A Current Overview of Two Viroids That Infect Chrysanthemums: Chrysanthemum stunt viroid and Chrysanthemum chlorotic mottle viroid
Abstract
:1. Introduction
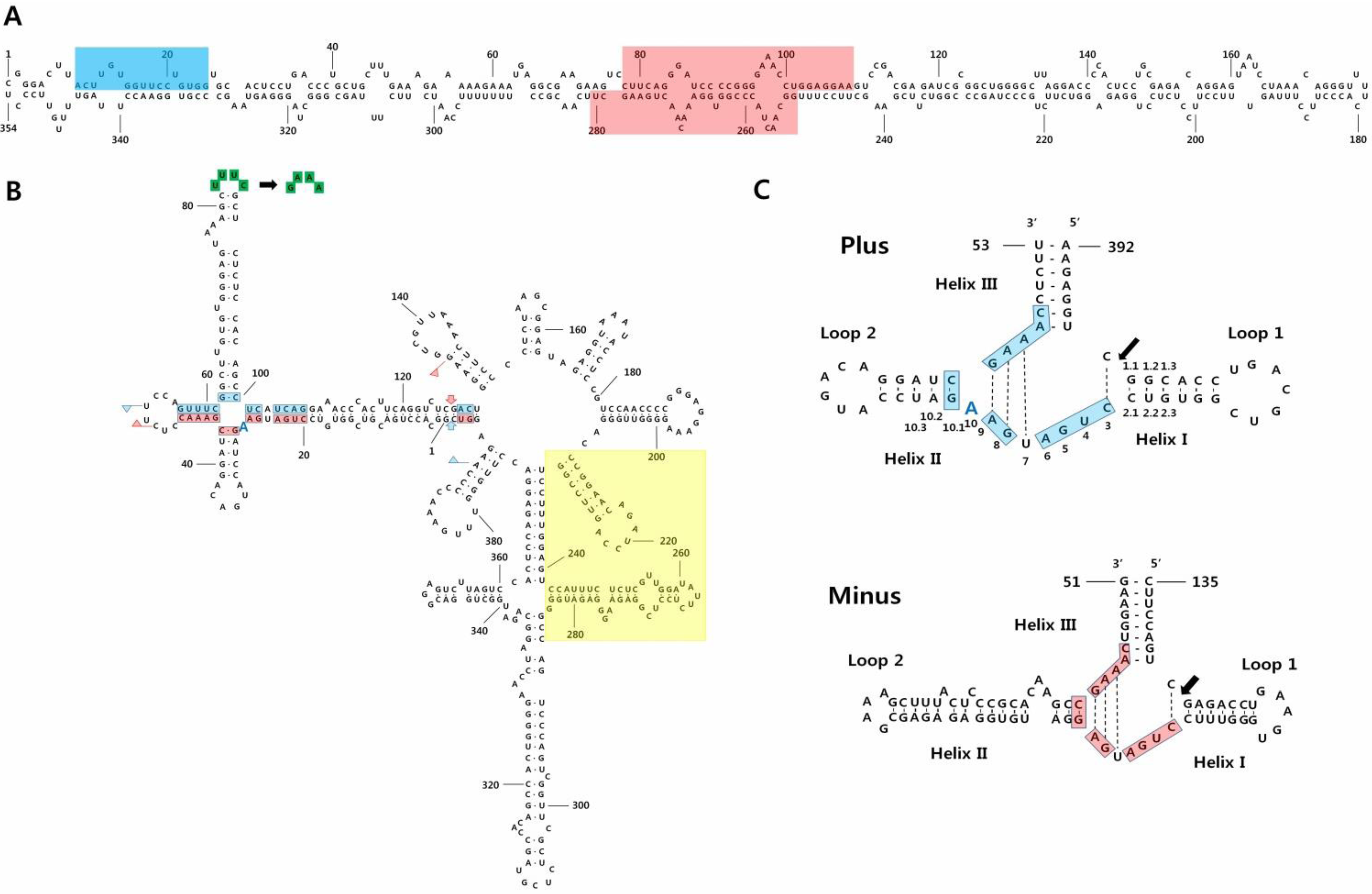
2. Symptoms, Isolation, Nucleotide Sequences, and Structures
| Characteristics | CSVd | CChMVd |
|---|---|---|
| Disease | Chrysanthemum stunt | Chrysanthemum chlorotic mottle |
| Symptoms | Light green young leaves, chlorotic spots, stunting, small leaves and flowers, and decreased rooting ability | Yellow-green mottling, chlorosis, and dwarfed size |
| Family and genus | Pospiviroidae, Pospiviroid | Avsunviroidae, Pelamoviroid |
| Genome size | 354–356 nt | 398–401 nt |
| Replication method | Asymmetric rolling circle mechanism | Symmetric rolling circle mechanism with the hammerhead ribozymes |
| Replication localization | Nucleus | Chloroplast |
| Structure | Rod-like structure including central conserved region (CCR) | Branched conformation including hammerhead ribozymes |
| Transmission | Sap, grafting, and seed | Sap, grafting |
| Host | Chrysanthemums, Petunia hybrida, tomato, Gynura aurantiaca, Ageratum, dahlia, Senecio, Vinca major, Argyranthemum frutescens and many plants belonging to the families Solanaceae and Asteraceae | Restricted to chrysanthemums |
3. Host Range
4. Identification of the Two Viroids
5. Replication Mechanisms
6. Symptom Determinants
7. Detection Methods
8. Efforts to Eliminate Viroids from Infected Plants
9. Identification and Development of Viroid-Resistant Chrysanthemum Cultivars
10. Other Studies Associated with CSVd and CChMVd
11. Future Directions in Host-Viroid Interaction Studies
Acknowledgments
Conflict of Interest
References and Notes
- Tomassoli, L.; Faggioli, F.; Zaccaria, A.; Caccia, R.; Albani, M.; Barba, M. Molecular diagnosis of Chrysanthemum stunt viroid for routine indexing. Phytopathol. Mediterr. 2004, 43, 285–288. [Google Scholar]
- Verma, N.; Sharma, A.; Ram, R.; Hallan, V.; Zaidi, A.A.; Garg, I.D. Detection, identification and incidence of Chrysanthemum B carlavirus in chrysanthemum in India. Crop Prot. 2003, 22, 425–429. [Google Scholar] [CrossRef]
- Ding, B. Viroids: Self-replicating, mobile, and fast-evolving noncoding regulatory RNAs. Wiley Interdiscip. Rev. RNA 2010, 1, 362–375. [Google Scholar] [CrossRef]
- Navarro, B.; Gisel, A.; Rodio, M.E.; Delgado, S.; Flores, R.; di Serio, F. Viroids: How to infect a host and cause disease without encoding proteins. Biochimie 2012, 94, 1474–1480. [Google Scholar] [CrossRef]
- Ding, B.; Wang, Y. Viroids: Uniquely simple and tractable models to elucidate regulation of cell-to-cell trafficking of RNA. DNA Cell. Biol. 2009, 28, 51–56. [Google Scholar] [CrossRef]
- Góra-Sochacka, A. Viroids: Unusual small pathogenic RNAs. Acta Biochim. Pol. 2004, 51, 587–608. [Google Scholar]
- Owens, R.A.; Flores, R.; Di Serio, F.; Li, S.F.; Pallás, V.; Randles, J.W.; Sano, T.; Vidalakis, G. Viroids. In Virus Taxonomy: Ninth Report of the International Committee on Taxonomy of Viruses; Andrew, M.Q.K., Michael, J.A., Eric, B.C., Elliot, J.L., Eds.; Elsevier Academic Press: London, UK, 2012; pp. 1221–1234. [Google Scholar]
- Keese, P.; Symons, R.H. Domains in viroids: Evidence of intermolecular RNA rearrangements and their contribution to viroid evolution. Proc. Natl. Acad. Sci. USA 1985, 82, 4582–4586. [Google Scholar] [CrossRef]
- McInnes, J.; Symons, R.H. Comparative Structure of Viroids and Their Rapid Detection using Radioactive and Nonradioactive Nucleic Acid Probes. In Viroids and Satellites: Molecular Parasites at the Frontier of Life; Maramorosch, K., Ed.; CRC Press: Boca Raton, FL, USA, 1991; pp. 21–58. [Google Scholar]
- Baumstark, T.; Schröder, A.R.; Riesner, D. Viroid processing: Switch from cleavage to ligation is driven by a change from a tetraloop to a loop E conformation. EMBO J. 1997, 16, 599–610. [Google Scholar] [CrossRef]
- Gas, M.-E.; Hernández, C.; Flores, R.; Daròs, J.-A. Processing of nuclear viroids in vivo: An interplay between RNA conformations. PLoS Pathog. 2007, 3, e182. [Google Scholar] [CrossRef]
- Gas, M.-E.; Molina-Serrano, D.; Hernández, C.; Flores, R.; Daròs, J.-A. Monomeric linear RNA of citrus exocortis viroid resulting from processing in vivo has 5'-phosphomonoester and 3'-hydroxyl termini: Implications for the RNase and RNA ligase involved in replication. J. Virol. 2008, 82, 10321–10325. [Google Scholar] [CrossRef]
- Koltunow, A.M.; Rezaian, M.A. Grapevine yellow speckle viroid: Structural features of a new viroid group. Nucleic Acids Res. 1988, 16, 849–864. [Google Scholar] [CrossRef]
- Puchta, H.; Ramm, K.; Sänger, H.L. The molecular structure of hop latent viroid (HLV), a new viroid occurring worldwide in hops. Nucleic Acids Res. 1988, 16, 4197–4216. [Google Scholar]
- Flores, R.; Di Serio, F.; Hernández, C. Viroids: The Noncoding Genomes, Seminars in Virology, 1997; Elsevier: Waltham, MA, USA, 1997; pp. 65–73. [Google Scholar]
- Ding, B. The biology of viroid-host interactions. Annu. Rev. Phytopathol. 2009, 47, 105–131. [Google Scholar] [CrossRef]
- Diener, T.; Lawson, R. Chrysanthemum stunt: A viroid disease. Virology 1973, 51, 94–101. [Google Scholar] [CrossRef]
- Diener, T.O. Viroids: The smallest known agents of infectious disease. Annu. Rev. Microbiol. 1974, 28, 23–40. [Google Scholar]
- Haseloff, J.; Symons, R.H. Chrysanthemum stunt viroid: Primary sequence and secondary structure. Nucleic Acids Res. 1981, 9, 2741–2752. [Google Scholar]
- Gross, H.J.; Krupp, G.; Domdey, H.; Raba, M.; Jank, P.; Lossow, C.; Alberty, H.; Ramm, K.; Sanger, H.L. Nucleotide sequence and secondary structure of citrus exocortis and chrysanthemum stunt viroid. Eur. J. Biochem. 1982, 121, 249–257. [Google Scholar] [CrossRef]
- Sieger, G.; Hofmann, H.; Förtsch, J.; Gross, H.J.; Randies, J.W.; Sänger, H.L.; Riesner, D. Conformational transitions in viroids and virusoids: Comparison of results from energy minimization algorithm and from experimental data. J. Biomol. Struct. Dyn. 1984, 2, 543–571. [Google Scholar] [CrossRef]
- Yoon, J.Y.; Palukaitis, P. Sequence comparisons of global Chrysanthemum stunt viroid variants: Multiple polymorphic positions scattered through the viroid genome. Virus Genes 2012, 46, 97–104. [Google Scholar]
- Yoon, J.; Baek, E.; Palukaitis, P. Are there strains of Chrysanthemum stunt viroid? J. Plant Pathol. 2012, 94, 697–701. [Google Scholar]
- Dimock, A.W.; Geissinger, C.M. A newly recognized disease of chrysanthemum caused by a graft-transmissible agent. Phytopathology 1969, 59, 1024 (abstr.). [Google Scholar]
- Dimock, A.W.; Geissinger, C.M.; Horst, R.K. Chlorotic mottle: A newly recognized disease oí chrysanthemum. Phytopathology 1971, 61, 415–419. [Google Scholar] [CrossRef]
- Navarro, B.; Flores, R. Chrysanthemum chlorotic mottle viroid: Unusual structural properties of a subgroup of self-cleaving viroids with hammerhead ribozymes. Proc. Natl. Acad. Sci. USA 1997, 94, 11262–11267. [Google Scholar] [CrossRef]
- Gago, S.; Elena, S.F.; Flores, R.; Sanjuán, R. Extremely high mutation rate of a hammerhead viroid. Science 2009, 323, 1308–1308. [Google Scholar]
- De la Peña, M.; Flores, R. An extra nucleotide in the consensus catalytic core of a viroid hammerhead ribozyme: Implication for the design of more efficient ribozymes. J. Biol. Chem. 2001, 276, 34586–34593. [Google Scholar]
- De la Pena, M.; Flores, R. Chrysanthemum chlorotic mottle viroid RNA: Dissection of the pathogenicity determinant and comparative fitness of symptomatic and non-symptomatic variants. J. Mol. Biol. 2002, 321, 411–421. [Google Scholar] [CrossRef]
- Dufour, D.; De La Peña, M.; Gago, S.; Flores, R.; Gallego, J. Structure-function analysis of the ribozymes of Chrysanthemum chlorotic mottle viroid: A loop-loop interaction motif conserved in most natural hammerheads. Nucleic Acids Res. 2009, 37, 368–381. [Google Scholar]
- Runia, W.T.; Peters, D. The response of plant species used in agriculture and horticulture to viroid infections. Eur. J. Plant. Pathol. 1980, 86, 135–146. [Google Scholar]
- Palukaitis, P.; Symons, R.H. Purification and characterization of the circular and linear forms of chrysanthemum stunt viroid. J. Gen. Virol. 1980, 46, 477–489. [Google Scholar]
- Matsushita, Y.; Penmetcha, K.K.R. In vitro-transcribed Chrysanthemum stunt viroid RNA is infectious to chrysanthemum and other plants. Phytopathology 2009, 99, 58–66. [Google Scholar] [CrossRef]
- Verhoeven, J.T.J.; Arts, M.S.J.; Owens, R.A.; Roenhorst, J.W. Natural infection of petunia by chrysanthemum stunt viroid. Eur. J. Plant. Pathol. 1998, 104, 383–386. [Google Scholar]
- Nakashima, A.; Hosokawa, M.; Maeda, S.; Yazawa, S. Natural infection of Chrysanthemum stunt viroid in dahlia plants. J. Gen. Plant. Pathol. 2007, 73, 225–227. [Google Scholar]
- Singh, D.; Pathania, M.; Ram, R.; Zaidi, A.A.; Verma, N. Screening of chrysanthemum cultivars for Chrysanthemum stunt viroid in an Indian scenario. Arch. Phytopathol. Plant. Protect. 2010, 43, 1517–1523. [Google Scholar] [CrossRef]
- Nie, X.; Singh, R.P.; Bostan, H. Molecular cloning, secondary structure, and phylogeny of three pospiviroids from ornamental plants. Can. J. Plant Pathol. 2005, 27, 592–602. [Google Scholar] [CrossRef]
- Torchetti, E.M.; Navarro, B.; Trisciuzzi, V.N.; Nuccitelli, L.; Silletti, M.R.; Di Serio, F. First report of Chrysanthemum stunt viroid in Argyranthemum frutescens in Italy. J. Plant. Pathol. 2012, 94, 451–454. [Google Scholar]
- Menzel, W.; Maiss, E. Detection of Chrysanthemum stunt viroid (CSVd) in cultivars of Argyranthemum frutescens by RT-PCR-ELISA. Z. Pflanzenkr. Pflanzenschutz 2000, 107, 548–552. [Google Scholar]
- Horst, R.K. Chrysanthemum chlorotic mottle. In The Viroids, The Viruses series; Plenum Press: New York, NY, USA, 1987; Volume 11, pp. 291–295. [Google Scholar]
- Chung, B.N.; Pak, H.S. Seed transmission of Chrysanthemum stunt viroid in chrysanthemum. Plant Pathol. J. 2008, 24, 31–35. [Google Scholar]
- Sugiura, H.; Hanada, K. Chrysanthemum stunt viroid, a disease of large-flowered chrysanthemum [Dendranthema] in Niigata prefecture [Japan]. J. Japan. Soc. Hort. Sci. 1998, 67, 432–438. [Google Scholar] [CrossRef]
- Hill, M.F.; Giles, R.J.; Moran, J.R.; Hepworth, G. The incidence of chrysanthemum stunt viroid, chrysanthemum B carlavirus, tomato aspermy cucumovirus and tomato spotted wilt tospovirus in Australian chrysanthemum crops. Australas. Plant Pathol. 1996, 25, 174–178. [Google Scholar]
- Monger, W.; Tomlinson, J.; Booonham, N.; Marn, M.V.; Plesko, I.M.; Molinero-Demilly, V.; Tassus, X.; Meekes, E.; Toonen, M.; Papayiannis, L. Development and inter-laboratory evaluation of real-time PCR assays for the detection of pospiviroids. J. Virol. Methods 2010, 169, 207–210. [Google Scholar]
- Baker, R.; Candresse, T.; Dormannsné Simon, E.; Gilioli, G.; Grégoire, J.-C.; Jeger, M.J.; Karajova, O.E.; Lövei, G.; Makowski, D.; Manceau, C.; et al. Scientific opinion on the assessment of the risk of solanaceous pospiviroids for the EU territory and the identification and evaluation of risk management options: Scientific Opinion; EFSA Panel on Plant Health (PLH). EFSA J. 2011, 9, 2330. [Google Scholar]
- Mehle, N.; Seljak, G.; Verhoeven, J.T.J.; Jansen, C.C.C.; Prezelj, N.; Ravnikar, M. Chrysanthemum stunt viroid newly reported in Slovenia. Plant Pathol. 2010, 59, 1159–1159. [Google Scholar]
- Bostan, H.; Gazel, M.; Elibuyuk, I.O. Occurrence of Pospiviroid in potato, tomato and some ornamental plants in Turkey. Afr. J. Biotechnol. 2012, 9, 2613–2617. [Google Scholar]
- El-Dougdoug, K.A.; Rezk, A.A.; Rehab, D.A.; Sofy, A.R. Partially nucleotide sequence and secondary structure of Chrysanthemum stunt viroid Egyptian isolate from infected-chrysanthemum plants. Int. J. Virol. 2012, 8, 133–150. [Google Scholar] [CrossRef]
- Matsushitali, Y. Nucleotide sequences and distribution of Chrysanthemum stunt viroid in Japan. J. Japan. Soc. Hort. Sci. 2007, 76, 333–337. [Google Scholar]
- Yamamoto, H.; Sano, T. Occurrence of Chrysanthemum chlorotic mottle viroid in Japan. J. Gen. Plant. Pathol. 2005, 71, 156–157. [Google Scholar] [CrossRef]
- Yamamoto, H.; Sano, T. An epidemiological survey of Chrysanthemum chlorotic mottle viroid in Akita Prefecture as a model region in Japan. J. Gen. Plant. Pathol. 2006, 72, 387–390. [Google Scholar] [CrossRef]
- Hosokawa, M.; Matsushita, Y.; Ohishi, K.; Yazawa, S. Elimination of Chrysanthemum chlorotic mottle viroid (CChMVd) recently detected in Japan by leaf-primordia free shoot apical meristem culture from infected cultivars. J. Japan. Soc. Hort. Sci. 2005, 74, 386–391. [Google Scholar] [CrossRef]
- Chung, B.N.; Choi, G.S.; Kim, H.R.; Kim, J.S. Chrysanthemum stunt viroid in Dendranthema grandiflorum. Plant Pathol. J. 2001, 17, 194–200. [Google Scholar]
- Chung, B.N.; Lim, J.H.; Choi, S.Y.; Kim, J.S.; Lee, E.J. Occurrence of Chrysanthemum stunt viroid in chrysanthemum in Korea. Plant Pathol. J. 2005, 21, 377–382. [Google Scholar] [CrossRef]
- Zhang, Z.X.; Pan, S.; Li, S.F. First Report of Chrysanthemum chlorotic mottle viroid in Chrysanthemum in China. Plant. Dis. 2011, 95, 1320. [Google Scholar]
- Flores, R.; Daròs, J.-A.; Hernández, C. Avsunviroidae family: Viroids containing hammerhead ribozymes. Adv. Virus Res. 2000, 55, 271–323. [Google Scholar] [CrossRef]
- Flores, R.; Hernández, C.; Alba, A.E.M.d.; Daròs, J.-A.; Serio, F.D. Viroids and viroid-host interactions. Annu. Rev. Phytopathol. 2005, 43, 117–139. [Google Scholar] [CrossRef]
- Branch, A.D.; Robertson, H.D. A replication cycle for viroids and other small infectious RNA’s. Science 1984, 223, 450–455. [Google Scholar]
- Flores, R.; Navarro, J.A.; Pena, M.; Navarro, B.; Ambrós, S.; Vera, A. Viroids with hammerhead ribozymes: Some unique structural and functional aspects with respect to other members of the group. Biol. Chem. 1999, 380, 849–854. [Google Scholar]
- Kaddour, H.; Vergne, J.; Hervé, G.; Maurel, M.C. High-pressure analysis of a hammerhead ribozyme from Chrysanthemum chlorotic mottle viroid reveals two different populations of self-cleaving molecule. FEBS J. 2011, 278, 3739–3747. [Google Scholar] [CrossRef]
- Gago, S.; De la Peña, M.; Flores, R. A kissing-loop interaction in a hammerhead viroid RNA critical for its in vitro folding and in vivo viability. RNA 2005, 11, 1073–1083. [Google Scholar] [CrossRef]
- De la Peña, M.; Navarro, B.; Flores, R. Mapping the molecular determinant of pathogenicity in a hammerhead viroid: A tetraloop within the in vivo branched RNA conformation. Proc. Natl. Acad. Sci. USA 1999, 96, 9960–9965. [Google Scholar] [CrossRef]
- Mosch, W.H.M.; Huttinga, H.; Hakkaart, F.A.; De Bokx, J.A. Detection of chrysanthemum stunt and potato spindle tuber viroids by polyacrylamide gelelectrophoresis. Eur. J. Plant Pathol. 1978, 84, 85–93. [Google Scholar]
- Morris, T.; Smith, E. Potato spindle tuber disease: Procedures for the detection of viroid RNA and certification of disease-free potato tubers. Phytopathology 1977, 67, 145–150. [Google Scholar] [CrossRef]
- Palukaitis, P.; Symons, R.H. Hybridization analysis of Chrysanthemum stunt viroid with complementary DNA and the quantitation of viroid RNA sequences in extracts of infected plants. Virology 1979, 98, 238–245. [Google Scholar] [CrossRef]
- Candresse, T.; Macquaire, G.; Monsion, M.; Dunez, J. Detection of Chrysanthemum stunt viroid (CSVd) using nick translated probes in a dot-blot hybridization assay. J. Virol. Methods 1988, 20, 185–193. [Google Scholar] [CrossRef]
- Macquaire, G.; Monsion, M.; Mouches, C.; Candresse, T.; Dunez, J. Spot hybridization: Application to viroid identification. Ann. Inst. Pasteur. Virol. 1984, 135, 219–230. [Google Scholar]
- McInnes, J.L.; Habili, N.; Symons, R.H. Nonradioactive, photobiotin-labelled DNA probes for routine diagnosis of viroids in plant extracts. J. Virol. Methods 1989, 23, 299–312. [Google Scholar] [CrossRef]
- Candresse, T.; Macquaire, G.; Brault, V.; Monsion, M.; Dunez, J. 32P-and biotin-labelled in vitro transcribed cRNA probes for the detection of Potato spindle tuber viroid and Chrysanthemum stunt viroid. Res. Virol. 1990, 141, 97–107. [Google Scholar] [CrossRef]
- Nakahara, K.; Hataya, T.; Uyeda, I. A simple, rapid method of nucleic acid extraction without tissue homogenization for detecting viroids by hybridization and RT-PCR. J. Virol. Methods 1999, 77, 47–58. [Google Scholar] [CrossRef]
- Bostan, H.; Nie, X.; Singh, R.P. An RT-PCR primer pair for the detection of Pospiviroid and its application in surveying ornamental plants for viroids. J. Virol. Methods 2004, 116, 189–193. [Google Scholar] [CrossRef]
- Torchetti, E.M.; Navarro, B.; Di Serio, F. A single polyprobe for detecting simultaneously eight pospiviroids infecting ornamentals and vegetables. J. Virol. Methods 2012, 186, 141–146. [Google Scholar] [CrossRef]
- Ragozzino, E.; Faggioli, F.; Barba, M. Development of a one tube-one step RT-PCR protocol for the detection of seven viroids in four genera: Apscaviroid, Hostuviroid, Pelamoviroid and Pospiviroi. J. Virol. Methods 2004, 121, 25–29. [Google Scholar] [CrossRef]
- Hosokawa, M.; Shiba, H.; Kawabe, T.; Nakashima, A.; Yazawa, S. A simple and simultaneous detection method for two different viroids infecting chrysanthemum by multiplex direct RT-PCR. J. Japan. Soc. Hort. Sci. 2007, 76, 60–65. [Google Scholar] [CrossRef]
- Hosokawa, M.; Matsushita, Y.; Uchida, H.; Yazawa, S. Direct RT-PCR method for detecting two chrysanthemum viroids using minimal amounts of plant tissue. J. Virol. Methods 2006, 131, 28–33. [Google Scholar] [CrossRef]
- Song, A.; You, Y.; Chen, F.; Li, P.; Jiang, J.; Chen, S. A multiplex RT-PCR for rapid and simultaneous detection of viruses and viroids in chrysanthemum. Lett. Appl. Microbiol. 2013, 56, 8–13. [Google Scholar] [CrossRef]
- Hollings, M.; Stone, O.M. Attempts to eliminate chrysanthemum stunt from chrysanthemum by meristem-tip culture after heat-treatment. Ann. Appl. Biol. 1970, 65, 311–315. [Google Scholar] [CrossRef]
- Paduch-Cichal, E.; Kryczyński, S. A low temperature therapy and meristem-tip culture for eliminating four viroids from infected plants. J. Phytopathol. 1987, 118, 341–346. [Google Scholar] [CrossRef]
- Chung, B.N.; Huh, E.J.; Kim, J.S. Effect of temperature on the concentration of Chrysanthemum stunt viroid in CSVd-infected chrysanthemum. Plant Pathol. J. 2006, 22, 152–154. [Google Scholar] [CrossRef]
- Hosokawa, M.; Otake, A.; Ohishi, K.; Ueda, E.; Hayashi, T.; Yazawa, S. Elimination of Chrysanthemum stunt viroid from an infected chrysanthemum cultivar by shoot regeneration from a leaf primordium-free shoot apical meristem dome attached to a root tip. Plant Cell Rep. 2004, 22, 859–863. [Google Scholar] [CrossRef]
- Hosokawa, M.; Ueda, E.; Ohishi, K.; Otake, A.; Yazawa, S. Chrysanthemum stunt viroid disturbs the photoperiodic response for flowering of chrysanthemum plants. Planta 2004, 220, 64–70. [Google Scholar] [CrossRef]
- Matsushita, Y.; Aoki, K.; Sumitomo, K. Selection and inheritance of resistance to Chrysanthemum stunt viroid. Crop Prot. 2012, 35, 1–4. [Google Scholar] [CrossRef]
- Nabeshima, T.; Hosokawa, M.; Yano, S.; Ohishi, K.; Doi, M. Screening of chrysanthemum cultivars with resistance to Chrysanthemum stunt viroid. J. Japan. Soc. Hort. Sci. 2012, 81, 285–294. [Google Scholar] [CrossRef]
- Omori, H.; Hosokawa, M.; Shiba, H.; Shitsukawa, N.; Murai, K.; Yazawa, S. Screening of chrysanthemum plants with strong resistance to chrysanthemum stunt viroid. J. Japan. Soc. Hort. Sci. 2009, 78, 350–355. [Google Scholar] [CrossRef]
- Ogawa, T.; Toguri, T.; Kudoh, H.; Okamura, M.; Momma, T.; Yoshioka, M.; Kato, K.; Hagiwara, Y.; Sano, T. Double-stranded RNA-specific ribonuclease confers tolerance against Chrysanthemum stunt viroid and Tomato spotted wilt virus in transgenic chrysanthemum plants. Breeding Sci. 2005, 55, 49–55. [Google Scholar] [CrossRef]
- Toguri, T.; Ogawa, T.; Kakitani, M.; Tukahara, M.; Yoshioka, M. Agrobacterium-mediated transformation of chrysanthemum (Dendranthema grandiflora) plants with a disease resistance gene (pac1). Plant Biotech. 2003, 20, 121–127. [Google Scholar] [CrossRef]
- Niblett, C.L.; Dickson, E.; Fernow, K.H.; Horst, R.K.; Zaitlin, M. Cross protection among four viroids. Virology 1978, 91, 198–203. [Google Scholar] [CrossRef]
- Chung, B.N.; Choi, G.S.; Kim, K.T. Chrysanthemum stunt viroid induces the accumulation of small RNAs associated with RNA silencing in infected chrysanthemum. Plant Pathol. J. 2006, 22, 235–238. [Google Scholar]
- Huh, E.J.; Lee, S.K.; Chung, B.N.; Lee, I.J.; Choi, S.Y. Changes of growth and gibberellin contents in chrysanthemum by infection of Chrysanthemum stunt viroid. Hort. Environ. Biotech. 2006, 47, 366–370. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cho, W.K.; Jo, Y.; Jo, K.-M.; Kim, K.-H. A Current Overview of Two Viroids That Infect Chrysanthemums: Chrysanthemum stunt viroid and Chrysanthemum chlorotic mottle viroid. Viruses 2013, 5, 1099-1113. https://doi.org/10.3390/v5041099
Cho WK, Jo Y, Jo K-M, Kim K-H. A Current Overview of Two Viroids That Infect Chrysanthemums: Chrysanthemum stunt viroid and Chrysanthemum chlorotic mottle viroid. Viruses. 2013; 5(4):1099-1113. https://doi.org/10.3390/v5041099
Chicago/Turabian StyleCho, Won Kyong, Yeonhwa Jo, Kyoung-Min Jo, and Kook-Hyung Kim. 2013. "A Current Overview of Two Viroids That Infect Chrysanthemums: Chrysanthemum stunt viroid and Chrysanthemum chlorotic mottle viroid" Viruses 5, no. 4: 1099-1113. https://doi.org/10.3390/v5041099
APA StyleCho, W. K., Jo, Y., Jo, K.-M., & Kim, K.-H. (2013). A Current Overview of Two Viroids That Infect Chrysanthemums: Chrysanthemum stunt viroid and Chrysanthemum chlorotic mottle viroid. Viruses, 5(4), 1099-1113. https://doi.org/10.3390/v5041099







