Gene Expression of Inflammatory and Oxidative Stress Markers
| Change between GRAPE and Placebo | Dyslipidemia ( n = 11) | Non-Dyslipidemia ( n = 13) | p Value |
|---|---|---|---|
| NOX2 (arbitrary units) | 0.179 ± 0.514 | −0.004 ± 0.726 | NS |
| SOD1 (arbitrary units) | 0.281 ± 1.335 | −0.355 ± 1.289 | NS |
| SOD2 (arbitrary units) | 0.927 ± 2.917 | −0.165 ± 1.383 | NS |
| GPX1 (arbitrary units) | 0.460 ± 1.793 | −0.152 ± 0.722 | NS |
| GPX4 (arbitrary units) | 0.196 ± 0.738 | −0.196 ± 0.798 | NS |
| iNOS (arbitrary units) | −0.460 ± 0.913 a | 0.449 ± 0.726 b | p < 0.025 |
3.4. Oxidative Stress Markers
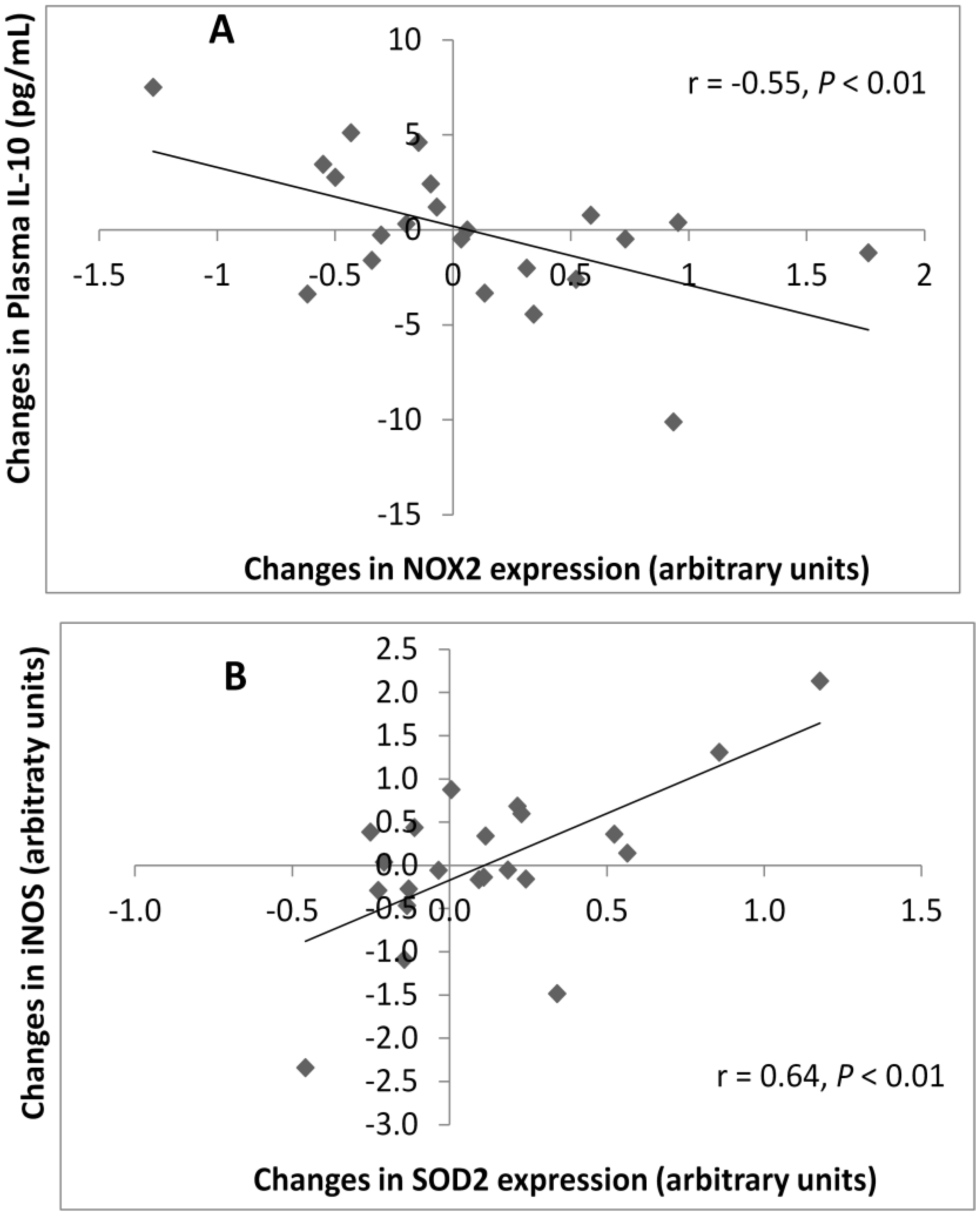
3.5. Correlations between Plasma and Gene Expression Markers
4. Discussion
5. Conclusions
Conflict of Interest
References
- Alberti, K.G.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.C.; James, W.P.; Loria, C.M.; Smith, S.C., Jr. Harmonizing the metabolic syndrome: A joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009, 120, 1640–1645. [Google Scholar]
- Grundy, S.M.; Brewer, H.B., Jr.; Cleeman, J.I.; Smith, S.C., Jr.; Lenfant, C. Definition of metabolic syndrome: Report of the National Heart, Lung, and Blood Institute/American Heart Association conference on scientific issues related to definition. Circulation 2004, 109, 433–438. [Google Scholar] [CrossRef]
- Furukawa, S.; Fujita, T.; Shimabukuro, M.; Iwaki, M.; Yamada, Y.; Nakajima, Y.; Nakayama, O.; Makishima, M.; Matsuda, M.; Shimomura, I. Increased oxidative stress in obesity and its impact on metabolic syndrome. J. Clin. Invest. 2004, 114, 1752–1761. [Google Scholar]
- Devaraj, S.; Rosenson, R.S.; Jialal, I. Metabolic syndrome: An appraisal of the pro-inflammatory and procoagulant status. Endocrinol. Metab. Clin. North Am. 2004, 33, 431–453. [Google Scholar] [CrossRef]
- Gustafson, B.; Hammarstedt, A.; Andersson, C.X.; Smith, U. Inflamed adipose tissue: A culprit underlying the metabolic syndrome and atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 2276–2283. [Google Scholar] [CrossRef]
- De Matthaeis, A.; Greco, A.; Serviddio, G.; Stramaglia, G.; Vendemiale, G. Endothelial dysfunction evaluated by flow mediated dilation is strongly associated to metabolic syndrome in the elderly. Aging Clin. Exp. Res. 2010, 22, 303–307. [Google Scholar]
- Choi, K.M.; Ryu, O.H.; Lee, K.W.; Kim, H.Y.; Seo, J.A.; Kim, S.G.; Kim, N.H.; Choi, D.S.; Baik, S.H. Serum adiponectin, interleukin-10 levels and inflammatory markers in the metabolic syndrome. Diabetes Res. Clin. Pract. 2007, 75, 235–240. [Google Scholar] [CrossRef]
- Schwartz, E.A.; Reaven, P.D. Lipolysis of triglyceride-rich lipoproteins, vascular inflammation, and atherosclerosis. Biochim. Biophys. Acta 2012, 1821, 858–866. [Google Scholar] [CrossRef]
- Feingold, K.R.; Grunfeld, C. Role of cytokines in inducing hyperlipidemia. Diabetes 1992, 41, 97–101. [Google Scholar]
- Esposito, K.; Pontillo, A.; Giugliano, F.; Giugliano, G.; Marfella, R.; Nicoletti, G.; Giugliano, D. Association of low interleukin-10 levels with the metabolic syndrome in obese women. J. Clin. Endocrinol. Metab. 2003, 88, 1055–1058. [Google Scholar] [CrossRef]
- Van Exel, E.; Gussekloo, J.; de Craen, A.J.; Frolich, M.; Bootsma-van der Wiel, A.; Westendorp, R.G. Low production capacity of interleukin-10 associates with the metabolic syndrome and type 2 diabetes: The Leiden 85-Plus Study. Diabetes 2002, 51, 1088–1092. [Google Scholar] [CrossRef]
- Zern, T.L.; Fernandez, M.L. Cardioprotective effects of dietary polyphenols. J. Nutr. 2005, 135, 2291–2294. [Google Scholar]
- Magrone, T.; Jirillo, E. Polyphenols from red wine are potent modulators of innate and adaptive immune responsiveness. Proc. Nutr. Soc. 2010, 69, 279–285. [Google Scholar] [CrossRef]
- Santangelo, C.; Vari, R.; Scazzocchio, B.; di Benedetto, R.; Filesi, C.; Masella, R. Polyphenols, intracellular signalling and inflammation. Ann. Ist. Super. Sanita 2007, 43, 394–405. [Google Scholar]
- Xia, E.Q.; Deng, G.F.; Guo, Y.J.; Li, H.B. Biological activities of polyphenols from grapes. Int. J. Mol. Sci. 2010, 11, 622–646. [Google Scholar] [CrossRef]
- Zern, T.L.; Wood, R.J.; Greene, C.; West, K.L.; Liu, Y.; Aggarwal, D.; Shachter, N.S.; Fernandez, M.L. Grape polyphenols exert a cardioprotective effect in pre- and postmenopausal women by lowering plasma lipids and reducing oxidative stress. J. Nutr. 2005, 135, 1911–1917. [Google Scholar]
- Barona, J.; Aristizabal, J.C.; Blesso, C.N.; Volek, J.S.; Fernandez, M.L. Grape polyphenols reduce blood pressure and increase flow-mediated vasodilation in men with metabolic syndrome. J. Nutr. 2012, 142, 1626–1632. [Google Scholar] [CrossRef]
- Holvoet, P.; Macy, E.; Landeloos, M.; Jones, D.; Jenny, N.S.; van de Werf, F.; Tracy, R.P. Analytical performance and diagnostic accuracy of immunometric assays for the measurement of circulating oxidized LDL. Clin. Chem. 2006, 52, 760–764. [Google Scholar]
- Böyum, A. Isolation of mononuclear cells and granulocytes from human blood. Isolation of monuclear cells by one centrifugation, and of granulocytes by combining centrifugation and sedimentation at 1 g. Scand. J. Clin. Lab. Invest. Suppl. 1968, 97, 77–89. [Google Scholar]
- Park, Y.K.; Rasmussen, H.E.; Ehlers, S.J.; Blobaum, K.R.; Lu, F.; Schlegal, V.L.; Carr, T.P.; Lee, J.Y. Repression of proinflammatory gene expression by lipid extract of Nostoc commune var sphaeroides Kutzing, a blue-green alga, via inhibition of nuclear factor-kappaB in RAW 26.7 macrophages. Nutr. Res. 2008, 28, 83–91. [Google Scholar] [CrossRef]
- Adiels, M.; Olofsson, S.O.; Taskinen, M.R.; Boren, J. Overproduction of very low-density lipoproteins is the hallmark of the dyslipidemia in the metabolic syndrome. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 1225–1236. [Google Scholar] [CrossRef]
- Raal, F.J. Pathogenesis and management of the dyslipidemia of the metabolic syndrome. Metab. Syndr. Relat. Disord. 2009, 7, 83–88. [Google Scholar] [CrossRef]
- Tedgui, A.; Mallat, Z. Anti-inflammatory mechanisms in the vascular wall. Circ. Res. 2001, 88, 877–887. [Google Scholar] [CrossRef]
- Decorde, K.; Teissedre, P.L.; Sutra, T.; Ventura, E.; Cristol, J.P.; Rouanet, J.M. Chardonnay grape seed procyanidin extract supplementation prevents high-fat diet-induced obesity in hamsters by improving adipokine imbalance and oxidative stress markers. Mol. Nutr. Food Res. 2009, 53, 659–666. [Google Scholar] [CrossRef]
- Kumada, M.; Kihara, S.; Ouchi, N.; Kobayashi, H.; Okamoto, Y.; Ohashi, K.; Maeda, K.; Nagaretani, H.; Kishida, K.; Maeda, N.; et al. Adiponectin specifically increased tissue inhibitor of metalloproteinase-1 through interleukin-10 expression in human macrophages. Circulation 2004, 109, 2046–2049. [Google Scholar] [CrossRef]
- Nishida, M.; Moriyama, T.; Sugita, Y.; Yamauchi-Takihara, K. Interleukin-10 associates with adiponectin predominantly in subjects with metabolic syndrome. Circ. J. 2007, 71, 1234–1238. [Google Scholar] [CrossRef]
- Tunon, M.J.; Garcia-Mediavilla, M.V.; Sanchez-Campos, S.; Gonzalez-Gallego, J. Potential of flavonoids as anti-inflammatory agents: Modulation of pro-inflammatory gene expression and signal transduction pathways. Curr. Drug Metab. 2009, 10, 256–271. [Google Scholar] [CrossRef]
- Mallat, Z.; Heymes, C.; Ohan, J.; Faggin, E.; Leseche, G.; Tedgui, A. Expression of interleukin-10 in advanced human atherosclerotic plaques: Relation to inducible nitric oxide synthase expression and cell death. Arterioscler. Thromb. Vasc. Biol. 1999, 19, 611–616. [Google Scholar] [CrossRef]
- Magrone, T.; Tafaro, A.; Jirillo, F.; Panaro, M.A.; Cuzzuol, P.; Cuzzuol, A.C.; Pugliese, V.; Amati, L.; Jirillo, E.; Covelli, V. Red wine consumption and prevention of atherosclerosis: An in vitro model using human peripheral blood mononuclear cells. Curr. Pharm. Des. 2007, 13, 3718–3725. [Google Scholar] [CrossRef]
- Fukai, T.; Ushio-Fukai, M. Superoxide dismutases: Role in redox signaling, vascular function, and diseases. Antioxid. Redox Signal. 2011, 15, 1583–1606. [Google Scholar] [CrossRef]
- Castilla, P.; Davalos, A.; Teruel, J.L.; Cerrato, F.; Fernandez-Lucas, M.; Merino, J.L.; Sanchez-Martin, C.C.; Ortuno, J.; Lasuncion, M.A. Comparative effects of dietary supplementation with red grape juice and vitamin E on production of superoxide by circulating neutrophil NADPH oxidase in hemodialysis patients. Am. J. Clin. Nutr. 2008, 87, 1053–1061. [Google Scholar]
- Davalos, A.; de la Pena, G.; Sanchez-Martin, C.C.; Teresa Guerra, M.; Bartolome, B.; Lasuncion, M.A. Effects of red grape juice polyphenols in NADPH oxidase subunit expression in human neutrophils and mononuclear blood cells. Br. J. Nutr. 2009, 102, 1125–1135. [Google Scholar] [CrossRef]
- Du, Y.; Guo, H.; Lou, H. Grape seed polyphenols protect cardiac cells from apoptosis via induction of endogenous antioxidant enzymes. J. Agric. Food Chem. 2007, 55, 1695–1701. [Google Scholar] [CrossRef]
- Borges, G.; Mullen, W.; Mullan, A.; Lean, M.E.; Roberts, S.A.; Crozier, A. Bioavailability of multiple components following acute ingestion of a polyphenol-rich juice drink. Mol. Nutr. Food Res. 2010, 54, S268–S277. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Barona, J.; Blesso, C.N.; Andersen, C.J.; Park, Y.; Lee, J.; Fernandez, M.L. Grape Consumption Increases Anti-Inflammatory Markers and Upregulates Peripheral Nitric Oxide Synthase in the Absence of Dyslipidemias in Men with Metabolic Syndrome. Nutrients 2012, 4, 1945-1957. https://doi.org/10.3390/nu4121945
Barona J, Blesso CN, Andersen CJ, Park Y, Lee J, Fernandez ML. Grape Consumption Increases Anti-Inflammatory Markers and Upregulates Peripheral Nitric Oxide Synthase in the Absence of Dyslipidemias in Men with Metabolic Syndrome. Nutrients. 2012; 4(12):1945-1957. https://doi.org/10.3390/nu4121945
Chicago/Turabian StyleBarona, Jacqueline, Christopher N. Blesso, Catherine J. Andersen, Youngki Park, Jiyoung Lee, and Maria Luz Fernandez. 2012. "Grape Consumption Increases Anti-Inflammatory Markers and Upregulates Peripheral Nitric Oxide Synthase in the Absence of Dyslipidemias in Men with Metabolic Syndrome" Nutrients 4, no. 12: 1945-1957. https://doi.org/10.3390/nu4121945
APA StyleBarona, J., Blesso, C. N., Andersen, C. J., Park, Y., Lee, J., & Fernandez, M. L. (2012). Grape Consumption Increases Anti-Inflammatory Markers and Upregulates Peripheral Nitric Oxide Synthase in the Absence of Dyslipidemias in Men with Metabolic Syndrome. Nutrients, 4(12), 1945-1957. https://doi.org/10.3390/nu4121945