Improving the Effectiveness of a Nutrient Removal System Composed of Microalgae and Daphnia by an Artificial Illumination
Abstract
:1. Introduction
2. Materials and Methods
2.1. Characteristics of Target Water
| Item * | Max | Min | Mean |
|---|---|---|---|
| Temp (°C) | 29.5 | 23.5 | 24.6 |
| pH (mg/L) | 6.92 | 6.10 | 6.38 |
| DO (mg/L) | 5.31 | 4.40 | 4.97 |
| TN (mg/L) | 8.88 | 8.13 | 8.37 |
| TP (mg/L) | 2.19 | 1.94 | 2.06 |
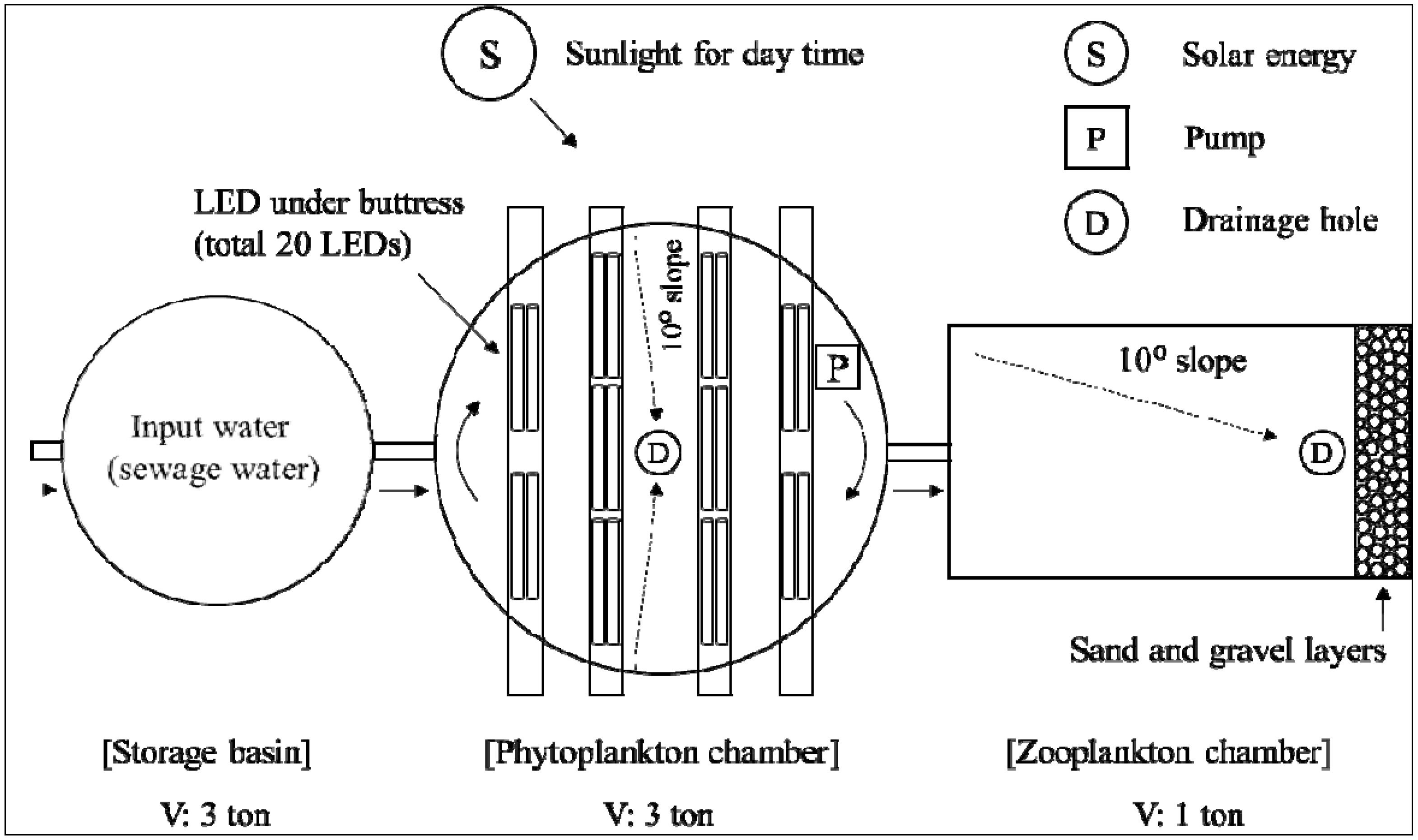
2.2. Facilities for AFW-LED System
2.3. Phytoplankton Culture Conditions
2.4. Zooplankton Culture Conditions
2.5. Continuous-Flow AFW-LED System
3. Results
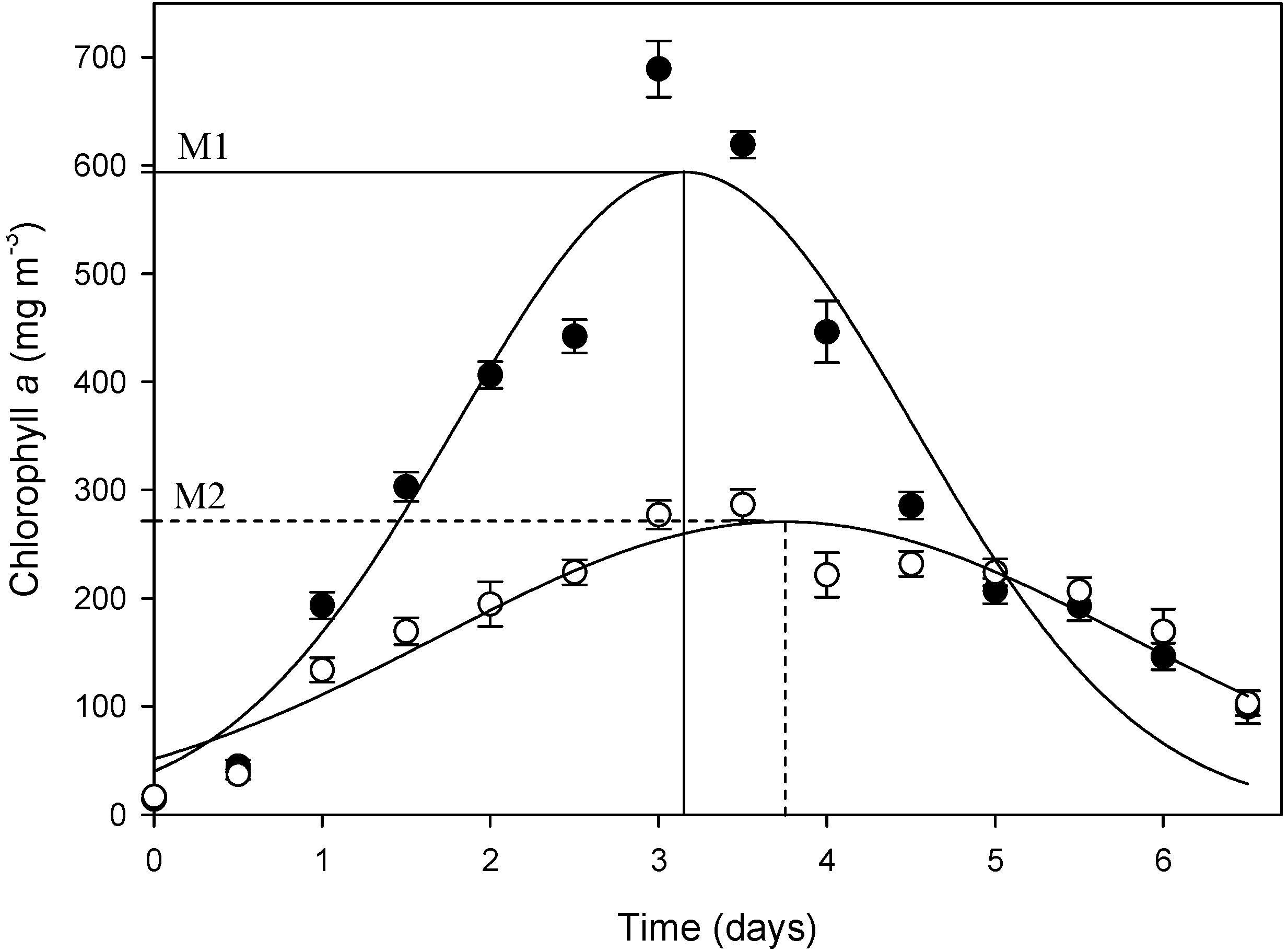
3.1. Growth of Phytoplankton
3.2. Growth of Zooplankton

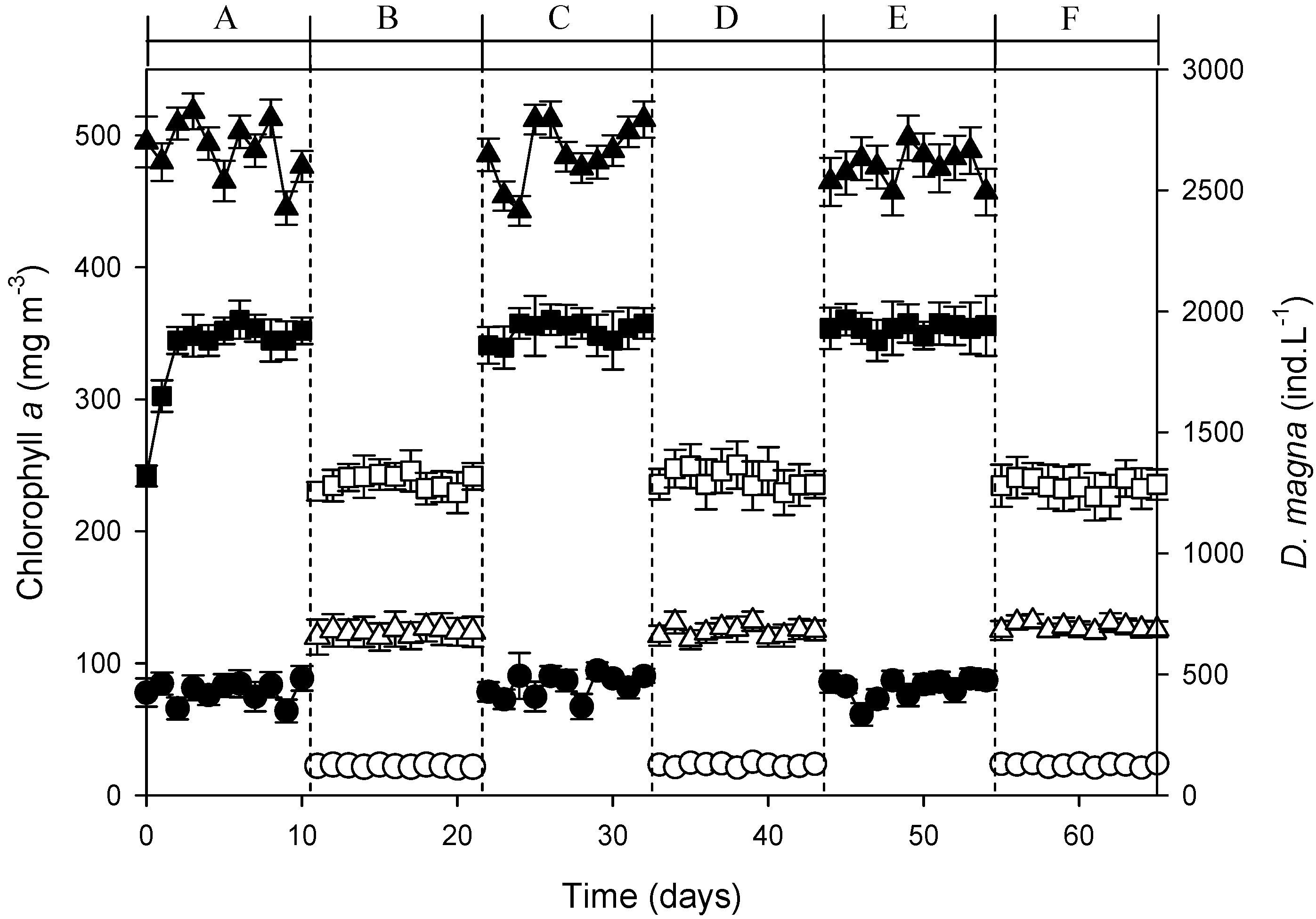
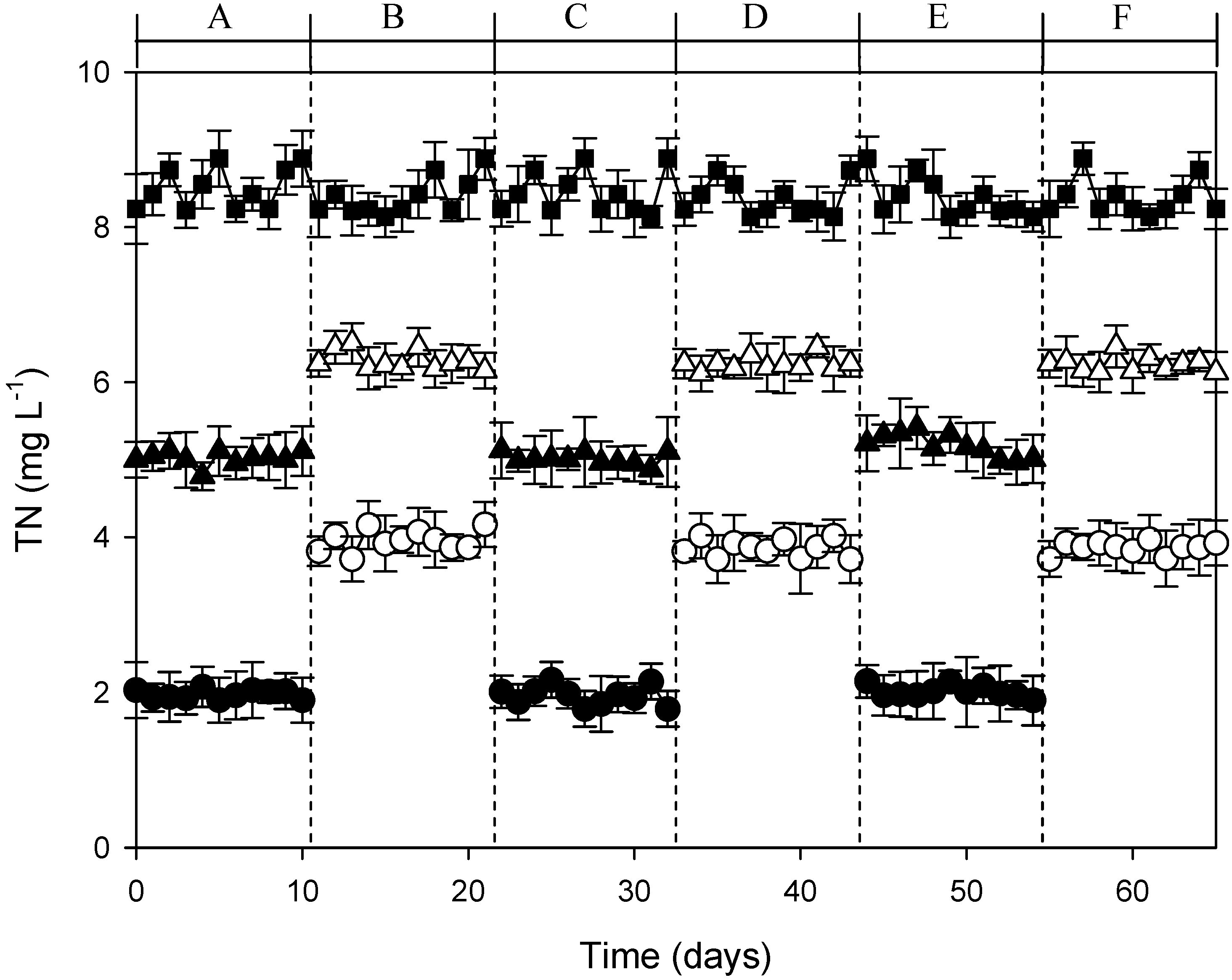
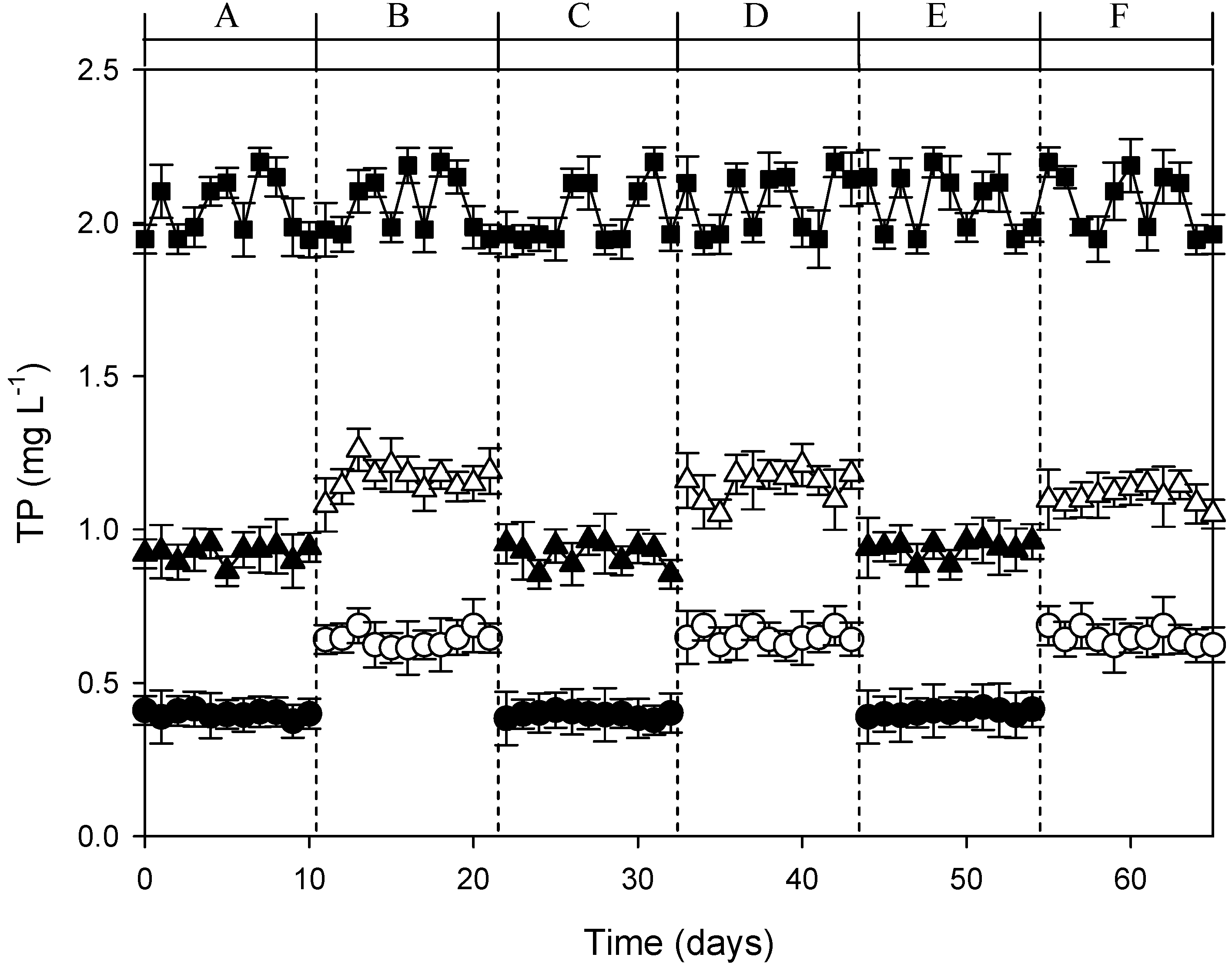
3.3. Continuous-Flow AFW-LED System
| LED-on/off | Item | Inflow | Removal in phytoplankton chamber | Removal in Daphnia magna chamber | Effluent | Total removal |
|---|---|---|---|---|---|---|
| LED-on | TN (g) | 84.7 (100%) | 34.3 (40%) | 30.9 (37%) | 19.5 (23%) | 65.2 (77%) |
| TP (g) | 20.4 (100%) | 11.1 (54%) | 5.3 (26%) | 4.0 (20%) | 16.4 (80%) | |
| LED-off | TN (g) | 83.8 (100%) | 21.5 (26%) | 23.5 (28%) | 38.8 (46%) | 45.0 (54%) |
| TP (g) | 20.6 (100%) | 8.9 (43%) | 4.9 (24%) | 6.8 (33%) | 13.8 (67%) |
4. Discussion
5. Conclusion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tawfik, A.; El-Gohary, F.; Ohashi, A.; Harada, H. Optimization of the performance of an integrated anaerobic-aerobic system for domestic wastewater treatment. Wat. Sci. Tech. 2008, 58, 320–328. [Google Scholar]
- Wu, C.-Y.; Peng, Y.-Z.; Wan, C.-L.; Wang, S.-Y. Performance and microbial population variation in a plug-flow A2O process treating domestic wastewater with low C/N ratio. J. Chem. Technol. Biotechnol. 2011, 86, 461–467. [Google Scholar] [CrossRef]
- Amini, M.; Younesi, H.; Najafpour, G.; Zinatizadeh-Lorestani, A.A. Application of response surface methodology for simultaneous carbon and nitrogen (SND) removal from dairy wastewater in batch systems. Int. J. Environ. Stud. 2012, 69, 962–986. [Google Scholar] [CrossRef]
- Li, Y.; Zhu, G.; Ng, W.J.; Tan, S.K. A review on removing pharmaceutical contaminants from wastewater by constructed wetlands: Design, performance and mechanism. Sci. Total Environ. 2012, 468, 908–932. [Google Scholar]
- Azza, N.; Denny, P.; Koppel, J.V.D.; Kansiime, F. Floating mats: Their occurrence and influence on shoreline distribution of emergent vegetation. Freshwater Biol. 2006, 51, 1286–1297. [Google Scholar] [CrossRef]
- Mohedano, R.A.; Costa, R.H.R.; Tavares, F.A.; Belli Filho, P. High nutrient removal rate from swine wastes and protein biomass production by full-scale duckweed ponds. Bioresour. Technol. 2006, 112, 98–104. [Google Scholar]
- Jung, D.; Cho, A.; Zo, Y.-G.; Choi, S.-I.; Ahn, T.-S. Nutrient removal from polluted stream water by artificial aquatic food web system. Hydrobiologia 2009, 630, 149–159. [Google Scholar] [CrossRef]
- Kim, S.-R.; Woo, S.-S.; Cheong, E.-H.; Ahn, T.-S. Nutrient removal from sewage by an artificial food web system composed of phytoplankton and Daphnia magna. Ecol. Eng. 2003, 21, 249–258. [Google Scholar] [CrossRef]
- Tam, N.F.Y.; Wong, Y.S. Wastewater nutrient removal by Chlorella pyrenoidosa and Scenedesmus sp. Environ. Pollut. 1989, 58, 19–34. [Google Scholar] [CrossRef]
- Bula, R.J.; Murrow, R.C.; Tibbitts, T.W.; Barta, D.J. Light-emitting diodes as a radiation source for plants. HortScience 1991, 26, 203–205. [Google Scholar]
- Oh, S.; Park, D.-S.; Yang, H-S.; Yoon, Y.; Honjo, T. Bioremediation on the benthic layer in polluted inner bay by promotion of microphytobenthos growth using light emitting diode (LED). J. Korean Soc. Mar. Sci. Eng. 2007, 10, 93–101. [Google Scholar]
- Lunka, A.; Bayless, D. Effects of flashing light-emitting diodes on algal biomass productivity. J. Appl. Phycol. 2013, 25, 1679–1685. [Google Scholar] [CrossRef]
- American Public Health Association. Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association: Washington, DC, USA, 2001. [Google Scholar]
- Kay, R.A.; Barton, L.L. Microalgae as food and supplement. Crit. Rev. Food Sci. Nutr. 1991, 30, 555–573. [Google Scholar] [CrossRef]
- Patil, V.; Källqvist, T.; Olsen, E.; Vogt, G.; Gislerød, H. Fatty acid composition of 12 microalgae for possible use in aquaculture feed. Aquacult. Int. 2007, 15, 1–9. [Google Scholar] [CrossRef]
- Mata, T.M.; Martins, A.A.; Caetano, N.S. Microalgae for biodiesel production and other applications: A review. Renew. Sust. Energ. Rev. 2010, 14, 217–232. [Google Scholar] [CrossRef]
- Van der Spiegel, M.; Noordam, M.Y.; Van der Fels-Klerx, H.J. Safety of Novel Protein Sources (Insects, Microalgae, Seaweed, Duckweed, and Rapeseed) and Legislative Aspects for Their Application in Food and Feed Production. Compr. Rev. Food Sci. F. 2013, 12, 662–678. [Google Scholar] [CrossRef]
- Ramos Tercero, E.A.; Sforza, E.; Morandini, M.; Bertucco, A. Cultivation of Chlorella protothecoides with urban wastewater in continuous photobioreactor: Biomass productivity and nutrient removal. Appl. Biochem. Biotechnol. 2014, 172, 1470–1485. [Google Scholar] [CrossRef]
- Lee, K.; Lee, C-G. Effect of light/dark cycles on wastewater treatments by microalgae. Biosci. Biotechnol. Biochem. 2001, 6, 194–199. [Google Scholar]
- Kawasaki, L.Y.; Tarifeňo-Silva, E.; Yu, D.P.; Gordon, M.S.; Chapman, D.J. Aquacultural approaches to recycling of dissolved nutrients in secondarily treated domestic wastewaters-I Nutrient uptake and release by artificial food chains. Water Res. 1982, 16, 37–49. [Google Scholar] [CrossRef]
- Persson, J.; Brett, M.T.; Vrede, T.; Ravet, J.L. Food quantity and quality regulation of trophic transfer between primary producers and a keystone grazer (Daphnia) in pelagic freshwater food webs. Oikos 2007, 116, 1152–1163. [Google Scholar] [CrossRef]
- Ahlgren, G.; Lundstedt, L.; Brett, M.; Forsberg, C. Lipid composition and food quality of some freshwater phytoplankton for cladoceran zooplankters. J. Plankton Res. 1990, 12, 809–818. [Google Scholar] [CrossRef]
- DeMott, W. Implications of element deficits for zooplankton growth. Hydrobiologia 2003, 491, 177–184. [Google Scholar] [CrossRef]
- Jacobi, A.; Steinweg, C.; Sastre, R.R.; Posten, C. Advanced photobioreactor LED illumination system: Scale-down approach to study microalgal growth kinetics. Eng. Life Sci. 2012, 12, 621–630. [Google Scholar] [CrossRef]
- Tennessen, D.; Singsaas, E.; Sharkey, T. Light-emitting diodes as a light source for photosynthesis research. Photosynth. Res. 1994, 39, 85–92. [Google Scholar] [CrossRef]
- Jeong, H.; Lee, J.; Cha, M. Energy efficient growth control of microalgae using photobiological methods. Renew. Energ. 2013, 54, 161–165. [Google Scholar] [CrossRef]
- Gris, B.; Morosinotto, T.; Giacometti, G.; Bertucco, A.; Sforza, E. Cultivation of Scenedesmus obliquus in photobioreactors: Effects of light intensities and light-dark cycles on growth, productivity, and biochemical composition. Appl. Biochem. Biotechnol. 2013, 172, 1–13. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chang, I.-H.; Jung, D.; Ahn, T.S. Improving the Effectiveness of a Nutrient Removal System Composed of Microalgae and Daphnia by an Artificial Illumination. Sustainability 2014, 6, 1346-1358. https://doi.org/10.3390/su6031346
Chang I-H, Jung D, Ahn TS. Improving the Effectiveness of a Nutrient Removal System Composed of Microalgae and Daphnia by an Artificial Illumination. Sustainability. 2014; 6(3):1346-1358. https://doi.org/10.3390/su6031346
Chicago/Turabian StyleChang, In-Ho, Dawoon Jung, and Tae Seok Ahn. 2014. "Improving the Effectiveness of a Nutrient Removal System Composed of Microalgae and Daphnia by an Artificial Illumination" Sustainability 6, no. 3: 1346-1358. https://doi.org/10.3390/su6031346




