Agronomic Management under Organic Farming May Affect the Bioactive Compounds of Lentil (Lens culinaris L.) and Grass Pea (Lathyrus communis L.)?
Abstract
:1. Introduction
2. Experimental Section
2.1. Field Experiment
2.2. Proximate Analysis
| Species | Phenotype of seed coat/cotyledon | TKW (g) | Seed size |
|---|---|---|---|
| Lentils | brown/red | 20.76 ± 1.56 | small |
| green/red | 21.98 ± 0.48 | small | |
| green/yellow | 65.01 ± 5.61 | large | |
| light brown/red | 40.30 ± 4.97 | medium | |
| Grass pea | beige/yellow | 314.19 ± 26.52 | large |
| beige/yellow | 136.78 ± 8.82 | small |
2.3. Extraction of Phenolic Compounds
2.4. Determination of Total Phenolic Content
2.5. Determination of Hydrolyzed Tannins
2.6. Determination of Condensed Tannin Content
2.7. Determination of Total Flavonoid Content
2.8. Trolox Equivalent Antioxidant Capacity
2.9. Statistical Analysis
3. Results
3.1. Proximate Composition
| Lentil | Variety | Row | Year | Year × Variety | Yera × Row | Variety × Row | Year × Row × Variety | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| df | MS | df | MS | df | MS | df | MS | df | MS | df | MS | df | MS | |
| Protein content | 3 | 13.06 *** | 1 | 4.44 * | 1 | 25.40 *** | 3 | 6.81 *** | 1 | 0.48 n.s. | 3 | 0.55 n.s. | 3 | 0.83 n.s. |
| IDF | 3 | 22.25 *** | 1 | 0.01 n.s. | 1 | 14.19 ** | 3 | 4.29 * | 1 | 3.69 n.s. | 3 | 1.00 n.s. | 3 | 4.23 * |
| SDF | 3 | 1.42 n.s. | 1 | 2.04 n.s. | 1 | 6.84 * | 3 | 2.02 n.s. | 1 | 0.23 n.s. | 3 | 3.24 * | 3 | 2.15 n.s. |
| TPC | 3 | 7.28 *** | 1 | 45.01 *** | 1 | 0.53 * | 3 | 0.66 ** | 1 | 14.40 *** | 3 | 0.47 * | 3 | 1.13 *** |
| HTC | 3 | 6.84 *** | 1 | 38.03 *** | 1 | 4.76 *** | 3 | 0.59 ** | 1 | 7.92 *** | 3 | 0.55 ** | 3 | 1.07 *** |
| TFC | 3 | 4.25 *** | 1 | 19.04 *** | 1 | 3.58 *** | 3 | 0.20 n.s. | 1 | 7.16 *** | 3 | 0.12 n.s. | 3 | 0.52 n.s. |
| CTC | 3 | 0.82 *** | 1 | 6.11 *** | 1 | 5.07 *** | 3 | 0.07 * | 1 | 0.66 *** | 3 | 0.08 ** | 3 | 0.06 * |
| Grass pea | Variety | Row | Year | Year × Variety | Year × Row | Variety × Row | Year × Row × Variety | |||||||
| df | MS | df | MS | df | MS | df | MS | df | MS | df | MS | df | MS | |
| Protein content | 1 | 2.34 * | 1 | 0.52 n.s. | 1 | 6.34 *** | 1 | 0.25 n.s. | 1 | 0.33 n.s. | 1 | 0.07 n.s. | 1 | 0.48 n.s. |
| IDF | 1 | 16.89 ** | 1 | 30.05 *** | 1 | 2.53 n.s. | 1 | 4.33 n.s. | 1 | 20.61 ** | 1 | 4.13 n.s. | 1 | 10.12 * |
| SDF | 1 | 0.07 n.s. | 1 | 0.32 n.s. | 1 | 0.05 n.s. | 1 | 0.11 n.s. | 1 | 0.17 n.s. | 1 | 1.51 * | 1 | 0.04 n.s. |
| TPC | 1 | 0.66 *** | 1 | 0.02 n.s. | 1 | 1.18 *** | 1 | 0.00 n.s. | 1 | 0.02 n.s. | 1 | 0.07 n.s. | 1 | 0.21 ** |
| HTC | 1 | 0.25 ** | 1 | 0.01 n.s. | 1 | 0.01 n.s. | 1 | 0.01 n.s. | 1 | 0.16 * | 1 | 0.01 n.s. | 1 | 0.12 * |
| TFC | 1 | 0.05 n.s. | 1 | 0.07 * | 1 | 0.28 *** | 1 | 0.08 * | 1 | 0.00 n.s. | 1 | 0.00 n.s. | 1 | 0.00 n.s. |
| CTC | 1 | 0.00 n.s. | 1 | 0.02 * | 1 | 0.06 ** | 1 | 0.00 n.s. | 1 | 0.06 *** | 1 | 0.05 ** | 1 | 0.01 n.s. |
| Protein content | IDF | SDF | TDF | TPC | HTC | TFC | CTC | ||
|---|---|---|---|---|---|---|---|---|---|
| Species | % d.m. | % d.m. | % d.m. | % d.m. | mg CE·g−1 d.m. | mg CE·g−1 d.m. | mg CE·g−1 d.m. | mg LE·g−1 d.m. | |
| Lentil | small (green) | 22.63 a | 26.05 a | 5.06 a | 31.11 a | 4.23 b | 2.95 b | 1.83 bc | 2.06 b |
| small (brown) | 20.91 b | 25.33 a | 4.51 a | 29.85 ab | 4.26 b | 2.94 b | 1.59 d | 2.05 b | |
| large (green) | 22.73 a | 22.75 c | 4.25 a | 27.00 c | 4.29 b | 2.92 b | 2.04 b | 1.76 c | |
| medium (light brown) | 20.85 b | 23.86 b | 4.74 a | 28.59 b | 5.82 a | 4.45 a | 2.95 a | 2.40 a | |
| mean | 21.78 | 24.50 | 4.64 | 29.14 | 4.65 | 3.32 | 2.10 | 2.07 | |
| Grass pea | large (beige) | 22.74 b | 23.69 b | 6.10 a | 29.80 b | 1.56 b | 0.28 b | 0.30 a | 0.40 a |
| small (beige) | 23.37 a | 25.37 a | 5.99 a | 31.37 a | 1.90 a | 0.48 a | 0.39 a | 0.39 a | |
| mean | 23.06 | 24.53 | 6.05 | 30.59 | 1.73 | 0.38 | 0.35 | 0.40 |
| Protein content | IDF | SDF | TDF | TPC | HTC | TFC | CTC | ||
|---|---|---|---|---|---|---|---|---|---|
| Species | % d.m. | % d.m. | % d.m. | % d.m. | mg CE·g−1 d.m. | mg CE·g−1 d.m. | mg CE·g−1 d.m. | mg LE·g−1 d.m. | |
| Lentil | 3 row | 21.48 b | 24.48 a | 4.44 a | 28.92 a | 3.68 b | 2.42 b | 1.47 b | 1.71 b |
| 8 row | 22.09 a | 24.51 a | 4.85 a | 29.36 a | 5.62 a | 4.20 a | 2.73 a | 2.42 a | |
| mean | 21.78 | 24.49 | 4.64 | 29.14 | 4.65 | 3.31 | 2.10 | 2.06 | |
| Grass pea | 3 row | 23.20 a | 25.65 a | 5.93 a | 31.81 a | 1.70 a | 0.37 a | 0.29 b | 0.37 b |
| 8 row | 22.91 a | 23.41 b | 6.16 a | 29.35 b | 1.76 a | 0.39 a | 0.40 a | 0.42 a | |
| mean | 23.05 | 24.53 | 6.04 | 30.58 | 1.73 | 0.38 | 0.34 | 0.39 |
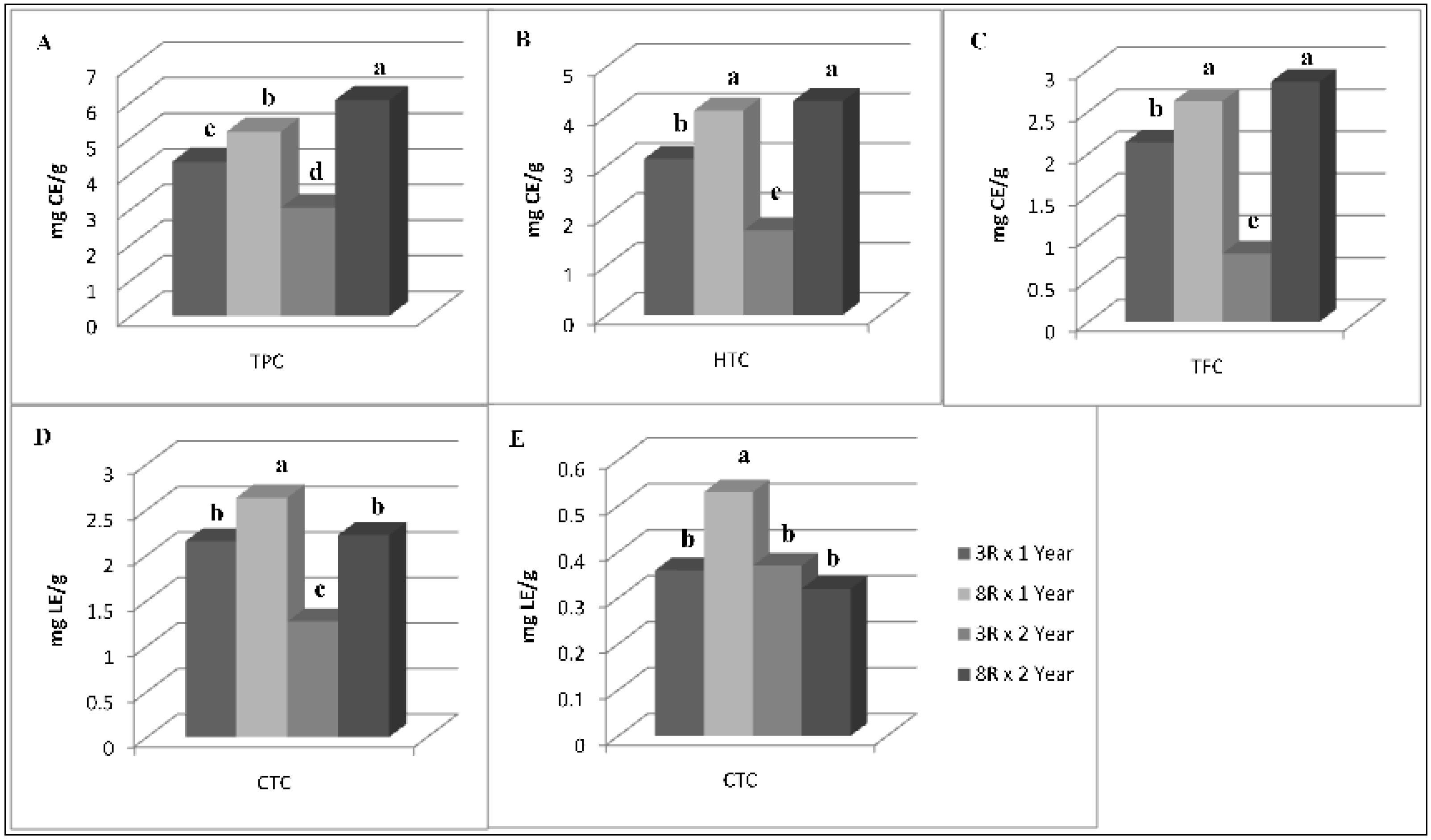
3.2. Antioxidant Compounds and Antioxidant Activity


| Total n = 72 | Correlation coefficient (r) | HTC | TFC | CTC | Antioxidant activity |
|---|---|---|---|---|---|
| TPC | 0.99 *** | 0.93 *** | 0.92 *** | 0.72 *** | |
| HTC | 0.94 *** | 0.95 *** | 0.70 *** | ||
| TFC | 0.98 *** | 0.67 *** | |||
| CTC | 0.59 *** | ||||
| Lentils n = 48 | |||||
| TPC | 0.98 *** | 0.88 *** | 0.79 *** | 0.45 ** | |
| HTC | 0.88 *** | 0.88 *** | 0.40 ** | ||
| TFC | 0.78 *** | 0.44 ** | |||
| CTC | 0.12 n.s. | ||||
| Grass pea n = 24 | |||||
| TPC | 0.71 *** | −0.07 n.s. | −0.12 n.s. | 0.69 *** | |
| HTC | 0.46 * | 0.35 n.s. | 0.15 n.s. | ||
| TFC | 0.53 ** | 0.23 n.s. | |||
| CTC | 0.47 * |
4. Discussion
4.1. Proximate Composition
4.2. Antioxidant Compounds and Antioxidant Activity
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Winter, C.K.; Davis, S.F. Organic foods. J. Food Sci. 2006, 71, R117–R124. [Google Scholar] [CrossRef]
- Worthington, W. Nutritional quality of organic versus conventional fruits, vegetables and grains. J. Altern. Complement. Med. 2001, 7, 161–173. [Google Scholar] [CrossRef]
- Del Amor, M.F.; Serrano-Martinez, A.; Fortea, I.; Nunez Delicado, E. Differential effect of organic cultivation on the levels of phenolics, peroxidase and capsidio in sweet peppers. J. Sci. Food Agric. 2008, 88, 770–777. [Google Scholar] [CrossRef]
- Brandt, K.; Mølgaard, J.P. Organic agriculture: Does it enhance or reduce the nutritional value of plant foods? J. Sci. Food Agric. 2001, 81, 924–931. [Google Scholar] [CrossRef]
- FiBL Excellence for Sustainability. Available online: http://www.fibl.org/ (accessed on 6 December 2013).
- Dykes, L.; Rooney, L.W.; Waniska, R.D.; Rooney, W.L. Phenolic compounds and antioxidant activity of sorghum grains of varying genotypes. J. Agric. Food Chem. 2005, 53, 6813–6818. [Google Scholar] [CrossRef]
- Vietmeyer, N.D. Lesser-known plants of potential use in agriculture and forestry. Science 1986, 232, 1379–1384. [Google Scholar]
- Xu, B.; Chang, S.K.C. Phenolic substance characterization and chemical and cell-based antioxidant activities of 11 lentils grown in the Northern United States. J. Agric. Food Chem. 2010, 58, 1509–1517. [Google Scholar] [CrossRef]
- Troszynska, A.; Bednarska, A.; Latosz, A.; Kozlowska, H. Polyphenolic compounds in the seed coat of legume seeds. Pol. Food Nutr. Sci. 1997, 6, 37–47. [Google Scholar]
- Xu, B.J.; Chang, S.K.C. Comparative analyses of phenolic composition, antioxidant capacity, and color of cool season legumes as affected by extraction solvents. J. Food Sci. 2007, 72, S159–S156. [Google Scholar] [CrossRef]
- Kenicer, G.J.; Kajita, T.; Pennington, R.T.; Murata, J. Systematics and biogeography of Lathyrus (Leguminosae) based on internal transcribed spacer and cpDNA sequence data. Am. J. Bot. 2005, 92, 1199–1209. [Google Scholar] [CrossRef]
- Abdel-Moneim, A.; van Dorrestein, B.; Baumc, M.; Mulugeta, W. Role of CARDA in Improving the Nutritional Qualityand Yield Potential of Grasspea for Subsistence Farmers in Developing Countries. In Proceedings of the CGIAR-Wide Conference on Agriculture-Nutrition, Los Banos, Philippines, 5–6 October 1999.
- Hanbury, C.D.; Siddique, K.H.M.; Galwey, N.W.; Cocks, P.S. Genotype environment interaction for seed yield and ODAP concentration of Lathyrus sativus L. and L. cicera L. in Mediterranean type environments. Euphytica 1999, 110, 45–60. [Google Scholar] [CrossRef]
- Sharm, R.N.; Chitale, M.W.; Ganvir, G.B.; Geda, A.K.; Pandey, R.L. Observations on the development of section criterion for high yield and low neurotoxin in grasspea based on genetic resources. Lathyrus Lathyrism Newsl. 2000, 1, 15–16. [Google Scholar]
- Lak, M.B.; Almassi, M. An analytical review of parameters and indices affecting decision making in agricultural mechanization. Aust. J. Agric. Eng. 2011, 2, 140–146. [Google Scholar]
- Tamburino, R.; Guida, V.; Pacifico, S.; Rocco, M.; Zarelli, A.; Parente, A.; di Maro, A. Nutritional values and radical scavenging capacities of grass pea (Lathyrus sativus L.) seeds in Valle Agricola district, Italy. Aust. J. Crop Sci. 2012, 6, 149–156. [Google Scholar]
- Yan, Z.Y.; Spencer, P.S.; Li, Z.X.; Liang, Y.M.; Wang, Y.F.; Wang, C.Y. Lathyrus sativus (grass pea) and its neurotoxin ODAP. Phytochemistry 2006, 67, 107–121. [Google Scholar] [CrossRef]
- Enneking, D. The nutritive value of grasspea (Lathyrus sativus) and allied species, their toxicity to animals and the role of malnutrition in neurolathyrism. Food Chem. Toxicol. 2011, 49, 694–709. [Google Scholar] [CrossRef]
- Shimelis, E.A.; Rakshit, S.K. Effect of processing on antinutrients and in vitro digestibility of kidney bean (Phaseolus vulgaris L) varieties grown in East Africa. Food Chem. 2007, 103, 161–172. [Google Scholar] [CrossRef]
- Soil Survey Staff. Soil Taxonomy; Agriculture Handbook 436; United States Department of Agriculture, Natural Resources Conservation Service (USDA-NRCS): Washington, DC, USA, 1999.
- Cunniff, P.; Association of Official Analytical Chemists. Official Methods of AOAC Analysis, 16th ed.; Gaithersburg, M.D., Ed.; Association of Official Analytical Chemists: Washington, DC, USA, 1997. [Google Scholar]
- Prosky, L.; Asp, N.G.; Schweizer, T.F.; de Vries, J.W.; Furda, I. Determination of insoluble and soluble dietary fiber in foods and food products: Collaborative study. J. Assoc. Offic. Anal. Chem. 1992, 75, 360–367. [Google Scholar]
- UNI, Ente Italiano di Unificazione. Method 10274 for protein content. Available online: http://www.uni.com/ (accessed on 6 December 2013).
- Beta, T.; Nam, S.; Dexter, J.E.; Sapirstein, H.D. Phenolic content and antioxidant activity of pearled wheat and roller-milled fractions. Cereal Chem. 2005, 82, 390–393. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Makkar, H.P.S.; Bluemmel, M.; Borowy, N.K.; Becker, R.K. Gravimetric determination of tannins and their correlat ions with chemical and protein precipitation methods. J. Sci. Food Agric. 1993, 61, 161–165. [Google Scholar] [CrossRef]
- Hagerman, A.E.; Harvey-Mueller, I.; Makker, H.P.S. Quantification of Tannins in tree Foliage—a Laboratory Manual; Food and Agriculture Organisation/International Atomic Energy Agency (FAO/IAEA): Vienna, Austria, 2000. [Google Scholar]
- Porter, L.J.; Hrstich, L.N.; Chan, B.G. The conversion of procyanidins and prodelphinidins to cyanidin and delphinidin. Phytochemistry 1986, 25, 223–230. [Google Scholar] [CrossRef]
- Eberhardt, M.V.; Lee, C.Y.; Liu, R.H. Antioxidant activity of fresh apples. Nature 2000, 405, 903–904. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 98, 1231–1237. [Google Scholar]
- StatSoft Italia Srl. STATISTICA, Version 7.1; StatSoft Italia Srl: Vigonza, Italy, 2005.
- Dalgetty, D.D.; Baik, B.K. Isolation and characterization of cotyledon fibres from peas lentils and chickpeas. Cereal Chem. 2003, 80, 310–315. [Google Scholar] [CrossRef]
- Wang, N.; Hatcher, D.W.; Tyler, R.T.; Toews, R.; Gawalko, E.J. Effect of cooking on the composition of beans (Phaseolus vulgaris L.) and chickpea (Cicer arietinum L.). Food Res. Int. 2010, 43, 589–594. [Google Scholar] [CrossRef]
- Cardador-Martinez, A.; Loarca-Pina, G.; Oomah, B.D. Antioxidant activity in common beans (Phaseolus vulgaris L.). J. Agric. Food Chem. 2002, 50, 6975–6980. [Google Scholar] [CrossRef]
- Harborne, J.B.; Baxter, H. The Handbook of Natural Flavonoids; Wiley: Chichester, UK, 1999; Volume 2. [Google Scholar]
- Wang, L.S.; Stoner, G.D. Anthocyanins and their role in cancer prevention. Cancer Lett. 2008, 269, 281–290. [Google Scholar] [CrossRef]
- Lee, K.W.; Lee, H.J. The role of polyphenols in cancer chemoprevention. BioFactors 2006, 26, 105–121. [Google Scholar] [CrossRef]
- You, Q.; Wang, B.; Chen, F.; Huang, Z.; Wang, X.; Luo, P.G. Comparisonof anthocyanins and phenolics in organically and conventionally grown blueberries in selected cultivars. Food Chem. 2011, 125, 201–208. [Google Scholar] [CrossRef]
- Bourn, D.; Prescott, J. A comparison of the nutritional value, sensory qualities, and food safety of organically and conventionally produced foods. Crit. Rev. Food Sci. Nutr. 2002, 42, 1–34. [Google Scholar] [CrossRef]
- Menga, V.; Fares, C.; Troccoli, A.; Cattivelli, L.; Baiano, A. Effects of genotype, location and baking on the phenolic content and some antioxidant properties of cereal species. Int. J. Food Sci. Technol. 2010, 45, 7–16. [Google Scholar]
- Yu, L.; Zhou, K. Antioxidant properties of bran extracts for “Platte” wheat grown at different locations. Food Chem. 2004, 90, 311–316. [Google Scholar]
- Naczk, M.; Shahidi, F. Phenolics in cereals, fruits and vegetables: Occurrence, extraction and analysis. J. Pharm. Biomed. Anal. 2006, l41, 1523–1542. [Google Scholar] [CrossRef]
- Zou, Y.; Chang, S.K.C.; Gu, Y.; Qian, S.Y. Antioxidant activity and phenolic compositions of lentil (Lens culinaris var. Morton) extract and its fractions. J. Agric. Food Chem. 2011, 59, 2268–2276. [Google Scholar] [CrossRef]
- Xu, B.J.; Yuan, S.H.; Chang, S.K.C. Comparative analyses of phenolic composition, antioxidant capacity, and color of cool season legumes and other selected food legumes. J. Food Sci. 2007, 72, S167–S177. [Google Scholar] [CrossRef]
- Takahata, Y.; Ohnishi-Kameyama, M.; Furuta, S.; Takahashi, M.; Suda, I. Highly polymerized procyanidins in brown soybean seed coat with a high radical-scavenging activity. J. Agric. Food Chem. 2001, 49, 5843–5847. [Google Scholar] [CrossRef]
- Troszynska, A.; Ciska, E. Phenolic compounds of seed coats of white and coloured varieties of pea (Pisum sativum L.) and their total antioxidant activity. Czech J. Food Sci. 2002, 20, 15–22. [Google Scholar]
- Pastor-Cavada, E.; Juan, R.; Pastor, J.E.; Manuel Alaiz, M.; Vioque, J. Antioxidant activity of seed polyphenols in fifteen wild Lathyrus species from South Spain. Food Sci. Technol. 2009, 42, 705–709. [Google Scholar]
- Harborne, J.B. Flavonoids in the environment: Structure-activity relationships. Prog. Clin. Biol. Res. 1998, 280, 17–27. [Google Scholar]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997, 2, 152–159. [Google Scholar]
- Makoi, J.H.J.R.; Ndakidemi, P.A. Changes in plant growth, nutrient dynamics and accumulation of flavonoids and anthocyanins by manipulating the cropping systems involving legumes and cereals—a review. Aust. J. Mech. Eng. 2011, 2, 56–65. [Google Scholar]
- Jensen, E.S. Grain yield, symbiotic N2-fixation and interspecific competition for inorganic N in pea-barley intercrops. Plant Soil 1996, 182, 25–38. [Google Scholar] [CrossRef]
- Rein, D.; Paglieroni, T.G.; Wun, T.; Pearson, D.A.; Schmitz, H.H.; Gosselin, R.; Keen, C.L. Cocoa inhibits platelet activation and function. Am. J. Clin. Nutr. 2000, 72, 30–35. [Google Scholar]
- Vitseva, O.; Varghese, S.; Chakrabarti, S.; Folts, J.D.; Freedman, J.E. Grape seed and skin extracts inhibit platelet function and release of reactive oxygen intermediates. J. Cardiovasc. Pharmacol. 2005, 46, 445–451. [Google Scholar] [CrossRef]
- Velayutham, P.; Babu, A.; Liu, D. Green tea catechins and cardiovascular health: An update. Curr. Med. Chem. 2008, 15, 1840–1850. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Menga, V.; Codianni, P.; Fares, C. Agronomic Management under Organic Farming May Affect the Bioactive Compounds of Lentil (Lens culinaris L.) and Grass Pea (Lathyrus communis L.)? Sustainability 2014, 6, 1059-1075. https://doi.org/10.3390/su6021059
Menga V, Codianni P, Fares C. Agronomic Management under Organic Farming May Affect the Bioactive Compounds of Lentil (Lens culinaris L.) and Grass Pea (Lathyrus communis L.)? Sustainability. 2014; 6(2):1059-1075. https://doi.org/10.3390/su6021059
Chicago/Turabian StyleMenga, Valeria, Pasquale Codianni, and Clara Fares. 2014. "Agronomic Management under Organic Farming May Affect the Bioactive Compounds of Lentil (Lens culinaris L.) and Grass Pea (Lathyrus communis L.)?" Sustainability 6, no. 2: 1059-1075. https://doi.org/10.3390/su6021059
APA StyleMenga, V., Codianni, P., & Fares, C. (2014). Agronomic Management under Organic Farming May Affect the Bioactive Compounds of Lentil (Lens culinaris L.) and Grass Pea (Lathyrus communis L.)? Sustainability, 6(2), 1059-1075. https://doi.org/10.3390/su6021059




