Exploring Polymeric Micelles for Improved Delivery of Anticancer Agents: Recent Developments in Preclinical Studies
Abstract
:1. Introduction
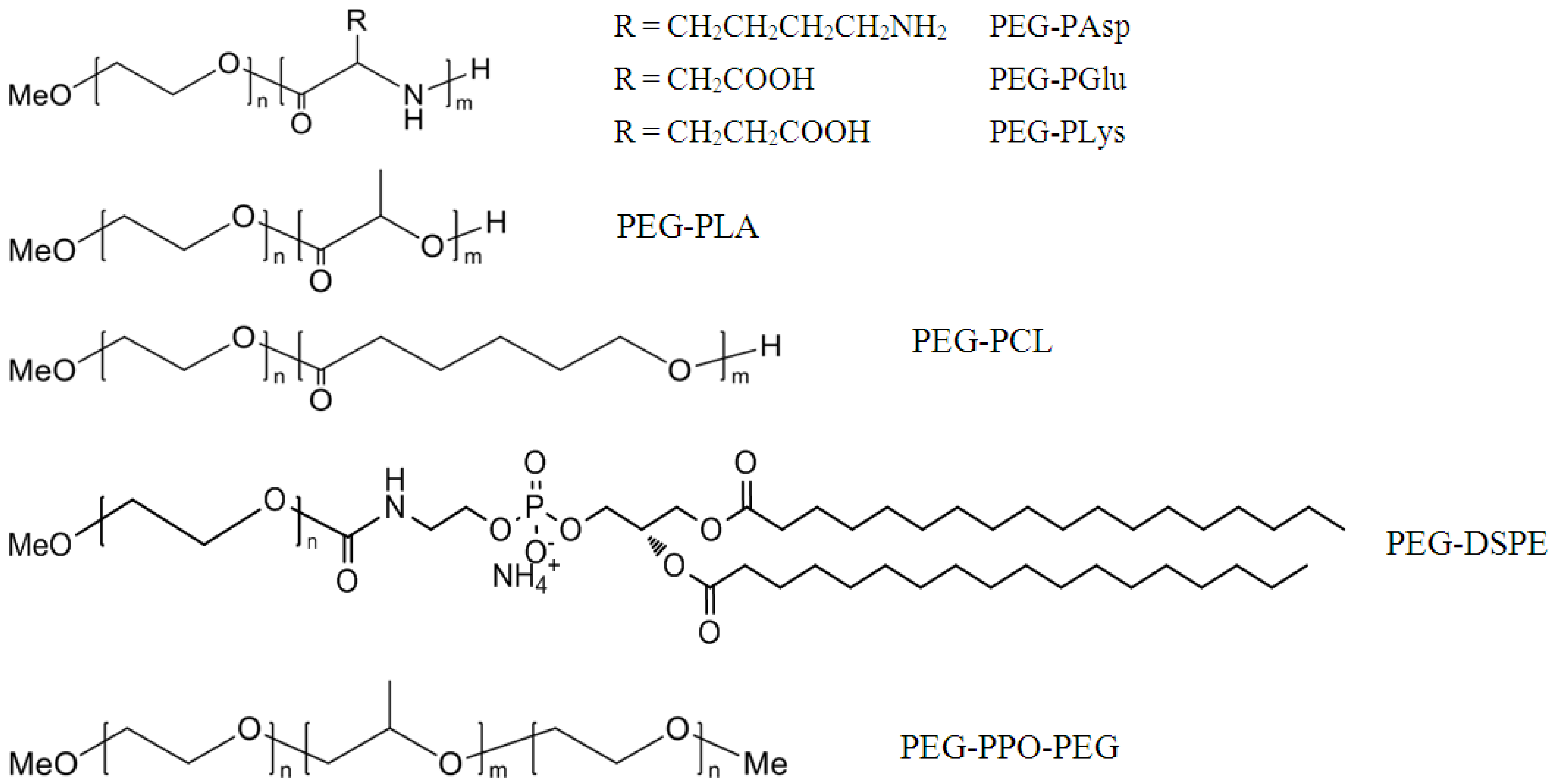
2. Polymeric Micelles Can Improve Anticancer Efficacy of Hydrophobic Drugs via Passive Targeting
| Polymer | Drug | Size (nm) | Tumor Model | Effects (compared to the free drug) | Ref. |
| PEG-PLA | β-lapachone | 30 | s.c. A549 | ↑ drug levels in plasma and the tumor ↓ tumor growth ↑ survival time | [7] |
| orthotopic LLC | ↓ metastatic tumor burden ↑ survival time | ||||
| PEG-PLA & P85 | docetaxel | 21 | s.c. KBv | ↑ drug levels in plasma and the tumor ↓ tumor growth | [8] |
| PEG-DSPE | doxorubicin | 10–20 | s.c. & orthotopic LLC | ↑ drug levels in plasma and the tumor | [9] |
| s.c. 4T1 | ↓ tumor growth ↑ survival time | ||||
| PEG-DSPE | docetaxel | 21 | s.c. MCF-7 | ↑ drug levels in plasma and the tumor ↓ tumor growth | [10] |
| PEG-DSPE | paclitaxel & 17-AAG | 11 | s.c. SKOV-3 | ↑ drug levels in plasma and the tumor ↓ tumor growth | [11] |
| PEG-poly(benzyl aspartate) | camptothecin | 192 | s.c. colon 26 | ↓ tumor growth | [12] |
| s.c. B16BL6 | ↑ drug levels in plasma | ||||
| fenretinide | 173 | ↓ tumor growth | [13] | ||
| PEG-PCL-PEG | paclitaxel | 93 | s.c. EMT6 | ↑ drug levels in plasma ↓ tumor growth | [14] |
| chlorin-conjugated PCL-PEG | SN-38 | 130 | s.c. HT-29 | ↑ drug levels in plasma and the tumor ↓ tumor growth ↑ survival time | [15] |
| P123/F127 | paclitaxel | 24 | ↑ drug levels in plasma and the tumor ↑ survival time | [16] | |
| P105/PCL50 | paclitaxel | 149 | s.c. SKOV-3 | ↓ tumor growth | [17] |
| CP750 | cis-(cha)2-Pt(NO3)2 | 7 | s.c. B16F10 | ↑ drug levels in plasma and the tumor without tumor growth inhibition | [18] |
| docetaxel | s.c. MDA-MB-231 & s.c. MNK-28 | no improvement in pharmacokinetics ↓ tumor growth | [19] | ||
| PEG-PAE | doxorubicin | 62 | s.c.B16F10 | ↓ tumor growth | [20,21] |
| camptothecin | 214 | s.c. MDA-MB-231 | ↑ survival time | ||
| PEG5K-CA8 | paclitaxel | 20–60 | s.c. & i.p.SKOV-3 | ↑ drug levels in plasma and the tumor ↓ tumor growth ↑ survival time | [22] |
| PEG2K-CA4 | doxorubicin | 12 | s.c. Raji lymphoma | ↓ tumor growth ↑ survival time | [23] |
2.1. PEG-PLA-Based Micelles
2.2. PEG-DSPE-Based Micelles
2.3. PEG-Poly(benzyl aspartate)-Based Micelles
2.4. PEG-PCL-Based Micelles
2.5. PEG-PPO-PEG-Based Micelles
2.6. Novel Micelle Constructs
3. Targeting Ligands Enhance the Anticancer Efficacy of Micellar Drugs
| Ligand | Polymer | Drug | Size (nm) | Tumor Model | Effects (compared to the nontargeted micelles) | Ref. |
| folate | PEG-PLLA &PEG-PHis | doxorubicin | 94 | s.c. 4T1 | ↓ tumor growth ↓ metastatic tumor burden ↑ survival time | [24] |
| folate | PEG-poly (His-co-Phe) & PEG-PLA | doxorubicin | 150 | s.c. A2780/DoxR | ↑ drug levels in the tumor ↓ tumor growth | [25] |
| folate | Poly(2-HEMA-co-His)-g-PLA & PEG-PLA | doxorubicin | ~200 | s.c. HeLa | ↓ drug levels in plasma (slightly) ↓ tumor growth | [26] |
| folate | P123 & F127 | paclitaxel | 20–50 | s.c. KBv | ↓ drug levels in plasma (slightly) ↓ tumor growth | [27] |
| folate | PEOz-PLA | m-THPC | ~100 | s.c. KB | ↑ drug levels in the tumor ↓ tumor growth | [28] |
| tarnsferrin | PCL-PEEP | paclitaxel | 88 | intracranial U87MG | ↑ drug levels in the brain ↑ survival time | [29] |
| cRGD | PEG-PLA | paclitaxel | 26 | s.c. & intracranial U87MG | ↓ tumor growth ↑ survival time | [30] |
| NGR | PEG-DSPE | paclitaxel | 54 | intracranial C6 | ↓ tumor growth | [31] |
| D-SP5 | PEG-DSPE | doxorubicin | 30 | s.c. KB | ↓ tumor growth | [32] |
| OA02 | PEG5K-CA8 | paclitaxel | 21 | s.c. SKOV-3 | ↓ tumor growth ↑ survival time | [33] |
| TAT | PEG-DSPE | paclitaxel | 8–25 | s.c. 4T1 | ↑ apoptosis in the tumor | [34] |
| lanreotide | PEG-PCL | paclitaxel | 43 | s.c. H446 | ↑ drug levels in the tumor | [35] |
| s.c. MCF-7 | ↓ tumor growth ↑ survival time | |||||
| LyP-1 | PEG-PCL | artemisinin | 30 | orthotopic MDA-MB-435 | home to lymphatic vessels ↓ tumor growth | [36] |
| AP peptide | MPEG-PAE/PEG-PLA | doxorubicin | 181 | s.c. MDA-MB-231 | more rapid accumulation in the tumor and longer retention ↓ tumor growth | [37] |
3.1. Folic Acid
3.2. Transferrin
3.3. Tumor-Targeting Peptides
4. Cross-Linked Micelles Improve the Stability and Drug Delivery Efficiency
| Polymer | Drug | Size (nm) | Tumor Model | Effect (compared to the free drug and/or non-crosslinked micelles) | Ref. |
| PEG-(Cys)4-PLA | doxorubicin | – | s.c. M109 | ↑ drug levels in the tumor ↓ tumor growth | [49] |
| PEG5K-Cys4-L8-CA8 | paclitaxel | 28 | s.c. SKOV-3 | ↓ tumor growth ↑ survival time | [50] |
| PEG-PLys-PPhe | docetaxel | 59 | s.c. MDA-MB-231 | ↑ drug levels in the tumor and normal organs ↓ tumor growth ↑ survival time | [51] |
| mPEG-b-p(HPMAm-Lacn) | doxorubicin | 80 | s.c. B16F10 | ↑ survival time | [52] |
5. Tumor-Specific Delivery of Plasmid DNA (pDNA) and Small Interference RNA (siRNA) Is Achievable via Polyplex Micelles
| pDNA or siRNA | Polymer | Size (nm) | Targeting Ligand | Tumor Model | Effects | Ref. |
|---|---|---|---|---|---|---|
| pDNA encoding sFlt-1 | thiolated PEG-PLL (disulfide cross-linked) | 116 | – | s.c. BxPC-3 | Compared to the non-cross-linked micelles, ↑ sFlt-1 expression in the tumor ↓ tumor growth ↓ vascular density | [57] |
| pDNA encoding sFlt-1 | thiolated PEG-PLL (disulfide cross-linked) | 104 | cRGD | s.c. BxPC-3 | Compared to the non-targeted micelles, ↓ pDNA level in blood ↑ pDNA level in the tumor ↓ tumor growth ↓ vascular density | [58] |
| pDNA encoding RGD4C-hTNF-α | PEG-SS-p[Asp(DET)] | 80–90 | – | i.p. SUIT-2 | ↑ hTNF-α expression in the tumor ↓ i.p. tumor burden ↑ survival time | [59] |
| VEGF/VEGFR2 siRNA | PEG-PLL(2IT) | 45 | cRGD | s.c. HeLa | ↑ siRNA levels in blood and the tumor ↓ tumor growth | [60] |
| VEGF siRNA | siRNA-conjugated PEG & PEI | 99 | – | s.c. PC-3 | ↑ VEGF siRNA levels in blood and the tumor ↓ VEGF mRNA and protein levels in the tumor ↓ microvessel density ↓ tumor growth | [61] |
| AC siRNA | PEG-PCL-PPEEA | 60 | – | s.c. BT474 | ↓ AC mRNA and protein levels in the tumor ↓ tumor growth | [62] |
| Plk1 siRNA with paclitaxel | PEG-PCL-PPEEA | 50 | – | s.c. MDA-MB-435 | co-localization of Plk1 siRNA and paclitaxel in the same tumor cells ↓ Plk1 protein in the tumor ↓ tumor growth | [63] |
5.1. pDNA Delivery
5.2. siRNA Delivery
6. Summary
Acknowledgments
Conflict of Interest
References
- Gong, J.; Chen, M.; Zheng, Y.; Wang, S.; Wang, Y. Polymeric micelles drug delivery system in oncology. J. Control. Release 2012, 159, 312–323. [Google Scholar] [CrossRef]
- Maeda, H.; Nakamura, H.; Fang, J. The EPR effect for macromolecular drug delivery to solid tumors: Improvement of tumor uptake, lowering of systemic toxicity, and distinct tumor imaging in vivo. Adv. Drug Deliv. Rev. 2013, 65, 71–79. [Google Scholar] [CrossRef]
- Xu, S.; Olenyuk, B.Z.; Okamoto, C.T.; Hamm-Alvarez, S.F. Targeting receptor-mediated endocytotic pathways with nanoparticles: rationale and advances. Adv. Drug Deliv. Rev. 2013, 65, 121–138. [Google Scholar] [CrossRef]
- Blanco, E.; Kessinger, C.W.; Sumer, B.D.; Gao, J. Multifunctional micellar nanomedicine for cancer therapy. Exp. Biol. Med. 2009, 234, 123–131. [Google Scholar]
- Kwon, G.S.; Kataoka, K. Block copolymer micelles as long-circulating drug vehicles. Adv. Drug Deliv. Rev. 2012, 64, 237–245. [Google Scholar] [CrossRef]
- Torchilin, V.P. Multifunctional nanocarriers. Adv. Drug Deliv. Rev. 2012, 64, 302–315. [Google Scholar] [CrossRef]
- Blanco, E.; Bey, E.A.; Khemtong, C.; Yang, S.G.; Setti-Guthi, J.; Chen, H.; Kessinger, C.W.; Carnevale, K.A.; Bornmann, W.G.; Boothman, D.A.; et al. β-lapachone micellar nanotherapeutics for non-small cell lung cancer therapy. Cancer Res. 2010, 70, 3896–3904. [Google Scholar] [CrossRef]
- Mu, C.F.; Balakrishnan, P.; Cui, F.D.; Yin, Y.M.; Lee, Y.B.; Choi, H.G.; Yong, C.S.; Chung, S.J.; Shim, C.K.; Kim, D.D. The effects of mixed MPEG–PLA/Pluronic® copolymer micelles on the bioavailability and multidrug resistance of docetaxeltaxel. Biomaterials 2010, 31, 2371–2379. [Google Scholar] [CrossRef]
- Tang, N.; Du, G.; Wang, N.; Liu, C.; Hang, H.; Liang, W. Improving penetration in tumors with nanoassemblies of phospholipids and doxorubicin. J. Natl. Cancer Inst. 2007, 99, 1004–1015. [Google Scholar] [CrossRef]
- Tong, S.W.; Xiang, B.; Dong, D.W.; Qi, X.R. Enhanced antitumor efficacy and decreased toxicity by self-associated docetaxel in phospholipid-based micelles. Int. J. Pharm. 2012, 434, 413–419. [Google Scholar] [CrossRef]
- Katragadda, U.; Fan, W.; Wang, Y.; Teng, Q.; Tan, C. Combined delivery of paclitaxel and tanespimycin via micellar nanocarriers: pharmacokinetics, efficacy and metabolomic analysis. PLoS One 2013, 8, e58619. [Google Scholar] [CrossRef]
- Kawano, K.; Watanabe, M.; Yamamoto, T.; Yokoyama, M.; Opanasopit, P.; Okano, T.; Maitani, Y. Enhanced antitumor effect of camptothecin loaded in long-circulating polymeric micelles. J. Control. Release 2006, 112, 329–332. [Google Scholar] [CrossRef]
- Okuda, T.; Kawakami, S.; Higuchi, Y.; Satoh, T.; Oka, Y.; Yokoyama, M.; Yamashita, F.; Hashida, M. Enhanced in vivo antitumor efficacy of fenretinide encapsulated in polymeric micelles. Int. J. Pharm. 2009, 373, 100–106. [Google Scholar] [CrossRef]
- Zhang, L.; He, Y.; Ma, G.; Song, C.; Sun, H. Paclitaxel-loaded polymeric micelles based on poly(ε-caprolactone)-poly(ethylene glycol)-poly(ε-caprolactone) triblock copolymers: In vitro and in vivo evaluation. Nanomedicine 2012, 8, 925–934. [Google Scholar] [CrossRef]
- Peng, C.L.; Lai, P.S.; Lin, F.H.; Wu, S.Y.-H.; Shieh, M.J. Dual chemotherapy and photodynamic therapy in an HT-29 human colon cancer xenograft model using SN-38-loaded chlorin-core star block copolymer micelles. Biomaterials 2009, 30, 3614–3625. [Google Scholar]
- Zhang, W.; Shi, Y.; Chen, Y.; Hao, J.; Sha, X.; Fang, X. The potential of Pluronic polymeric micelles encapsulated with paclitaxel for the treatment of melanoma using subcutaneous and pulmonary metastatic mice models. Biomaterials 2011, 32, 5934–5944. [Google Scholar] [CrossRef]
- Wang, Y.; Hao, J.; Li, Y.; Zhang, Z.; Sha, X.; Han, L.; Fang, X. Poly(caprolactone)-modified Pluronic P105 micelles for reversal of paclitaxcel-resistance in SKOV-3 tumors. Biomaterials 2012, 33, 4741–4751. [Google Scholar] [CrossRef]
- Jadhav, V.B.; Jun, Y.J.; Song, J.H.; Park, M.K.; Oh, J.H.; Chae, S.W.; Kim, I.S.; Choi, S.J.; Lee, H.J.; Sohn, Y.S. A novel micelle-encapsulated platinum (II) anticancer agent. J. Control. Release 2010, 147, 144–150. [Google Scholar] [CrossRef]
- Jun, Y.J.; Jadhav, V.B.; Min, J.H.; Cui, J.X.; Chae, S.W.; Choi, J.M.; Kim, I.S.; Choi, S.J.; Lee, H.J.; Sohn, Y.S. Stable and efficient delivery of docetaxel by micelle-encapsulation using a tripodal cyclotriphosphazene amphiphile. Int. J. Pharm. 2012, 31, 374–380. [Google Scholar]
- Ko, J.; Park, K.; Kim, Y.-S.; Kim, M.S.; Han, J.K.; Kim, K.; Park, R.-W.; Kim, I.-S.; Song, H.Y.; Lee, D.S.; et al. Tumoral acidic extracellular pH targeting of pH-responsive MPEG-poly(β-amino ester) block copolymer micelles for cancer therapy. J. Control. Release 2007, 123, 109–115. [Google Scholar]
- Min, K.H.; Kim, J.-H.; Bae, S.M.; Shin, H.; Kim, M.S.; Park, S.; Lee, H.; Park, R.-W.; Kim, I.-S.; Kim, K.; et al. Tumoral acidic pH-responsive MPEG-poly(β-amino ester) polymeric micelles for cancer targeting therapy. J. Control. Release 2010, 144, 259–266. [Google Scholar] [CrossRef]
- Xiao, K.; Luo, J.; Fowler, W.L.; Li, Y.; Lee, J.S.; Xing, L.; Cheng, R.H.; Wang, L.; Lam, K.S. A self-assembling nanoparticle for paclitaxel delivery in ovarian cancer. Biomaterials 2009, 30, 6006–6016. [Google Scholar] [CrossRef]
- Xiao, K.; Li, Y.; Luo, J.; Lee, J.S.; Xiao, W.; Gonik, A.M.; Agarwal, R.G.; Lam, K.S. PEG-oligocholic acid telodendrimer micelles for the targeted delivery of doxorubicin to B-cell lymphoma. J. Control. Release 2011, 155, 272–281. [Google Scholar] [CrossRef]
- Gao, Z.G.; Tian, L.; Hu, J.; Park, I.S.; Bae, Y.H. Prevention of metastasis in a 4T1 murine breast cancer model by doxorubicin carried by folate conjugated pH sensitive polymeric micelles. J. Control. Release 2011, 1521, 84–89. [Google Scholar]
- Kim, D.; Gao, Z.G.; Lee, E.S.; Bae, Y.H. In vivo evaluation of doxorubicin-loaded polymeric micelles targeting folate receptors and early endosomal pH in drug-resistant ovarian cancer. Mol. Pharm. 2009, 6, 1353–1362. [Google Scholar] [CrossRef]
- Tsai, H.C.; Chang, W.H.; Lo, C.L.; Tsai, C.H.; Chang, C.H.; Ou, T.W.; Yen, T.C.; Hsiue, G.H. Graft and diblock copolymer multifunctional micelles for cancer chemotherapy and imaging. Biomaterials 2010, 31, 2293–2301. [Google Scholar]
- Zhang, W.; Shi, Y.; Chen, Y.; Ye, J.; Sha, X.; Fang, X. Multifunctional Pluronic P123/F127 mixed polymeric micelles loaded with paclitaxel for the treatment of multidrug resistant tumors. Biomaterials 2011, 32, 2894–2906. [Google Scholar] [CrossRef]
- Syu, W.J.; Yu, H.P.; Hsu, C.Y.; Rajan, Y.C.; Hsu, Y.H.; Chang, Y.C.; Hsieh, W.Y.; Wang, C.H.; Lai, P.S. Improved photodynamic cancer treatment by folate-conjugated polymeric micelles in a KB xenografted animal model. Small 2012, 8, 2060–2069. [Google Scholar] [CrossRef]
- Zhang, P.; Hu, L.; Yin, Q.; Zhang, Z.; Feng, L.; Li, Y. Transferrin-conjugated polyphosphoester hybrid micelle loading paclitaxel for brain-targeting delivery: synthesis, preparation and in vivo evaluation. J. Control. Release 2012, 159, 429–434. [Google Scholar] [CrossRef]
- Zhan, C.; Gu, B.; Xie, C.; Li, J.; Liu, Y.; Lu, W. Cyclic RGD conjugated poly (ethylene glycol)-co-poly (lactic acid) micelle enhances paclitaxel anti-glioblastoma effect. J. Control. Release 2010, 143, 136–142. [Google Scholar] [CrossRef]
- Zhao, B.J.; Ke, X.Y.; Huang, Y.; Chen, X.M.; Zhao, X.; Zhao, B.X.; Lu, W.L.; Lou, J.N.; Zhang, X.; Zhang, Q. The antiangiogenic efficacy of NGR-modified PEG-DSPE micelles containing paclitaxel (NGR-M-PTX) for the treatment of glioma in rats. J. Drug Target. 2011, 19, 382–390. [Google Scholar] [CrossRef]
- Ying, L.; Lei, Y.; Wagner, E.; Xie, C.; Lu, W.; Zhu, J.; Shen, J.; Wang, J.; Liu, M. Potent retro-inverso d-peptide for simultaneous targeting of angiogenic blood vasculature and tumor cells. Bioconjug. Chem. 2013, 24, 133–143. [Google Scholar] [CrossRef]
- Xiao, K.; Li, Y.; Lee, J.S.; Gonik, A.M.; Dong, T.; Fung, G.; Sanchez, E.; Xing, L.; Cheng, H.R.; Luo, J.; et al. “OA02” peptide facilitates the precise targeting of paclitaxel-loaded micellar nanoparticles to ovarian cancer in vivo. Cancer Res. 2012, 72, 2100–2110. [Google Scholar] [CrossRef]
- Sawant, R.R.; Torchilin, V.P. Enhanced cytotoxicity of TATp-bearing paclitaxel-loaded micelles in vitro and in vivo. Int. J. Pharm. 2009, 31, 114–118. [Google Scholar] [CrossRef]
- Zheng, N.; Dai, W.; Du, W.; Zhang, H.; Lei, L.; Zhang, H.; Wang, X.; Wang, J.; Zhang, X.; Gao, J.; et al. A novel lanreotide-encoded micelle system targets paclitaxel to the tumors with overexpression of somatostatin receptors. Mol. Pharm. 2012, 9, 1175–1188. [Google Scholar]
- Wang, Z.; Yu, Y.; Ma, J.; Zhang, H.; Zhang, H.; Wang, X.; Wang, J.; Zhang, X.; Zhang, Q. LyP-1 modification to enhance delivery of artemisinin or fluorescent probe loaded polymeric micelles to highly metastatic tumor and its lymphatics. Mol. Pharm. 2012, 9, 2646–2657. [Google Scholar] [CrossRef]
- Wu, X.L.; Kim, J.H.; Koo, H.; Bae, S.M.; Shin, H.; Kim, M.S.; Lee, B.H.; Park, R.W.; Kim, I.S.; Choi, K.; et al. Tumor-targeting peptide conjugated pH-responsive micelles as a potential drug carrier for cancer therapy. Bioconjug. Chem. 2010, 21, 208–213. [Google Scholar] [CrossRef]
- Lu, Y.; Low, P.S. Folate-mediated delivery of macromolecular anticancer therapeutic agents. Adv. Drug Deliv. Rev. 2012, 64, 342–352. [Google Scholar] [CrossRef]
- Daniels, T.R.; Bernabeu, E.; Rodríguez, J.A.; Patel, S.; Kozman, M.; Chiappetta, D.A.; Holler, E.; Ljubimova, J.Y.; Helguera, G.; Penichet, M.L. The transferrin receptor and the targeted delivery of therapeutic agents against cancer. Biochim. Biophys. Acta 2012, 1820, 291–317. [Google Scholar] [CrossRef]
- Desgrosellier, J.S.; Cheresh, D.A. Integrins in cancer: biological implications and therapeutic opportunities. Nat. Rev. Cancer 2010, 10, 9–22. [Google Scholar] [CrossRef]
- Curnis, F.; Arrigoni, G.; Sacchi, A.; Fischetti, L.; Arap, W.; Pasqualini, R.; Corti, A. Differential binding of drugs containing the NGR motif to CD13 isoforms in tumor vessels, epithelia, and myeloid cells. Cancer Res. 2002, 62, 867–874. [Google Scholar]
- Lee, T.Y.; Lin, C.T.; Kuo, S.Y.; Chang, D.K.; Wu, H.C. Peptide-mediated targeting to tumor blood vessels of lung cancer for drug delivery. Cancer Res. 2007, 67, 10958–10965. [Google Scholar] [CrossRef]
- Bolhassani, A. Potential efficacy of cell-penetrating peptides for nucleic acid and drug delivery in cancer. Biochim. Biophys. Acta 2011, 1816, 232–246. [Google Scholar]
- Sun, L.C.; Coy, D.H. Somatostatin receptor-targeted anti-cancer therapy. Curr. Drug Deliv. 2011, 8, 2–10. [Google Scholar] [CrossRef]
- Laakkonen, P.; Porkka, K.; Hoffman, J.A.; Ruoslahti, E. A tumor-homing peptide with a targeting specificity related to lymphatic vessels. Nat. Med. 2002, 8, 751–755. [Google Scholar]
- Leland, P.; Taguchi, J.; Husain, S.R.; Kreitman, R.J.; Pastan, I.; Puri, R.K. Human breast carcinoma cells express type II IL-4 receptors and are sensitive to antitumor activity of a chimeric IL-4-Pseudomonas exotoxin fusion protein in vitro and in vivo. Mol. Med. 2000, 6, 165–178. [Google Scholar]
- Van Nostrum, C.F. Covalently cross-linked amphiphilic block copolymer micelles. Soft Matter 2011, 7, 3246–3259. [Google Scholar] [CrossRef]
- Cheng, R.; Feng, F.; Meng, F.; Deng, C.; Feijen, J.; Zhong, Z. Glutathione-responsive nano-vehicles as a promising platform for targeted intracellular drug and gene delivery. J. Control. Release 2011, 152, 2–12. [Google Scholar] [CrossRef]
- Lee, S.Y.; Kim, S.; Tyler, J.Y.; Park, K.; Cheng, J.X. Blood-stable, tumor-adaptable disulfide bonded mPEG-(Cys)4-PDLLA micelles for chemotherapy. Biomaterials 2013, 34, 552–561. [Google Scholar] [CrossRef]
- Li, Y.; Xiao, K.; Luo, J.; Xiao, W.; Lee, J.S.; Gonik, A.M.; Kato, J.; Dong, T.A.; Lam, K.S. Well-defined, reversible disulfide cross-linked micelles for on-demand paclitaxel delivery. Biomaterials 2011, 32, 6633–6645. [Google Scholar]
- Koo, A.N.; Min, K.H.; Lee, H.J.; Lee, S.U.; Kim, K.; Kwon, I.C.; Cho, S.H.; Jeong, S.Y.; Lee, S.C. Tumor accumulation and antitumor efficacy of docetaxel-loaded core-shell-corona micelles with shell-specific redox-responsive cross-links. Biomaterials 2012, 33, 1489–1499. [Google Scholar]
- Talelli, M.; Iman, M.; Varkouhi, A.K.; Rijcken, C.J.; Schiffelers, R.M.; Etrych, T.; Ulbrich, K.; van Nostrum, C.F.; Lammers, T.; Storm, G.; et al. Core-crosslinked polymeric micelles with controlled release of covalently entrapped doxorubicin. Biomaterials 2010, 31, 7797–7804. [Google Scholar] [CrossRef]
- Fernandez, C.A.; Rice, K.G. Engineered nanoscaled polyplex gene delivery systems. Mol. Pharm. 2009, 6, 1277–1289. [Google Scholar] [CrossRef]
- Osada, K.; Christie, R.J.; Kataoka, K. Polymeric micelles from poly (ethylene glycol)–poly (amino acid) block copolymer for drug and gene delivery. J. R. Soc. Interface 2009, 6, S325–S339. [Google Scholar] [CrossRef]
- Lee, S.H.; Chung, B.H.; Park, T.G.; Nam, Y.S.; Mok, H. Small-interfering RNA (siRNA)-based functional micro-and nanostructures for efficient and selective gene silencing. Acc. Chem. Res. 2012, 45, 1014–1025. [Google Scholar] [CrossRef]
- Davidson, B.L.; McCray, P.B., Jr. Current prospects for RNA interference-based therapies. Nat. Rev. Genet. 2011, 329–340. [Google Scholar] [CrossRef]
- Oba, M.; Vachutinsky, Y.; Miyata, K.; Kano, M.R.; Ikeda, S.; Nishiyama, N.; Itaka, K.; Miyazono, K.; Koyama, H.; Kataoka, K. Antiangiogenic gene therapy of solid tumor by systemic injection of polyplex micelles loading plasmid DNA encoding soluble Flt-1. Mol. Pharm. 2010, 7, 501–509. [Google Scholar]
- Vachutinsky, Y.; Oba, M.; Miyata, K.; Hiki, S.; Kano, M.R.; Nishiyama, N.; Koyama, H.; Miyazono, K.; Kataoka, K. Antiangiogenic gene therapy of experimental pancreatic tumor by sFlt-1 plasmid DNA carried by RGD-modified crosslinked polyplex micelles. J. Control. Release 2011, 149, 51–57. [Google Scholar] [CrossRef]
- Kumagai, M.; Shimoda, S.; Wakabayashi, R.; Kunisawa, Y.; Ishii, T.; Osada, K.; Itaka, K.; Nishiyama, N.; Kataoka, K.; Nakano, K. Effective transgene expression without toxicity by intraperitoneal administration of PEG-detachable polyplex micelles in mice with peritoneal dissemination. J. Control. Release 2012, 2012, 542–551. [Google Scholar]
- Christie, R.J.; Matsumoto, Y.; Miyata, K.; Nomoto, T.; Fukushima, S.; Osada, K.; Halnaut, J.; Pittella, F.; Kim, H.J.; Nishiyama, N.; et al. Targeted polymeric micelles for siRNA treatment of experimental cancer by intravenous injection. ACS Nano 2012, 6, 5174–5189. [Google Scholar] [CrossRef]
- Kim, S.H.; Jeong, J.H.; Lee, S.H.; Kim, S.W.; Park, T.G. Local and systemic delivery of VEGF siRNA using polyelectrolyte complex micelles for effective treatment of cancer. J. Control. Release 2008, 129, 107–116. [Google Scholar] [CrossRef]
- Mao, C.Q.; Du, J.Z.; Sun, T.M.; Yao, Y.D.; Zhang, P.Z.; Song, E.W.; Wang, J. A biodegradable amphiphilic and cationic triblock copolymer for the delivery of siRNA targeting the acid ceramidase gene for cancer therapy. Biomaterials 2011, 32, 3124–3133. [Google Scholar] [CrossRef]
- Sun, T.M.; Du, J.Z.; Yao, Y.D.; Mao, C.Q.; Dou, S.; Huang, S.Y.; Zhang, P.Z.; Leong, K.W.; Song, E.W.; Wang, J. Simultaneous delivery of siRNA and paclitaxel via a “two-in-one” micelleplex promotes synergistic tumor suppression. ACS Nano 2011, 5, 1483–1494. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tan, C.; Wang, Y.; Fan, W. Exploring Polymeric Micelles for Improved Delivery of Anticancer Agents: Recent Developments in Preclinical Studies. Pharmaceutics 2013, 5, 201-219. https://doi.org/10.3390/pharmaceutics5010201
Tan C, Wang Y, Fan W. Exploring Polymeric Micelles for Improved Delivery of Anticancer Agents: Recent Developments in Preclinical Studies. Pharmaceutics. 2013; 5(1):201-219. https://doi.org/10.3390/pharmaceutics5010201
Chicago/Turabian StyleTan, Chalet, Yingzhe Wang, and Wei Fan. 2013. "Exploring Polymeric Micelles for Improved Delivery of Anticancer Agents: Recent Developments in Preclinical Studies" Pharmaceutics 5, no. 1: 201-219. https://doi.org/10.3390/pharmaceutics5010201




