Experimental Infections of Wild Birds with West Nile Virus
Abstract
:1. Introduction
2. Viral Factors
2.1. Lineages and Strains
2.2. Pathogenicity Determinants
2.3. Viral Dose
3. Host Factors
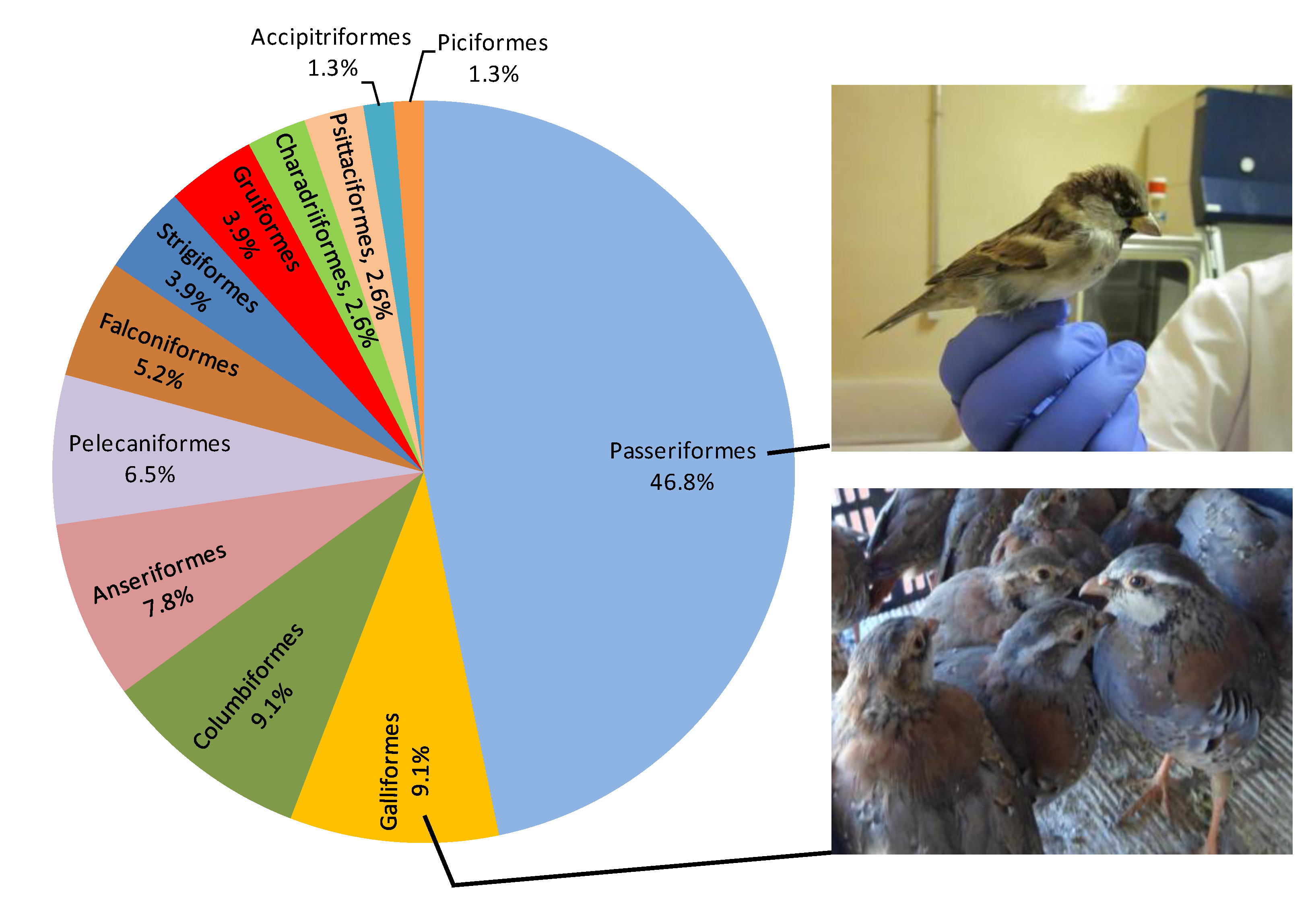
3.1. Taxonomic Classification
3.2. Geographical Origin (Paleartic vs. Neartic Species)

3.3. Age
3.4. Other Life History Traits
| Order | Family | Species | Strain/Mortality+ | Viremia | Distribution | Ref. |
|---|---|---|---|---|---|---|
| Passeriformes | Turdidae | American robin (Turdus migratorius) | NY | H | AM | [5,54] |
| Swainson’s thrush (Catharus ustulatus) | NY | M | AM | [82] | ||
| Clay-colored thrush (Turdus grayi) |  | M | AM | [88] | ||
| Corvidae | Carrion crow (Corvus corone) |  | L | EUR/ASIA | [34] | |
| American crow (Corvus brachyrhynchos) |  | H | AM | [5,25,26,27,35,89,90,91,92] | ||
 | M | |||||
| Fish crow (Corvus ossifragus) |  | H | AM | [5,89,92] | ||
| Little raven (Corvus mellori) | NY | M | OCE | [59] | ||
| KUN | L | |||||
| Hooded crow (Corvus cornix) |  | H | EUR/ASIA/AFR | [13] | ||
| Western scrub-jay (Aphelocoma californica) |  | H | AM | [53] | ||
| Blue jay (Cyanocitta cristata) |  | H | AM | [5,91] | ||
| Black-billed magpie (Pica hudsonia) |  | H | AM | [5] | ||
| Jungle crow (Corvus macrorhynchos) |  | H | ASIA | [93] | ||
| Passeridae | House sparrow (Passer domesticus) |  | H | WORLDWIDE | [5,13,27,28,36,38,53,67,76,80,81,88,94] | |
| TEX/KUN/IT08 | M | |||||
| MEX | L | |||||
| Cape sparrow (Passer melanurus) |  | L | AFR | [21] | ||
| Icteridae | Red-winged blackbird (Agelaius phoeniceus) | NY | M/L | AM | [5,77,95] | |
| Brown-headed cowbird (Molothrus ater) | NY | L | AM | [67,77] | ||
| Brewer’s blackbird (Euphagus cyanocephalus) | NY | H | AM | [67,77] | ||
| Tricolored blackbird (Agelaius tricolor) | NY | H | AM | [77] | ||
| Common grackle (Quiscalus quiscula) |  | H | AM | [5] | ||
| Great-tailed grackle (Quiscalus mexicanus) |  | H | AM | [88] | ||
| Bay-winged cowbird (Agelaioides badius) | ARG | L | AM | [30] | ||
| Shiny cowbird (Molothrus bonariensis) | ARG | L | AM | [30] | ||
| Emberizidae | Song sparrow (Melospiza melodia) | NY | M | AM | [96] | |
| White-crowned sparrow(Zonotrichia leucophrys) |  | na | AM | [67] | ||
| Fringillidae | Hawai’i ’amakihi (Hemignathus virens) |  | H | AM | [81] | |
| House finch (Haemorhous mexicanus) |  | H | AM | [5,53,67,87] | ||
| Passeriformes | Ploceidae | African masked weaver (Ploceus velatus) |  | M | AFR | [21] |
| Red-billed quelea (Quelea quelea) |  | L | AFR | [21] | ||
| Red bishop (Euplectes orix) |  | M | AFR | [21] | ||
| Hirundinidae | Cliff swallow (Petrochelidon pyrrhonota) | NY | M | AM | [56,97] | |
| Mimidae | Gray catbird (Dumetella carolinensis) | NY | M | AM | [82] | |
| Northern mockingbird (Mimuspolyglottos) | NY | H | AM | [94] | ||
| Sturnidae | European starling (Sturnus vulgaris) | NY | M | WORLDWIDE | [5,67] | |
| Cardinalidae | Northern cardinal (Cardinalis cardinalis) | NY | H | AM | [79,94] | |
| Paridae | Tufted titmouse (Baeolophus bicolor) |  | H | AM | [98] | |
| Troglodytidae | Carolina wren (Thryothorus ludovicianus) |  | H | AM | [98] | |
| Falconiformes | Falconidae | Gyrfalcon (Falco rusticolus) |  | H | AM/EUR/AS | [43] |
| NY | M | |||||
| Hybrid falcon (Falco rusticolus x Falco cherrug) |  | L | WORLDWIDE | [99] | ||
| American kestrel (Falco sparverius) | NY | H | AM | [5,66] | ||
| Common kestrel (Falco tinnunculus) | EGY | L | EUR/AS/AFR | [13] | ||
| Accipitriformes | Accipitridae | Red-tailed hawk (Buteo jamaicensis) | NY | H | AM | [66] |
| Strigiformes | Tytonidae | Barn owl (Tyto alba) | NY | L | WORLDWIDE | [66] |
| Strigidae | Great horned owl (Bubo virginianus) | NY | H | AM | [5,66] | |
| Eastern screech-owl (Megascops asio) |  | H | AM | [100] | ||
| Galliformes | Odontophoridae | California quail (Callipepla californica) | NY | L | AM | [53,68] |
| Gambel’s quail (Callipepla gambelii) | NY | L | AM | [68] | ||
| Northern bobwhite (Colinus virginianus) | NY | L | AM | [5] | ||
| Phasianidae | Red-legged partridge (Alectoris rufa) |  | H | EUR | [37,101] | |
 | L | |||||
| Japanese quail (Coturnix japonica) | NY | L | WORLDWIDE | [5] | ||
| Ring-necked pheasant (Phasianus colchicus) | NY | L | WORLDWIDE | [5] | ||
| Greater sage-grouse (Centrocercus urophasianus) |  | M | AM | [102] | ||
| Pelecaniformes | Ardeidae | Rufous night-heron (Nycticorax caledonicus) | KUN | L | OCE | [23,24] |
| Little egret (Egretta garzetta) | KUN | L | EUR/AS/AFR/OCE | [23,24] | ||
| Intermediate heron (Mesophoyx intermedia) | KUN | L | AFR/AS | [23,24] | ||
| Cattle egret (Bubulcus ibis) |  /EGY /EGY | L | WORLDWIDE | [13,21] | ||
| Threskiornithidae | African sacred ibis (Threskiornis aethiopicus) |  | L | AFR/AS | [21] | |
| Columbiformes | Columbidae | Rock pigeon (Columba livia) |  /NY/TEC/TAB /NY/TEC/TAB | L | WORLDWIDE | [21,88] |
| Ring-necked dove (Streptopelia capicola) |  | L | AFR | [21] | ||
| Eurasian collared-dove (Streptopelia decaocto) | NY/CO | M | AM/EUR/AS/AFR | [29] | ||
| Laughing dove (Spilopelia senegalensis) |  /EGY /EGY | L | AFR/AS | [13,21] | ||
| Common ground-dove (Columbina passerina) |  | na | AM | [67] | ||
| Mourning dove (Zenaida macroura) | NY | M | AM | [5,53,67] | ||
| Picui ground-dove (Columbina picui) | ARG | M | AM | [30] | ||
| Gruiformes | Rallidae | American coot (Fulica americana) | NY | L | AM | [5] |
| Crested coot (Fulica cristata) |  | L | AFR/EUR | [21] | ||
| Gruidae | Sandhill crane (Grus canadensis) | NY | L | AM | [103] | |
| Anseriformes | Anatidae | Common goose (Anser anser) |  | M | WORLDWIDE | [33] |
| Canada goose (Branta canadensis) | NY | M | AM/EUR | [5] | ||
| Mallard (Anas platyrhynchos) | NY | H | WORLDWIDE | [5] | ||
| Yellow-billed duck(Anas undulata) |  | L | AFR | [21] | ||
| Red-billed teal (Anas erythrorhyncha) |  | L | AFR | [21] | ||
| Southern pochard (Netta erythrophthalma) |  | L | AFR | [21] | ||
| Charadriiformes | Charadriidae | Killdeer (Charadrius vociferus) | NY | H | AM | [5] |
| Laridae | Ring-billed gull (Larus delawarensis) |  | H | AM | [5] | |
| Psittaciformes | Psittacidae | Monk parakeet (Myiopsitta monachus) | NY | L | AM | [5] |
| Budgerigar (Melopsittacus undulatus) | NY | L | OCE | [5] | ||
| Piciformes | Picidae | Northern flicker (Colaptes auratus) | NY | M | AM | [5] |
 . L: Low viremia (mean peak viremia ≤ 104 PFU/mL); M: Medium viremia (mean peak viremia 104–106 PFU/mL); H: High viremia (mean peak viremia > 106 PFU/mL); na: Data not available. AFR: Africa; AM: America; AS: Asia; EUR: Europe; OCE: Oceania.
. L: Low viremia (mean peak viremia ≤ 104 PFU/mL); M: Medium viremia (mean peak viremia 104–106 PFU/mL); H: High viremia (mean peak viremia > 106 PFU/mL); na: Data not available. AFR: Africa; AM: America; AS: Asia; EUR: Europe; OCE: Oceania.4. Pathogenesis
5. Immune Response
5.1. Duration of Humoral Immunity
5.2. Effect of Previous Exposure to WNV or Other Flaviviruses (Cross-Protection)
6. Transmission
6.1. Arthropod-Borne Transmission
| Vector species | Host | Transmission arthropod-bird | Transmission bird-arthropod-vertebrate * | Ref. | ||
|---|---|---|---|---|---|---|
| Order | Species | |||||
| Class: Insecta | ||||||
| Order: Diptera | Culex pipiens | Passeriformes | House sparrow, Hooded crow | Yes | Yes (mouse) | [13] |
| Falconiformes | Common kestrel | Yes | Yes (mouse) | [13] | ||
| Pelecaniformes | Buff-backed heron | Yes | Yes (mouse) | [13] | ||
| Columbiformes | Palm dove | Yes | No (mouse) | [13] | ||
| Culex univittatus | Passeriformes | House sparrow, Hooded crow | Yes | Yes (mouse) | [13] | |
| Pelecaniformes | Buff-backed heron | Yes | - | [13] | ||
| Columbiformes | Palm dove | - | Yes (mouse) | [13] | ||
| Culex antennatus | Passeriformes | House sparrow | - | Yes (mouse) | [13] | |
| Culex tritaeniorhynchus | Passeriformes | American robin, American crow, Fish crow, Blue jay, Black-billed magpie, House sparrow, Red-winged blackbird, Common grackle, House finch, European starling | Yes | - | [5] | |
| Falconiformes | American kestrel | Yes | - | [5,66] | ||
| Strigiformes | Great horned owl | Yes | - | [5,66] | ||
| Galliformes | Northern bobwhite, Japanese quail, Ring-necked pheasant | Yes | - | [5] | ||
| Columbiformes | Mourning dove, Rock pigeon | Yes | - | [5] | ||
| Gruiformes | American coot | Yes | - | [5] | ||
| Anseriformes | Canada goose, Mallard | Yes | - | [5] | ||
| Charadriiformes | Killdeer, Ring-billed gull | Yes | - | [5] | ||
| Psittaciformes | Monk parakeet, Budgerigar | Yes | - | [5] | ||
| Piciformes | Northern flicker | Yes | - | [5] | ||
| Culex quincefasciatus | Passeriformes | Cape sparrow, Red bishop | - | Yes (mouse) | [117] | |
| Passeriformes | Hawai`i `amakihi | Yes | Yes, (Hawai`i `amakihi) | [81] | ||
| Culex tarsalis | Passeriformes | House finch | - | Yes (House finch) | [116] | |
| Order: Hemiptera | Oeciacus vicarius | Passeriformes | Cliff swallow | - | No (Cliff swallow) | [97] |
| Class: Arachnida | ||||||
| Order: Acari | Ixodes pacificus | Passeriformes | Song sparrow | - | No (Song sparrow) | [120] |
6.2. Contact and Oral Transmission
6.3. Persistence of Infection
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Beck, C.; Jiménez-Clavero, M.A.; Leblond, A.; Durand, B.; Nowotny, N.; Leparc-Goffart, I.; Zientara, S.; Jourdain, E.; Lecollinet, S. Flaviviruses in Europe: complex circulation patterns and their consequences for the diagnosis and control of west nile disease. Int. J. Environ. Res. Public Health 2013, 10, 6049–6083. [Google Scholar]
- Smithburn, K.C.; Hughes, T.P.; Burke, A.W.; Paul, J.H.; African, A. A neurotropic virus isolated from the blood of a native of Uganda. Am. J. Trop. Med. Hyg. 1940, 20, 471–492. [Google Scholar]
- Kramer, L.D.; Li, J.; Shi, P. West Nile virus. Lancet Neurol. 2007, 6, 171–181. [Google Scholar] [CrossRef]
- McLean, R.G.; Ubico, S.R.; Bourne, D.; Komar, N. West Nile virus in livestock and wildlife. Curr. Top. Microbiol. Immunol. 2002, 267, 271–308. [Google Scholar]
- Komar, N.; Langevin, S.; Hinten, S.; Nemeth, N.; Edwards, E.; Hettler, D.; Davis, B.; Bowen, R.; Bunning, M. Experimental infection of North American birds with the New York 1999 strain of West Nile virus. Emerg. Infect. Dis. 2003, 9, 311–322. [Google Scholar] [CrossRef]
- Van der Meulen, K.M.; Pensaert, M.B.; Nauwynck, H.J. West Nile virus in the vertebrate world. Arch. Virol. 2005, 150, 637–57. [Google Scholar] [CrossRef]
- Hayes, E.B.; Komar, N.; Nasci, R.S.; Montgomery, S.P.; O’Leary, D.R.; Campbell, G.L. Epidemiology and transmission dynamics of West Nile virus disease. Emerg. Infect. Dis. 2005, 11, 1167–1173. [Google Scholar] [CrossRef]
- Engler, O.; Savini, G.; Papa, A.; Figuerola, J.; Groschup, M.H.; Kampen, H.; Medlock, J.; Vaux, A.; Wilson, A.J.; Werner, D.; et al. European surveillance for West Nile virus in mosquito populations. Int. J. Environ. Res. Public Health 2013, 10, 4869–4895. [Google Scholar] [CrossRef]
- Lwande, O.W.; Lutomiah, J.; Obanda, V.; Gakuya, F.; Mutisya, J.; Mulwa, F.; Michuki, G.; Chepkorir, E.; Fischer, A.; Venter, M.; Sang, R. Isolation of tick and mosquito-borne arboviruses from ticks sampled from livestock and wild animal hosts in Ijara District, Kenya. Vector Borne Zoonotic Dis. 2013, 13, 637–642. [Google Scholar] [CrossRef]
- Weissenböck, H.; Hubálek, Z.; Bakonyi, T.; Nowotny, N. Zoonotic mosquito-borne flaviviruses: Worldwide presence of agents with proven pathogenicity and potential candidates of future emerging diseases. Vet. Microbiol. 2010, 140, 271–280. [Google Scholar]
- Petersen, L.R.; Marfin, A.A. Review West Nile Virus: A Primer for the Clinician. Ann. Intern. Med. 2002, 137, 173–179. [Google Scholar] [CrossRef]
- Pradier, S.; Lecollinet, S.; Leblond, A. West Nile virus epidemiology and factors triggering change in its distribution in Europe. Rev. Sci. Tech. 2012, 31, 829–844. [Google Scholar]
- Work, T.; Huribut, H.; Taylor, R. Indigenous wild birds of the Nile delta as potential WNV circulating reservoirs. Am. J. Trop. Med. Hyg. 1955, 4, 872–888. [Google Scholar]
- Bin, H.; Grossman, Z.; Pokamunski, S.; Malkinson, M.; Weiss, L.; Duvdevani, P.; Banet, C.; Weisman, Y.; Annis, E.; Gandaku, D.; et al. West Nile fever in Israel 1999–2000: From geese to humans. Ann. N. Y. Acad. Sci. 2001, 951, 127–142. [Google Scholar]
- Brault, A.C. Changing patterns of West Nile virus transmission: altered vector competence and host susceptibility. Vet. Res. 2009, 40, 43. [Google Scholar]
- Pauli, G.; Bauerfeind, U.; Blümel, J.; Burger, R.; Drosten, C.; Gröner, A.; Gürtler, L.; Heiden, M.; Hildebrandt, M.; Jansen, B.; et al. West nile virus. Transfus. Med. hemotherapy 2013, 40, 265–284. [Google Scholar]
- Gamino, V.; Höfle, U. Pathology and tissue tropism of natural West Nile virus infection in birds: A review. Vet. Res. 2013, 44, 39. [Google Scholar] [CrossRef] [Green Version]
- Bowen, R.A.; Nemeth, N.M. Experimental infections with West Nile virus. Curr. Opin. Infect. Dis. 2007, 20, 293–297. [Google Scholar] [CrossRef]
- Ben-Nathan, D. Stress and Virulence: West Nile virus encephalitis. Isr. J. Vet. Med. 2013, 68, 135–140. [Google Scholar]
- Vazquez, A.; Sanchez-Seco, M.P.; Ruiz, S.; Molero, F.; Hernandez, L.; Moreno, J.; Magallanes, A.; Tejedor, C.G.; Tenorio, A. Putative new lineage of west nile virus, Spain. Emerg. Infect. Dis. 2010, 16, 549–552. [Google Scholar] [CrossRef]
- McIntosh, B.; Dickinson, D.; McGillivray, G. Ecological studies on Sinbis and WNV in South Africa. V. The response of birds to inoculation of virus. South African J. Med. Sci. 1969, 77–82. [Google Scholar]
- Burt, F.J.; Grobbelaar, A.A.; Leman, P.A.; Anthony, F.S.; Gibson, G.V; Swanepoel, R. Phylogenetic relationships of southern African West Nile virus isolates. Emerg. Infect. Dis. 2002, 8, 820–826. [Google Scholar] [CrossRef]
- Boyle, D.B.; Dickerman, R.W.; Marshall, I.D. Primary viraemia responses of herons to experimental infection with Murray Valley encephalitis, Kunjin and Japanese encephalitis viruses. Aust. J. Exp. Biol. Med. Sci. 1983, 61, 655–664. [Google Scholar] [CrossRef]
- Boyle, D.B.; Marshall, I.D.; Dickerman, R.W. Primary antibody responses of herons to experimental infection with Murray Valley encephalitis and Kunjin viruses. Aust. J. Exp. Biol. Med. Sci. 1983, 61, 665–674. [Google Scholar]
- McLean, R.G.; Ubico, S.R.; Docherty, D.E.; Hansen, W.R.; Sileo, L.; McNamara, T.S. West Nile virus transmission and ecology in birds. Ann. N. Y. Acad. Sci. 2001, 951, 54–57. [Google Scholar]
- Brault, A.C.; Langevin, S.A.; Bowen, R.A.; Panella, N.A.; Biggerstaff, B.J.; Miller, B.R.; Komar, N. Differential virulence of West Nile strains for American crows. Emerg. Infect. Dis. 2004, 10, 2161–2168. [Google Scholar] [CrossRef]
- Brault, A.C.; Langevin, S.A.; Ramey, W.N.; Fang, Y.; Beasley, D.W.C.; Barker, C.M.; Sanders, T.A.; Reisen, W.K.; Barrett, A.D.T.; Bowen, R.A. Reduced avian virulence and viremia of West Nile virus isolates from Mexico and Texas. Am. J. Trop. Med. Hyg. 2011, 85, 758–767. [Google Scholar] [CrossRef]
- Wheeler, S.S.; Vineyard, M.P.; Woods, L.W.; Reisen, W.K. Dynamics of West Nile Virus Persistence in House Sparrows (Passer domesticus). PLoS Negl. Trop. Dis. 2012, 6, e1860. [Google Scholar] [CrossRef]
- Panella, N.A.; Young, G.; Komar, N. Experimental infection of Eurasian collared-dove (Streptopelia decaocto) with West Nile virus. J. Vector Ecol. 2013, 38, 210–214. [Google Scholar] [CrossRef]
- Diaz, L.A.; Flores, F.S.; Contigiani, M.S. Viremia profiles and host competence index for West Nile virus (Flavivirus, Flaviviridae) in three autochthonous birds species from Argentina. J. Ornithol. 2010, 152, 21–25. [Google Scholar]
- Lanciotti, R.; Roehrig, J.; Deubel, V.; Smith, J.; Parker, M.; Steele, K.; Crise, B.; Volpe, K.E.; Crabtree, M.B.; Scherret, J.H.; et al. Origin of the West Nile virus responsible for an outbreak of encephalitis in the northeastern United States. Science 1999, 286, 2333–2337. [Google Scholar] [CrossRef]
- Malkinson, M.; Banet, C.; Weisman, Y.; Pokamunski, S.; King, R.; Drouet, M.-T.; Deubel, V. Introduction of West Nile virus in the Middle East by migrating white storks. Emerg. Infect. Dis. 2002, 8, 392–397. [Google Scholar] [CrossRef]
- Banet-Noach, C.; Simanov, L.; Malkinson, M. Direct (non-vector) transmission of West Nile virus in geese. Avian Pathol. 2003, 32, 489–894. [Google Scholar] [CrossRef]
- Dridi, M.; Vangeluwe, D.; Lecollinet, S.; van den Berg, T.; Lambrecht, B. Experimental infection of Carrion crows (Corvuscorone) with two European West Nile virus (WNV) strains. Vet. Microbiol. 2013, 165, 160–166. [Google Scholar] [CrossRef]
- Brault, A.; Huang, C.; Langevin, S. A single positively selected West Nile viral mutation confers increased virogenesis in American crows. Nat. Genet. 2007, 39, 1162–1166. [Google Scholar]
- Langevin, S.A.; Brault, A.C.; Panella, N.A.; Bowen, R.A.; Komar, N. Variation in virulence of West Nile virus strains for house sparrows (Passer domesticus). Am. J. Trop. Med. Hyg. 2005, 72, 99–102. [Google Scholar]
- Sotelo, E.; Gutierrez-Guzmán, A.V.; Del Amo, J.; Llorente, F.; El-Harrak, M.; Pérez-Ramírez, E.; Blanco, J.M.; Höfle, U.; Jiménez-Clavero, M.Á. Pathogenicity of two recent Western Mediterranean West Nile virus isolates in a wild bird species indigenous to Southern Europe: The red-legged partridge. Vet. Res. 2011, 42, 11. [Google Scholar]
- Del Amo, J.; Llorente, F.; Figuerola, J.; Soriguer, R.; Moreno, A.; Cordioli, P.; Weissenböck, H.; Jimenez-Clavero, M.A. Experimental infection of house sparrows (Passer domesticus) with West Nile virus isolates of Euro-Mediterranean and North American origins. Vet. Res. 2014, in press. [Google Scholar]
- Ciccozzi, M.; Peletto, S.; Cella, E.; Giovanetti, M.; Lai, A.; Gabanelli, E.; Acutis, P.L.; Modesto, P.; Rezza, G.; Platonov, A.E.; et al. Epidemiological history and phylogeography of West Nile Virus lineage 2. Infect. Genet. Evol. 2013, 17, 46–50. [Google Scholar] [CrossRef]
- Bakonyi, T.; Ivanics, E.; Erdélyi, K.; Ursu, K.; Ferenczi, E.; Weissenböck, H.; Nowotny, N. Lineage 1 and 2 strains of encephalitic West Nile virus, central Europe. Emerg. Infect. Dis. 2006, 12, 618–623. [Google Scholar]
- Platonov, A.E.; Fedorova, M.V.; Karan, L.S.; Shopenskaya, T.A.; Platonova, O.V.; Zhuravlev, V.I. Epidemiology of West Nile infection in Volgograd, Russia, in relation to climate change and mosquito (Diptera: Culicidae) bionomics. Parasitol. Res. 2008, 103, S45–S53. [Google Scholar] [CrossRef]
- Sirbu, A.; Ceianu, C.S.; Panculescu-Gatej, R.I.; Vazquez, A.; Tenorio, A.; Rebreanu, R.; Niedrig, M.; Nicolescu, G.; Pistol, A. Outbreak of West Nile virus infection in humans, Romania, July to October 2010. Euro Surveill. 2011, 16, 19762. [Google Scholar]
- Ziegler, U.; Angenvoort, J.; Fischer, D.; Fast, C.; Eiden, M.; Rodriguez, A.V.; Revilla-Fernández, S.; Nowotny, N.; de la Fuente, J.G.; Lierz, M.; et al. Pathogenesis of West Nile virus lineage 1 and 2 in experimentally infected large falcons. Vet. Microbiol. 2013, 161, 263–273. [Google Scholar] [CrossRef]
- Beasley, D.W.C.; Li, L.; Suderman, M.T.; Barrett, A.D.T. Mouse neuroinvasive phenotype of West Nile virus strains varies depending upon virus genotype. Virology 2002, 296, 17–23. [Google Scholar] [CrossRef]
- Puig-Basagoiti, F.; Tilgner, M.; Bennett, C.J.; Zhou, Y.; Muñoz-Jordán, J.L.; García-Sastre, A.; Bernard, K.A.; Shi, P.-Y. A mouse cell-adapted NS4B mutation attenuates West Nile virus RNA synthesis. Virology 2007, 361, 229–241. [Google Scholar]
- Audsley, M.; Edmonds, J.; Liu, W.; Mokhonov, V.; Mokhonova, E.; Melian, E.B.; Prow, N.; Hall, R.A.; Khromykh, A.A. Virulence determinants between New York 99 and Kunjin strains of West Nile virus. Virology 2011, 414, 63–73. [Google Scholar] [CrossRef]
- Pijlman, G.P.; Funk, A.; Kondratieva, N.; Leung, J.; Torres, S.; van der Aa, L.; Liu, W.J.; Palmenberg, A.C.; Shi, P.-Y.; Hall, R.A.; et al. A highly structured, nuclease-resistant, noncoding RNA produced by flaviviruses is required for pathogenicity. Cell Host Microbe 2008, 4, 579–591. [Google Scholar]
- Shirato, K.; Miyoshi, H.; Goto, A.; Ako, Y.; Ueki, T.; Kariwa, H.; Takashima, I. Viral envelope protein glycosylation is a molecular determinant of the neuroinvasiveness of the New York strain of West Nile virus. J. Gen. Virol. 2004, 85, 3637–3645. [Google Scholar] [CrossRef]
- Beasley, D.W.C.; Whiteman, M.C.; Zhang, S.; Huang, C.Y.-H.; Schneider, B.S.; Smith, D.R.; Gromowski, G.D.; Higgs, S.; Kinney, R.M.; Barrett, A.D.T. Envelope protein glycosylation status influences mouse neuroinvasion phenotype of genetic lineage 1 West Nile virus strains. J. Virol. 2005, 79, 8339–8347. [Google Scholar] [CrossRef]
- Murata, R.; Eshita, Y.; Maeda, A.; Maeda, J.; Akita, S.; Tanaka, T.; Yoshii, K.; Kariwa, H.; Umemura, T.; Takashima, I. Glycosylation of the West Nile Virus envelope protein increases in vivo and in vitro viral multiplication in birds. Am. J. Trop. Med. Hyg. 2010, 82, 696–704. [Google Scholar] [CrossRef]
- Totani, M.; Yoshii, K.; Kariwa, H.; Takashima, I. Glycosylation of the envelope protein of West Nile Virus affects its replication in chicks. Avian Dis. 2011, 55, 561–568. [Google Scholar] [CrossRef]
- Sotelo, E.; Fernandez-Pinero, J.; Llorente, F.; Agüero, M.; Hoefle, U.; Blanco, J.M.; Jiménez-Clavero, M.Á. Characterization of West Nile virus isolates from Spain: New insights into the distinct West Nile virus eco-epidemiology in the Western Mediterranean. Virology 2009, 395, 289–297. [Google Scholar] [CrossRef]
- Reisen, W.K.; Fang, Y.; Martinez, V.M. Avian Host and Mosquito (Diptera: Culicidae) Vector competence determines the efficiency of West Nile and St. Louis encephalitis virus transmission. J. Med. Entomol. 2005, 42, 367–375. [Google Scholar] [CrossRef]
- VanDalen, K.K.; Hall, J.S.; Clark, L.; McLean, R.G.; Smeraski, C. West Nile virus infection in American robins: new insights on dose response. PLoS One 2013, 8, e68537. [Google Scholar]
- Styer, L.M.; Bernard, K.A.; Kramer, L.D. Enhanced early West Nile virus infection in young chickens infected by mosquito bite: Effect of viral dose. Am. J. Trop. Med. Hyg. 2006, 75, 337–345. [Google Scholar]
- Oesterle, P.T.; Nemeth, N.M.; VanDalen, K.; Sullivan, H.; Bentler, K.T.; Young, G.R.; McLean, R.G.; Clark, L.; Smeraski, C.; Hall, J.S. Experimental infection of Cliff swallows (Petrochelidon pyrrhonota) with varying doses of West Nile virus. Am. J. Trop. Med. Hyg. 2009, 81, 1159–1164. [Google Scholar]
- Styer, L.M.; Lim, P.-Y.; Louie, K.L.; Albright, R.G.; Kramer, L.D.; Bernard, K.A. Mosquito saliva causes enhancement of West Nile virus infection in mice. J. Virol. 2011, 85, 1517–1527. [Google Scholar] [CrossRef]
- Schneider, B.S.; Soong, L.; Girard, Y.A.; Campbell, G.; Mason, P.; Higgs, S. Potentiation of West Nile encephalitis by mosquito feeding. Viral Immunol. 2006, 19, 74–82. [Google Scholar] [CrossRef]
- Bingham, J.; Lunt, R.A.; Green, D.J.; Davies, K.R.; Stevens, V.; Wong, F.Y.K. Experimental studies of the role of the little raven (Corvus mellori) in surveillance for West Nile virus in Australia. Aust. Vet. J. 2010, 88, 204–210. [Google Scholar] [CrossRef]
- Komar, N.; Dohm, D.J.; Turell, M.J. Spielman, a Eastern equine encephalitis virus in birds: Relative competence of European starlings (Sturnus vulgaris). Am. J. Trop. Med. Hyg. 1999, 60, 387–391. [Google Scholar]
- Turell, M.J.; O’Guinn, M.; Oliver, J. Potential for New York mosquitoes to transmit West Nile virus. Am. J. Trop. Med. Hyg. 2000, 62, 413–414. [Google Scholar]
- Kilpatrick, A.M. Globalization, land use, and the invasion of West Nile virus. Science 2011, 334, 323–327. [Google Scholar] [CrossRef]
- Komar, N. West Nile virus: Epidemiology and ecology in North America. Adv. Virus Res. 2003, 61, 185–234. [Google Scholar] [CrossRef]
- Kilpatrick, A.; LaDeau, S.; Marra, P. Ecology of West Nile virus transmission and its impact on birds in the western hemisphere. Auk 2007, 124, 1121–1136. [Google Scholar] [CrossRef]
- Johnson, G.; Panella, N.; Hale, K.; Komar, N. Detection of West Nile Virus in Stable Flies (Diptera: Muscidae) Parasitizing Juvenile American White Pelicans. J. Med. Entomol. 2010, 47, 1205–1211. [Google Scholar] [CrossRef]
- Nemeth, N.; Gould, D.; Bowen, R.; Komar, N. Natural and experimental West Nile virus infection in five raptor species. J. Wildl. Dis. 2006, 42, 1–13. [Google Scholar] [CrossRef]
- Reisen, W.K.; Fang, Y.; Lothrop, H.D.; Martinez, V.M.; Wilson, J.; Oconnor, P.; Carney, R.; Cahoon-Young, B.; Shafii, M.; Brault, A.C. Overwintering of West Nile virus in Southern California. J. Med. Entomol. 2006, 43, 344–355. [Google Scholar] [CrossRef]
- Reisen, W.K.; Martinez, V. Role of California (Callipepla californica) and Gambel’s (Callipepla gambelii) quail in the ecology of mosquito-borne encephalitis viruses in California, USA. Vector Borne Zoonotic Dis. 2006, 6, 248–260. [Google Scholar] [CrossRef]
- McLean, R.; Shriner, R. Western equine encephalitis in avian populations in North Dakota, 1975. J. Wildl. Dis. 1989, 25, 481–489. [Google Scholar] [CrossRef]
- Mahmood, F.; Chiles, R.E.; Fang, Y.; Barker, C.M.; Reisen, W.K. Role of nestling Mourning Doves and House Finches as amplifying hosts of St.Louis encephalitis virus. J. Med. Entomol. 2004, 41, 965–972. [Google Scholar] [CrossRef]
- Blackmore, J.S.; Dow, R.P. Differential feeding of Culex tarsalis on nestling and adult birds. Mosq. News 1958, 18, 15–17. [Google Scholar]
- Kale, H.; Edman, J.; Webber, L. Effect of behavior and age of individual ciconiiform birds on mosquito feeding success. Mosq. News 1972, 32, 343–350. [Google Scholar]
- Swayne, D.E.; Beck, J.R.; Smith, C.S.; Shieh, W.J.; Zaki, S.R. Fatal encephalitis and myocarditis in young domestic geese (Anser anser domesticus) caused by West Nile virus. Emerg. Infect. Dis. 2001, 7, 751–753. [Google Scholar] [CrossRef]
- Sovada, M.; Pietz, P.; Converse, K.; Tommyking, D.; Hofmeister, E.; Scherr, P.; Ip, H. Impact of West Nile virus and other mortality factors on American white pelicans at breeding colonies in the northern plains of North America. Biol. Conserv. 2008, 141, 1021–1031. [Google Scholar] [CrossRef]
- Wünschmann, A.; Ziegler, A. West Nile virus-associated mortality events in domestic chukar partridges (Alectoris chukar) and domestic Impeyan pheasants (Lophophorus impeyanus). Avian Dis. 2006, 50, 456–459. [Google Scholar] [CrossRef]
- Nemeth, N.; Young, G.; Ndaluka, C.; Bielefeldt-Ohmann, H.; Komar, N.; Bowen, R. Persistent West Nile virus infection in the house sparrow (Passer domesticus). Arch. Virol. 2009, 154, 783–789. [Google Scholar] [CrossRef]
- Reisen, W.K.; Hahn, D.C. Comparison of immune responses of brown-headed cowbird and related blackbirds to west Nile and other mosquito-borne encephalitis viruses. J. Wildl. Dis. 2007, 43, 439–449. [Google Scholar] [CrossRef]
- Marsland, A.L.; Bachen, E.A.; Cohen, S.; Rabin, B.; Manuck, S.B. Stress, immune reactivity and susceptibility to infectious disease. Physiol. Behav. 2002, 77, 711–716. [Google Scholar] [CrossRef]
- Owen, J.C.; Nakamura, A.; Coon, C.A.; Martin, L.B. The effect of exogenous corticosterone on West Nile virus infection in Northern Cardinals (Cardinalis cardinalis). Vet. Res. 2012, 43, 34. [Google Scholar] [CrossRef]
- Nemeth, N.M.; Oesterle, P.T.; Bowen, R.A. Passive immunity to West Nile virus provides limited protection in a common passerine species. Am. J. Trop. Med. Hyg. 2008, 79, 283–290. [Google Scholar]
- Lapointe, D.A.; Hofmeister, E.K.; Atkinson, C.T.; Porter, R.E.; Dusek, R.J. Experimental infection of hawai`i`amakihi (Hemignathus virens) with West Nile Virus and competence of a co-occurring vector, Culex quinquefasciatus: Potential impacts on endemic hawaiian avifauna. J. Wildl. Dis. 2009, 45, 257–271. [Google Scholar] [CrossRef]
- Owen, J.; Moore, F.; Panella, N.; Edwards, E.; Bru, R.; Hughes, M.; Komar, N. Migrating birds as dispersal vehicles for West Nile virus. Ecohealth 2006, 3, 79–85. [Google Scholar] [CrossRef]
- Beaudoin, R.L.; Applegate, J.E.; Davis, D.E.; McLean, R.G. A model for the ecology of avian malaria. J. Wildl. Dis. 1971, 7, 5–13. [Google Scholar] [CrossRef]
- Gylfe, A.; Bergstrom, S.; Lundstrom, J.O.; Olsen, B. Reactivation of Borrelia infection in birds. Nature 2000, 403, 724–725. [Google Scholar] [CrossRef]
- Reisen, W.K.; Chiles, R.E.; Green, E.N.; Fang, Y.; Mahmood, F.; Martinez, V.M.; Laver, T. Effects of immunosuppression on encephalitis virus infection in the house finch, Carpodacus mexicanus. J. Med. Entomol. 2003, 40, 206–214. [Google Scholar] [CrossRef]
- Reisen, W.K.; Chiles, R.E.; Martinez, V.M.; Fang, Y.; Green, E.N. Encephalitis virus persistence in California birds: Experimental infections in mourning doves (Zenaidura macroura). J. Med. Entomol. 2004, 41, 462–466. [Google Scholar] [CrossRef]
- Fang, Y.; Reisen, W.K. Previous infection with West Nile or St. Louis encephalitis viruses provides cross protection during reinfection in house finches. Am. J. Trop. Med. Hyg. 2006, 75, 480–485. [Google Scholar]
- Guerrero-Sánchez, S.; Cuevas-Romero, S.; Nemeth, N.M.; Teresa, M.; Trujillo-Olivera, J.; Worwa, G.; Dupuis, A.; Brault, A.C.; Kramer, L.D.; Komar, N.; et al. West Nile Virus Infection of Birds, Mexico. Emerg. Infect. Dis. 2011, 17, 2245–2252. [Google Scholar]
- Kipp, A.M.; Lehman, J.A.; Bowen, R.A.; Fox, P.E.; Stephens, M.R.; Klenk, K.; Komar, N.; Bunning, M.L. West Nile virus quantification in feces of experimentally infected American and fish crows. Am. J. Trop. Med. Hyg. 2006, 75, 688–690. [Google Scholar]
- Kinney, R.M.; Huang, C.Y.-H.; Whiteman, M.C.; Bowen, R.A.; Langevin, S.A.; Miller, B.R.; Brault, A.C. Avian virulence and thermostable replication of the North American strain of West Nile virus. J. Gen. Virol. 2006, 87, 3611–3622. [Google Scholar]
- Weingartl, H.M.; Neufeld, J.L.; Copps, J.; Marszal, P. Experimental West Nile virus infection in blue jays (Cyanocitta cristata) and crows (Corvus brachyrhynchos). Vet. Pathol. 2004, 41, 362–370. [Google Scholar]
- Nemeth, N.M.; Thomsen, B.V; Spraker, T.R.; Benson, J.M.; Bosco-Lauth, A.M.; Oesterle, P.T.; Bright, J.M.; Muth, J.P.; Campbell, T.W.; Gidlewski, T.L.; Bowen, R.A. Clinical and pathologic responses of American crows (Corvus brachyrhynchos) and fish crows (C ossifragus) to experimental West Nile virus infection. Vet. Pathol. 2011, 48, 1061–1074. [Google Scholar] [CrossRef]
- Shirafuji, H.; Kanehira, K.; Kubo, M.; Shibahara, T.; Kamio, T. Experimental West Nile virus infection in jungle crows (Corvus macrorhynchos). Am. J. Trop. Med. Hyg. 2008, 78, 838–842. [Google Scholar]
- Komar, N.; Panella, N.A; Langevin, S.A; Brault, A.C.; Amador, M.; Edwards, E.; Owen, J.C. Avian hosts for West Nile virus in St. Tammany Parish, Louisiana, 2002. Am. J. Trop. Med. Hyg. 2005, 73, 1031–1037. [Google Scholar]
- Nemeth, N.M.; Bosco-Lauth, A.M.; Bowen, R.A. Cross-protection between West Nile and Japanese encephalitis viruses in red-winged blackbirds (Agelaius phoeniceus). Avian Dis. 2009, 53, 421–425. [Google Scholar] [CrossRef]
- Reisen, W.K.; Fang, Y. Does feeding on infected mosquitoes (Diptera: Culicidae) enhance the role of Song sparrows in the transmission of arboviruses in California? J. Med. Entomol. 2007, 44, 316–319. [Google Scholar]
- Oesterle, P.; Nemeth, N.; Young, G.; Mooers, N.; Elmore, S.; Bowen, R.; Doherty, P.; Hall, J.; Mclean, R.; Clark, L. Cliff Swallows, Swallow Bugs, and West Nile Virus: An unlikely transmission mechanis. Vector Borne Zoonotic Dis. 2010, 10, 507–513. [Google Scholar] [CrossRef]
- Kilpatrick, A.M.; Peters, R.J.; Dupuis, A.P.; Jones, M.J.; Marra, P.P.; Kramer, L.D. Predicted and observed mortality from vector-borne disease in small songbirds. Biol. Conserv. 2013, 165, 79–85. [Google Scholar] [CrossRef]
- Busquets, N.; Bertran, K.; Costa, T.P.; Rivas, R.; de la Fuente, J.G.; Villalba, R.; Solanes, D.; Bensaid, A.; Majó, N.; Pagès, N. Experimental West Nile virus infection in Gyr-Saker hybrid falcons. Vector Borne Zoonotic Dis. 2012, 12, 482–489. [Google Scholar] [CrossRef]
- Nemeth, N.M.; Hahn, D.C.; Gould, D.H.; Bowen, R.A. Experimental West Nile virus infection in Eastern Screech Owls (Megascops asio). Avian Dis. 2006, 50, 252–258. [Google Scholar] [CrossRef]
- Escribano-Romero, E.; Gamino, V.; Merino-Ramos, T.; Blázquez, A.B.; Martín-Acebes, M.A.; de Oya, N.J.; Gutiérrez-Guzmán, A.V; Escribano, J.M.; Höfle, U.; Saiz, J.C. Protection of red-legged partridges (Alectoris rufa) against West Nile virus (WNV) infection after immunization with WNV recombinant envelope protein E (rE). Vaccine 2013, 31, 4523–4527. [Google Scholar]
- Clark, L.; Hall, J.; McLean, R.; Dunbar, M.; Klenk, K.; Bowen, R.; Smeraski, C.A. Susceptibility of greater sage-grouse to experimental infection with West Nile virus. J. Wildl. Dis. 2006, 42, 14–22. [Google Scholar]
- Olsen, G.H.; Miller, K.J.; Docherty, D.E.; Bochsler, V.S.; Sileo, L. Pathogenicity of West Nile virus and response to vaccination in sandhill cranes (Grus canadensis) using a killed vaccine. J. Zoo Wildl. Med. 2009, 40, 263–271. [Google Scholar]
- Wheeler, S.S.; Langevin, S.A.; Brault, A.C.; Woods, L.; Carroll, B.D.; Reisen, W.K. Detection of persistent West Nile virus RNA in experimentally and naturally infected avian hosts. Am. J. Trop. Med. Hyg. 2012, 559–564. [Google Scholar]
- Kilpatrick, A.M.; Daszak, P.; Jones, M.J.; Marra, P.P.; Kramer, L.D. Host heterogeneity dominates West Nile virus transmission. Proc. Biol. Sci. 2006, 273, 2327–2333. [Google Scholar] [CrossRef]
- Langevin, S.A.; Bunning, M.; Davis, B.; Komar, N. Experimental infection of chickens as candidate sentinels for West Nile virus. Emerg. Infect. Dis. 2001, 7, 726–729. [Google Scholar] [CrossRef]
- Nemeth, N.M.; Oesterle, P.T.; Bowen, R.A. Humoral immunity to West Nile virus is long-lasting and protective in the house sparrow (Passer domesticus). Am. J. Trop. Med. Hyg. 2009, 80, 864–869. [Google Scholar]
- Nemeth, N.M.; Bowen, R.A. Dynamics of passive immunity to West Nile virus in domestic chickens (Gallus gallus domesticus). Am. J. Trop. Med. Hyg. 2007, 76, 310–317. [Google Scholar]
- Gibbs, S.E.J.; Hoffman, D.M.; Stark, L.M.; Marlenee, N.L.; Blitvich, B.J.; Beaty, B.J.; Stallknecht, D.E. Persistence of antibodies to West Nile virus in naturally infected rock pigeons (Columba livia). Clin. Diagn. Lab. Immunol. 2005, 12, 665–667. [Google Scholar]
- Hahn, D.C.; Nemeth, N.M.; Edwards, E.; Bright, P.R.; Komar, N. Passive West Nile virus antibody transfer from maternal Eastern screech-owls (Megascops asio) to progeny. Avian Dis. 2006, 50, 454–455. [Google Scholar] [CrossRef]
- Figuerola, J.; Jiménez-Clavero, M.A.; Rojo, G.; Gómez-Tejedor, C.; Soriguer, R. Prevalence of West Nile virus neutralizing antibodies in colonial aquatic birds in southern Spain. Avian Pathol. 2007, 36, 209–212. [Google Scholar] [CrossRef]
- Mackenzie, J.S.; Gubler, D.J.; Petersen, L.R. Emerging flaviviruses: The spread and resurgence of Japanese encephalitis, West Nile and dengue viruses. Nat. Med. 2004, 10, S98–S109. [Google Scholar] [CrossRef]
- Llorente, F.; Pérez-Ramírez, E.; Fernández-Pinero, J.; Soriguer, R.; Figuerola, J.; Jiménez-Clavero, M.Á. Flaviviruses in game birds, southern Spain, 2011–2012. Emerg. Infect. Dis. 2013, 19, 1023–1025. [Google Scholar]
- Vázquez, A.; Jiménez-Clavero, M.A.; Franco, L.; Donoso-Mantke, O.; Sambri, V.; Niedrig, M.; Zeller, H.; Tenorio, A. Usutu virus: Potential risk of human disease in Europe. Euro Surveill. 2011, 16, 22–26. [Google Scholar]
- Schneider, B.; Higgs, S. The enhancement of arbovirus transmission and disease by mosquito saliva is associated with modulation of the host immune response. Trans. R. Soc. Trop. Med. Hyg. 2008, 102, 400–408. [Google Scholar] [CrossRef]
- Reisen, W.K.; Fang, Y.; Martinez, V. Is nonviremic transmission of West Nile virus by Culex mosquitoes (Diptera: Culicidae) nonviremic? J. Med. Entomol. 2007, 44, 299–302. [Google Scholar] [CrossRef]
- Donaldson, J.M. An assessment of Culex pipiens quinquefasciatus Say as a vector of viruses in the witwatersrand region of the transvaal. I. West Nile virus. South African J. Med. Sci. 1966, 31, 1–10. [Google Scholar]
- Abbassy, M.M.; Osman, M.; Marzouk, A.S. West Nile virus (Flaviviridae: Flavivirus) in experimentally infected argas ticks (Acari:Argasidae). Am. J. Trop. Med. Hyg. 1993, 48, 726–737. [Google Scholar]
- Hutcheson, H.J.; Gorham, C.H.; Machain-Williams, C.; Loroño-Pino, M.A.; James, A.M.; Marlenee, N.L.; Winn, B.; Beaty, B.J.; Blair, C.D. Experimental transmission of West Nile virus (Flaviviridae: Flavivirus) by Carios capensis ticks from North America. Vector Borne Zoonotic Dis. 2005, 5, 293–295. [Google Scholar] [CrossRef]
- Reisen, W.K.; Brault, A.C.; Martinez, V.M.; Fang, Y.; Simmons, K.; Garcia, S.; Omi-Olsen, E.; Lane, R.S. Ability of transstadially infected Ixodes pacificus (Acari: Ixodidae) to transmit West Nile virus to song sparrows or western fence lizards. J. Med. Entomol. 2007, 44, 320–327. [Google Scholar]
- Garmendia, A.E.; van Kruiningen, H.J.; French, R.A.; Anderson, J.F.; Andreadis, T.G.; Kumar, A.; West, A.B. Recovery and identification of West Nile Virus from a hawk in winter. J. Clin. Microbiol. 2000, 38, 3110–3111. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pérez-Ramírez, E.; Llorente, F.; Jiménez-Clavero, M.Á. Experimental Infections of Wild Birds with West Nile Virus. Viruses 2014, 6, 752-781. https://doi.org/10.3390/v6020752
Pérez-Ramírez E, Llorente F, Jiménez-Clavero MÁ. Experimental Infections of Wild Birds with West Nile Virus. Viruses. 2014; 6(2):752-781. https://doi.org/10.3390/v6020752
Chicago/Turabian StylePérez-Ramírez, Elisa, Francisco Llorente, and Miguel Ángel Jiménez-Clavero. 2014. "Experimental Infections of Wild Birds with West Nile Virus" Viruses 6, no. 2: 752-781. https://doi.org/10.3390/v6020752







