Life Cycle Impact Assessment in the Arctic: Challenges and Research Needs
Abstract
:1. Introduction
2. Characteristics of the Arctic Region
2.1. Environment
2.2. Biology
2.3. Population
3. Life Cycle Impact Assessment for the Arctic Region
3.1. Climate Change
3.2. Photochemical Oxidant Formation
3.3. Particulate Matter Formation
3.4. Acidification and Eutrophication
3.5. Human Toxicity and Ecotoxicity
4. Arctic Mining LCA Case Study
4.1. Materials and Methods
4.2. Results of the Mining LCA Case Study
5. Discussion and Concluding Remarks
5.1. Influences of the Arctic Context on Impact Assessment in the Mining LCA Case Study
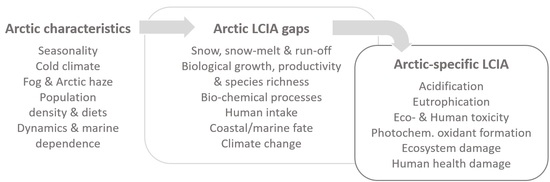
5.2. Research Gaps and Demand for Future Arctic LCA
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Huntington, H.; Weller, G.; Bush, E.; Callaghan, T.V.; Kattsov, V.M.; Nuttall, M. An Introduction to the Arctic Climate Impact. In Arctic Climate Impact Assessment; Symon, C., Arris, L., Hea, B., Eds.; Cambridge University Press: Cambridge, UK, 2005; pp. 1–20. [Google Scholar]
- Jeffries, M.O.; Overland, J.E.; Perovich, D.K. The Arctic shifts to a new normal. Phys. Today 2013, 66, 35–40. [Google Scholar] [CrossRef]
- Arctic Monitoring and Assessment Programme (AMAP). Summary for Policy-Makers: Arctic Pollution Issues 2015; Arctic Monitoring and Assessment Programme (AMAP): Oslo, Norway, 2015. [Google Scholar]
- Quinn, P.K.; Shaw, G.; Andrews, E.; Dutton, E.G.; Ruoho-Airola, T.; Gong, S.L. Arctic haze: Current trends and knowledge gaps. Tellus B 2007, 59, 99–114. [Google Scholar] [CrossRef]
- Gautier, D.L.; Bird, K.J.; Charpentier, R.R.; Grantz, A.; Houseknecht, D.W.; Klett, T.R.; Moore, T.E.; Pitman, J.K.; Schenk, C.J.; Schuenemeyer, J.H.; et al. Assessment of Undiscovered Oil and Gas in the Arctic. Science 2009, 324, 1175–1179. [Google Scholar] [CrossRef] [PubMed]
- Van Dam, K.; Scheepstra, A.; Gille, J.; Stępień, A.; Koivurova, T. Mining in The European Arctic. In Strategic Assessment of Development of the Arctic: Assessment Conducted for the European Union; Stępień, A., Koivurova, T., Kankaanpää, P., Eds.; The Arctic Center, University of Lapland: Rovaniemi, Finland, 2014; pp. 87–99. [Google Scholar]
- Nørregaard, R.D.; Nielsen, T.G.; Møller, E.F.; Strand, J.; Espersen, L.; Møhl, M. Evaluating pyrene toxicity on Arctic key copepod species Calanus hyperboreus. Ecotoxicology 2014, 23, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Curran, M.A. Life Cycle Assessment Student Handbook; Wiley: Hoboken, NJ, USA, 2015; ISBN 978-1-119-08354-2. [Google Scholar]
- International Organization for Standardization (ISO). ISO 14040: Environmental Management—Life Cycle Assessment—Principles and Framework; International Organization for Standardization (ISO): Geneva, Switzerland, 2006. [Google Scholar]
- Curran, M.A. Life Cycle Assessment: A review of the methodology and its application to sustainability. Curr. Opin. Chem. Eng. 2013, 2, 273–277. [Google Scholar] [CrossRef]
- Margni, M.; Curran, M.A. Life Cycle Impact Assessment. In Life Cycle Assessment Handbook: A Guide for Environmentally Sustainable Products; Curran, M.A., Ed.; John Wiley & Sons, Inc.: Somerset, NJ, USA, 2012; pp. 67–103. [Google Scholar]
- Woods, J.S.; Veltman, K.; Huijbregts, M.A.J.; Verones, F.; Hertwich, E.G. Towards a meaningful assessment of marine ecological impacts in life cycle assessment (LCA). Environ. Int. 2016, 89–90, 48–61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fava, J.; Consoli, F.; Denison, R.; Dickson, K.; Mohin, T.; Vigon, B. A Conceptual Framework for Life Cycle Impact Assessment; Society of Environmental Toxicology and Chemistry (SETAC): Brussels, Belgium, 1993. [Google Scholar]
- Potting, J.; Hauschild, M. Predicted Environmental Impact and Expected Occurance of Actual Enviornmental Impact, Part I: The linear nature of environmental impact from emissions in life-cycle assessment. Int. J. Life Cycle Assess. 1997, 2, 171–177. [Google Scholar] [CrossRef]
- Potting, J.; Hauschild, M.; Hauschild, M. Spatial Differentiation in Life Cycle Impact Assessment: A decade of method development to increase the environmental realism of LCIA. Int. J. Life Cycle Assess. 2006, 11, 11–13. [Google Scholar] [CrossRef]
- United Nations Environment Programme (UNEP). Global Guidance for Life Cycle Impact Assessment Indicators Volume 1; Frischknecht, R., Jolliet, O., Eds.; United Nations Environment Programme (UNEP): Nairobi, Kenya, 2016. [Google Scholar]
- European Commission-Joint Research Centre (EC-JRC). International Reference Life Cycle Data System (ILCD) Handbook-Recommendations for Life Cycle Impact Assessment in the European Context; European Commission-Joint Research Centre (EC-JRC): Luxemburg, 2011. [Google Scholar]
- Rosenbaum, R.K.; Bachmann, T.M.; Gold, L.S.; Huijbregts, M.A.J.; Jolliet, O.; Juraske, R.; Koehler, A.; Larsen, H.F.; MacLeod, M.; Margni, M.; et al. USEtox—The UNEP-SETAC toxicity model: Recommended characterisation factors for human toxicity and freshwater ecotoxicity in life cycle impact assessment. Int. J. Life Cycle Assess. 2008, 13, 532–546. [Google Scholar] [CrossRef]
- Bennett, D.; McKone, T.E.; Evans, J.S.; Nazaroff, W.W.; Margni, M.D.; Smith, K.R. Defining Intake Fraction. Environ. Sci. Technol. 2002, 36, 206A–211A. [Google Scholar] [CrossRef]
- Hofstetter, P. Perspectives in Life Cycle Impact Assessment: A Structured Approach to Combine Models of the Technosphere, Ecosphere and Valuesphere; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1998. [Google Scholar]
- Potting, J.; Schöpp, W.; Blok, K.; Hauschild, M. Site-Dependent Life-Cycle Impact Assessment of Acidification. J. Ind. Ecol. 1998, 2, 63–87. [Google Scholar] [CrossRef]
- Van Zelm, R.; Huijbregts, M.A.J.; van De Meent, D. USES-LCA 2.0-a global nested multi-media fate, exposure, and effects model. Int. J. Life Cycle Assess. 2009, 14, 282–284. [Google Scholar] [CrossRef]
- Owsianiak, M.; Rosenbaum, R.K.; Huijbregts, M.A.J.; Hauschild, M.Z. Addressing geographic variability in the comparative toxicity potential of copper and nickel in soils. Environ. Sci. Technol. 2013, 47, 3241–3250. [Google Scholar] [CrossRef] [PubMed]
- Posch, M.; Seppälä, J.; Hettelingh, J.P.; Johansson, M.; Margni, M.; Jolliet, O. The role of atmospheric dispersion models and ecosystem sensitivity in the determination of characterisation factors for acidifying and eutrophying emissions in LCIA. Int. J. Life Cycle Assess. 2008, 13, 477–486. [Google Scholar] [CrossRef]
- Toffoletto, L.; Bulle, C.; Godin, J.; Reid, C.; Deschênes, L. LUCAS—A New LCIA Method Used for a Canadian-Specific Context. Int. J. Life Cycle Assess. 2007, 12, 93–102. [Google Scholar] [CrossRef]
- Finnveden, G.; Nilsson, M. Site-dependent life-cycle impact assessment in Sweden. Int. J. Life Cycle Assess. 2005, 10, 235–239. [Google Scholar] [CrossRef]
- Verones, F.; Henderson, A.D.; Laurent, A.; Ridoutt, B.; Ugaya, C.; Hellweg, S. LCIA framework and modelling guidance. In Global Guidance for Life Cycle Impact Assessment Indicators Volume 1; Frischknecht, R., Jolliet, O., Eds.; United Nations Environment Programme (UNEP): Nairobi, Kenya, 2016. [Google Scholar]
- Johnsen, F.M. Bridging Arctic environmental science and life cycle assessment: A preliminary assessment of regional scaling factors. Clean Technol. Environ. Policy 2014, 16, 1713–1724. [Google Scholar] [CrossRef]
- Arctic Monitoring and Assessment Programme (AMAP). Physical/Geographical Characteristics of the Arctic. In AMAP Assessment Report: Arctic Pollution Issues; Arctic Monitoring and Assessment Programme (AMAP): Oslo, Norway, 1998; pp. 9–24. [Google Scholar]
- Huntington, H.P. Arctic Flora and Fauna: Status and Conservation; Edita: Helsinki, Finland, 2001. [Google Scholar]
- Prowse, T.D.; Wrona, F.J.; Reist, J.D.; Hobbie, J.E.; Lévesque, L.M.J.; Vincent, W.F.; Gibson, J.J.; Hobbie, J.E.; Lévesque, L.M.J.; Vincent, W.F. General Features of the Arctic Relevant to Climate Change in Freshwater Ecosystems. Ambio 2006, 35, 330–338. [Google Scholar] [CrossRef]
- Przybylak, R. Cloudiness. In The Climate of the Arctic; Springer International Publishing: Basel, Switzerland, 2016; pp. 111–126. [Google Scholar]
- Holmes, R.M.; McClelland, J.W.; Peterson, B.J.; Tank, S.E.; Bulygina, E.; Eglinton, T.I.; Gordeev, V.V.; Gurtovaya, T.Y.; Raymond, P.A.; Repeta, D.J.; et al. Seasonal and Annual Fluxes of Nutrients and Organic Matter from Large Rivers to the Arctic Ocean and Surrounding Seas. Estuaries Coasts 2012, 35, 369–382. [Google Scholar] [CrossRef]
- Siron, R.; Sherman, K.; Skjoldal, H.R.; Hiltz, E. Ecosystem-Based Management in the Arctic Ocean: A Multi-Level Spatial Approach. Arctic 2008, 61, 86–102. [Google Scholar] [CrossRef]
- Bring, A.; Fedorova, I.; Dibike, Y.; Hinzman, L.; Mård, J.; Mernild, S.H.; Prowse, T.; Semenova, O.; Stuefer, S.L.; Woo, M.K. Arctic terrestrial hydrology: A synthesis of processes, regional effects, and research challenges. J. Geophys. Res. Biogeosci. 2016, 121, 621–649. [Google Scholar] [CrossRef]
- Kaste, Ø.; Rankinen, K.; Lepistö, A. Modelling impacts of climate and deposition changes on nitrogen fluxes in northern catchments of Norway and Finland. Hydrol. Earth Syst. Sci. 2004, 8, 778–792. [Google Scholar] [CrossRef]
- Arctic Monitoring and Assessment Programme (AMAP). Snow, Water, Ice and Permafrost. Summary for Policy-Makers; Arctic Monitoring and Assessment Programme (AMAP): Oslo, Norway, 2017. [Google Scholar]
- Chen, M.; Huang, Y.P.; Guo, L.D.; Cai, P.H.; Yang, W.F.; Liu, G.S.; Qiu, Y.S. Biological productivity and carbon cycling in the Arctic Ocean. Chin. Sci. Bull. 2002, 47, 1037–1040. [Google Scholar] [CrossRef]
- Loeng, H.; Brander, K.; Carmack, E.; Denisenko, S.; Drinkwater, K.; Hansen, B.; Kovacs, K.; Livingston, P.; McLaughlin, F.; Sakshaug, E. Marine Systems. In Arctic Cimate Impact Assessment; Cambridge University Press: Cambridge, UK, 2005; pp. 452–538. ISBN 0521865093. [Google Scholar]
- Darnis, G.; Robert, D.; Pomerleau, C.; Link, H.; Archambault, P.; Nelson, R.J.; Geoffroy, M.; Tremblay, J.E.; Lovejoy, C.; Ferguson, S.H.; et al. Current state and trends in Canadian Arctic marine ecosystems: II. Heterotrophic food web, pelagic-benthic coupling, and biodiversity. Clim. Chang. 2012, 115, 179–205. [Google Scholar] [CrossRef]
- Chapman, P.M.; Riddle, M.J. Toxic Effects of Contaminants in Polar Marine Environments. Environ. Sci. Technol. 2005, 39, 200A–207A. [Google Scholar] [CrossRef] [PubMed]
- Murray, J.L. Ecological Characteristics of the Arctic. In AMAP Assessment Report: Arctic Pollution Issues; Arctic Monitoring and Assessment Programme (AMAP): Oslo, Norway, 1998; pp. 117–140. [Google Scholar]
- Bejarano, A.C.; Gardiner, W.W.; Barron, M.G.; Word, J.Q. Relative sensitivity of Arctic species to physically and chemically dispersed oil determined from three hydrocarbon measures of aquatic toxicity. Mar. Pollut. Bull. 2017, 122, 316–322. [Google Scholar] [CrossRef] [PubMed]
- Chapman, P.M.; McDonald, B.G.; Kickham, P.E.; McKinnon, S. Global geographic differences in marine metals toxicity. Mar. Pollut. Bull. 2006, 52, 1081–1084. [Google Scholar] [CrossRef] [PubMed]
- Marcus Zamora, L.; King, C.K.; Payne, S.J.; Virtue, P. Sensitivity and response time of three common Antarctic marine copepods to metal exposure. Chemosphere 2015, 120, 267–272. [Google Scholar] [CrossRef] [PubMed]
- McMeans, B.C.; McCann, K.S.; Humphries, M.; Rooney, N.; Fisk, A.T. Food Web Structure in Temporally-Forced Ecosystems. Trends Ecol. Evol. 2015, 30, 662–672. [Google Scholar] [CrossRef] [PubMed]
- Wirta, H.K.; Vesterinen, E.J.; Hambäck, P.A.; Weingartner, E.; Rasmussen, C.; Reneerkens, J.; Schmidt, N.M.; Gilg, O.; Roslin, T. Exposing the structure of an Arctic food web. Ecol. Evol. 2015, 5, 3842–3856. [Google Scholar] [CrossRef] [PubMed]
- Heleniak, T.; Bogoyavlenskiy, D. Arctic Populations and Migration. In Arctic Human Development Report: Regional Processes and Global Linkages; Larsen, J.N., Fondahl, G., Eds.; Nordic Council of Ministers: Copenhagen, Denmark, 2013; pp. 53–104. [Google Scholar]
- Petrov, A.N.; BurnSilver, S.; Chapin, F.S.; Fondahl, G.; Graybill, J.; Keil, K.; Nilsson, A.E.; Riedlsperger, R.; Schweitzer, P.; Chapin, F.S., III; et al. Arctic sustainability research: Toward a new agenda. Polar Geogr. 2016, 39, 165–178. [Google Scholar] [CrossRef]
- Rasmussen, R.O.; Hovelsrud, G.K.; Gearheard, S. Community viability and adaptation. In Arctic Human Development Report: Regional Processes and Global Linkages; Larsen, J.N., Fondahl, G., Eds.; Nordic Council of Ministers: Copenhagen, Denmark, 2015. [Google Scholar]
- ArcticStat. Available online: http://www.arcticstat.org/ (accessed on 6 July 2017).
- Huntington, H.P. Peoples of the Arctic: Characteristics of Human Populations Relevant to Pollution Issues. In AMAP Assessment Report: Arctic Pollution Issues; Arctic Monitoring and Assessment Programme (AMAP): Oslo, Norway, 1998; pp. 141–183. [Google Scholar]
- Bjerregaard, P.; Aidt, E.C. Levevilkår, Livsstil og Helbred. Befolkningsundersøgelser i Grønland 2005–2009; Statens Institut for Folkesundhet: Copenhagen, Denmark, 2010; ISBN 9788778991676. [Google Scholar]
- Bjerregaard, P.; Curtis, T.; Senderovitz, F.; Christensen, U.; Pars, T. Levevilkår, Livsstil og Helbred i Grønland; Dansk Institut for Klinisk Epidemiologi (DIKE): Copenhagen, Denmark, 1995; ISBN 8789662741. [Google Scholar]
- Nobmann, E.D.; Byers, T.; Lanier, A.P.; Hankin, J.H.; Jackson, M.Y. The diet of Alaska Native adults: 1987–1988. Am. J. Clin. Nutr. 1992, 55, 1024–1032. [Google Scholar] [PubMed]
- Huijbregts, M.A.J.; Steinmann, Z.J.N.; Elshout, P.M.F.; Stam, G.; Verones, F.; Vieira, M.; Zijp, M.; Hollander, A.; van Zelm, R. ReCiPe2016: A harmonised life cycle impact assessment method at midpoint and endpoint level. Int. J. Life Cycle Assess. 2017, 22, 138–147. [Google Scholar] [CrossRef]
- Jolliet, O.; Margni, M.; Charles, R.; Humbert, S.; Payet, J.; Rebitzer, G.; Robenbaum, R.K. IMPACT 2002+: A New Life Cycle Impact Assessment Methodology. Int. J. Life Cycle Assess. 2003, 8, 324–330. [Google Scholar] [CrossRef]
- IMPACT World+. Available online: http://www.impactworldplus.org/en/ (accessed on 10 June 2017).
- LC-Impact: A Spatially Differentiated Life Cycle Impact Assessment Method. Available online: http://www.lc-impact.eu/ (accessed on 10 May 2017).
- Itsubo, N.; Sakagami, M.; Washida, T.; Kokubu, K.; Inaba, A. Weighting across safeguard subjects for LCIA through the application of conjoint analysis. Int. J. Life Cycle Assess. 2004, 9, 196–205. [Google Scholar] [CrossRef]
- Bare, J. TRACI 2.0: The tool for the reduction and assessment of chemical and other environmental impacts 2.0. Clean Technol. Environ. Policy 2011, 13, 687–696. [Google Scholar] [CrossRef]
- Cherubini, F.; Fuglestvedt, J.; Gasser, T.; Reisinger, A.; Cavalett, O.; Huijbregts, M.A.J.; Johansson, D.J.A.; Jørgensen, S.V.; Raugei, M.; Schivley, G.; et al. Bridging the gap between impact assessment methods and climate science. Environ. Sci. Policy 2016, 64, 129–140. [Google Scholar] [CrossRef]
- Levasseur, A. Climate Change. In Life Cycle Impact Assessment; Hauschild, M.Z., Huijbregts, M.A.J., Eds.; Springer: Dordrecht, The Netherlands, 2015; pp. 257–259. [Google Scholar]
- Addressing Climate Change in the Near Term: Short-Lived Climate Pollutants. Available online: http://www.c2es.org/science-impacts/short-lived-climate-pollutants (accessed on 26 July 2017).
- Myhre, G.; Shindell, D.; Bréon, F.M.; Collins, W.; Fuglestvedt, J.; Huang, J.; Koch, D.; Lamarque, J.F.; Lee, D.; Mendoza, B.; et al. Anthropogenic and Natural Radiative Forcing. In Climate Change 2013—The Physical Science Basis; Cambridge University Press: Cambridge, UK, 2013; pp. 659–740. [Google Scholar]
- Ødemark, K.; Dalsøren, S.B.; Samset, B.H.; Berntsen, T.K.; Fuglestvedt, J.S.; Myhre, G. Short-lived climate forcers from current shipping and petroleum activities in the Arctic. Atmos. Chem. Phys. 2012, 12, 1979–1993. [Google Scholar] [CrossRef] [Green Version]
- Quinn, P.K.; Stohl, A.; Arneth, A.; Berntsen, T.; Burkhart, J.F.; Christensen, J.; Flanner, M.; Kupiainen, K.; Lihavainen, H.; Shepherd, M.; et al. The Impact of Black Carbon on Arctic Climate; Arctic Monitoring and Assessment Programme (AMAP): Oslo, Norway, 2011. [Google Scholar]
- Flanner, M.G.; Zender, C.S.; Randerson, J.T.; Rasch, P.J. Present-day climate forcing and response from black carbon in snow. J. Geophys. Res. Atmos. 2007, 112. [Google Scholar] [CrossRef]
- Fiore, A.M.; Jacob, D.J.; Bey, I.; Yantosca, R.M.; Field, B.D.; Fusco, A.C.; Wilkinson, J.G. Background ozone over the United States in summer: Origin, trend, and contribution to pollution episodes. J. Geophys. Res. Atmos. 2002, 107. [Google Scholar] [CrossRef]
- Preiss, P. Photochemical Ozone Formation. In Life Cycle Impact Assessment; Hauschild, M.Z., Huijbregts, M.A.J., Eds.; Springer: Dordrecht, The Netherlands, 2015; pp. 115–138. [Google Scholar]
- Schaap, M.; Timmermans, R.M.A.; Roemer, M.; Boersen, G.A.C.; Builtjes, P.J.H.; Sauter, F.J.; Velders, G.J.M.; Beck, J.P. The LOTOS-EUROS model: Description, validation and latest developments. Int. J. Environ. Pollut. 2008, 32, 270–290. [Google Scholar] [CrossRef]
- Van Zelm, R.; Preiss, P.; van Goethem, T.; van Dingenen, R.; Huijbregts, M. Regionalized life cycle impact assessment of air pollution on the global scale: Damage to human health and vegetation. Atmos. Environ. 2016, 134, 129–137. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization. Health Risks of Particulate Matter from Long-Range Transboundary Air Pollution; World Health Organization: Copenhagen, Denmark, 2006. [Google Scholar]
- Pijnappel, A. Statistical Post Processing of Model Output from the Air Quality Model LOTOS-EUROS; The Royal Netherlands Meteorological Institute (KNMI): De Bilt, The Netherlands, 2011. [Google Scholar]
- Humbert, S.; Fantke, P.; Jolliet, O. Particulate Matter Formation. In Life Cycle Impact Assessment; Hauschild, M.Z., Huijbregts, M.A.J., Eds.; Springer: Dordrecht, The Netherlands, 2015; pp. 97–113. [Google Scholar]
- Van Zelm, R.; Huijbregts, M.A.J.; den Hollander, H.A.; van Jaarsveld, H.A.; Sauter, F.J.; Struijs, J.; van Wijnen, H.J.; van de Meent, D. European characterization factors for human health damage of PM10 and ozone in life cycle impact assessment. Atmos. Environ. 2008, 42, 441–453. [Google Scholar] [CrossRef]
- Goedkoop, M.; Heijungs, R.; De Schryver, A.; Struijs, J.; van Zelm, R. ReCiPe 2008: A Life Cycle Impact Assessment Method Which Comprises Harmonised Category Indicators at the Midpoint and the Endpoint Level. Report I: Characterisation; Ministry of Housing, Spatial Planning and Environment: The Hague, The Netherlands, 2013. [Google Scholar]
- Humbert, S.; Marshall, J.D.; Shaked, S.; Spadaro, J.V.; Nishioka, Y.; Preiss, P.; McKone, T.E.; Horvath, A.; Jolliet, O. Intake fraction for particulate matter: Recommendations for life cycle impact assessment. Environ. Sci. Technol. 2011, 45, 4808–4816. [Google Scholar] [CrossRef] [PubMed]
- Van Zelm, R. Damage Modeling in Life Cycle Impact Assessment; Radboud University: Nijmegen, The Netherlands, 2010. [Google Scholar]
- Van Zelm, R.; Huijbregts, M.A.J.; van Jaarsveld, H.A.; Reinds, G.J.; De Zwart, D.; Struijs, J.; Van De Meent, D. Time horizon dependent characterization factors for acidification in life-cycle assessment based on forest plant species occurrence in Europe. Environ. Sci. Technol. 2007, 41, 922–927. [Google Scholar] [CrossRef] [PubMed]
- Seppälä, J.; Posch, M.; Johansson, M.; Hettelingh, J.-P. Country-dependent Characterisation Factors for Acidification and Terrestrial Eutrophication Based on Accumulated Exceedance as an Impact Category Indicator. Int. J. Life Cycle Assess. 2006, 11, 403–416. [Google Scholar] [CrossRef]
- Forsius, M.; Posch, M.; Aherne, J.; Reinds, G.J.; Christensen, J.; Hole, L. Assessing the impacts of long-range sulfur and nitrogen deposition on arctic and sub-arctic ecosystems. Ambio 2010, 39, 136–147. [Google Scholar] [CrossRef] [PubMed]
- Huijbregts, M.A.J.; Schöpp, W.; Verkuijlen, E.; Heijungs, R.; Reijnders, L. Spatially excplicit characterisation of acidifying and eutrophying air pollution in life-cycle assessment. J. Ind. Ecol. 2001, 4, 75–92. [Google Scholar] [CrossRef]
- Roy, P.O.; Deschênes, L.; Margni, M. Life cycle impact assessment of terrestrial acidification: Modeling spatially explicit soil sensitivity at the global scale. Environ. Sci. Technol. 2012, 46, 8270–8278. [Google Scholar] [CrossRef] [PubMed]
- Wet Deposition. Available online: http://wiki.seas.harvard.edu/geos-chem/index.php/Wet_deposition (accessed on 3 July 2017).
- Azevedo, L.B.; Roy, P.O.; Verones, F.; van Zelm, R.; Huijbregts, M.A.J. Terrestrial acidification. In LC-Impact Version 0.5: A Spatially Differentiated Life Cycle Impact Assessment Approach; Verones, F., Hellweg, S., Azevedo, L.B., Chaudhary, A., Cosme, N., Fantke, P., Goedkoop, M., Hauschild, M., Laurent, A., Mutel, C.L., et al., Eds.; European LC-IMPACT Project Report: Trondheim, Norway, 2016; Available online: http://www.lc-impact.eu (accessed on 7 September 2017).
- Roy, P.O.; Deschênes, L.; Margni, M. Uncertainty and spatial variability in characterization factors for aquatic acidification at the global scale. Int. J. Life Cycle Assess. 2014, 19, 882–890. [Google Scholar] [CrossRef]
- Hauschild, M.; Potting, J. Spatial Differentiation in LCA Impact Assessment—The EDIP2003 Methodology; Danish Environmental Protection Agency: Copenhagen, Denmark, 2005. [Google Scholar]
- Beusen, A.H.W.; Klepper, O.; Meinardi, C.R. Modelling the flow of nitrogen and phosphorus in Europe: From loads to coastal seas. Water Sci. Technol. 1995, 31, 141–145. [Google Scholar]
- Verones, F.; Hellweg, S.; Azevedo, L.B.; Laurent, A.; Mutel, C.L.; Pfister, S. Freshwater eutrophication. In LC-Impact Version 0.5: A Spatially Differentiated Life Cycle Impact Assessment Approach; Verones, F., Hellweg, S., Azevedo, L.B., Chaudhary, A., Cosme, N., Fantke, P., Goedkoop, M., Hauschild, M., Laurent, A., Mutel, C.L., et al., Eds.; European LC-IMPACT Project Report: Trondheim, Norway, 2016; Available online: http://www.lc-impact.eu/ (accessed on 7 September 2017).
- Azevedo, L.B.; Henderson, A.D.; van Zelm, R.; Jolliet, O.; Huijbregts, M.A.J. Assessing the importance of spatial variability versus model choices in life cycle impact assessment: The case of freshwater eutrophication in Europe. Environ. Sci. Technol. 2013, 47, 13565–13570. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, N.; Huijbregts, M.A.J.; van de Meent, D.; Peijnenburg, W.J.G.M.; Guinee, J.; Diamond, M.L. Implications of geographic variability on Comparative Toxicity Potentials of Cu, Ni and Zn in freshwaters of Canadian ecoregions. Chemosphere 2011, 82, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Henderson, A.D.; Hauschild, M.Z.; van De Meent, D.; Huijbregts, M.A.J.; Larsen, H.F.; Margni, M.; McKone, T.E.; Payet, J.; Rosenbaum, R.K.; Jolliet, O. USEtox fate and ecotoxicity factors for comparative assessment of toxic emissions in life cycle analysis: Sensitivity to key chemical properties. Int. J. Life Cycle Assess. 2011, 16, 701–709. [Google Scholar] [CrossRef]
- Rosenbaum, R.K. Ecotoxicity. In Life Cycle Impact Assessment; Hauschild, Z.M., Huijbregts, A.J.M., Eds.; Springer: Dordrecht, The Netherlands, 2015; pp. 139–162. [Google Scholar]
- Stewart, D.M.; Holt, D.P.; Rouwette, M.R. Application of LCA in Mining and Minerals Processing-Current Programs and Noticeable Gaps. In Life Cycle Assessment Handbook; Curran, M.A., Ed.; John Wiley & Sons, Inc.: Somerset, NJ, USA, 2012; pp. 267–289. [Google Scholar]
- Hauschild, M.Z.; Goedkoop, M.; Guinée, J.; Heijungs, R.; Huijbregts, M.; Jolliet, O.; Margni, M.; De Schryver, A.; Humbert, S.; Laurent, A.; et al. Identifying best existing practice for characterization modeling in life cycle impact assessment. Int. J. Life Cycle Assess. 2013, 18, 683–697. [Google Scholar] [CrossRef]
- Mcfarlin, K.M.; Perkins, R.A.; Gardiner, W.W.; Word, J.D.; Word, J.Q. Toxicity of Physically and Chemically Dispersed Oil to Selected Arctic Species. Int. Oil Spill Conf. Proc. 2011, 2011, abs149. [Google Scholar] [CrossRef]
- Chapman, P.M. Toxicity delayed in cold freshwaters? J. Great Lakes Res. 2016, 42, 286–289. [Google Scholar] [CrossRef]
- Hjorth, M.; Nielsen, T.G. Oil exposure in a warmer Arctic: Potential impacts on key zooplankton species. Mar. Biol. 2011, 158, 1339–1347. [Google Scholar] [CrossRef]
- Payne, S.J.; King, C.K.; Zamora, L.M.; Virtue, P. Temporal changes in the sensitivity of coastal Antarctic zooplankton communities to diesel fuel: A comparison between single- and multi-species toxicity tests. Environ. Toxicol. Chem. 2014, 33, 882–890. [Google Scholar] [CrossRef] [PubMed]
- Nussir ASA. Nussir Mine and Ulveryggen Mine Preliminary Economic Assessment (PEA); Nussir ASA: Kvalsund, Norway, 2014. [Google Scholar]
- Song, X.; Pettersen, J.B.; Pedersen, K.B.; Røberg, S. Comparative life cycle assessment of tailings management and energy scenarios for a copper ore mine: A case study in Northern Norway. J. Clean. Prod. 2017, 164, 892–904. [Google Scholar] [CrossRef]
- Shindell, D.; Faluvegi, G. Climate response to regional radiative forcing during the twentieth century. Nat. Geosci. 2009, 2, 294–300. [Google Scholar] [CrossRef]
- Svendsen, H. Physical oceanography of coupled fjord-coast systems in northern Norawy with special focus on frontal dynamics and tides. In Ecology of Fjords and Coastal Waters; Elsevier: New York, NY, USA, 1995; pp. 149–164. [Google Scholar]
- Roy, P.O.; Huijbregts, M.; Deschênes, L.; Margni, M. Spatially-differentiated atmospheric source-receptor relationships for nitrogen oxides, sulfur oxides and ammonia emissions at the global scale for life cycle impact assessment. Atmos. Environ. 2012, 62, 74–81. [Google Scholar] [CrossRef]
- Gandhi, N.; Diamond, M.L.; Huijbregts, M.A.J.; Guinée, J.B.; Peijnenburg, W.J.G.M.; van De Meent, D. Implications of considering metal bioavailability in estimates of freshwater ecotoxicity: Examination of two case studies. Int. J. Life Cycle Assess. 2011, 16, 774–787. [Google Scholar] [CrossRef]


| Arctic Characteristics | Gaps in Causality Chains (Impact Pathways) | Arctic-Dependent Impacts |
|---|---|---|
| Seasonality | Snow melt and spring run-off | ACID, EUTR, metal toxicity |
| Short growth season | ACID, EUTR, ECOTOX | |
| Seasonal lipid storage | ACID, EUTR, ECOTOX, HUMTOX | |
| Photochemistry (sun-hours and UV) | Photochemical oxidant formation | |
| Migratory behavior | Ecosystem damage, toxicity | |
| Seasonal food webs | Ecosystem damage, toxicity | |
| Cold climate | BC and PM on snow (albedo) | Climate change |
| Bio-chemical process rates | ACID, EUTR, ECOTOX | |
| Biological productivity | ECOTOX | |
| Species richness Ecosystem sensitivity | Ecosystem damage Ecosystem damage | |
| Fog and Arctic haze | Photochemistry (sun-hours and UV) | Photochemical oxidant formation |
| Precipitation | Precipitation rate, snow vs. rain | ACID, EUTR, toxicity |
| Population density | Human intake fraction | Human toxicity |
| Diets | Human intake fraction | Human toxicity |
| Marine dependence | Marine-freshwater interaction and near-shore/coastal fate processes | ACID, EUTR, ECOTOX, HUMTOX |
| Dynamics | Climate change | Climate change, ACID, EUTR, ECOTOX, HUMTOX |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pettersen, J.B.; Song, X. Life Cycle Impact Assessment in the Arctic: Challenges and Research Needs. Sustainability 2017, 9, 1605. https://doi.org/10.3390/su9091605
Pettersen JB, Song X. Life Cycle Impact Assessment in the Arctic: Challenges and Research Needs. Sustainability. 2017; 9(9):1605. https://doi.org/10.3390/su9091605
Chicago/Turabian StylePettersen, Johan Berg, and Xingqiang Song. 2017. "Life Cycle Impact Assessment in the Arctic: Challenges and Research Needs" Sustainability 9, no. 9: 1605. https://doi.org/10.3390/su9091605