Use of Novel Composite Coagulants for Arsenic Removal from Waters—Experimental Insight for the Application of Polyferric Sulfate (PFS)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis of Coagulants
2.2. Jar-Tests
2.3. Arsenic Speciation
2.4. Arsenic, Aluminum, and Iron Determination
3. Results and Discussion
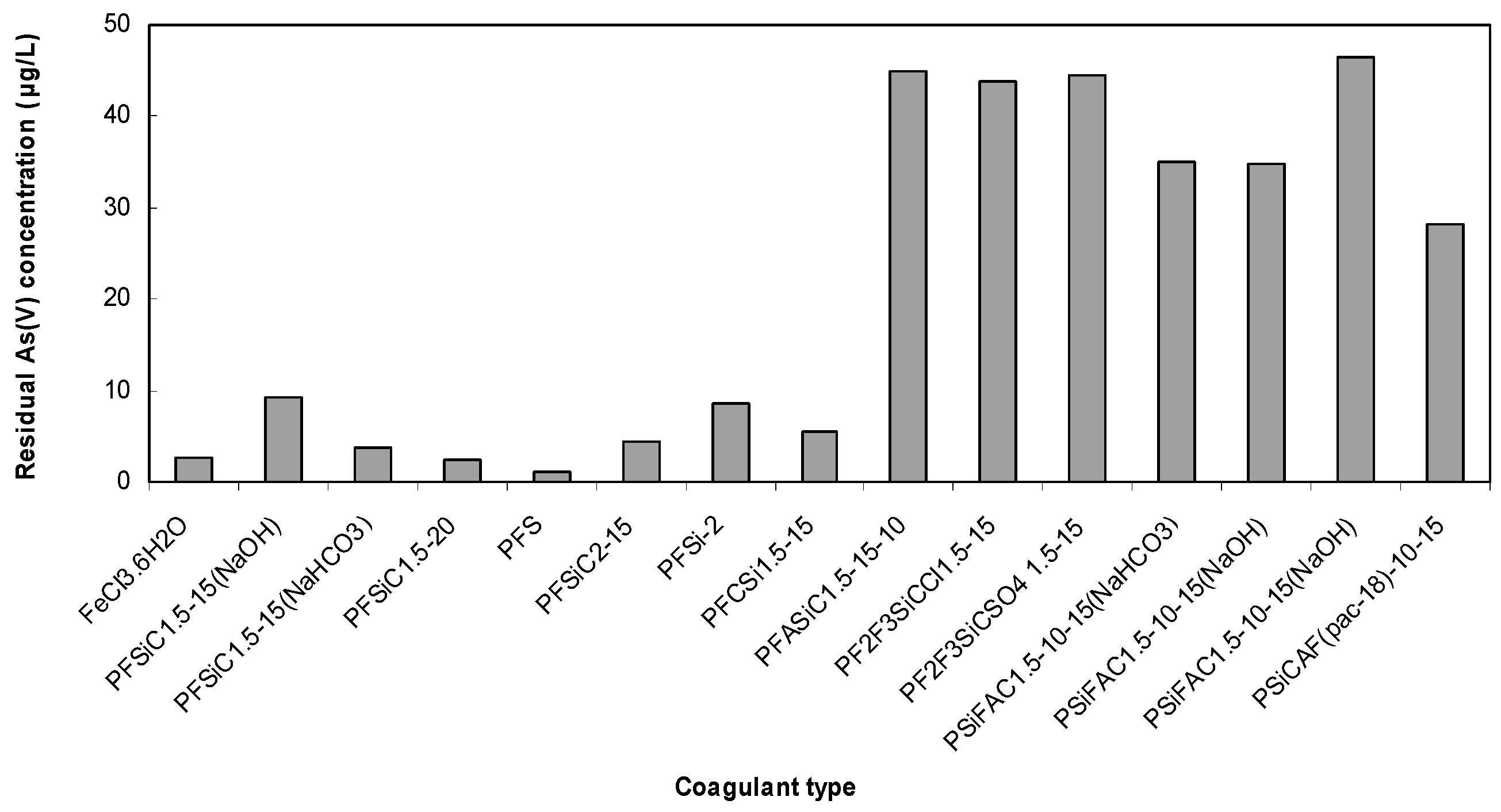
3.1. As(V) Removal by Pre-Polymerized Coagulants of Iron and Aluminum
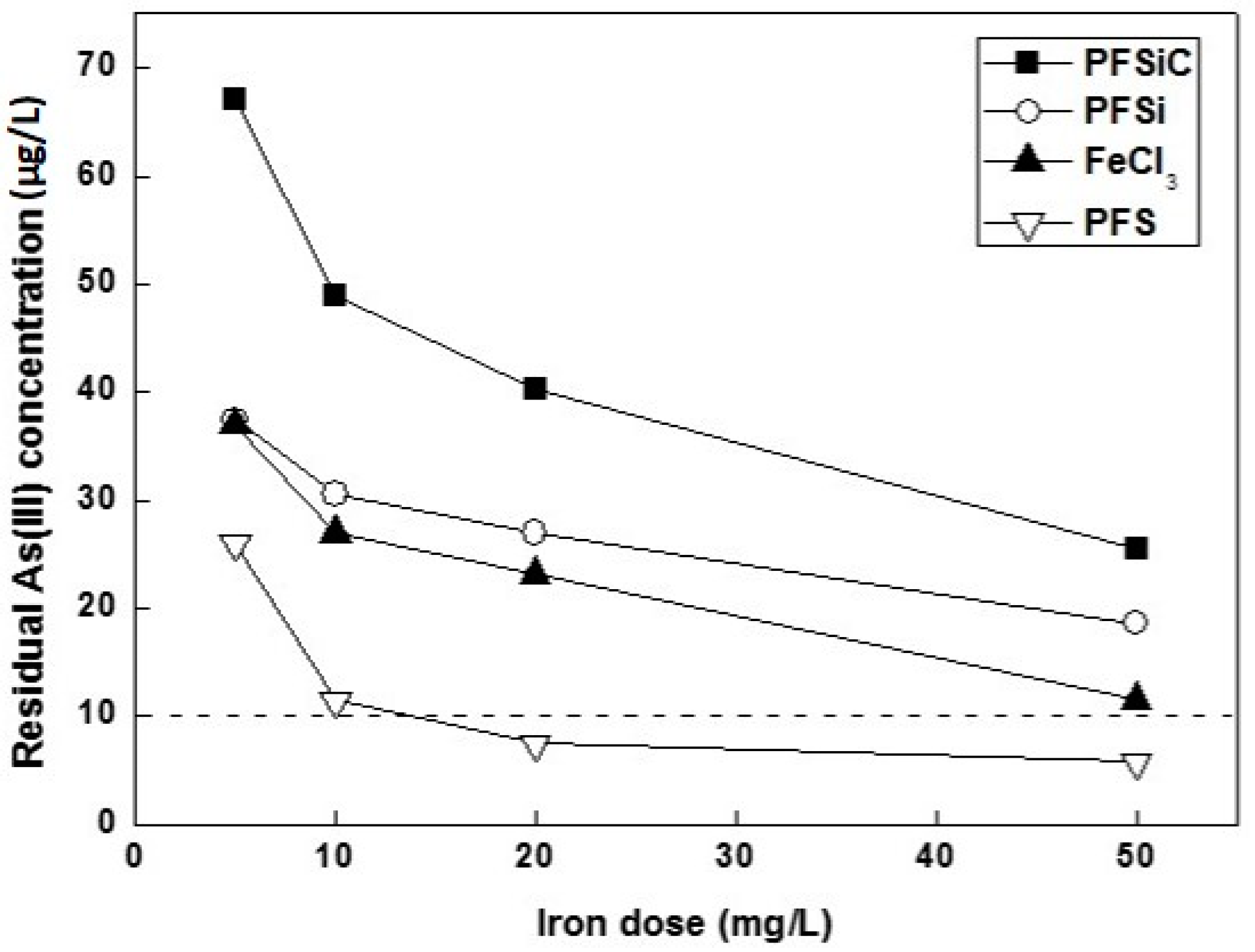
3.2. As(III) Removal by Use of Pre-Polymerized Coagulants of Iron
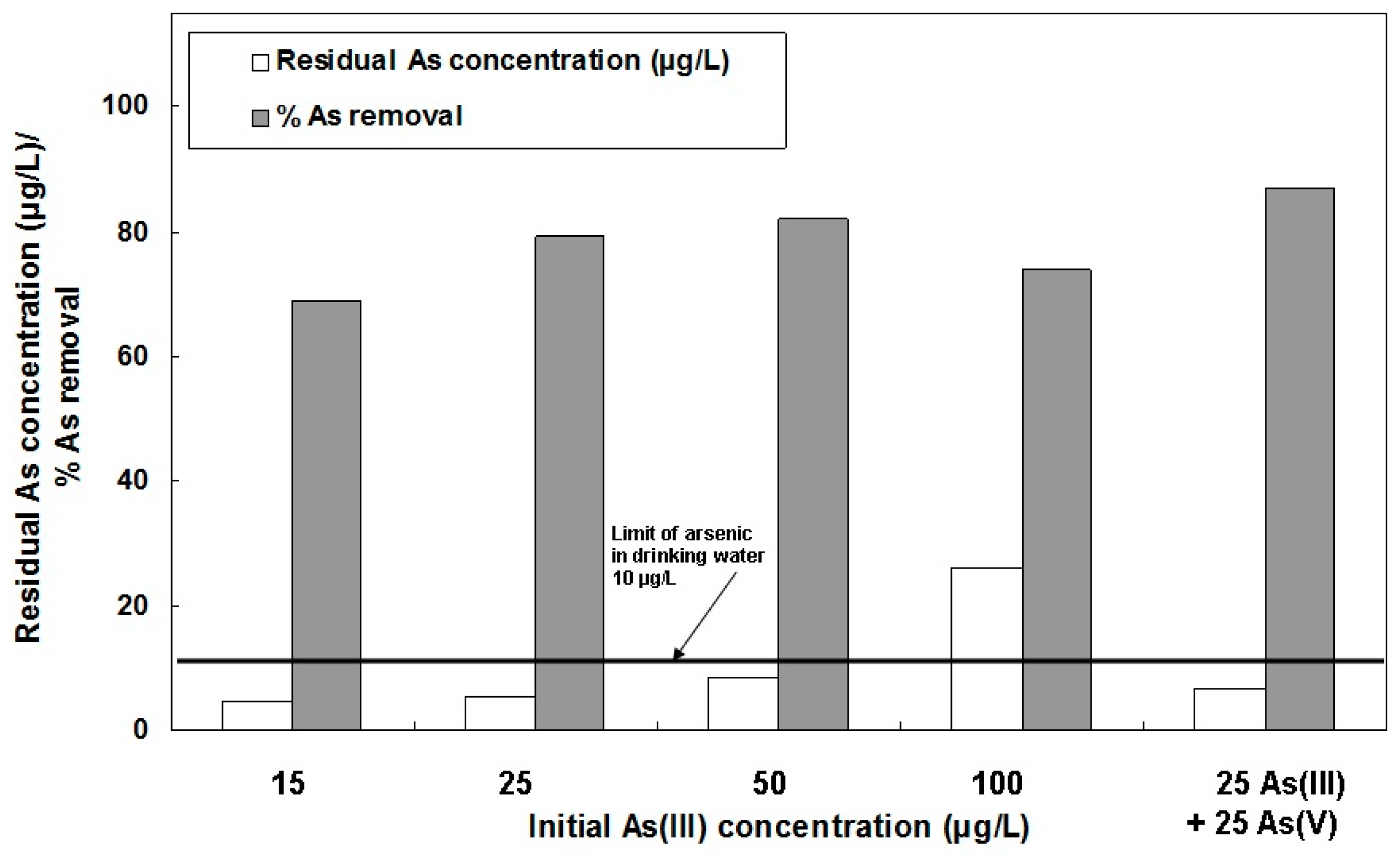
3.3. As(III) and As(V) Removal by PFS
3.4. Effect of Carbonate Concentration on As(III) Removal by the Use of PFS
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mitrakas, M. A survey of arsenic levels in tap, underground, and thermal mineral waters of Greece. Fresenius Environ. Bull. 2001, 10, 717–721. [Google Scholar]
- Katsoyiannis, I.A.; Mitrakas, M.; Zouboulis, A.I. Arsenic occurrence in Europe: Emphasis in Greece and description of the applied full-scale treatment plants. Desalin. Water Treat. 2015, 54, 2100–2107. [Google Scholar] [CrossRef]
- Sorlini, S.; Gialdini, F.; Collivignarelli, M.C. Survey on full-scale drinking water treatment plants for arsenic removal in Italy. Water Pract. Technol. 2014, 9, 42–51. [Google Scholar] [CrossRef]
- Hering, J.G.; Katsoyiannis, I.A.; Ahumada Theoduloz, G.; Berg, M.; Hug, S.J. Arsenic removal from drinking water: Experiences with technologies and constraints in practice. J. Environ. Eng. 2017. [Google Scholar] [CrossRef]
- Katsoyiannis, I.A.; Hug, S.J.; Ammann, A.; Zikoudi, A.; Hatziliontos, Ch. Arsenic speciation and uranium concentrations in drinking water supply wells in Northern Greece: Correlations with redox indicative parameters and implications for groundwater treatment. Sci. Total Environ. 2007, 383, 128–140. [Google Scholar] [CrossRef] [PubMed]
- Cullen, W.R.; Reimer, K.J. Arsenic speciation in the environment. Chem. Rev. 1989, 89, 713–764. [Google Scholar] [CrossRef]
- Tresintsi, S.; Simeonidis, K.; Estradé, S.; Martinez-Boubeta, C.; Vourlias, G.; Pinakidou, F.; Katsikini, M.; Paloura, E.C.; Stavropoulos, G.; Mitrakas, M. Tetravalent manganese feroxyhyte: A novel nanoadsorbent equally selective for As(III) and As(V) removal from drinking water. Environ. Sci. Technol. 2013, 47, 9699–9705. [Google Scholar] [CrossRef] [PubMed]
- Tresintsi, S.; Simeonidis, K.; Zouboulis, A.; Mitrakas, M. Comparative study of As(V) removal by ferric coagulation and oxy-hydroxides adsorption: Laboratory and full scale case studies. Desalin. Water Treat. 2013, 51, 2872–2880. [Google Scholar] [CrossRef]
- Katsoyiannis, I.A.; Zouboulis, A.I. Comparative evaluation of conventional and alternative methods for the removal of arsenic from contaminated groundwaters. Rev. Environ. Health 2006, 21, 1–25. [Google Scholar] [CrossRef]
- Katsoyiannis, I.A.; Ruettimann, T.; Hug, S.J. Response to comment on “pH dependence of Fenton reagent generation and As(III) oxidation and removal by corrosion of zero valent iron in aerated water”. Environ. Sci. Technol. 2009, 43, 3980–3981. [Google Scholar] [CrossRef]
- Ouzounis, K.; Katsoyiannis, I.A.; Zouboulis, A.; Mitrakas, M. Is the coagulation-filtration process with Fe(III) efficient for As(III) removal from groundwaters? Sep. Sci. Technol. 2015, 50, 1587–1592. [Google Scholar] [CrossRef]
- Sinha, S.; Yoon, Y.; Amy, G.; Yoon, J. Determining the effectiveness of conventional and alternative coagulants through effective characterization schemes. Chemosphere 2004, 57, 1115–1122. [Google Scholar] [CrossRef] [PubMed]
- Mertens, J.; Casentini, B.; Masion, A.; Pöthig, R.; Wehrli, B.; Furrer, G. Polyaluminum chloride with high Al30 content as removal agent for arsenic-contaminated well water. Water Res. 2012, 46, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Liu, Q.; Zhu, X. Studies on removal of As(III) by several types of ferrated salt in aqueous medium. In Proceedings of the World Congress on Engineering 2009 Volume I (WCE 2009), London, UK, 1–3 July 2009. [Google Scholar]
- Cui, J.; Jung, C.; Che, D.; Zhang, J.; Duan, S. Groundwater arsenic removal by coagulation using ferric(III) sulfate and polyferric sulfate: A comparative and mechanistic study. J. Environ. Sci. 2015, 32, 42–53. [Google Scholar] [CrossRef] [PubMed]
- Tolkou, A.K.; Zouboulis, A.I. Synthesis and coagulation performance of composite poly-aluminum-ferric-silicate-chloride coagulants in water and wastewater. Desalin. Water Treat. 2014, 53, 3309–3318. [Google Scholar] [CrossRef]
- Zouboulis, A.; Tzoupanos, N. Poly-aluminum silicate chloride—A systematic study for the preparation and application of an efficient coagulant for water or wastewater treatment. J. Hazard. Mater. 2009, 162, 1379–1389. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.-Q.; Graham, N.J.D. Preparation and characterisation of an optimal polyferric sulphate (PFS) as a coagulant for water treatment. J. Chem. Technol. Biotechnol. 1998, 73, 351–358. [Google Scholar] [CrossRef]
- Meng, X.; Wang, W. Speciation of arsenic by disposable cartridges. In Proceedings of the Third International Conference on Arsenic Exposure and Health Effects, San Diego, CA, USA, 12–15 July 1998. [Google Scholar]
- Clesceri, L.; Greenberg, A.; Trussel, R.L. Standard Methods for the Examination of Water and Wastewater, 17th ed.; APHA–AWWA–WEF: Washington, DC, USA, 1989. [Google Scholar]
- Mitrakas, Μ.; Panteliadis, P.; Keramidas, V.; Tzimou-Tsitouridou, R.; Sikalidis, C. Predicting Fe3+ dose for As(V) removal at pHs and temperatures commonly encountered in natural waters. Chem. Eng. J. 2009, 155, 716–721. [Google Scholar] [CrossRef]
- Hug, S.J.; Leupin, O.X. Iron-catalyzed oxidation of Arsenic(III) by oxygen and by hydrogen peroxide: pH-dependent formation of oxidants in the Fenton reaction. Environ. Sci. Technol. 2003, 37, 2734–2742. [Google Scholar] [CrossRef] [PubMed]
- Zouboulis, A.I.; Katsoyiannis, I.A. Removal of arsenates from contaminated water by coagulation-direct filtration. Sep. Sci. Technol. 2002, 37, 2859–2873. [Google Scholar] [CrossRef]
- Bhattacharya, P.; Claesson, M.; Bundschuh, J.; Sracek, O.; Fagerberg, J.; Jacks, G.; Martin, R.A.; Storniolo, A.; Thir, J.M. Distribution and mobility of arsenic in the Rıo Dulce alluvial aquifers in Santiago del Estero Province, Argentina. Sci. Total Environ. 2006, 358, 97–120. [Google Scholar] [CrossRef] [PubMed]
- Rowland, H.A.L.; Omoregie, E.O.; Millot, R.; Jimenez, C.; Mertens, J.; Baciu, C.; Hug, S.J.; Berg, M. Geochemistry and arsenic behaviour in groundwater resources of the Pannonian Basin (Hungary and Romania). Appl. Geochem. 2011, 26, 1–17. [Google Scholar] [CrossRef]
- Rasool, A.; Xiao, T.; Farooqi, A.; Shafeeque, M.; Liu, Y.; Kamran, M.A.; Katsoyiannis, I.A.; Eqani, S.A.M.A.S. Quality of tube well water intended for irrigation and human consumption with special emphasis on arsenic contamination at the area of Punjab, Pakistan. Environ. Geochem. Health 2016. [Google Scholar] [CrossRef] [PubMed]

| Coagulant Type | Molar Ratios | Description | Procedure |
|---|---|---|---|
| PFS0.3 | [OH]/[Fe] = 0.3 | Fe2(SO4)3 + H2SO4 + H2O2 + NaHCO3 | Oxidation of ferrous sulfate to ferric sulfate in highly acidic conditions (H2SO4, 96 wt %). The oxidizing agent was H2O2 (30%) and then, the alkaline solution of NaHCO3 was slowly added up to pH 1.3 |
| PFSiC1.5-15 | [OH]/[Fe] = 1.5 [Fe]/[Si] = 15 | FeCl3 + pSi + NaOH | Appropriate amount of pSi solution was added in FeCl3 solution under vigorous stirring and then, the alkaline solution of NaOH was slowly added up to pH 1.4 |
| PSiFAC1.5-10-15 | [OH]/[Al] = 1.5 [Al]/[Fe] = 10 [Al + Fe]/[Si] = 15 | (AlCl3 + FeCl3) + pSi + NaOH | Appropriate amount of FeCl3 solution was added in AlCl3 solution under vigorous stirring. Then, the pSi solution was added to the resulted Fe/Al solution at desired ratios, followed by the alkaline solution of (NaOH) addition up to pH 3.5 |
| Sample | Rapid Mixing Period | Slow Mixing Period | Sedimentation |
|---|---|---|---|
| Artificial water | 2 min at 160 rpm | 10 min at 45 rpm | 45 min without mixing |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Katsoyiannis, I.A.; Tzollas, N.M.; Tolkou, A.K.; Mitrakas, M.; Ernst, M.; Zouboulis, A.I. Use of Novel Composite Coagulants for Arsenic Removal from Waters—Experimental Insight for the Application of Polyferric Sulfate (PFS). Sustainability 2017, 9, 590. https://doi.org/10.3390/su9040590
Katsoyiannis IA, Tzollas NM, Tolkou AK, Mitrakas M, Ernst M, Zouboulis AI. Use of Novel Composite Coagulants for Arsenic Removal from Waters—Experimental Insight for the Application of Polyferric Sulfate (PFS). Sustainability. 2017; 9(4):590. https://doi.org/10.3390/su9040590
Chicago/Turabian StyleKatsoyiannis, Ioannis A., Nikolaos M. Tzollas, Athanasia K. Tolkou, Manassis Mitrakas, Mathias Ernst, and Anastasios I. Zouboulis. 2017. "Use of Novel Composite Coagulants for Arsenic Removal from Waters—Experimental Insight for the Application of Polyferric Sulfate (PFS)" Sustainability 9, no. 4: 590. https://doi.org/10.3390/su9040590











