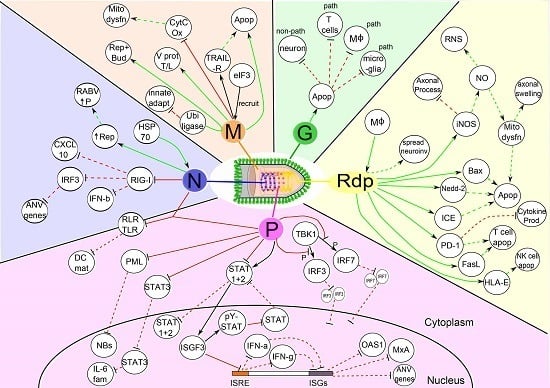
Subversion of the Immune Response by Rabies Virus
Abstract
:1. Introduction
2. Experimental and Non-Experimental Factors Influencing the Immune Response
2.1. Age
2.2. Timing
2.3. Sex
2.4. Temperature
2.5. Genetic Polymorphisms
2.6. The Appropriate Animal Model and Other Influential Factors
2.7. Cerebral Lateralization
3. Rabies Entry into the Nervous System: The Use of Stealth
4. Apoptosis: Detrimental or Beneficial to the Host?
4.1. Neuronal Apoptosis as a Host Defence Mechanism
4.2. Apoptosis as a Mechanism for Immune Subversion
4.3. Regulation of Immunosubversive Molecules Involved in Apoptosis
4.4. Caspase-Dependent and -Independent Apoptotic Pathways
5. Contributing Factors and Mechanisms to RABV Pathogenesis
5.1. Nitric Oxide
5.2. Mitochondrial Dysfunction
5.3. Heat Shock Proteins
5.4. Glucocorticoids
6. The “Early Death” Phenomenon
7. How Does RABV Do It?—the Mechanisms by Which RABV Sequesters the Immune System
7.1. Phosphoprotein
7.1.1. Interaction of Phosphoprotein and Nuclear Bodies—Effects on Antiviral Responses
7.1.2. The Importance of the Nucleocytoplasmic Shuttling Abilities of RABV P Protein
7.1.3. RABV P Protein Impairs Phosphorylation of Interferon Regulatory Factor 3 and 7
7.1.4. Indirect Consequences of P Protein on the Innate and Adaptive Immune Response
7.2. Nucleoprotein
7.3. Matrix Protein
7.4. Inhibition of the Host’s Immune Response by RABV—The Bigger Picture
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Adamson, P. The spread of rabies into Europe and the probable origin of this disease in antiquity. J. R. Asiat. Soc. Gt. Br. Irel. 1977, 2, 140–144. [Google Scholar] [CrossRef]
- Pasteur, M.L. Méthode pour prévenir la rage après morsure. C. R. Seances L’Acad. Sci. 1885, 17, 765–772. [Google Scholar]
- Dreesen, D.W. A global review of rabies vaccines for human use. Vaccine 1997, 15, S2–S6. [Google Scholar] [CrossRef]
- Hemachudha, T.; Laothamatas, J.; Rupprecht, C.E. Human rabies: A disease of complex neuropathogenetic mechanisms and diagnostic challenges. Lancet 2002, 1, 101–109. [Google Scholar] [CrossRef]
- Turner, G.S.; Ballard, R. Interaction of mouse peritoneal macrophages with fixed rabies virus in vivo and in vitro. J. Gen. Virol. 1976, 30, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.C.; Park, H. Apoptotic cell death in experimental rabies in suckling mice. Acta Neuropathol. 1998, 95, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Habel, K. Evaluation of a mouse test for the standardization of the immunizing power of anti-rabies vaccines. Public Health Rep. 1940, 55, 1473–1487. [Google Scholar] [CrossRef]
- Casals, J. Influence of age factors on susceptibility of mice to rabies virus. J. Exp. Med. 1940, 72, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Aubert, M.F. Practical significance of rabies antibodies in cats and dogs. Rev. Sci. Tech. 1992, 11, 735–760. [Google Scholar] [CrossRef] [PubMed]
- Mansfield, K.L.; Burr, P.D.; Snodgrass, D.R.; Sayers, R.; Fooks, A.R. Factors affecting the serological response of dogs and cats to rabies vaccination. Vet. Rec. 2004, 154, 423–426. [Google Scholar] [CrossRef] [PubMed]
- HogenEsch, H.; Thompson, S.; Dunham, A.; Ceddia, M.; Hayek, M. Effect of age on immune parameters and the immune response of dogs to vaccines: A cross-sectional study. Vet. Immunol. Immunopathol. 2004, 97, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Mastroeni, I.; Vescia, N.; Pompa, M.G.; Cattaruzza, M.S.; Marini, G.P.; Fara, G.M. Immune response of the elderly to rabies vaccines. Vaccine 1994, 12, 518–520. [Google Scholar] [CrossRef]
- Jackson, A.C.; Warrell, M.J.; Rupprecht, C.E.; Ertl, H.C.J.; Dietzschold, B.; O’Reilly, M.; Leach, R.P.; Fu, Z.F.; Wunner, W.H.; Bleck, T.P.; et al. Management of rabies in humans. Clin. Infect. Dis. 2003, 36, 60–63. [Google Scholar] [CrossRef] [PubMed]
- Willoughby, R.E. “Early death” and the contraindication of vaccine during treatment of rabies. Vaccine 2009, 27, 7173–7177. [Google Scholar] [CrossRef] [PubMed]
- Klein, S.L. The effects of hormones on sex differences in infection: From genes to behavior. Neurosci. Behav. Rev. 2000, 24, 627–638. [Google Scholar] [CrossRef]
- Lodmell, D.L. Genetic control of resistance to street rabies virus in mice. J. Exp. Med. 1983, 157, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Wilde, H.; Chomchey, P.; Punyaratabandhu, P.; Phanupak, P.; Chutivongse, S. Purified equine rabies immune globulin: A safe and affordable alternative to human rabies immune globulin. Bull. World Health Organ. 1989, 67, 731–736. [Google Scholar] [PubMed]
- Suwansrinon, K.; Jaijareonsup, W.; Wilde, H.; Benjavongkulchai, M.; Sriaroon, C.; Sitprija, V. Sex- and age-related differences in rabies immunoglobulin hypersensitivity. Trans. R. Soc. Trop. Med. Hyg. 2007, 101, 206–208. [Google Scholar] [CrossRef] [PubMed]
- Karp, C.L. Unstressing intemperate models: How cold stress undermines mouse modeling. J. Exp. Infect. 2012, 209, 1069–1074. [Google Scholar]
- Bell, J.F.; Moore, G.J. Effects of high ambient temperature on various stages of rabies virus infection in mice. Infect. Immun. 1974, 10, 510–515. [Google Scholar] [PubMed]
- Wells, C.A.; Ravasi, T.; Faulkner, G.J.; Carninci, P.; Okazaki, Y.; Hayashizaki, Y.; Sweet, M.; Wainwright, B.J.; Hume, D.A. Genetic control of the innate immune response. BMC Immunol. 2003, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noverr, M.C.; Huffnagle, G.B. Does the microbiota regulate immune responses outside the gut? Trends Microbiol. 2004, 12, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Suja, M.S.; Mahadevan, A.; Madhusudana, S.N.; Shankar, S.K. Role of apoptosis in rabies viral encephalitis: A comparative study in mice, canine, and human brain with a review of literature. Pathol. Res. Int. 2011, 2011, 374286. [Google Scholar] [CrossRef] [PubMed]
- Consales, C.; Bolzan, V. Rabies review: Immunopathology, clinical aspects and treatment. J. Venom. Anim. Toxins Incl. Trop. Dis. 2007, 13, 35–38. [Google Scholar] [CrossRef]
- Jackson, A.C. Rabies virus infection: An update. J. Neurovirol. 2003, 9, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Murphy, F.A. Rabies pathogenesis. Arch. Virol. 1977, 54, 279–297. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.; Cunningham, A.F.; Fooks, A.R. The immune response to rabies virus infection and vaccination. Vaccine 2010, 28, 3896–3901. [Google Scholar] [CrossRef] [PubMed]
- Kelly, R.M.; Strick, P.L. Rabies as a transneuronal tracer of circuits in the central nervous system. J. Neurosci. Methods 2000, 103, 63–71. [Google Scholar] [CrossRef]
- Smart, N.L.; Charlton, K.M. The distribution of challenge virus standard rabies virus versus skunk street rabies virus in the brains of experimentally infected rabid skunks. Acta Neuropathol. 1992, 84, 501–508. [Google Scholar] [CrossRef] [PubMed]
- Srithayakumar, V.; Sribalachandran, H.; Rosatte, R.; Nadin-Davis, S.A.; Kyle, C.J. Innate immune responses in raccoons after raccoon rabies virus infection. J. Gen. Virol. 2014, 95 Pt 1, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Kucera, P.; Dolivo, M.; Coulon, P.; Flamand, A. Pathways of the early propagation of virulent and avirulent rabies strains from the eye to the brain. J. Virol. 1985, 55, 158–162. [Google Scholar] [PubMed]
- Lycke, E.; Tsiang, H. Rabies virus infection of cultured rat sensory neurons. J. Virol. 1987, 61, 2733–2741. [Google Scholar] [PubMed]
- Tsiang, H.; Ceccaldi, P.E.; Lycke, E. Rabies virus infection and transport in human sensory dorsal root ganglia neurons. J. Gen. Virol. 1991, 72, 1191–1194. [Google Scholar] [CrossRef] [PubMed]
- Siniscalchi, M.; Sasso, R.; Pepe, A.M.; Vallortigara, G.; Quaranta, A. Dogs turn left to emotional stimuli. Behav. Brain Res. 2010, 208, 516–521. [Google Scholar] [CrossRef] [PubMed]
- Siniscalchi, M.; Sasso, R.; Pepe, A.M.; Dimatteo, S.; Vallortigara, G.; Quaranta, A. Sniffing with the right nostril: Lateralization of response to odour stimuli by dogs. Anim. Behav. 2011, 82, 399–404. [Google Scholar] [CrossRef]
- Quaranta, A.; Siniscalchi, M.; Vallortigara, G. Asymmetric tail-wagging responses by dogs to different emotive stimuli. Curr. Biol. 2007, 17, R199–R201. [Google Scholar] [CrossRef] [PubMed]
- Reimchen, T.E.; Spoljaric, M.A. Right paw foraging bias in wild black bear (Ursus americanus kermodei). Laterality 2011, 16, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Komárková, M.; Bartošová, J. Lateralized suckling in domestic horses (Equus caballus). Anim. Cogn. 2013, 16, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Walker, S.F. Lateralization of functions in the vertebrate brain: A review. Br. J. Psychol. 1980, 71, 329–367. [Google Scholar] [CrossRef] [PubMed]
- Ghirlanda, S.; Vallortigara, G. The evolution of brain lateralization: A game-theoretical analysis of population structure. Proc. R. Soc. Lond. Biol. Sci. 2004, 271, 853–857. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Carlson, J.N.; Seegal, R.F.; Lawrence, D.A. Differential immune responses in mice with left- and right-turning preference. J. Neuroimmunol. 1999, 93, 164–171. [Google Scholar] [CrossRef]
- Neveu, P.J. Lateralization and stress responses in mice: Interindividual differences in the association of brain, neuroendocrine, and immune responses. Behav. Genet. 1996, 26, 373–377. [Google Scholar] [CrossRef] [PubMed]
- Neveu, P.J. Brain asymmetry in neural-immune interactions. Eur. Neuropsychopharmacol. 1991, S18–1, 367–369. [Google Scholar] [CrossRef]
- Quaranta, A.; Siniscalchi, M.; Frate, A.; Vallortigara, G. Paw preference in dogs: Relations between lateralised behaviour and immunity. Behav. Brain Res. 2004, 153, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Siniscalchi, M.; Sasso, R.; Pepe, A.M.; Dimatteo, S.; Vallortigara, G.; Quaranta, A. Catecholamine plasma levels following immune stimulation with rabies vaccine in dogs selected for their paw preferences. Neurosci. Lett. 2010, 476, 142–145. [Google Scholar] [CrossRef] [PubMed]
- Siniscalchi, M.; Cirone, F.; Guaricci, A.C.; Quaranta, A. Catecholamine plasma levels, IFN-γ serum levels and antibodies production induced by rabies vaccine in dogs selected for their paw preference. Laterality 2014, 19, 522–532. [Google Scholar] [PubMed]
- Elenkov, I.J.; Wilder, R.L.; Chrousos, G.P.; Vizi, E.S. The sympathetic nerve—An integrative interface between two supersystems: The brain and the immune system. Pharmacol. Rev. 2000, 52, 595–638. [Google Scholar] [PubMed]
- Baloul, L.; Camelo, S.; Lafon, M. Up-regulation of Fas ligand (FasL) in the central nervous system: A mechanism of immune evasion by rabies virus. J. Neurovirol. 2004, 10, 372–382. [Google Scholar] [CrossRef] [PubMed]
- Carson, M.J.; Doose, J.M.; Melchior, B.; Schmid, C.D.; Ploix, C.C. CNS immune privilege: Hiding in plain sight. Immunol. Rev. 2006, 213, 48–65. [Google Scholar] [CrossRef] [PubMed]
- Lewis, P.; Fu, Y.; Lentz, T.L. Rabies virus entry at the neuromuscular junction in nerve-muscle cocultures. Muscle Nerve 2000, 23, 720–730. [Google Scholar] [CrossRef]
- Hemachudha, T.; Ugolini, G.; Wacharapluesadee, S.; Sungkarat, W.; Shuangshoti, S.; Laothamatas, J. Human rabies: Neuropathogenesis, diagnosis, and management. Lancet Neurol. 2013, 12, 498–513. [Google Scholar] [CrossRef]
- Charlton, K.M.; Nadin-Davis, S.; Casey, G.A.; Wandeler, A.I. The long incubation period in rabies: Delayed progression of infection in muscle at the site of exposure. Acta Neuropathol. 1997, 94, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Yamaoka, S.; Ito, N.; Ohka, S.; Kaneda, S.; Nakamura, H.; Agari, T.; Masatani, T.; Nakagawa, K.; Okada, K.; Okadera, K.; et al. Involvement of the rabies virus phosphoprotein gene in neuroinvasiveness. J. Virol. 2013, 87, 12327–12338. [Google Scholar] [CrossRef] [PubMed]
- Charlton, K.M.; Casey, G.A. Experimental rabies in skunks: Persistence of virus in denervated muscle at the inoculation site. Can. J. Comp. Med. 1981, 45, 357–362. [Google Scholar] [PubMed]
- Mentis, G.Z.; Gravell, M.; Hamilton, R.; Shneider, N.A.; O’Donovan, M.J.; Schubert, M. Transduction of motor neurons and muscle fibers by intramuscular injection of HIV-1-based vectors pseudotyped with select rabies virus glycoproteins. J. Neurosci. Methods 2006, 157, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Lafon, M. Modulation of the immune response in the nervous system by rabies virus. Curr. Top. Microbiol. Immunol. 2005, 289, 239–258. [Google Scholar] [PubMed]
- Lentz, T.L.; Burrage, T.G.; Smith, A.L.; Tignor, G.H. The acetylcholine receptor as a cellular receptor for rabies virus. Yale J. Biol. Med. 1983, 56, 315–322. [Google Scholar] [PubMed]
- Lafon, M. Evasive Strategies in Rabies Virus Infection, 1st ed.; Elsevier Inc.: Oxford, UK, 2011; Volume 79. [Google Scholar]
- Shankar, V.; Dietzschold, B.; Koprowski, H. Direct entry of rabies virus into the central nervous system without prior local replication. J. Virol. 1991, 65, 2736–2738. [Google Scholar] [PubMed]
- Thoulouze, M.; Lafage, M.; Montano-hirose, J.A.; Lafon, M. Rabies virus infects mouse and human lymphocytes and induces apoptosis. J. Virol. 1997, 71, 7372–7380. [Google Scholar] [PubMed]
- Senba, K.; Matsumoto, T.; Yamada, K.; Shiota, S.; Iha, H.; Date, Y.; Ohtsubo, M.; Nishizono, A. Passive carriage of rabies virus by dendritic cells. Springerplus 2013, 2, 419. [Google Scholar] [CrossRef] [PubMed]
- King, A.A.; Sands, J.J.; Porterfield, J.S. Antibody-mediated enhancement of rabies virus infection in a mouse macrophage cell line (P388D1). J. Gen. Virol. 1984, 65 Pt 6, 1091–1093. [Google Scholar] [CrossRef] [PubMed]
- Ray, N.B.; Ewalt, L.C.; Lodmell, D.L. Rabies virus replication in primary murine bone marrow macrophages and in human and murine macrophage-like cell lines: Implications for viral persistence. J. Virol. 1995, 69, 764–772. [Google Scholar] [PubMed]
- Benedict, C.A.; Norris, P.S.; Ware, C.F. To kill or be killed: Viral evasion of apoptosis. Nat. Immunol. 2002, 3, 1013–1018. [Google Scholar] [CrossRef] [PubMed]
- Koyama, A.H. Induction of apoptotic DNA fragmentation by the infection of vesicular stomatitis virus. Virus Res. 1995, 37, 285–290. [Google Scholar] [CrossRef]
- Gougeon, M.-L. Apoptosis as an HIV strategy to escape immune attack. Nat. Rev. Immunol. 2003, 3, 392–404. [Google Scholar] [CrossRef] [PubMed]
- Faber, M.; Pulmanausahakul, R.; Suchita, S.H.; Spitsin, S.; Mcgettigan, J.P.; Schnell, M.J.; Dietzschold, B. Overexpression of the rabies virus glycoprotein results in enhancement of apoptosis and antiviral immune response. J. Virol. 2002, 76, 3374–3381. [Google Scholar] [CrossRef] [PubMed]
- Sarmento, L.; Li, X.; Howerth, E.; Jackson, A.C.; Fu, Z.F. Glycoprotein-mediated induction of apoptosis limits the spread of attenuated rabies viruses in the central nervous system of mice. J. Neurovirol. 2005, 11, 571–581. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, K.; Hooper, D.C.; Spitsin, S.; Koprowski, H.; Dietzschold, B. Pathogenicity of different rabies virus variants inversely correlates with apoptosis and rabies virus glycoprotein expression in infected primary neuron cultures. J. Virol. 1999, 73, 510–518. [Google Scholar] [PubMed]
- Prehaud, C.; Lay, S.; Dietzschold, B.; Lafon, M. Glycoprotein of nonpathogenic rabies viruses is a key determinant of human cell apoptosis. J. Virol. 2003, 77, 10537–10547. [Google Scholar] [CrossRef] [PubMed]
- Dietzschold, B.; Li, J.; Faber, M.; Schnell, M. Concepts in the pathogenesis of rabies. Future Virol. 2009, 3, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Wirblich, C.; Schnell, M.J. Rabies virus (RV) glycoprotein expression levels are not critical for pathogenicity of RV. J. Virol. 2011, 85, 697–704. [Google Scholar] [CrossRef] [PubMed]
- Préhaud, C.; Wolff, N.; Terrien, E.; Lafage, M.; Mégret, F.; Babault, N.; Cordier, F.; Tan, G.S.; Maitrepierre, E.; Ménager, P.; et al. Attenuation of rabies virulence: Takeover by the cytoplasmic domain of its envelope protein. Sci. Signal. 2010, 3, ra5. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.F.; Jackson, A.C. Neuronal dysfunction and death in rabies virus infection. J. Neurovirol. 2005, 11, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Thoulouze, M.; Lafage, M.; Schachner, M.; Hartmann, U.; Cremer, H.; Lafon, M. The neural cell adhesion molecule is a receptor for rabies virus. J. Virol. 1998, 72, 7181–7190. [Google Scholar] [PubMed]
- Jackson, A.C. Apoptosis in experimental rabies in Bax-deficient mice. Acta Neuropathol. 1999, 98, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.C.; Rossiter, J.P. Apoptosis plays an important role in experimental rabies virus infection. J. Virol. 1997, 71, 5603–5607. [Google Scholar] [PubMed]
- Rutherford, M.; Jackson, A.C. Neuronal apoptosis in immunodeficient mice infected with the challenge virus standard strain of rabies virus by intracerebral inoculation. J. Neurovirol. 2004, 10, 409–413. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.C.; Randle, E.; Lawrance, G.; Rossiter, J.P. Neuronal apoptosis does not play an important role in human rabies encephalitis. J. Neurovirol. 2008, 14, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Reid, J.E.; Jackson, A.C. Experimental rabies virus infection in Artibeus jamaicensis bats with CVS-24 variants. J. Neurovirol. 2001, 7, 511–517. [Google Scholar] [PubMed]
- Theerasurakarn, S.; Ubol, S. Apoptosis induction in brain during the fixed strain of rabies virus infection correlates with onset and severity of illness. J. Neurovirol. 1998, 4, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Li, X.-Q.; Sarmento, L.; Fu, Z.F. Degeneration of neuronal processes after infection with pathogenic, but not attenuated, rabies viruses. J. Virol. 2005, 79, 10063–10068. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.C. Diabolical effects of rabies encephalitis. J. Neurovirol. 2016, 22, 1, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Scott, C.A.; Rossiter, J.P.; Andrew, R.D.; Jackson, A.C. Structural abnormalities in neurons are sufficient to explain the clinical disease and fatal outcome of experimental rabies in yellow fluorescent protein-expressing transgenic mice. J. Virol. 2008, 82, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Kammouni, W.; Wood, H.; Saleh, A.; Appolinario, C.M.; Fernyhough, P.; Jackson, A.C. Rabies virus phosphoprotein interacts with mitochondrial complex I and induces mitochondrial dysfunction and oxidative stress. J. Neurovirol. 2015, 21, 370–382. [Google Scholar] [CrossRef] [PubMed]
- Gholami, A.; Kassis, R.; Real, E.; Delmas, O.; Guadagnini, S.; Larrous, F.; Obach, D.; Prevost, M.; Jacob, Y.; Bourhy, H. Mitochondrial dysfunction in lyssavirus-induced apoptosis. J. Virol. 2008, 82, 4774–4784. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, E.; de Andrade, H.; Lancellotti, C. In situ apoptosis of adaptive immune cells and the cellular escape of rabies virus in CNS from patients with human rabies transmitted by Desmodus rotundus. Virus Res. 2011, 156, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Baloul, L.; Lafon, M. Apoptosis and rabies virus neuroinvasion. Biochimie 2003, 85, 777–788. [Google Scholar] [CrossRef]
- Adle-Biassette, H.; Bourhy, H.; Gisselbrecht, M.; Chrétien, F.; Wingertsmann, L.; Baudrimont, M.; Rotivel, Y.; Godeau, B.; Gray, F. Rabies encephalitis in a patient with AIDS: A clinicopathological study. Acta Neuropathol. 1996, 92, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Kasempimolporn, S.; Tirawatnapong, T.; Saengseesom, W.; Nookhai, S.; Sitprija, V. Immunosuppression in rabies virus infection mediated by lymphocyte apoptosis. Jpn. J. Infect. Dis. 2001, 54, 144–147. [Google Scholar] [PubMed]
- Lafon, M. Immune Evasion, a Critical Strategy for Rabies Virus. In Towards the Elimination of Rabies in Eurasia; Karger: Basel, Switzerland, 2008; pp. 413–419. [Google Scholar]
- Wang, Z.W.; Sarmento, L.; Wang, Y.; Li, X.; Dhingra, V.; Tseggai, T.; Jiang, B.; Zhen, F.; Fu, Z.F. Attenuated rabies virus activates, while pathogenic rabies virus evades, the host innate immune responses in the central nervous system. J. Virol. 2005, 79, 12554–12565. [Google Scholar] [CrossRef] [PubMed]
- Westendorp, M.O.; Frank, R.; Ochsenbauer, C.; Stricker, K.; Dhein, J.; Walczak, H.; Debating, K.M.; Krammer, P.H. Sensitization of T cells to CD95-mediated apoptosis by HIV-1 Tat and gp120. Nature 1995, 375, 497–500. [Google Scholar] [CrossRef] [PubMed]
- Flügel, A.; Schwaiger, F.; Neumann, H.; Medana, I.; Willem, M.; Wekerle, H.; Kreutzberg, G.; Graeber, M. Neuronal FasL induces cell death of encephalitogenic T lymphocytes. Brain Pathol. 2000, 10, 353–364. [Google Scholar] [CrossRef] [PubMed]
- Lafon, M.; Prehaud, C.; Megret, F.; Lafage, M.; Mouillot, G.; Roa, M.; Moreau, P.; Rouas-Freiss, N.; Carosella, E.D. Modulation of HLA-G expression in human neural cells after neurotropic viral infections. J. Virol. 2005, 79, 15226–15237. [Google Scholar] [CrossRef] [PubMed]
- Mégret, F.; Prehaud, C.; Lafage, M.; Moreau, P.; Rouas-Freiss, N.; Carosella, E.D.; Lafon, M. Modulation of HLA-G and HLA-E expression in human neuronal cells after rabies virus or herpes virus simplex type 1 infections. Hum. Immunol. 2007, 68, 294–302. [Google Scholar] [CrossRef] [PubMed]
- Wiendl, H.; Mitsdoerffer, M.; Weller, M. Hide-and-seek in the brain: A role for HLA-G mediating immune privilege for glioma cells. Semin. Cancer Biol. 2003, 13, 343–351. [Google Scholar] [CrossRef]
- Panpanich, T.; Hemachudha, T.; Piyasirisilp, S.; Manatsathit, S.; Wilde, H.; Phanuphak, P. Cells with natural killer activity in human rabies. Clin. Exp. Immunol. 1992, 89, 414–418. [Google Scholar] [CrossRef] [PubMed]
- Freeman, G.J.; Long, A.J.; Iwai, Y.; Bourque, K.; Chernova, T.; Nishimura, H.; Fitz, L.J.; Malenkovich, N.; Okazaki, T.; Byrne, M.C.; et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J. Exp. Med. 2000, 192, 1027–1034. [Google Scholar] [CrossRef] [PubMed]
- Lafon, M.; Mégret, F.; Meuth, S.G.; Simon, O.; Romero, M.L.V.; Lafage, M.; Chen, L.; Alexopoulou, L.; Flavell, R.A.; Prehaud, C.; et al. Detrimental contribution of the immuno-inhibitor B7-H1 to Rabies Virus Encephalitis. J. Immunol. 2008, 180, 7506–7515. [Google Scholar] [CrossRef] [PubMed]
- Ubol, S.; Sukwattanapan, C.; Maneerat, Y. Inducible nitric oxide synthase inhibition delays death of rabies virus-infected mice. J. Med. Microbiol. 2001, 50, 238–242. [Google Scholar] [CrossRef] [PubMed]
- Ubol, S.; Sukwattanapan, C.; Utaisincharoen, P. Rabies virus replication induces Bax-related, caspase dependent apoptosis in mouse neuroblastoma cells. Virus Res. 1998, 56, 207–215. [Google Scholar] [CrossRef]
- Sarmento, L.; Tseggai, T.; Dhingra, V.; Fu, Z.F. Rabies virus-induced apoptosis involves caspase-dependent and caspase-independent pathways. Virus Res. 2006, 121, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Ubol, S.; Kasisith, J. Reactivation of Nedd-2, a developmentally down-regulated apoptotic gene, in apoptosis induced by a street strain of rabies virus. J. Med. Microbiol. 2000, 49, 1043–1046. [Google Scholar] [CrossRef] [PubMed]
- Kassis, R.; Larrous, F.; Estaquier, J.; Bourhy, H. Lyssavirus Matrix Protein Induces Apoptosis by a TRAIL-Dependent Mechanism Involving Caspase-8 Activation. J. Virol. 2004, 78, 6543–6555. [Google Scholar] [CrossRef] [PubMed]
- Larrous, F.; Gholami, A.; Mouhamad, S.; Estaquier, J.; Bourhy, H. Two overlapping domains of a lyssavirus matrix protein that acts on different cell death pathways. J. Virol. 2010, 84, 9897–9906. [Google Scholar] [CrossRef] [PubMed]
- Zhao, P.; Zhao, L.; Zhang, T.; Qi, Y.; Wang, T.; Liu, K.; Wang, H.; Feng, H.; Jin, H.; Qin, C.; et al. Innate immune response gene expression profiles in central nervous system of mice infected with rabies virus. Comp. Immunol. Microbiol. Infect. Dis. 2011, 34, 503–512. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, V.; Mancuso, C.; Calvani, M.; Rizzarelli, E.; Butterfield, D.A.; Stella, A.M.G. Nitric oxide in the central nervous system: Neuroprotection versus neurotoxicity. Nat. Rev. Neurosci. 2007, 8, 766–775. [Google Scholar] [CrossRef] [PubMed]
- Drechsel, D.A.; Estévez, A.G.; Barbeito, L.; Beckman, J.S. Nitric oxide-mediated oxidative damage and the progressive demise of motor neurons in ALS. Neurotox. Res. 2012, 22, 251–264. [Google Scholar] [CrossRef] [PubMed]
- Ubol, S.; Hiriote, W.; Anuntagool, N.; Utaisincharoen, P. A radical form of nitric oxide suppresses RNA synthesis of rabies virus. Virus Res. 2001, 81, 125–132. [Google Scholar] [CrossRef]
- Hooper, D.C.; Ohnishi, S.T.; Kean, R.; Numagami, Y.; Dietzschold, B.; Koprowski, H. Local nitric oxide production in viral and autoimmune diseases of the central nervous system. Proc. Natl. Acad. Sci. USA 1995, 92, 5312–5316. [Google Scholar] [CrossRef] [PubMed]
- Nakamichi, K.; Inoue, S.; Takasaki, T.; Morimoto, K.; Kurane, I. Rabies Virus Stimulates Nitric Oxide Production and CXC Chemokine Ligand 10 Expression in Macrophages through Activation of Extracellular Signal-Regulated Kinases 1 and 2. J. Virol. 2004, 78, 9376–9388. [Google Scholar] [CrossRef] [PubMed]
- Liao, P.; Hsu, Y.; Yang, H.; Wang, M.; Chen, L. Involvement of extraneural tissues and upregulation of inducible nitric oxide synthase after experimental infection with rabies virus in BALB/c mice and LEW/SsN rats. Pathol. Int. 2012, 62, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.C.; Kammouni, W.; Zherebitskaya, E.; Fernyhough, P. Role of oxidative stress in rabies virus infection of adult mouse dorsal root ganglion neurons. J. Virol. 2010, 84, 4697–4705. [Google Scholar] [CrossRef] [PubMed]
- Kammouni, W.; Hasan, L.; Saleh, A.; Wood, H.; Fernyhough, P.; Jackson, A.C. Role of nuclear factor-B in oxidative stress associated with rabies virus infection of adult rat dorsal root ganglion neurons. J. Virol. 2012, 86, 8139–8146. [Google Scholar] [CrossRef] [PubMed]
- Shin, T.; Weinstock, D.; Castro, M.D.; Hamir, A.N.; Wampler, T.; Walter, M.; Kim, H.Y.; Acland, H. Immunohistochemical localization of endothelial and inducible nitric oxide synthase within neurons of cattle with rabies. J. Vet. Med. Sci. 2004, 66, 539–541. [Google Scholar] [CrossRef] [PubMed]
- Van Dam, A.-M.; Bauer, J.; Man-A-Hing, W.K.H.; Marquette, C.; Tilders, F.J.H.; Berkenbosch, F. Appearance of inducible nitric oxide synthase in the rat central nervous system after rabies virus infection and during experimental allergic encephalomyelitis but not after peripheral administration of endotoxin. J. Neurosci. Res. 1995, 40, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Prosniak, M.; Curtis, M.T.; Weiss, M.L.; Faber, M.; Dietzschold, B.; Fu, Z.F. Silver-haired bat rabies virus variant does not induce apoptosis in the brain of experimentally infected mice. J. Neurovirol. 2001, 7, 518–527. [Google Scholar] [PubMed]
- Guigoni, C.; Coulon, P. Rabies virus is not cytolytic for rat spinal motoneurons in vitro. J. Neurovirol. 2002, 8, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Koprowski, H.; Zheng, Y.M.; Heber-Katz, E.; Fraser, N.; Rorke, L.; Fu, Z.F.; Hanlon, C.; Dietzschold, B. In vivo expression of inducible nitric oxide synthase in experimentally induced neurologic diseases. Proc. Natl. Acad. Sci. USA 1993, 90, 3024–3027. [Google Scholar] [CrossRef] [PubMed]
- Alandijany, T.; Kammouni, W.; Roy Chowdhury, S.K.; Fernyhough, P.; Jackson, A.C. Mitochondrial dysfunction in rabies virus infection of neurons. J. Neurovirol. 2013, 19, 537–549. [Google Scholar] [CrossRef] [PubMed]
- Teepker, M.; Anthes, N.; Fischer, S.; Krieg, J.C.; Vedder, H. Effects of oxidative challenge and calcium on ATP-levels in neuronal cells. Neurotoxicology 2007, 28, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Lambert, A.; Brand, M. Reactive Oxygen Species Production by Mitochondria. In Mitochondrial DNA SE—11; Stuart, J., Ed.; Methods in Molecular Biology™, Humana Press: Clifton, NJ, USA, 2009; Volume 554, pp. 165–181. [Google Scholar]
- Szczepanek, K.; Chen, Q.; Larner, A.C.; Lesnefsky, E.J. Cytoprotection by the modulation of mitochondrial electron transport chain: The emerging role of mitochondrial STAT3. Mitochondrion 2012, 12, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Lieu, K.G.; Brice, A.; Wiltzer, L.; Hirst, B.; Jans, D.A.; Blondel, D.; Moseley, G.W. The rabies virus interferon antagonist P-protein interacts with activated STAT3 and inhibits Gp130 receptor signaling. J. Virol. 2013, 81, 8261–8265. [Google Scholar] [CrossRef] [PubMed]
- Mayer, M.P. Gymnastics of molecular chaperones. Mol. Cell 2010, 39, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Mayer, M.P.; Bukau, B. Hsp70 chaperones: Cellular functions and molecular mechanism. Cell. Mol. Life Sci. 2005, 62, 670–684. [Google Scholar] [CrossRef] [PubMed]
- Lahaye, X.; Vidy, A.; Fouquet, B.; Blondel, D. Hsp70 protein positively regulates rabies virus infection. J. Virol. 2012, 86, 4743–4751. [Google Scholar] [CrossRef] [PubMed]
- Silverman, M.N.; Sternberg, E.M. Glucocorticoid regulation of inflammation and its functional correlates: From HPA axis to glucocorticoid receptor dysfunction. Ann. N. Y. Acad. Sci. 2012, 1261, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Schutsky, K.; Portocarrero, C.; Hooper, D.C.; Dietzschold, B.; Faber, M. Limited brain metabolism changes differentiate between the progression and clearance of rabies virus. PLoS ONE 2014, 9, e87180. [Google Scholar] [CrossRef] [PubMed]
- Burns, K.F.; Shelton, D.F.; Lukeman, J.M.; Grogan, E.W. Cortisone and ACTH impairment of response to rabies vaccine. Public Health Rep. 1960, 75, 441–445. [Google Scholar] [CrossRef] [PubMed]
- Enright, J.B.; Franti, C.E.; Frye, F.L.; Behymer, D.E. The effects of corticosteroids on rabies in mice. Can. J. Microbiol. 1970, 16, 667–675. [Google Scholar] [CrossRef] [PubMed]
- Enright, J.B.; Goggin, J.E.; Frye, F.L.; Franti, C.E.; Behymer, D.E. Effects of corticosteroids on rabies virus infections in various animal species. J. Am. Vet. Med. Assoc. 1970, 156, 765–769. [Google Scholar] [PubMed]
- Ciuchini, F.; Pestalozza, S.; Buonavoglia, C.; Di Trani, L.; Tollis, M.; Orfei, Z. Effects of corticosteroids mediated immunosuppression on the distribution of rabies vaccine virus in red foxes orally immunized against rabies. Zentralbl. Veterinarmed. B 1986, 33, 628–631. [Google Scholar] [CrossRef] [PubMed]
- Wiktor, T.J.; Doherty, P.C.; Koprowski, H. In vitro evidence of cell-mediated immunity after exposure of mice to both live and inactivated rabies virus. Proc. Natl. Acad. Sci. USA Immun. 1977, 74, 334–338. [Google Scholar] [CrossRef]
- Prabhakar, B.S.; Nathanson, N. Acute rabies death mediated by antibody. Nature 1981, 290, 590–591. [Google Scholar] [CrossRef] [PubMed]
- Andral, B.; Blancou, J. Study of the mechanisms of early death occurring after vaccination in mice inoculated with street rabies virus. Ann. L’inst. Pasteur Virol. 1981, 132, 503–517. [Google Scholar] [CrossRef]
- Sikes, R.K.; Cleary, W.F.; Koprowski, H.; Wiktor, T.J.; Kaplan, M.M. Effective protection of monkeys against death from street virus by post-exposure administration of tissue-culture rabies vaccine. Bull. World Health Organ. 1971, 45, 1–11. [Google Scholar] [PubMed]
- Prabhakar, B.S.; Fischman, H.R.; Nathanson, N. Recovery from experimental rabies by adoptive transfer of immune cells. J. Gen. Virol. 1981, 56, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Blancou, J.; Andral, B.; Andral, L. A model in mice for the study of the early death phenomenon after vaccination and challenge with rabies virus. J. Gen. Virol. 1980, 50, 433–435. [Google Scholar] [CrossRef] [PubMed]
- Vennema, H.; de Groot, R.J.; Harbour, D.A.; Dalderup, M.; Gruffydd-Jones, T.; Horzinek, M.C.; Spaan, W.J. Early death after feline infectious peritonitis virus challenge due to recombinant vaccinia virus immunization. J. Virol. 1990, 64, 1407–1409. [Google Scholar] [PubMed]
- Barrett, A.D.T.; Gould, E.A. Antibody-mediated early death in vivo after infection with yellow fever virus. J. Gen. Virol. 1986, 67, 2539–2542. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, K.; Ito, N.; Sugiyama, M.; Minamoto, N. Sensitivity of rabies virus to type I interferon is determined by the phosphoprotein gene. Microbiol. Immunol. 2006, 50, 975–978. [Google Scholar] [CrossRef] [PubMed]
- Niu, X.; Tang, L.; Tseggai, T.; Guo, Y.; Fu, Z.F. Wild-type rabies virus phosphoprotein is associated with viral sensitivity to type I interferon treatment. Arch. Virol. 2013, 158, 2297–2305. [Google Scholar] [CrossRef] [PubMed]
- Chelbi-Alix, M.K.; Vidy, A.; El Bougrini, J.; Blondel, D. Rabies viral mechanisms to escape the IFN system: The viral protein P interferes with IRF-3, Stat1, and PML nuclear bodies. J. Interferon Cytokine Res. 2006, 26, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Everett, R.D.; Chelbi-Alix, M.K. PML and PML nuclear bodies: Implications in antiviral defence. Biochimie 2007, 89, 819–830. [Google Scholar] [CrossRef] [PubMed]
- Regad, T.; Saib, A.; Lallemand-Breitenbach, V.; Pandolfi, P.P.; de Thé, H.; Chelbi-Alix, M.K. PML mediates the interferon-induced antiviral state against a complex retrovirus via its association with the viral transactivator. EMBO J. 2001, 20, 3495–3505. [Google Scholar] [CrossRef] [PubMed]
- Chenik, M.; Chebli, K.; Blondel, D. Translation initiation at alternate in-frame AUG codons in the rabies virus phosphoprotein mRNA is mediated by a ribosomal leaky scanning mechanism. J. Virol. 1995, 69, 707–712. [Google Scholar] [PubMed]
- Blondel, D.; Regad, T.; Poisson, N.; Pavie, B.; Harper, F.; Pandolfi, P.P.; De Thé, H.; Chelbi-Alix, M.K. Rabies virus P and small P products interact directly with PML and reorganize PML nuclear bodies. Oncogene 2002, 21, 7957–7970. [Google Scholar] [CrossRef] [PubMed]
- Blondel, D.; Kheddache, S.; Lahaye, X.; Dianoux, L.; Chelbi-Alix, M.K. Resistance to rabies virus infection conferred by the PMLIV isoform. J. Virol. 2010, 84, 10719–10726. [Google Scholar] [CrossRef] [PubMed]
- Goodbourn, S.; Didcock, L.; Randall, R.E. Interferons: Cell signalling, immune modulation, antiviral responses and virus countermeasures. J. Gen. Virol. 2000, 81, 2341–2364. [Google Scholar] [CrossRef] [PubMed]
- Pasdeloup, D.; Poisson, N.; Raux, H.; Gaudin, Y.; Ruigrok, R.W.H.; Blondel, D. Nucleocytoplasmic shuttling of the rabies virus P protein requires a nuclear localization signal and a CRM1-dependent nuclear export signal. Virology 2005, 334, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Wiltzer, L.; Larrous, F.; Oksayan, S.; Ito, N.; Marsh, G.A.; Wang, L.F.; Blondel, D.; Bourhy, H.; Jans, D.A.; Moseley, G.W. Conservation of a unique mechanism of immune evasion across the Lyssavirus genus. J. Virol. 2012, 86, 10194–10199. [Google Scholar] [CrossRef] [PubMed]
- Brzózka, K.; Finke, S.; Conzelmann, K.K. Inhibition of interferon signaling by rabies virus phosphoprotein P: Activation-dependent binding of STAT1 and STAT2. J. Virol. 2006, 80, 2675–2683. [Google Scholar] [CrossRef] [PubMed]
- Vidy, A.; Chelbi-alix, M.; Blondel, D. Rabies virus P protein interacts with STAT1 and inhibits interferon signal transduction pathways. J. Virol. 2005, 79, 14411–14420. [Google Scholar] [CrossRef] [PubMed]
- Wiltzer, L.; Okada, K.; Yamaoka, S.; Larrous, F.; Kuusisto, H.V.; Sugiyama, M.; Blondel, D.; Bourhy, H.; Jans, D.A.; Ito, N.; et al. Interaction of rabies virus P-protein with STAT proteins is critical to lethal rabies disease. J. Infect. Dis. 2014, 209, 1744–1753. [Google Scholar] [CrossRef] [PubMed]
- Ito, N.; Moseley, G.W.; Blondel, D.; Shimizu, K.; Rowe, C.L.; Ito, Y.; Masatani, T.; Nakagawa, K.; Jans, D.A.; Sugiyama, M. Role of interferon antagonist activity of rabies virus phosphoprotein in viral pathogenicity. J. Virol. 2010, 84, 6699–6710. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, P.C.; Behrmann, I.; Müller-Newen, G.; Schaper, F.; Graeve, L. Interleukin-6-type cytokine signalling through the gp130/Jak/STAT pathway. Biochem. J. 1998, 334 Pt 2, 297–314. [Google Scholar] [CrossRef] [PubMed]
- Marschalek, A.; Drechsel, L.; Conzelmann, K.K. The importance of being short: The role of rabies virus phosphoprotein isoforms assessed by differential IRES translation initiation. Eur. J. Cell Biol. 2011, 20, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Vidy, A.; El Bougrini, J.; Chelbi-alix, M.K.; Blondel, D. The Nucleocytoplasmic rabies virus P protein counteracts interferon signaling by inhibiting both nuclear accumulation and DNA binding of STAT1. J. Virol. 2007, 81, 4255–4263. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.; Yoneyama, M.; Ito, T.; Takahashi, K.; Inagaki, F.; Fujita, T. Identification of Ser-386 of interferon regulatory factor 3 as critical target for inducible phosphorylation that determines activation. J. Biol. Chem. Mol. Biol. 2004, 279, 9698–9702. [Google Scholar] [CrossRef] [PubMed]
- Brzozka, K.; Finke, S.; Conzelmann, K.K. Identification of the rabies virus alpha/beta interferon antagonist: Phosphoprotein P interferes with phosphorylation of interferon regulatory factor 3. J. Virol. 2005, 79, 7673–7681. [Google Scholar] [CrossRef] [PubMed]
- Rieder, M.; Brzózka, K.; Pfaller, C.K.; Cox, J.H.; Stitz, L.; Conzelmann, K.K. Genetic dissection of interferon-antagonistic functions of rabies virus phosphoprotein: Inhibition of interferon regulatory factor 3 activation is important for pathogenicity. J. Virol. 2011, 85, 842–852. [Google Scholar] [CrossRef] [PubMed]
- Faul, E.J.; Wanjalla, C.N.; Suthar, M.S.; Gale, M.; Wirblich, C.; Schnell, M.J. Rabies virus infection induces type I interferon production in an IPS-1 dependent manner while dendritic cell activation relies on IFNAR signaling. PLoS Pathog. 2010, 6, e1001016. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; McGettigan, J.P.; Faber, M.; Schnell, M.J.; Dietzschold, B. Infection of monocytes or immature dendritic cells (DCs) with an attenuated rabies virus results in DC maturation and a strong activation of the NFkappaB signaling pathway. Vaccine 2008, 26, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Albertini, A.A.V.; Wernimont, A.K.; Muziol, T.; Ravelli, R.B.G.; Clapier, C.R.; Schoehn, G.; Weissenhorn, W.; Ruigrok, R.W.H. Crystal structure of the rabies virus nucleoprotein-RNA complex. Science 2006, 313, 360–363. [Google Scholar] [CrossRef] [PubMed]
- Masatani, T.; Ito, N.; Shimizu, K.; Ito, Y.; Nakagawa, K.; Abe, M.; Yamaoka, S.; Sugiyama, M. Amino acids at positions 273 and 394 in rabies virus nucleoprotein are important for both evasion of host RIG-I-mediated antiviral response and pathogenicity. Virus Res. 2011, 155, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Masatani, T.; Ito, N.; Shimizu, K.; Ito, Y.; Nakagawa, K.; Sawaki, Y.; Koyama, H.; Sugiyama, M. Rabies virus nucleoprotein functions to evade activation of the RIG-I-mediated antiviral response. J. Virol. 2010, 84, 4002–4012. [Google Scholar] [CrossRef]
- Masatani, T.; Ito, N.; Ito, Y.; Nakagawa, K.; Abe, M.; Yamaoka, S.; Okadera, K.; Sugiyama, M. Importance of rabies virus nucleoprotein in viral evasion of interferon response in the brain. Microbiol. Immunol. 2013, 57, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Mebatsion, T.; Weiland, F.; Conzelmann, K.K. Matrix protein of rabies virus is responsible for the assembly and budding of bullet-shaped particles and interacts with the transmembrane spike glycoprotein G. J. Virol. 1999, 73, 242–250. [Google Scholar] [PubMed]
- Komarova, A.V.; Real, E.; Borman, A.M.; Brocard, M.; England, P.; Tordo, N.; Hershey, J.W.B.; Kean, K.M.; Jacob, Y. Rabies virus matrix protein interplay with eIF3, new insights into rabies virus pathogenesis. Nucleic Acids Res. 2007, 35, 1522–1532. [Google Scholar] [CrossRef] [PubMed]
- Wirblich, C.; Tan, G.S.; Papaneri, A.; Godlewski, P.J.; Orenstein, J.M.; Harty, R.N.; Schnell, M.J. PPEY motif within the rabies virus (RV) matrix protein is essential for efficient virion release and RV pathogenicity. J. Virol. 2008, 82, 9730–9738. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-C. Ubiquitin Ligases and the Immune Response. Annu. Rev. Immunol. 2004, 22, 81–127. [Google Scholar] [CrossRef] [PubMed]
- Sonwane, A.A.; Dahiya, S.S.; Saini, M.; Chaturvedi, V.K.; Singh, R.P.; Gupta, P.K. Inhibition of rabies virus multiplication by siRNA delivered through adenoviral vector in vitro in BHK-21 cells and in vivo in mice. Res. Vet. Sci. 2012, 93, 498–503. [Google Scholar] [CrossRef] [PubMed]
- Meshram, C.D.; Singh, N.K.; Sonwane, A.A.; Pawar, S.S.; Mishra, B.P.; Chaturvedi, V.K.; Saini, M.; Singh, R.P.; Gupta, P.K. Evaluation of single and dual siRNAs targeting rabies virus glycoprotein and nucleoprotein genes for inhibition of virus multiplication in vitro. Arch. Virol. 2013, 158, 2323–2332. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.K.; Meshram, C.D.; Sonwane, A.A.; Dahiya, S.S.; Pawar, S.S.; Chaurvedi, V.K.; Saini, M.; Singh, R.P.; Gupta, P.K. Protection of mice against lethal rabies virus challenge using ihort interfering RNAs (siRNAs) delivered through lentiviral vector. Mol. Biotechnol. 2013, 56, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Israsena, N.; Mahavihakanont, A.; Hemachudha, T. Rabies virus infection and microRNAs. Adv. Virus Res. 2011, 79, 329–344. [Google Scholar] [PubMed]
- Gupta, P.K.; Sonwane, A.A.; Singh, N.K.; Meshram, C.D.; Dahiya, S.S.; Pawar, S.S.; Gupta, S.P.; Chaturvedi, V.K.; Saini, M. Intracerebral delivery of small interfering RNAs (siRNAs) using adenoviral vector protects mice against lethal peripheral rabies challenge. Virus Res. 2012, 163, 11–18. [Google Scholar] [CrossRef]
- Brandão, P.E.; Castilho, J.G.; Fahl, W.; Carnieli, P., Jr.; Oliveira, R.D.N. Short-interfering RNAs as antivirals Against Rabies. Braz. J. Infect. Dis. 2007, 11, 224–225. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.X.; Wang, H.L.; Guo, X.F.; Yang, Y.J.; Ma, J.Z.; Wang, T.C.; Gao, Y.W.; Zhao, Y.K.; Yang, S.T.; Xia, X.Z. Adeno-associated viruses serotype 2-mediated RNA interference efficiently inhibits rabies virus Replication In Vitro and In Vivo. J. Vet. Med. Sci. 2013, 75, 1355–1361. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Rossi, J.J. Strategies for silencing human disease using RNA interference. Nat. Rev. Genet. 2007, 8, 173–184. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Hu, G.; Zhang, T.; Yang, Y.; Zhao, L.; Qi, Y.; Wang, H.; Goa, Y.; Yang, S.; Xia, X. Isolation of ssDNA aptamers that inhibit rabies virus. Int. Immunopharmacol. 2012, 14, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Israsena, N.; Supavonwong, P.; Ratanasetyuth, N.; Khawplod, P.; Hemachudha, T. Inhibition of rabies virus replication by multiple artificial microRNAs. Antivir. Res. 2009, 84, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Zhao, P.; Zhao, L.; Zhang, T.; Wang, H.; Qin, C.; Yang, S.; Xia, X. Changes in microRNA expression induced by rabies virus infection in mouse brains. Microb. Pathog. 2011, 52, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Taylor, L.H.; Nel, L.H. Global epidemiology of canine rabies: Past, present, and future prospects. Vet. Med. Res. Rep. 2015, 6, 361–371. [Google Scholar] [CrossRef]
- McFadden, G. Killing a killer: What next for smallpox? PLoS Pathog. 2010, 6, e1000727. [Google Scholar] [CrossRef] [PubMed]
- Guerra, M.A.; Curns, A.T.; Rupprecht, C.E.; Hanlon, C.A.; Krebs, J.W.; Childs, J.E. Skunk and raccoon rabies in the eastern United States: Temporal and spatial analysis. Emerg. Infect. Dis. 2003, 9, 1143–1150. [Google Scholar] [CrossRef] [PubMed]
- Hass, C.C.; Dragoo, J.W. Rabies in hooded and striped skunks in Arizona. J. Wildl. Dis. 2006, 42, 825–829. [Google Scholar] [CrossRef] [PubMed]
- Scott, T.P.; Fischer, M.; Khaiseb, S.; Freuling, C.M.; Hoper, D.; Hoffmann, B.; Markotter, W.; Muller, T.; Nel, L.H. Complete genome and molecular epidemiological data infer the maintenance of rabies among kudu (Tragelaphus strepsiceros) in Namibia. PLoS ONE 2013, 8, e58739. [Google Scholar] [CrossRef] [PubMed]
- Baby, J.; Mani, R.S.; Abraham, S.S.; Thankappan, A.T.; Pillai, P.M.; Anand, A.M.; Madhusudana, S.N.; Ramachandran, J.; Sreekumar, S. Natural rabies infection in a domestic fowl (Gallus domesticus): A Report from India. PLoS Negl. Trop. Dis. 2015, 9, e0003942. [Google Scholar] [CrossRef] [PubMed]
- Banyard, A.C.; Evans, J.S.; Luo, T.R.; Fooks, A.R. Lyssaviruses and bats: Emergence and zoonotic threat. Viruses 2014, 6, 2974–2990. [Google Scholar] [CrossRef] [PubMed]
- Wickersham, I.R.; Finke, S.; Conzelmann, K.K.; Callaway, E.M. Retrograde neuronal tracing with a deletion-mutant rabies virus. Nat. Meth. 2007, 4, 47–49. [Google Scholar] [CrossRef] [PubMed]
- Osakada, F.; Mori, T.; Cetin, A.H.; Marshel, J.H.; Virgen, B.; Callaway, E.M. New Rabies virus variants for monitoring and manipulating activity and gene expression in defined neural circuits. Neuron 2011, 71, 617–631. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scott, T.P.; Nel, L.H. Subversion of the Immune Response by Rabies Virus. Viruses 2016, 8, 231. https://doi.org/10.3390/v8080231
Scott TP, Nel LH. Subversion of the Immune Response by Rabies Virus. Viruses. 2016; 8(8):231. https://doi.org/10.3390/v8080231
Chicago/Turabian StyleScott, Terence P., and Louis H. Nel. 2016. "Subversion of the Immune Response by Rabies Virus" Viruses 8, no. 8: 231. https://doi.org/10.3390/v8080231