Synthesis and Antimicrobial Activity of Some New 1,3,4-Thiadiazole and 1,2,4-Triazole Compounds Having a D,L-Methionine Moiety
Abstract
:Introduction
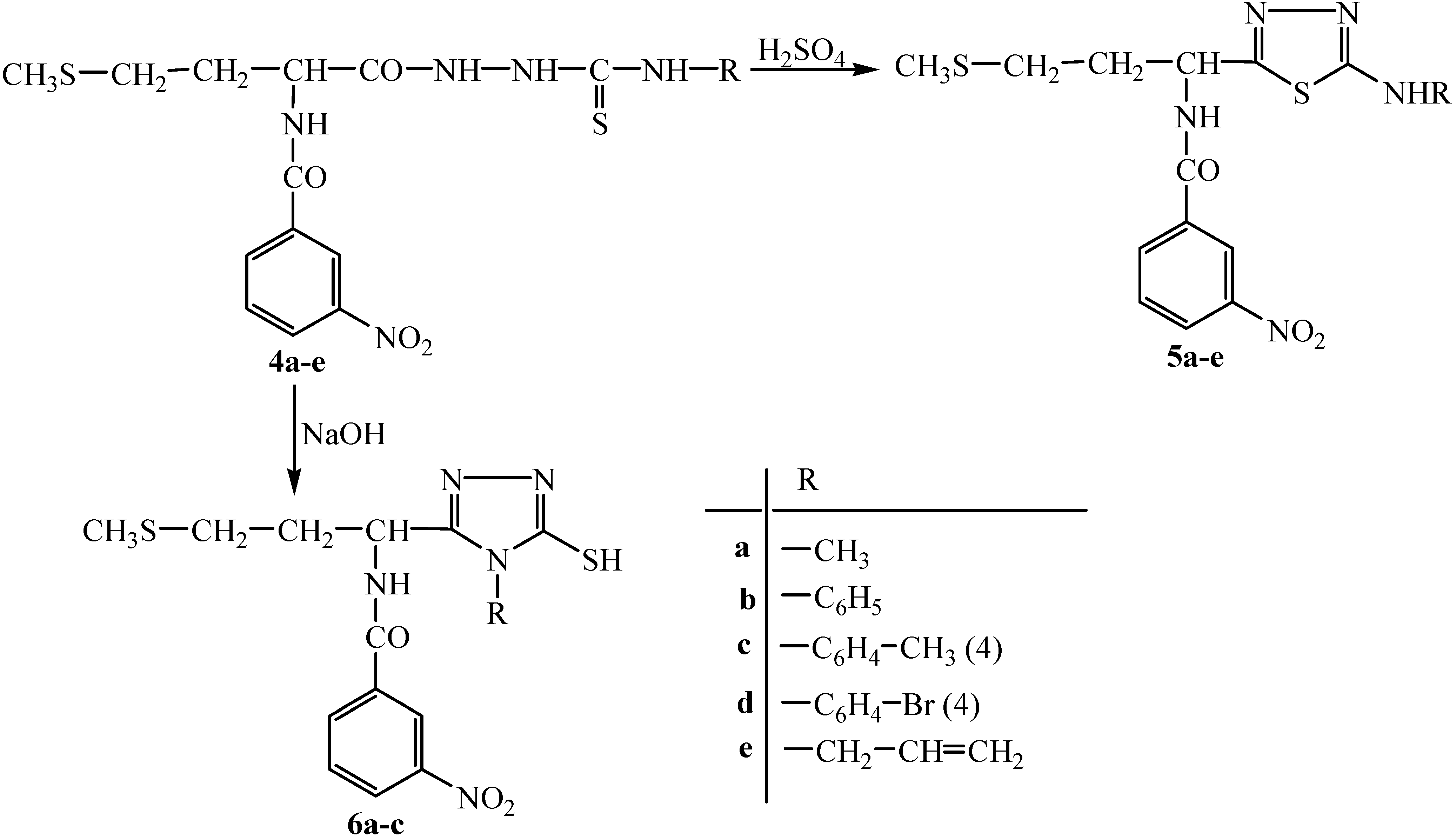
Results and Discussion

Biological activity
| Comp. no. | Minimum Inhibitory Concentration (MIC) (μg/mL) | ||||
|---|---|---|---|---|---|
| Sa | Ba | Bc | Sl | Ec | |
| 4a | 171 | 618 | 714 | 638 | 193 |
| Comp. | DL50 mg/Kg body | Comp. | DL50 mg/Kg body |
|---|---|---|---|
| 4a | 1465 | 5c | 3100 |
| 4b | 1275 | 5d | 2025 |
| 4c | 1625 | 5e | 2816 |
| 4d | 1315 | 6a | 4620 |
| 4e | 1260 | 6b | 5010 |
| 5a | 1825 | 6c | 4920 |
| 5b | 2110 |
Conclusions
Experimental Section
General
Synthesis of 2-(3-nitrophenyl)-4-(2-methylthioethyl)-Δ2-5-oxazolinone (2).
N-(3-nitrobenzoyl)-D,L-methionyl-hydrazide (3).
General procedure for the synthesis of 1,4-disubstituted thiosemicarbazides 4a-e.
General procedure for the synthesis of 1,3,4-thiadiazole compounds 5a-e.
General procedure for the synthesis of 1,2,4-triazole compounds 6a-c.
Antimicrobial activity assessment
Toxicity study
References
- Palaska, E.; Sahin, G.; Kelicen, P.; Durlu, N. T.; Altinok, G. Synthesis and anti-inflammatory activity of 1-acylthiosemicarbazides, 1,3,4-oxadiazoles, 1,3,4-thiadiazoles and 1,2,4-triazole-3-thiones. Farmaco 2002, 57, 101–107. [Google Scholar] [CrossRef]
- Labanauskas, L.; Kalcas, V.; Udrenaite, E.; Gaidelis, P.; Brukstus, A.; Dauksas, V. Synthesis of 3-(3,4-dimethoxyphenyl)-1 H-1,2,4-triazole-5-thiol and 2-amino-5-(3,4-dimethoxyphenyl)-1,3,4-thiadiazole derivatives exhibiting anti-inflammatory activity. Pharmazie 2001, 56, 617–619. [Google Scholar]
- Onkol, T.; Cakir, B.; Sahin, M. F. Synthesis and Antinociceptive Activity of 2-[(2-Oxobenzothiazolin-3-yl)methyl]-5-aminoalkyl / aryl-1,3,4-thiadiazole. Turk. J. Chem. 2004, 28, 461–466. [Google Scholar]
- Schenone, S.; Bruno, O.; Ranise, A.; Bondavalli, F.; Filippeli, W.; Falcone, G.; Giordano, L.; Vitelli, M. R. 3-Arylsulphonyl-5-arylamino-1,3,4-thiadiazol-2(3H)ones as anti-inflammatory and analgesic agents. Bioorg. Med. Chem. 2001, 9, 2149–2153. [Google Scholar] [CrossRef]
- Gokce, M.; Cakir, B.; Erol, K.; Sahin, M. F. Synthesis and antinociceptive activity of [(2-oxobenzothiazolin-3-yl)methyl]-4-alkyl/aryl-1,2,4-triazoline-5-thiones. Arch. Pharm. 2001, 334, 279–283. [Google Scholar] [CrossRef]
- Baldwin, J. J.; Engelhardt, E. L.; Hirschmann, R.; Ponticello, G. S.; Atkinson, J. G.; Wasson, B. K.; Sweet, C. S.; Scriabine, A. Heterocyclic analogues of the antihypertensive beta-adrenergic blocking agents (S)-2-[3-(ter-butylamino)-2-hydroxypropoxy]-3-cyanopyridine. J. Med. Chem. 1980, 23, 65–70. [Google Scholar] [CrossRef]
- Varvaresou, A.; Tsantili-Kakoulidou, A.; Siatra-Papastasikoudi, T.; Tiligada, E. Synthesis and biological evaluation of indole containing derivatives of thiosemicarbazide and their cyclic 1,2,4-triazole and 1,3,4-thiadiazole analogs. Arzneimittelforschung 2000, 50, 48–54. [Google Scholar] Varvaresou, A.; Siatra-Papastasikoudi, T.; Tsontinis, A.; Tsantili-Kakoulidou, A.; Vamvakides, A. Synthesis, lipophilicity and biological evaluation of indole-containing derivatives of 1,3,4-thiadiazole and 1,2, 4-triazole. Farmaco 1998, 53, 320–326. [Google Scholar]
- Foroumadi, A.; Mirzaei, M.; Shafiee, A. Antituberculosis agents, I: Synthesis and antituberculosis activity of 2-aryl-1,3,4-thiadiazole derivatives. Pharmazie 2001, 56, 610–612. [Google Scholar]
- Mamolo, M. G.; Falagiani, V.; Zanpieir, D.; Vio, L.; Banfi, F. Synthesis and antimycobacterial activity of [5-(pyridin-2-yl)-1,3,4-thiadiazol-2-ylthio]acetic acid arylidene-hydrazide derivatives. Farmaco 2001, 56, 587–592. [Google Scholar]
- Wujec, M.; Pitucha, M.; Dobosz, M.; Kosikowska, U.; Malm, A. Synthesis and potential antimycotic activity of 4-substituted-3-(thiophene-2-yl-methyl)-Delta2-1,2,4-triazoline-5-thiones. Acta Pharm. 2004, 54, 251–260. [Google Scholar]
- Zamani, K.; Faghifi, K.; Tefighi, I.; Sharlatzadeh, M. R. Synthesis and potential antimycotic activity of 4-substituted 3-(thiophene-2-yl-methyl)-Δ2-1,2,4-triazoline-5-thiones. Turk. J. Chem. 2004, 28, 95–101. [Google Scholar]
- Chen, H.; Li, Z.; Han, Y. Synthesis and fungicidal activity against Rhizoctonia solani of 2-alkyl (Alkylthio)-5-pyrazolyl-1,3,4-oxadiazoles (Thiadiazoles). J. Agric. Food Chem. 2000, 48, 5312–5315. [Google Scholar] [CrossRef]
- Zou, X. J.; Jin, G. Y.; Zhang, Z. X. Synthesis, fungicidal activity, and QSAR of pyridazinonethiadiazoles. J. Agric. Food Chem. 2002, 50, 1451–1454. [Google Scholar] Zou, X. J.; Lai, L. H.; Jin, G. Y.; Zhang, Z. X. Synthesis, fungicidal activity, and 3D-QSAR of pyridazinone-substituted 1,3,4-oxadiazoles and 1,3,4-thiadiazoles. J. Agric. Food Chem. 2002, 50, 3757–3760. [Google Scholar]
- Clerici, F.; Pocar, D.; Guido, M.; Loche, A.; Perlini, V.; Brufani, M. Synthesis of 2-amino-5-sulfanyl-1,3,4-thiadiazole derivatives and evaluation of their antidepressant and anxiolytic activity. J. Med. Chem. 2001, 44, 931–936. [Google Scholar] [CrossRef]
- Sunel, V.; Lionte, C.; Popa, M.; Pintilie, O.; Mungiu, O.; Teleman, S. Synthesis of new methionine derivatives for the treatment of paracetamol-induced hepatic injury. Eur. Chem. Tech. J. 2002, 4, 201–205. [Google Scholar]
- Rhee, S. G.; Chock, P.; Boon, W.; Sugiyama, Y. Subunit interaction in unadenylylated glutamine synthetase from Escherichia coli. Evidence from methionine sulfoximine. J. Biol. Chem. 1981, 256, 644–648. [Google Scholar]
- National Committee for Clinical Laboratory Standard. Methods for diluation antimicrobial susceptibility tests for bacteria that grow aerobically, Approved standard M7-A2; NCCLS: Vilanova, PA, 1990. [Google Scholar]
- Czajkowska, T.; Graczyk, J.; Krysiak, B.; Stetkiewicz, J. Acute toxic effects of trimethyl and triethyl phosphates. J. Med. Pr. 1978, 29, 393–398. [Google Scholar]
- Sample Availability: Samples of the compounds 5a-e and 6a-c are available from authors.
© 2007 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Pintilie, O.; Profire, L.; Sunel, V.; Popa, M.; Pui, A. Synthesis and Antimicrobial Activity of Some New 1,3,4-Thiadiazole and 1,2,4-Triazole Compounds Having a D,L-Methionine Moiety. Molecules 2007, 12, 103-113. https://doi.org/10.3390/12010103
Pintilie O, Profire L, Sunel V, Popa M, Pui A. Synthesis and Antimicrobial Activity of Some New 1,3,4-Thiadiazole and 1,2,4-Triazole Compounds Having a D,L-Methionine Moiety. Molecules. 2007; 12(1):103-113. https://doi.org/10.3390/12010103
Chicago/Turabian StylePintilie, Otilia, Lenuta Profire, Valeriu Sunel, Marcel Popa, and Aurel Pui. 2007. "Synthesis and Antimicrobial Activity of Some New 1,3,4-Thiadiazole and 1,2,4-Triazole Compounds Having a D,L-Methionine Moiety" Molecules 12, no. 1: 103-113. https://doi.org/10.3390/12010103
APA StylePintilie, O., Profire, L., Sunel, V., Popa, M., & Pui, A. (2007). Synthesis and Antimicrobial Activity of Some New 1,3,4-Thiadiazole and 1,2,4-Triazole Compounds Having a D,L-Methionine Moiety. Molecules, 12(1), 103-113. https://doi.org/10.3390/12010103





