Blackcurrant Anthocyanins Increase the Levels of Collagen, Elastin, and Hyaluronic Acid in Human Skin Fibroblasts and Ovariectomized Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Cell Culture
2.2. Microarray Gene Expression Profiling
2.3. IPA
2.4. RT-qPCR
2.5. Immunofluorescence Staining
2.6. Animals and Treatments
2.7. Collagen Thickness Measurement
2.8. Elastic Fiber Staining
2.9. Immunohistochemical Staining for Hyaluronic Acid
2.10. Statistical Analysis
3. Results
3.1. Microarray Profiling of TIG113 Cells Exposed to BCE
3.2. RT-qPCR Analysis of ECM Gene mRNA Levels
3.3. Induction of ECM Proteins by BCE and Anthocyanins
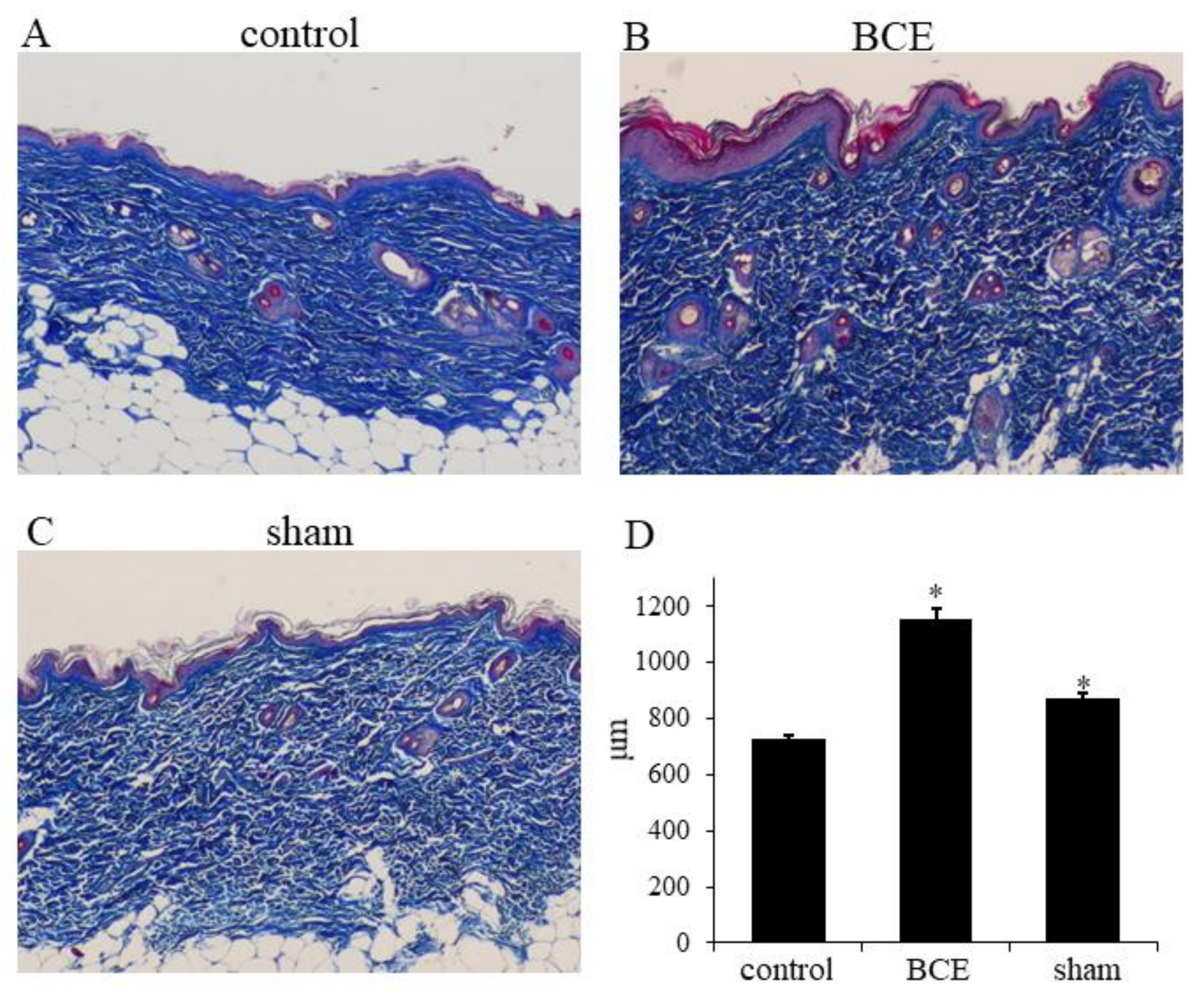
3.4. Increased Collagen Thickness in BCE-Treated OVX Rats
3.5. Induction of Elastic Fibers in BCE-Treated OVX Rats
3.6. Induction of Hyaluronic Acid Expression in BCE-Treated OVX Rats
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| BCE | blackcurrant extract |
| E2 | 17β-estradiol |
| C3G | cyanidin-3-glucoside |
| C3R | cyanidin-3-rutinoside |
| D3G | delphinidin-3-glucoside |
| D3R | delphinidin-3-rutinoside |
| ER | estrogen receptor |
| ECM | extracellular matrix |
| GAPDH | glyceraldehyde 3-phosphate dehydrogenase |
| HAS | hyaluronan synthase |
| HYAL | hyaluronoglucosaminidase |
| IGF | insulin-like growth factor |
| IGFBP | insulin-like growth factor binding protein |
| IPA | Ingenuity® Pathway Analysis |
| MMP | matrix metalloproteinase |
| TGF | transforming growth factor |
| TIMP | tissue inhibitor of metalloproteinase |
| OVX | ovariectomized |
References
- Gopalan, A.; Reuben, S.C.; Ahmed, S.; Darvesh, A.S.; Hohmann, J.; Bishayee, A. The health benefits of blackcurrants. Food Funct. 2012, 3, 795–809. [Google Scholar] [CrossRef] [PubMed]
- Nanashima, N.; Horie, K.; Chiba, M.; Nakano, M.; Maeda, H.; Nakamura, T. Anthocyanin-rich blackcurrant extract inhibits proliferation of the MCF10A healthy human breast epithelial cell line through induction of G0/G1 arrest and apoptosis. Mol. Med. Rep. 2017, 16, 6134–6141. [Google Scholar] [CrossRef] [PubMed]
- Brincat, M.P.; Baron, Y.M.; Galea, R. Estrogens and the skin. Climacteric 2005, 8, 110–123. [Google Scholar] [CrossRef] [PubMed]
- Thornton, M.J. The biological actions of estrogens on skin. Exp. Dermatol. 2002, 11, 487–502. [Google Scholar] [CrossRef] [PubMed]
- Thornton, M.J. Estrogens and aging skin. Dermatoendocrinology 2013, 5, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Verdier-Sevrain, S. Effect of estrogens on skin aging and the potential role of selective estrogen receptor modulators. Climacteric 2007, 10, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Murphy, G. Tissue inhibitors of metalloproteinases. Genome Biol. 2011, 12, 233. [Google Scholar] [CrossRef] [PubMed]
- Stern, R.; Maibach, H.I. Hyaluronan in skin: Aspects of aging and its pharmacologic modulation. Clin. Dermatol. 2008, 26, 106–122. [Google Scholar] [CrossRef] [PubMed]
- Itano, N.; Kimata, K. Mammalian hyaluronan synthases. IUBMB Life 2002, 54, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Affinito, P.; Palomba, S.; Sorrentino, C.; Di Carlo, C.; Bifulco, G.; Arienzo, M.P.; Nappi, C. Effects of postmenopausal hypoestrogenism on skin collagen. Maturitas 1999, 33, 239–247. [Google Scholar] [CrossRef]
- Brincat, M.; Moniz, C.J.; Studd, J.W.; Darby, A.; Magos, A.; Emburey, G.; Versi, E. Long-term effects of the menopause and sex hormones on skin thickness. Br. J. Obstet. Gynaecol. 1985, 92, 256–259. [Google Scholar] [CrossRef] [PubMed]
- Surazynski, A.; Jarzabek, K.; Haczynski, J.; Laudanski, P.; Palka, J.; Wolczynski, S. Differential effects of estradiol and raloxifene on collagen biosynthesis in cultured human skin fibroblasts. Int. J. Mol. Med. 2003, 12, 803–809. [Google Scholar] [CrossRef] [PubMed]
- Bentley, J.P.; Brenner, R.M.; Linstedt, A.D.; West, N.B.; Carlisle, K.S.; Rokosova, B.C.; MacDonald, N. Increased hyaluronate and collagen biosynthesis and fibroblast estrogen receptors in macaque sex skin. J. Investig. Dermatol. 1986, 87, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Patriarca, M.T.; Barbosa de Moraes, A.R.; Nader, H.B.; Petri, V.; Martins, J.R.; Gomes, R.C.; Soares, J.M., Jr. Hyaluronic acid concentration in postmenopausal facial skin after topical estradiol and genistein treatment: A double-blind, randomized clinical trial of efficacy. Menopause 2013, 20, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Sobel, H.; Cohen, R.A. Effect of estradion on hyaluronic acid in the skin of aging mice. Steroids 1970, 16, 1–3. [Google Scholar] [CrossRef]
- Son, E.D.; Lee, J.Y.; Lee, S.; Kim, M.S.; Lee, B.G.; Chang, I.S.; Chung, J.H. Topical application of 17beta-estradiol increases extracellular matrix protein synthesis by stimulating TGF-Beta signaling in aged human skin in vivo. J. Investig. Dermatol. 2005, 124, 1149–1161. [Google Scholar] [CrossRef] [PubMed]
- Maheux, R.; Naud, F.; Rioux, M.; Grenier, R.; Lemay, A.; Guy, J.; Langevin, M. A randomized, double-blind, placebo-controlled study on the effect of conjugated estrogens on skin thickness. Am. J. Obstet. Gynecol. 1994, 170, 642–649. [Google Scholar] [CrossRef]
- Guo, D.; Wang, J.; Wang, X.; Luo, H.; Zhang, H.; Cao, D.; Chen, L.; Huang, N. Double directional adjusting estrogenic effect of naringin from Rhizoma drynariae (Gusuibu). J. Ethnopharmacology 2011, 138, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.M.; Kim, J.B.; Bae, J.H.; Lee, J.S.; Kim, P.S.; Jang, H.H.; Kim, H.R. Estrogen-like activity of aqueous extract from Agrimonia pilosa Ledeb. in MCF-7 cells. BMC Complement. Altern. Med. 2012, 12, 260. [Google Scholar] [CrossRef] [PubMed]
- Limer, J.L.; Speirs, V. Phyto-oestrogens and breast cancer chemoprevention. Breast Cancer Res. 2004, 6, 119–127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahmoud, A.M.; Yang, W.; Bosland, M.C. Soy isoflavones and prostate cancer: A review of molecular mechanisms. J. Steroid Biochem. Mol. Biol. 2014, 140, 116–132. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, E.; Stopper, H. Estrogenic activity of naturally occurring anthocyanidins. Nutr. Cancer 2001, 41, 145–149. [Google Scholar] [CrossRef] [PubMed]
- Nanashima, N.; Horie, K.; Maeda, H. Phytoestrogenic activity of blackcurrant anthocyanins is partially mediated through estrogen receptor beta. Molecules 2018, 23, 74. [Google Scholar] [CrossRef] [PubMed]
- Nanashima, N.; Horie, K.; Tomisawa, T.; Chiba, M.; Nakano, M.; Fujita, T.; Maeda, H.; Kitajima, M.; Takamagi, S.; Uchiyama, D.; et al. Phytoestrogenic activity of blackcurrant (Ribes nigrum) anthocyanins is mediated through estrogen receptor alpha. Mol. Nutr. Food Res. 2015, 59, 2419–2431. [Google Scholar] [CrossRef] [PubMed]
- Gopaul, R.; Knaggs, H.E.; Lephart, E.D. Biochemical investigation and gene analysis of equol: A plant and soy-derived isoflavonoid with antiaging and antioxidant properties with potential human skin applications. Biofactors 2012, 38, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Mao, S.; Wang, Y.; Zhang, M.; Hinek, A. Phytoestrogen, tanshinone IIA diminishes collagen deposition and stimulates new elastogenesis in cultures of human cardiac fibroblasts. Exp. Cell Res. 2014, 323, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Polito, F.; Marini, H.; Bitto, A.; Irrera, N.; Vaccaro, M.; Adamo, E.B.; Micali, A.; Squadrito, F.; Minutoli, L.; Altavilla, D. Genistein aglycone, a soy-derived isoflavone, improves skin changes induced by ovariectomy in rats. Br. J. Pharmacol. 2012, 165, 994–1005. [Google Scholar] [CrossRef] [PubMed]
- Chua, L.S.; Lee, S.Y.; Abdullah, N.; Sarmidi, M.R. Review on Labisia pumila (Kacip Fatimah): Bioactive phytochemicals and skin collagen synthesis promoting herb. Fitoterapia 2012, 83, 1322–1335. [Google Scholar] [CrossRef] [PubMed]
- Lephart, E.D. Resveratrol, 4′ acetoxy resveratrol, R-equol, racemic equol or S-equol as cosmeceuticals to improve dermal health. Int. J. Mol. Sci. 2017, 18, 1193. [Google Scholar] [CrossRef] [PubMed]
- Haczynski, J.; Tarkowski, R.; Jarzabek, K.; Wolczynski, S.; Magoffin, D.A.; Czarnocki, K.J.; Ziegert, M.; Jakowicki, J.; Jakimiuk, A.J. Differential effects of estradiol, raloxifene and tamoxifen on estrogen receptor expression in cultured human skin fibroblasts. Int. J. Mol. Med. 2004, 13, 903–908. [Google Scholar] [CrossRef] [PubMed]
- Carnesecchi, J.; Malbouyres, M.; de Mets, R.; Balland, M.; Beauchef, G.; Vie, K.; Chamot, C.; Lionnet, C.; Ruggiero, F.; Vanacker, J.M. Estrogens induce rapid cytoskeleton re-organization in human dermal fibroblasts via the non-classical receptor GPR30. PLoS ONE 2015, 10, e0120672. [Google Scholar] [CrossRef] [PubMed]
- Voloshenyuk, T.G.; Gardner, J.D. Estrogen improves TIMP-MMP balance and collagen distribution in volume-overloaded hearts of ovariectomized females. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 299, R683–R693. [Google Scholar] [CrossRef] [PubMed]
- Van Doren, S.R. Matrix metalloproteinase interactions with collagen and elastin. Matrix Biol. 2015, 44–46, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Mecham, R.P.; Broekelmann, T.J.; Fliszar, C.J.; Shapiro, S.D.; Welgus, H.G.; Senior, R.M. Elastin degradation by matrix metalloproteinases. Cleavage site specificity and mechanisms of elastolysis. J. Biol. Chem. 1997, 272, 18071–18076. [Google Scholar] [CrossRef] [PubMed]
- Massague, J. The transforming growth factor-beta family. Annu. Rev. Cell Biol. 1990, 6, 597–641. [Google Scholar] [CrossRef] [PubMed]
- Massague, J. TGF-beta signal transduction. Annu. Rev. Biochem. 1998, 67, 753–791. [Google Scholar] [CrossRef] [PubMed]
- Piek, E.; Heldin, C.H.; Ten Dijke, P. Specificity, diversity, and regulation in TGF-beta superfamily signaling. FASEB J. 1999, 13, 2105–2124. [Google Scholar] [CrossRef] [PubMed]
- Band, A.M.; Laiho, M. Crosstalk of TGF-beta and estrogen receptor signaling in breast cancer. J. Mammary Gland Biol. Neoplasia 2011, 16, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Rinderknecht, E.; Humbel, R.E. The amino acid sequence of human insulin-like growth factor I and its structural homology with proinsulin. J. Biol. Chem. 1978, 253, 2769–2776. [Google Scholar] [PubMed]
- Noordam, R.; Gunn, D.A.; Tomlin, C.C.; Maier, A.B.; Griffiths, T.; Catt, S.D.; Ogden, S.; Slagboom, P.E.; Westendorp, R.G.; Griffiths, C.E.; et al. Serum insulin-like growth factor 1 and facial ageing: High levels associate with reduced skin wrinkling in a cross-sectional study. Br. J. Dermatol. 2013, 168, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama-Nakagiri, Y.; Naoe, A.; Ohuchi, A.; Kitahara, T. Serum levels of IGF-1 are related to human skin characteristics including the conspicuousness of facial pores. Int. J. Cosmet. Sci. 2011, 33, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Povoa, G.; Diniz, L.M. Growth hormone system: Skin interactions. An. Bras. Dermatol. 2011, 86, 1159–1165. [Google Scholar] [PubMed]
- Brandt, K.; Grunler, J.; Brismar, K.; Wang, J. Effects of IGFBP-1 and IGFBP-2 and their fragments on migration and IGF-induced proliferation of human dermal fibroblasts. Growth Horm. IGF Res. 2015, 25, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Edmondson, S.R.; Thumiger, S.P.; Werther, G.A.; Wraight, C.J. Epidermal homeostasis: The role of the growth hormone and insulin-like growth factor systems. Endocr. Rev. 2003, 24, 737–764. [Google Scholar] [CrossRef] [PubMed]
- Takeo, C.; Ikeda, K.; Horie-Inoue, K.; Inoue, S. Identification of Igf2, Igfbp2 and Enpp2 as estrogen-responsive genes in rat hippocampus. Endocr. J. 2009, 56, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Skandalis, S.S.; Afratis, N.; Smirlaki, G.; Nikitovic, D.; Theocharis, A.D.; Tzanakakis, G.N.; Karamanos, N.K. Cross-talk between estradiol receptor and EGFR/IGF-IR signaling pathways in estrogen-responsive breast cancers: Focus on the role and impact of proteoglycans. Matrix Biol. 2014, 35, 182–193. [Google Scholar] [CrossRef] [PubMed]
- Rock, K.; Meusch, M.; Fuchs, N.; Tigges, J.; Zipper, P.; Fritsche, E.; Krutmann, J.; Homey, B.; Reifenberger, J.; Fischer, J.W. Estradiol protects dermal hyaluronan/versican matrix during photoaging by release of epidermal growth factor from keratinocytes. J. Biol. Chem. 2012, 287, 20056–20069. [Google Scholar] [CrossRef] [PubMed]
- Suetsugi, M.; Su, L.; Karlsberg, K.; Yuan, Y.C.; Chen, S. Flavone and isoflavone phytoestrogens are agonists of estrogen-related receptors. Mol. Cancer Res. 2003, 1, 981–991. [Google Scholar] [PubMed]
- Pomari, E.; Dalla Valle, L.; Pertile, P.; Colombo, L.; Thornton, M.J. Intracrine sex steroid synthesis and signaling in human epidermal keratinocytes and dermal fibroblasts. FASEB J. 2015, 29, 508–524. [Google Scholar] [CrossRef] [PubMed]
- Moriwaki, S.; Suzuki, K.; Muramatsu, M.; Nomura, A.; Inoue, F.; Into, T.; Yoshiko, Y.; Niida, S. Delphinidin, one of the major anthocyanidins, prevents bone loss through the inhibition of excessive osteoclastogenesis in osteoporosis model mice. PLoS ONE 2014, 9, e97177. [Google Scholar] [CrossRef] [PubMed]





| Predicted Upstream Regulator | z-Score |
|---|---|
| Estradiol | 2.68 |
| ERα | 1.68 |
| ERβ | 0.89 |
| Abbreviation | Full Name | Fold Change * |
|---|---|---|
| COL1A1 | collagen, type I, alpha 1 | 1.0 |
| COL1A2 | collagen, type I, alpha 2 | 1.0 |
| COL3A1 | collagen, type III, alpha 1 | 1.8 |
| ELN | elastin | −1.4 |
| HAS2 | hyaluronan synthase 2 | 1.3 |
| HAS3 | hyaluronan synthase 3 | 1.5 |
| HYAL2 | hyaluronoglucosaminidase 2 | −1.3 |
| HYAL3 | hyaluronoglucosaminidase 3 | −1.6 |
| HYAL4 | hyaluronoglucosaminidase 4 | −1.1 |
| MMP1 | matrix metalloproteinase 1 | −1.4 |
| MMP2 | matrix metalloproteinase 2 | −1.0 |
| MMP3 | matrix metalloproteinase 3 | 1.3 |
| MMP9 | matrix metalloproteinase 9 | −1.1 |
| MMP12 | matrix metalloproteinase 12 | −1.5 |
| TIMP1 | tissue inhibitor of metalloproteinase 1 | −1.1 |
| TIMP2 | tissue inhibitor of metalloproteinase 2 | 1.1 |
| TIMP3 | tissue inhibitor of metalloproteinase 3 | 5.7 |
| TIMP4 | tissue inhibitor of metalloproteinase 4 | 1.2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nanashima, N.; Horie, K.; Maeda, H.; Tomisawa, T.; Kitajima, M.; Nakamura, T. Blackcurrant Anthocyanins Increase the Levels of Collagen, Elastin, and Hyaluronic Acid in Human Skin Fibroblasts and Ovariectomized Rats. Nutrients 2018, 10, 495. https://doi.org/10.3390/nu10040495
Nanashima N, Horie K, Maeda H, Tomisawa T, Kitajima M, Nakamura T. Blackcurrant Anthocyanins Increase the Levels of Collagen, Elastin, and Hyaluronic Acid in Human Skin Fibroblasts and Ovariectomized Rats. Nutrients. 2018; 10(4):495. https://doi.org/10.3390/nu10040495
Chicago/Turabian StyleNanashima, Naoki, Kayo Horie, Hayato Maeda, Toshiko Tomisawa, Maiko Kitajima, and Toshiya Nakamura. 2018. "Blackcurrant Anthocyanins Increase the Levels of Collagen, Elastin, and Hyaluronic Acid in Human Skin Fibroblasts and Ovariectomized Rats" Nutrients 10, no. 4: 495. https://doi.org/10.3390/nu10040495
APA StyleNanashima, N., Horie, K., Maeda, H., Tomisawa, T., Kitajima, M., & Nakamura, T. (2018). Blackcurrant Anthocyanins Increase the Levels of Collagen, Elastin, and Hyaluronic Acid in Human Skin Fibroblasts and Ovariectomized Rats. Nutrients, 10(4), 495. https://doi.org/10.3390/nu10040495






