Tissue Engineered Human Skin Equivalents
Abstract
:1. Human Skin and Skin Barrier
- (a)
- through stratum corneum via intercellular route and/or intracellular route, and then through the viable epidermis and dermis via partitioning/diffusion;
- (b)
- through the appendageal pathway.
2. Tissue Engineered Human Skin Equivalents
2.1. Tissue Engineering and Regenerative Medicine
2.2. Need and Criteria for Human Skin Equivalents
| Brand | Company | FDA Approval | Product Description |
|---|---|---|---|
| 1. Clinical skin replacements and grafts | |||
| Integra® DRT (Dermal Regeneration Template) | Integra Lifesciences | 1996 | Thin silicone film covering a porous matrix of cow collagen and glycosaminoglycan |
| Apligraf® | Organogenisis | 1998 | Fibroblasts and collagen combined in dermal matrix onto which keratinocytes are seeded to form an epidermal layer |
| Epicel® | Genzyme | 2007 | Autologous keratinocytes grown ex vivo in the presence of proliferation-arrested mouse fibroblasts |
| Transcyte®/Dermagraft® | Advanced Tissue Sciences/Advanced Biohealing | 1997/2001 | Cryopreserved dermal substitute: human fibroblast seeded onto polymer mesh and cultured ex vivo |
| Orcel® | FortiCell Bioscience | 2001/2008 | Human epidermal keratinocytes and dermal fibroblasts are cultured in separate layers into a Type I bovine collagen sponge |
| Alloderm®/Strattice® | LifeCell Co. | None | Acellular cadaver skin matrix |
| StrataGraft® | StrataTech | None | Full thickness skin substitute where a near-diploid human keratinocytes cell line, NIKS, was utilized. |
| 2. In Vitro Permeation and Toxicity Screening Models | |||
| SkinEthic Rhe (Reconstructed Human Epidermis) | SkinEthic | Human keratinocytes cultured on an inert polycarbonate filter at the air-liquid interface in chemically defined medium | |
| Episkin | SkinEthic | Human keratinocytes cultured on a collagen base which permit terminal differentiation and reconstruction of the epidermis with a functional stratum corneum | |
| Epiderm | MatTek | Neonatal human-derived epidermal keratinocytes (NHEK) cultured to form a multi-layered, highly differentiated model of the human epidermis | |
| EpidermFT | MatTek | Neonatal human-derived dermal fibroblasts (NHFB) and NHEK co-cultured to form a multi-layered, highly differentiated model of the human dermis and epidermis | |
| StrataTest | StrataTech | Full thickness skin model where a near-diploid human keratinocytes cell line, NIKS, was utilized. | |
| Epidermal Skin Test 1000 (EST1000) | CellSystems Biotechnologie GmbH | Reconstructed epidermal model made from primary human keratinocytes; it comprises a fully differentiated epidermis with viable and cornifiedcell layers | |
| Advanced Skin Test 2000 (AST2000) | CellSystems Biotechnologie GmbH | It comprises a dermal equivalent with embedded fibroblasts as a basis and epidermal layer of keratinocytes on top; it is a full thickness model. | |
2.3. Commercially Available Human Skin Equivalents for in Vivo Applications: Clinical Skin Replacements and Skin Grafts
2.3.1. Epidermal Models
2.3.2. Dermal Models
2.3.3. Full-Thickness Models
2.4. Commercially Available Human Skin Equivalents for in Vitro Applications: Models for Drug Permeability Tests and Toxicity Screening
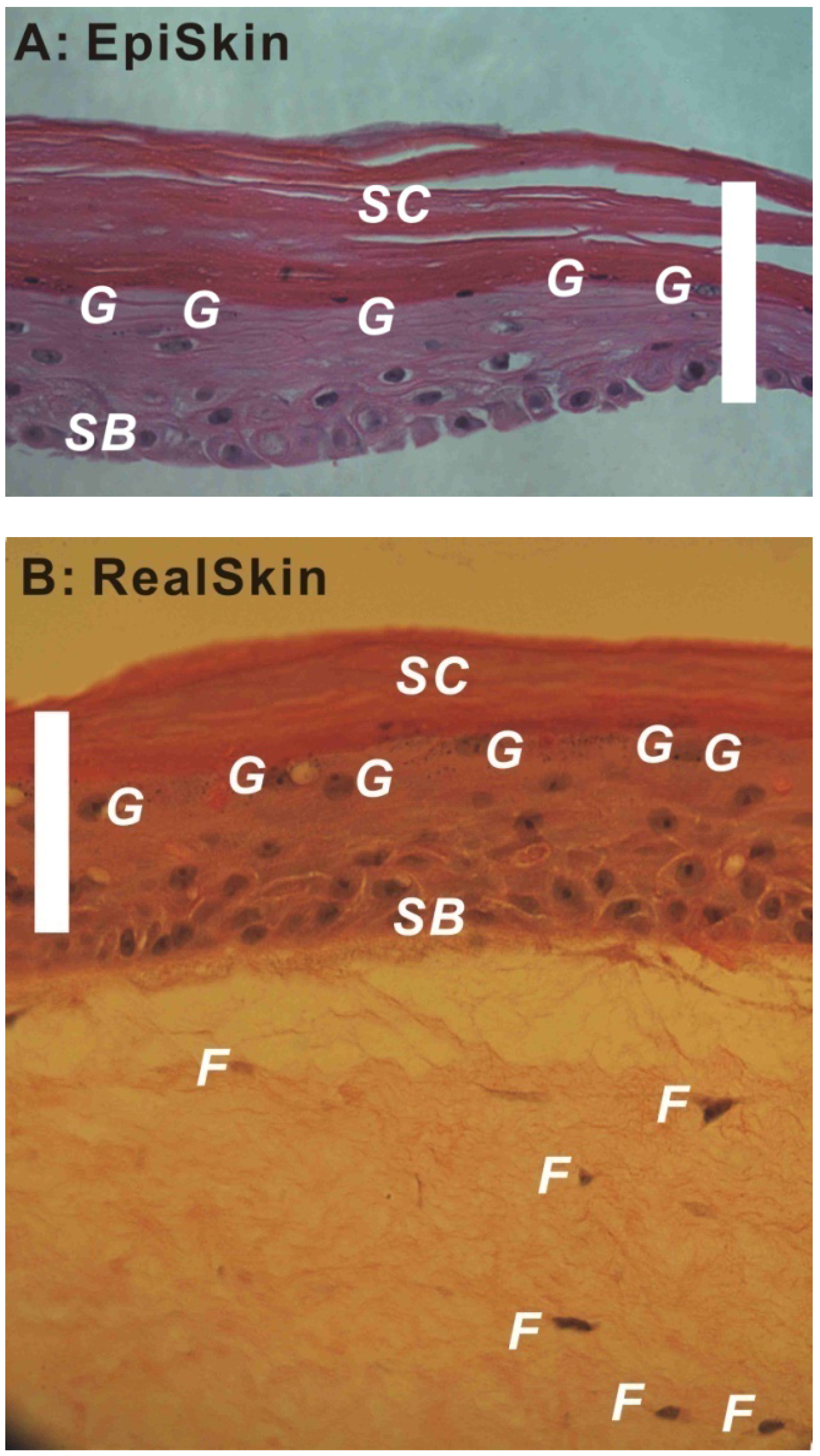
| Brand of HSE Models | Company | US Regulatory Acceptance/Endorsement by NICEATM-ICCVAM [58] | EU Regulatory Acceptance/Endorsement by ECVAM [59] |
|---|---|---|---|
| Skin Corrosivity Test | |||
| EpiSkinTM | SkinEthic | OECD Test Guideline 431 accepted in 2004 | Commission Regulation (EC) No 440/2008; OECD Test Guideline 431 (April 1998) |
| EpidermTM | MatTek | OECD Test Guideline 431 accepted in 2004 | Commission Regulation (EC) No 440/2008; OECD Test Guideline 431 (March 2000) |
| SkinEthicTM Rhe | SkinEthic | OECD Test Guideline 431 (meets performance standards 2006) | Commission Regulation (EC) No 440/2008; OECD Test Guideline 431 (November 2006) |
| EST1000 | CellSystems Biotechnologie GmbH | OECD Test Guideline 431 (meets performance standards 2009) | Commission Regulation (EC) No 440/2008; OECD Test Guideline 431 (June 2009) |
| Skin Irritation Test | |||
| EpiSkinTM | SkinEthic | OECD Test Guideline 439 accepted in 2010 | Commission Regulation (EC) Nr 761/2009; OECD Test Guideline 439 (April 2007) |
| EpidermTM | MatTek | OECD Test Guideline 439 accepted in 2010 | Commission Regulation (EC) Nr 761/2009; OECD Test Guideline 439 (April 2007; modified Skin Irritation Test Method validated in November 2008) |
| SkinEthicTM Rhe | SkinEthic | OECD Test Guideline 439 accepted in 2010 | Commission Regulation (EC) Nr 761/2009; OECD Test Guideline 439 (November 2008) |
2.5. Development of Non-Commercial Human Skin Equivalents
4. Perspectives
Acknowledgements
Conflict of Interest
References
- Holbrook, K.A.; Wolff, K. The Structure and Development of Skin. In Dermatology in General Medicine, 6th; Fitzpatrick, T.B., Eisen, A.Z., Wolff, K., Freedberg, I.M., Austen, K.F., Eds.; McGraw-Hill: New York, NY, USA, 1993. [Google Scholar]
- Scheuplein, R.J.; Blank, I.H. Permeability of the skin. Physiol. Rev. 1971, 51, 702–747. [Google Scholar]
- Briggaman, R.A.; Wheeler, C.E., Jr. The epidermal-dermal junction. J. Invest. Dermatol. 1975, 65, 71–84. [Google Scholar]
- Parenteau, N.L.; Hardin-Young, J.; Ross, R.N. Skin. In Principles of Tissue Engineering, 2nd; Lanza, R.P., Langer, R., Vacanti, J.P., Eds.; Academic Press: San Diego, CA, USA, 2000; pp. 879–890. [Google Scholar]
- Hadgraft, J.; Guy, R.H. Feasibility Assessment in Topical and Transdermal Delivery: Mathematical Models and in Vitro Studies. In Transdermal Drug Delivery, 2nd; Guy, R.H., Hadgraft, J., Eds.; Marcel Dekker, Inc.: New York, NY, USA, 2003; Chapter 1. [Google Scholar]
- Zhang, Q.; Grice, J.E.; Li, P.; Jepps, O.G.; Wang, G.-J.; Roberts, M.S. Skin solubility determines maximum transepidermal flux for similar size molecules. Pharm. Res. 2009, 26, 1974–1985. [Google Scholar] [CrossRef]
- Vacanti, J.P.; Vacanti, C.A. The History and Scope of Tissue Engineering. In Principles of Tissue Engineering, 2nd; Lanza, R.P., Langer, R., Vacanti, J.P., Eds.; Academic Press: San Diego, CA, USA, 2000; pp. 3–7. [Google Scholar]
- Langer, R.; Vacanti, J.P. Tissue engineering. Science 1993, 260, 920–926. [Google Scholar]
- Rheinwald, J.G.; Green, H. Serial cultivation of strains of human epidermal keratinocytes: The Formation of keratinizing colonies from single cells. Cell 1975, 6, 331–337. [Google Scholar] [CrossRef]
- Rheinwald, J.G.; Green, H. Epidermal growth factor and the multiplication of cultured human epidermal keratinocytes. Nature 1977, 264, 421–424. [Google Scholar] [CrossRef]
- Green, H.; Kehinde, O.; Thomas, J. Growth of cultured human epidermal cells into multiple epithelia suitable for grafting. Proc. Natl. Acad. Sci. USA 1979, 76, 5665–5668. [Google Scholar] [CrossRef]
- Bell, E.; Ehrlich, H.P.; Buttle, D.J.; Nakatsuji, T. Living tissue formed in vitro and accepted as skin-equivalent tissue of full thickness. Science 1981, 211, 1052–1054. [Google Scholar]
- Deuel, T.F.; Kawahara, R.S.; Mustoe, T.A.; Pierce, A.F. Growth factors and wound healing: Platelet-derived growth factor as a model cytokine. Annu. Rev. Med. 1991, 42, 567–584. [Google Scholar] [CrossRef]
- Freed, L.E.; Hollander, A.P.; Martin, I.; Barry, J.R.; Langer, R.; Vunjak-Novakovic, G. Chondrogenesis in a cell-polymer-bioreactor system. Exp. Cell Res. 1998, 240, 58–65. [Google Scholar] [CrossRef]
- Freed, L.E.; Vunjak-Novakovic, G. Tissue Engineering Bioreactors. In Principles of Tissue Engineering, 2nd; Lanza, R.P., Langer, R., Vacanti, J.P., Eds.; Academic Press: San Diego, CA, USA, 2000; pp. 143–156, Chapter 13. [Google Scholar]
- Peppas, N.A.; Langer, R. New challenges in biomaterials. Science 1994, 263, 1715–1720. [Google Scholar]
- Kohn, J.; Langer, R. Polymerization reactions involving the side chains of α-l-amino acids. J. Am. Chem. Soc. 1987, 109, 817–820. [Google Scholar] [CrossRef]
- Pachence, J.M.; Kohn, J. Biodegradable Polymers. In Principles of Tissue Engineering, 2nd; Lanza, R.P., Langer, R., Vacanti, J.P., Eds.; Academic Press: San Diego, CA, USA, 2000; pp. 263–277. [Google Scholar]
- Lee, K.Y.; Mooney, D.J. Hydrogels for tissue engineering. Chem. Rev. 2001, 101, 1869–1879. [Google Scholar] [CrossRef]
- Drury, J.L.; Mooney, D.J. Hydrogels for tissue engineering: Scaffold design variables and applications. Biomaterials 2003, 24, 4337–4351. [Google Scholar] [CrossRef]
- Peppas, N.A.; Carr, D.A. Impact of absorption and transport on intelligent therapeutics and nano-scale delivery of protein therapeutic agents. Chem. Eng. Sci. 2009, 64, 4553–4565. [Google Scholar] [CrossRef]
- Baptista, P.M.; Orlando, G.; Mirmalek-Sani, S.H.; Siddiqui, M.; Atala, A.; Soker, S. Whole Organ Decellularization- a Tool for Bioscaffold Fabrication and Organ Bioengineering. In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC ’09), Minneapolis, MN, USA, 3–6 September 2009; pp. 6526–6529.
- Atala, A.; Bauer, S.B.; Soker, S.; Yoo, J.J.; Retik, A.B. Tissue-engineered autologous bladders for patients needing cystoplasty. Lancet 2006, 367, 1241–1246. [Google Scholar] [CrossRef]
- Tissue Engineering and Cell Transplantation: US Markets for Skin Replacements and Substitutes; Report #A426; Medtech Insight: Bridgeport, PA, USA, August 2010.
- Guidance Document for the Conduct of Skin Absorption Studies. In OECD Environmental Health and Safety Publications, Series on Testing and Assessment, No. 28; Organisation for Economic Co-Operation and Development: Paris, France, 2004.
- Panchagnula, R.; Stemmer, K.; Ritschel, W.A. Animal models for transdermal drug delivery. Methods Find. Exp. Clin. Pharmacol. 1997, 19, 335–341. [Google Scholar]
- Kiowski, G.; Biedermann, T.; Widmer, D.S.; Civenni, G.; Burger, C.; Dummer, R.; Sommer, L.; Reichmann, E. Engineering melanoma progression in a humanized environment in vivo. J. Invest. Dermatol. 2011, 132, 144–153. [Google Scholar]
- Commandeur, S.; Ho, S.H.; de Gruijl, F.R.; Willemze, R.; Tensen, C.P.; EI-Ghalbzouri, A. Functional characterization of cancer-associated fibroblasts of human cutaneous squamous cell carcinoma. Exp. Dermatol. 2011, 20, 737–742. [Google Scholar]
- Tjabringa, G.; Bergers, M.; van Rens, D.; de Boer, R.; Lamme, E.; Schalkwijk, J. Development and validation of human psoriatic skin equivalents. Am. J. Pathol. 2008, 173, 815–823. [Google Scholar] [CrossRef] [Green Version]
- MacNeil, S. Progress and opportunities for tissue-engineered skin. Nature 2007, 445, 874–880. [Google Scholar] [CrossRef]
- Groen, D.; Poole, D.S.; Gooris, G.S.; Bouwstra, J.A. Is an orthorhombic lateral packing and a proper lamellar organization important for the skin barrier function? Biochim. Biophys. Acta 2011, 1808, 1529–1537. [Google Scholar] [CrossRef]
- Nowak, J.A.; Fuchs, E. Isolation and culture of epithelial stem cells. Methods Mol. Biol. 2009, 482, 215–232. [Google Scholar] [CrossRef]
- Midwood, K.S.; Williams, L.V.; Schwarzbauer, J.E. Tissue repair and the dynamics of the extracellular matrix. Int. J. Biochem. Cell Biol. 2004, 36, 1031–1037. [Google Scholar] [CrossRef]
- Herndon, D.N.; Barrow, R.E.; Rutan, R.L.; Rutan, T.C.; Desai, M.H.; Abston, S. A comparison of conservative versus early excision. Therapies in severely burned patients. Ann. Surg. 1989, 209, 547–553. [Google Scholar] [CrossRef]
- Gallico, G.G.; O’Connor, N.E. Engineering a skin replacement. Tissue Eng. 1995, 1, 231–240. [Google Scholar] [CrossRef]
- Groeber, F.; Holeiter, M.; Hampel, M.; Hinderer, S.; Schenke-Layland, K. Skin tissue engineering- in vivo and in vitro applications. Adv. Drug Deliv. Rev. 2011, 63, 352–366. [Google Scholar] [CrossRef]
- O’Conner, N.E.; Mulliken, J.B.; Banks-Schlegel, S.; Kehinde, O.; Green, H. Grafting of burns with cultured epithelium prepared from autologous epidermal cells. Lancet 1981, 317, 75–78. [Google Scholar]
- Purdue, G.F. Dermagraft-TC pivotal efficacy and safety study. J. Burn Care Rehabil. 1997, 18, S13–S14. [Google Scholar] [CrossRef]
- Gentzkow, G.D.; Iwasaki, S.D.; Hershon, K.S.; Mengel, M.; Prendergast, J.J.; Ricotta, J.J.; Steed, D.P.; Lipkin, S. Use of dermagraft: A cultured human dermis to treat diabetic foot ulcers. Diabetes Care 1996, 19, 350–354. [Google Scholar]
- Cuono, C.; Langdon, R.; McGuire, J. Use of cultured epidermal autografts and dermal allografts as skin replacement after burn injury. Lancet 1986, 327, 1123–1124. [Google Scholar] [CrossRef]
- Heimbach, D.; Luterman, A.; Burke, J.; Cram, A.; Herndon, D.; Hunt, J.; Jordan, M.; McManus, W.; Solem, L.; Warden, G.; Zawacki, B. Artificial dermis for major burns: A multi-center randomized clinical trial. Ann. Surg. 1988, 208, 313–320. [Google Scholar] [CrossRef]
- Shevchenko, R.V.; James, S.L.; James, S.E. A review of tissue-engineered skin bioconstructs available for skin reconstruction. J. R. Soc. Interface 2010, 7, 229–258. [Google Scholar] [CrossRef]
- Maruguchi, T.; Maruguchi, Y.; Suzuki, S.; Matsuda, K.; Toda, K.; Isshiki, N. A new skin equivalent: Keratinocytes proliferated and differentiated on collagen sponge containing fibroblasts. Plast. Reconstr. Surg. 1994, 93, 537–544. [Google Scholar]
- Eaglstein, W.H.; Iriondo, M.; Laszlo, K. A composite skin substitute (graftskin) for surgical wounds: A clinical experience. Dermatol. Surg. 1995, 21, 839–843. [Google Scholar] [CrossRef]
- Sabolinski, M.L.; Alvarez, O.; Auletta, M.; Mulder, G.; Parenteau, N.L. Cultured skin as a “smart material” for healing wounds: Experience in venous ulcers. Biomaterials 1996, 17, 311–320. [Google Scholar] [CrossRef]
- Bello, Y.M.; Falabella, A.F. The role of graftskin (apligraf) in difficult-to-heal venous leg ulcers. J. Wound Care 2002, 11, 182–183. [Google Scholar]
- Boyce, S.T.; Kagen, R.J.; Greenhalgh, D.G.; Warner, P.; Yakuboff, K.P.; Palmieri, T.; Warden, G.D. Cultured skin substitutes reduce requirements for harvesting of skin autograft for closure of excised, full-thickness burns. J. Trauma 2006, 60, 821–829. [Google Scholar]
- Ponec, M. Skin constructs for replacement of skin tissues for in vitro testing. Adv. Drug Deliv. Rev. 2002, 54, S19–S30. [Google Scholar] [CrossRef]
- Johsnon, E.W.; Meunier, S.F.; Roy, C.J.; Parenteau, N.L. Serial cultivation of normal human keratinocytes: A defined system for studying the regulation of growth and differentiation. In Vitro Cell. Dev. Biol. 1992, 28, 429–435. [Google Scholar] [CrossRef]
- Boehnke, K.; Mirancea, N.; Pavesio, A.; Fusenig, N.E.; Boukamp, P.; Stark, H.J. Effects of fibroblasts and microenvironment on epidermal regeneration and tissue function in long-term skin equivalents. Eur. J. Cell Biol. 2007, 86, 731–746. [Google Scholar] [CrossRef]
- Xie, Y.; Rizzi, S.C.; Dawson, R.; Lynam, E.; Richards, S.; Leavesley, D.I.; Upton, Z. Development of a three-dimensional human skin equivalent wound model for investigating novel wound healing therapies. Tissue Eng. Part C Method 2010, 16, 1111–1123. [Google Scholar] [CrossRef] [Green Version]
- Oliver, G.J.; Pemberton, M.A.; Rhodes, C. An in vitro model for identifying skin-corrosive chemicals. 1. Initial validation. Toxicol. in Vitro 1988, 2, 7–17. [Google Scholar] [CrossRef]
- Lelievre, D.; Justine, P.; Christiaens, F.; Bonaventure, N.; Coutet, J.; Marrot, L.; Cotovio, J. The Episkin Phototoxicity Assay (EPA): Development of an in vitro tiered strategy using 17 reference chemicals to predict phototoxic potency. Toxicol. in Vitro 2007, 21, 977–995. [Google Scholar] [CrossRef]
- Gabbanini, S.; Lucchi, E.; Carli, M.; Berlini, E.; Minghetti, A.; Valgimigli, L. In vitro evaluation of the permeation through reconstructed human epidermis of essential oils from cosmetic formulations. J. Pharm. Biomed. Anal. 2009, 50, 370–376. [Google Scholar] [CrossRef]
- de Jager, M.; Groenink, W.; van der Spek, J.; Janmaat, C.; Gooris, G.; Ponec, M.; Bouwstra, J. Preparation and characterization of a stratum corneum substitute for in vitro precutaneuous penetration studies. Biochim. Biophys. Acta 1758, 636–644. [Google Scholar]
- Wertz, P.W. Epidermal lipids. Semin. Dermatol. 1992, 11, 106–113. [Google Scholar]
- Vicanova, J.; Boelsma, E.; Mommaas, A.M.; Kempenaar, J.A.; Forslind, B.; Pallon, J.; Egelrud, T.; Koerten, H.K.; Ponec, M. Normalization of epidermal calcium distribution profile in reconstructed human epidermis is related to improvement of terminal differentiation and stratum corneum barrier formation. J. Invest. Dermatol. 1998, 111, 97–106. [Google Scholar]
- US and International Acceptance of Alternative Methods, 1998–2011 Chronological List, published by NICEATM-ICCVAM. Available online: http://iccvam.niehs.nih.gov/about/accept.htm (accessed on 23 December 2011).
- The Regulatory Acceptance for Skin Corrosion and Skin Irritation can be found at the Method Validation website of ECVAM. Available online: http://ecvam.jrc.it/ (accessed on 23 December 2011).
- Stark, H.J.; Boehnke, K.; Mirancea, N.; Willhauck, M.J.; Pavesio, A.; Fuesnig, N.E.; Boukamp, P. Epidermal homeostasis in long-term scaffold-enforced skin equivalents. J. Investig. Dermatol. Symp. Proc. 2006, 11, 93–105. [Google Scholar] [CrossRef]
- EI-Ghalbzouri, A.; Lamme, E.N.; van Blitterswijk, C.; Koopman, J.; Ponec, M. The use of PEGT/PBT as a dermal scaffold for skin tissue engineering. Biomaterials 2004, 25, 2987–2996. [Google Scholar] [CrossRef]
- Thakoersing, V.S.; Ponec, M.; Bouwstra, J.A. Generating of human skin equivalents under submerged conditions- mimicking the in utero environment. Tissue Eng. Part A 2010, 16, 1433–1441. [Google Scholar] [CrossRef]
- Ponec, M.; Weerheim, A.; Kempenaar, J.; Mulder, A.; Gooris, G.S.; Bouwstra, J.; Mommaas, A.M. The formation of competent barrier lipids in reconstructed human epidermis requires the presence of vitamin C. J. Invest. Dermatol. 1997, 109, 348–355. [Google Scholar]
- Thakoersing, V.S.; Gooris, G.S.; Mulder, A.; Rietveld, M.; EI-Ghalbzouri, A.; Bouwstra, J.A. Unraveling barrier properties of three different in-house human skin equivalents. Tissue Eng. Part C 2012, 18, 1–11. [Google Scholar]
- Andreadis, S.T.; Hamoen, K.E.; Yarmush, M.L.; Morgan, J.R. Keratinocyte growth factor induces hyperproliferation and delays differentiation in a skin equivalent model system. FASEB J. 2001, 15, 898–906. [Google Scholar] [CrossRef]
- Schmook, F.P.; Meingassner, J.G.; Billich, A. Comparison of human skin or epidermis models with human and animal skin in in-vitro percutaneous absorption. Int. J. Pharm. 2001, 215, 51–56. [Google Scholar] [CrossRef]
- Roy, S.D.; Fujiki, J.; Fleitman, J.S. Permeabilities of alkyl p-amino-benzoates through living skin equivalent and cadaver skin. J. Pharm. Sci. 1993, 82, 1266–1268. [Google Scholar] [CrossRef]
- Batheja, P.; Song, Y.; Wertz, P.; Michniak-Kohn, B. Effects of growth conditions on the barrier properties of a human skin equivalent. Pharm. Res. 2009, 26, 1689–1700. [Google Scholar] [CrossRef]
- Wilkins, L.M.; Watson, S.R.; Prosky, S.J.; Meunier, S.F.; Parenteau, N.L. Development of a bilayered living skin construct for clinical applications. Biotechnol. Bioeng. 1994, 43, 747–756. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, Z.; Michniak-Kohn, B.B. Tissue Engineered Human Skin Equivalents. Pharmaceutics 2012, 4, 26-41. https://doi.org/10.3390/pharmaceutics4010026
Zhang Z, Michniak-Kohn BB. Tissue Engineered Human Skin Equivalents. Pharmaceutics. 2012; 4(1):26-41. https://doi.org/10.3390/pharmaceutics4010026
Chicago/Turabian StyleZhang, Zheng, and Bozena B. Michniak-Kohn. 2012. "Tissue Engineered Human Skin Equivalents" Pharmaceutics 4, no. 1: 26-41. https://doi.org/10.3390/pharmaceutics4010026




