Phosphorus Fertilizer Rate, Soil P Availability, and Long-Term Growth Response in a Loblolly Pine Plantation on a Weathered Ultisol
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Location and Establishment
2.2. Vegetation Response to Fertilization
2.3. Soil Sampling and Chemical Analysis
2.4. Hydrology Estimates
2.5. Statistical Analysis
3. Results
3.1. Soil and Stand Characteristics
| Depth | Bulk density (Mg m−3) | Clay | Sand | pH | C | N |
|---|---|---|---|---|---|---|
| (%) | (mg kg−1) | |||||
| 0–10 | 1.27 (0.10) | 9 | 33 | 5.04 | 16.91 | 0.99 |
| 10–20 | 1.46 (0.08) | 11 | 31 | 5.02 | 11.53 | 0.60 |
| 20–30 | 1.54 (0.09) | 17 | 33 | 4.75 | 6.07 | 0.44 |
| 30–50 | 1.53 (0.11) | 22 | 25 | 4.76 | 3.16 | 0.45 |
| 50–75 | 1.61 (0.07) | 25 | 29 | 4.73 | 2.34 | 0.32 |
| 75–100 | 1.70 (0.08) | 21 | 29 | 5.10 | 1.44 | 0.23 |
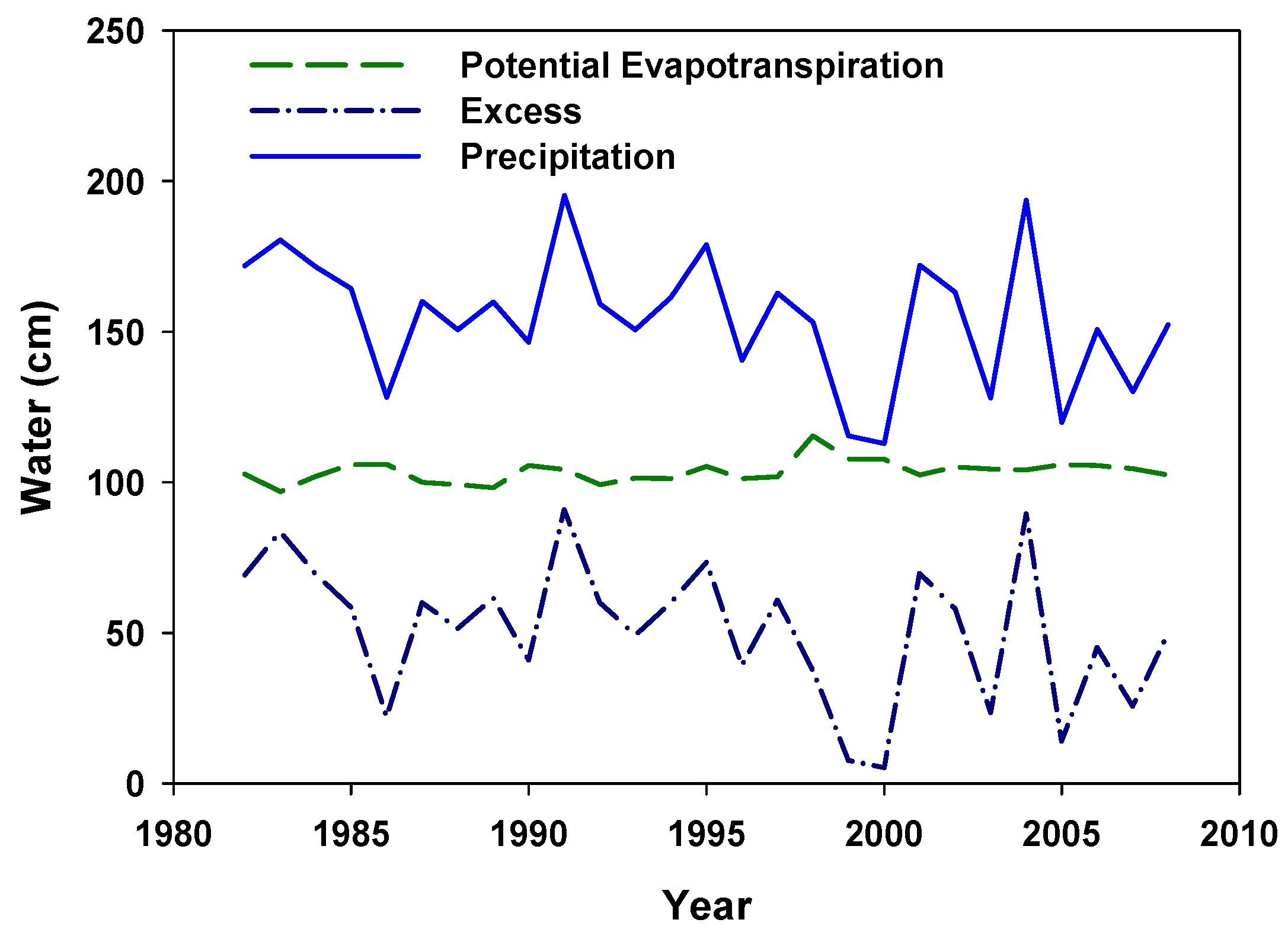
3.2. Vegetative Response to Fertilization
| Effect | Df | Height | Dbh | Density | Volume | Pine biomass | Other vegetation biomass |
|---|---|---|---|---|---|---|---|
| F | 3 | 0.0016 | 0.0120 | 0.1884 | 0.0080 | 0.0074 | 1.76 |
| CS | 2 | 0.0625 | 0.7386 | 0.5707 | 0.0270 | 0.0271 | 0.55 |
| F X CS | 6 | 0.1245 | 0.7137 | 0.6123 | 0.2603 | 0.2516 | 1.69 |
| Treatment | Height (m) | Dbh (cm) | Density (Trees ha−1) | Pine biomass (Mg ha−1) | Understory biomass (Mg ha−1) | Forest floor biomass (Mg ha−1) |
|---|---|---|---|---|---|---|
| Fertilization | ||||||
| 0 | 22.0 b | 20.5 b | 1321 a | 230 b | 3.1 a | 14.4 a |
| 81 | 24.3 a | 22.7 a | 1235 ab | 311 a | 2.5 a | 18.0 a |
| 162 | 25.5 a | 23.7 a | 1160 b | 318 a | 2.2 a | 17.5 a |
| 324 | 25.1 a | 23.1 a | 1259 ab | 329 a | 1.3 a | 18.9 a |
3.3. Phosphorus Availability and Pools
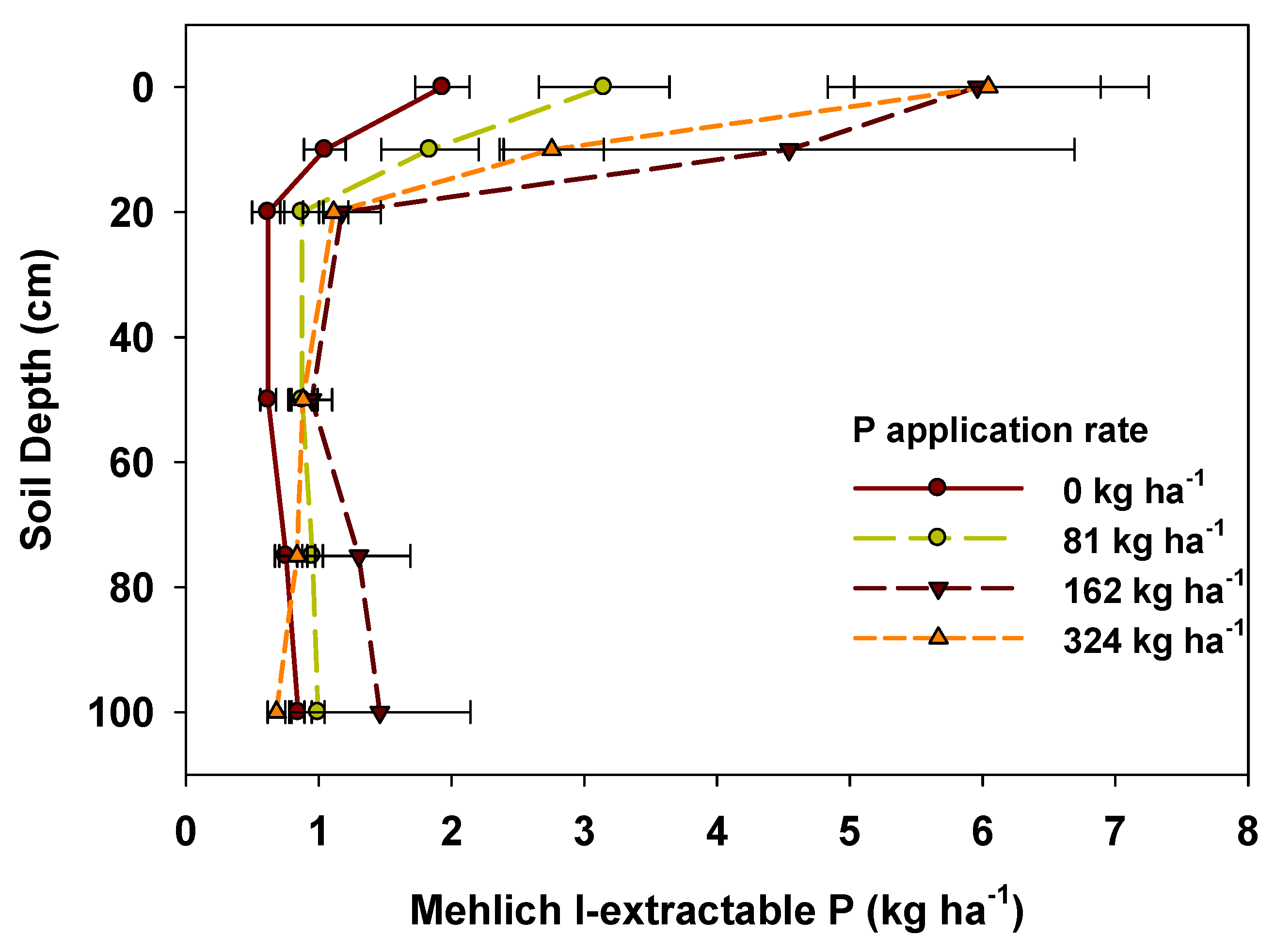

| Total P (kg ha−1) | Fertilizer rate (kg ha−1) | |||
|---|---|---|---|---|
| Soil Pool | 0 | 81 | 162 | 324 |
| Forest floor | 4.4 (0.55) a | 5.5 (0.57) a | 6.9 (0.58) a | 6.5 (0.76) a |
| Surface soil (0–20 cm) | 219.7 (6.0) a | 254.8 (6.5) ab | 292.0 (34.3) b | 312.2 (22.6) b |
| Subsoil (20–100 cm) | 898.4 (26.7) a | 874.2 (32.6) a | 936.8 (97.4) a | 849.7 (38.2) a |
| Total mineral soil (0–100 cm) | 1118.1 (30.6) a | 1129.1 (36.2) a | 1228.8 (96.8) a | 1162.0 (56.6) a |
| Potentially available P (kg ha−1) 1 | ||||
| Surface soil (0–20 cm) | 57.0 (2.56) b | 80.7 (4.20) ab | 126.1 (18.16) a | 118.4 (11.24) a |
| Subsoil (20–100 cm) | 154.6 (7.82) a | 186.0 (9.16) a | 183.1 (22.0) a | 143.0 (12.70) a |
| Total (forest floor + mineral soil) | 215.9 (9.05) a | 272.2 (12.44) a | 316.2 (34.06) a | 272.3 (16.51) a |
4. Discussion
4.1. Vegetative Response
4.2. Fertilizer P Fate
4.3. P Availability
5. Conclusions
Acknowledgments
Conflict of Interest
References
- Cordell, D.; Drangert, J.-O.; White, S. The story of phosphorus: Global food security and food for thought. Glob. Environ. Change 2009, 19, 292–305. [Google Scholar] [CrossRef]
- Jokela, E.J. Nutrient Management of Southern Pines. In Slash Pine: Still Growing and Growing; Dickens, E.D., Barnett, J.P., Hubbard, W.G., Jokela, E.J., Eds.; USDA Forest Service, Southern Research Station: Asheville, NC, USA, 2004; pp. 27–35, General Technical Report SRS-6. [Google Scholar]
- Wells, C.G.; Crutchfield, D.M.; Berenyi, N.M.; Davey, C.B. Soil and Foliar Guidelines for Phosphorus Fertilization of Loblolly pine; Research Paper SE-110; USDA Forest Service, Southeastern Forest Experiment Station: Asheville, NC, USA, 1973. [Google Scholar]
- Allen, H.L.; Dougherty, P.M.; Campbell, R.G. Manipulation of water and nutrients—Practice and opportunity in southern U.S. pine forests. For. Ecol. Manag. 1990, 30, 437–453. [Google Scholar] [CrossRef]
- Gregoire, N.; Fisher, R.F. Nutritional diagnoses in loblolly pine (Pinus taeda L.) established stands using three different approaches. For. Ecol. Manag. 2004, 203, 195–208. [Google Scholar] [CrossRef]
- Albaugh, T.J.; Allen, H.L.; Fox, T.R. Historical patterns of forest fertilization in the southeastern United States from 1969 to 2004. South. J. Appl. For. 2007, 31, 129–137. [Google Scholar]
- Scott, D.A.; Dean, T.J. Energy trade-offs between intensive biomass utilization, site productivity loss, and ameliorative treatments in Loblolly pine plantations. Biomass Bioenerg. 2006, 30, 1001–1010. [Google Scholar]
- Scott, D.A.; Novosad, J.; Goldsmith, G. Ten-Year Results from the North American Long-Term Soil Productivity Study in the Western Gulf Coastal Plain. In Advancing the Fundamental Sciences: Proceedings of the Forest Service National Earth Sciences Conference, San Diego, CA, USA, 18–22 October 2004; Furniss, M., Clifton, C., Ronnenburg, K., Eds.; USDA Forest Service, Pacific Northwest Research Statio: Portland, OR, USA, 2007; pp. 331–340, General Technical Report PNW-689. [Google Scholar]
- Turner, J.; Lambert, M.J.; Humphreys, F.R. Continuing growth response to phosphate fertilizers by a Pinus radiata plantation over fifty years. For. Sci. 2002, 48, 556–568. [Google Scholar]
- Ballard, R.; Fiskell, J.G.A. Phosphorus retention in the coastal plain forest soils: I. Relationship to soil properties. Soil Sci. Soc. Am. Proc. 1974, 38, 250–255. [Google Scholar]
- Ballard, R.; Pritchett, W.L. Phosphorus retention in the coastal plain forest soils: II. Significance to forest fertilization. Soil Sci. Soc. Am. Proc. 1974, 38, 363–366. [Google Scholar]
- Snowdon, P. Modeling Type 1 and Type 2 growth responses in plantations after application of fertilizer or other silvicultural treatments. For. Ecol. Manag. 2002, 163, 229–244. [Google Scholar]
- Comerford, N.B.; McLeod, M.; Skinner, M. Phosphorus form and bioavailability in the pine rotation following fertilization. For. Ecol. Manag. 2002, 169, 203–211. [Google Scholar]
- Gentle, S.W.; Humphreys, F.R.; Lambert, M.J. Continuing response on Pinus radiata to phosphatic fertilizers over two rotations. For. Sci. 1986, 32, 822–829. [Google Scholar]
- Everett, C.J.; Palm-Leis, H. Availability of residual phosphorus fertilizer for Loblolly pine. For. Ecol. Manag. 2009, 258, 2207–2213. [Google Scholar] [CrossRef]
- Gentle, W.; Humphreys, F.R.; Lambert, M.J. An examination of a Pinus radiata phosphate fertilizer trial fifteen years after treatment. For. Sci. 1965, 11, 315–324. [Google Scholar]
- Fransson, A.-M.; Bergvist, B. Phosphorus fertilisation causes durable enhancement of phosphorus concentrations in forest soil. For. Ecol. Manag. 2000, 130, 69–76. [Google Scholar]
- Torbert, J.L.; Burger, J.A. Long-term availability of applied phosphorus to Loblolly pine on a Piedmont soil. Soil Sci. Soc. Am. J. 1984, 48, 1174–1178. [Google Scholar] [CrossRef]
- Pritchett, W.L.; Comerford, N.B. Long-term response to phosphorus fertilization on selected southeastern coastal plain soils. Soil Sci. Soc. Am. J. 1982, 46, 640–644. [Google Scholar] [CrossRef]
- Tiarks, A.E. Phosphorus sorption curves for evaluating phosphorus requirements of Loblolly pine (Pinus taeda). Commun. Soil Sci. Plan. 1982, 13, 619–631. [Google Scholar] [CrossRef]
- Sheoran, V.; Sheoran, A.S.; Poonia, P. Soil reclamation of abandoned mine land by revegetation: A review. Int. J. Soil Sediment Water. 2010, 3, p. Article 13. Available online: http://scholarworks.umass.edu/intljssw/vol3/iss2/13/ (accessed on 29 August 2012).
- Gray, C.A.; Schwab, A.P. Phosphorus-fixing ability of high pH, high calcium, coal-combustion, waste materials. Water Air Soil Poll. 1993, 69, 309–320. [Google Scholar] [CrossRef]
- Tiarks, A.E. Phosphorus Requirements for Establishment of Dual-Cropped Loblolly pine. In Proceedings of the Third Biennial Southern Silvicultural Research Conference, Atlanta, GA, USA, 7–8 November 1984; houlders, E., Ed.; USDA Forest Service, Southern Forest Experiment Station: New Orleans, LA, USA, 1985; pp. 390–394, General Technical Report SO-54. [Google Scholar]
- Tiarks, A.E. Biomass production of Loblolly pine seeded between rows of planted Loblolly pine. Biomass Bioenerg. 1993, 4, 61–67. [Google Scholar] [CrossRef]
- Kerr, A., Jr.; Griffis, B.J.; Powell, J.W.; Edwards, J.P.; Venson, R.L.; Long, J.K.; Kilpatrick, W.W. Soil Survey of Rapides Parish, Louisiana; USDA Soil Conservation Service: Washington, DC, USA, 1980. [Google Scholar]
- Scott, D.A.; Tiarks, A.E. Dual-cropping Loblolly pine for biomass energy and conventional wood products. South. J. Appl. For. 2008, 32, 33–37. [Google Scholar]
- Baldwin, V.C., Jr.; Feduccia, D.P. Loblolly pine Growth and Yield Prediction for Managed West Gulf Plantations; Research Paper SO-236; USDA Forest Service, Southern Forest Experiment Station: New Orleans, LA, USA, 1987. [Google Scholar]
- Scott, D.A.; Stagg, R.H.; Smith, M.A., Jr. A Non-Destructive Method for Quantifying Small-Diameter Woody Biomass in Southern Pine Forests. In Proceedings of the 13th Biennial Southern Silvicultural Research Conference, Memphis, TN, USA, 28 February–3 March 2005; Connor, K.F., Ed.; USDA Forest Service, Southern Research Station: Asheville, NC, USA, 2006; p. 358, General Technical Report SRS-92. [Google Scholar]
- Murphy, J.; Riley, J.P. A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta. 1962, 27, 31–36. [Google Scholar]
- Gee, G.W.; Bauder, J.W. Particle-Size Analysis. In Methods of Soil Analysis, 2nd; Klute, A., Ed.; American Society of Agronomy and Soil Science Society of America: Madison, WI, USA, 1986; pp. 383–411, Agronomy Monograph 9, Part 1. [Google Scholar]
- Mehlich, A. Determination of P, Ca, Mg, K, Na and NH4; North Carolina Soil Testing Division Mimeo: Raleigh, NC, USA, 1953. [Google Scholar]
- Tiessen, H.; Moir, J.O. Characterization of Available P by Sequential Extraction. In Soil Sampling and Methods of Analysis, 2nd; Carter, M.R., Gregorich, E.G., Eds.; CRC Press: Boca Raton, FL, USA, 2007; pp. 293–306. [Google Scholar]
- Fox, T.R.; Comerford, N.B.; McFee, W.W. Phosphorus and aluminum release from a spodic horizon mediated by organic acids. Soil Sci. Soc. Am. J. 1990, 54, 1763–1767. [Google Scholar] [CrossRef]
- Thornthwaite, C.W. An approach toward a rational classification of climate. Geogr. Rev. 1948, 38, 55–94. [Google Scholar] [CrossRef]
- Conner, W.H.; Toliver, J.R.; Sklar, F.H. Natural regeneration of baldcypress (Taxodium distichum (L.) Rich.) in a Louisiana swamp. For. Ecol. Manag. 1986, 14, 305–317. [Google Scholar] [CrossRef]
- National Climatic Data Center. Available online: http://www.ncdc.noaa.gov/oa/ncdc.html (accessed on 20 August 2012).
- SAS Institute, SAS/STAT Users Guide; SAS Institute, Inc.: Cary, NC, USA, 2004; pp. 2659–2851.
- Fox, T.R.; Allen, H.L.; Albaugh, T.J.; Rubilar, R.; Carlson, C.A. Tree nutrition and forest fertilization of pine plantations in the southern United States. South. J. Appl. For. 2007, 31, 5–11. [Google Scholar]
- Hynynen, J.; Burkhart, H.E.; Allen, H.L. Modeling tree growth in fertilized midrotation Loblolly pine plantations. For. Ecol. Manag. 1998, 107, 213–229. [Google Scholar] [CrossRef]
- Pote, D.H.; Daniel, T.C.; Sharpley, A.N.; Moore, P.A., Jr.; Edwards, D.R.; Nichols, D.J. Relating extractable soil phosphorus to phosphorus losses in runoff. Soil Sci. Soc. Am. J. 1996, 60, 855–859. [Google Scholar] [CrossRef]
- Shepherd, M.A.; Withers, P.J. Phosphorus leaching from liquid digested sewage sludge applied to sandy soils. J. Agr. Sci. 2001, 136, 433–441. [Google Scholar]
- Turtola, E.; Yli-Halla, M. Fate of phosphorus applied in slurry and mineral fertilizer: Accumulation in soil and release into surface runoff water. Nutr. Cycl. Agroecosys. 1999, 55, 165–174. [Google Scholar] [CrossRef]
- Shoulders, E.; Tiarks, A.E. Fertilizer fate in a 13-year-old slash pine plantation. Soil Sci. Soc. Am. J. 1980, 44, 1085–1089. [Google Scholar] [CrossRef]
- Djodjic, F.; Börling, K.; Bergström, L. Phosphorus leaching in relation to soil type and soil phosphorus content. J. Environ. Qual. 2004, 33, 678–684. [Google Scholar] [CrossRef]
- Fraser, A.I.; Harrod, T.R.; Haygarth, P.M. The effect of rainfall intensity on soil erosion and particulate phosphorus transfer from arable soils. Water Sci. Technol. 1999, 39, 41–45. [Google Scholar]
- Nash, D.L.; Clemow, L.; Hannah, M.; Barlow, K.; Gangaiya, P. Modelling phosphorus exports from rain-fed and irrigated pastures in southern Australia. Aust. J. Soil Res. 2005, 43, 745–755. [Google Scholar] [CrossRef]
- Preedy, N.; McTiernan, K.; Matthews, R.; Heathwaite, L.; Haygarth, P. Rapid incidental phosphorus transfers from grassland. J. Environ. Qual. 2001, 30, 2105–2112. [Google Scholar] [CrossRef]
- Haygarth, P.M.; Jarvis, S.C. Transfer of phosphorus from agricultural soils. Adv. Agron. 1999, 66, 195–249. [Google Scholar] [CrossRef]
- Withers, P.J.A.; Ulen, B.; Stamm, C.; Bechmann, M. Incidental phosphorus losses—Are they significant and can they be predicted? J. Plant Nutr. Soil Sci. 2003, 166, 459–468. [Google Scholar] [CrossRef]
- Hart, M.R.; Quin, B.F.; Nguyen, M.L. Phosphorus runoff from agricultural land and direct fertilizer effects: A review. J. Environ. Qual. 2004, 33, 1954–1972. [Google Scholar] [CrossRef]
- Sharpley, A.N.; McDowell, R.W.; Kleinman, P.J.A. Phosphorus loss from land to water: Integrating agricultural and environmental management. Plant Soil 2004, 237, 287–307. [Google Scholar]
- Kleinman, P.J.A.; Sharpley, A.N.; Moyer, B.G.; Elwinger, G.F. Effect of mineral and manure phosphorus sources on runoff phosphorus. J. Environ. Qual. 2002, 31, 2026–2033. [Google Scholar]
- Fox, T.R.; Comerford, N.B. Rhizosphere phosphatase activity and phosphatase hydrolyzable organic phosphorus in two forested spodosols. Soil Biol. Biochem. 1992, 24, 579–583. [Google Scholar] [CrossRef]
- Richter, D.D.; Allen, H.L.; Li, J.; Markewitz, D.; Raikes, J. Bioavailability of slowly cycling soil phosphorus: Major restructuring of soil P fractions over four decades in an aggrading forest. Oecologia 2006, 150, 259–271. [Google Scholar] [CrossRef]
- Polglase, P.J.; Jokela, E.J.; Comerford, N.B. Nitrogen and phosphorus release from decomposing needles of southern pine plantations. Soil Sci. Soc. Am. J. 1992, 56, 914–920. [Google Scholar] [CrossRef]
- Beardsley, T.M. Peak phosphorus. BioScience 2011, 61, 91. [Google Scholar]
- McDowell, R.W.; Monaghan, R.M.; Carey, P.L. Potential phosphorus losses in overland flow from pastoral soils receiving long-term applications of either superphosphate or reactive phosphate rock. New Zeal. J. Agr. Res. 2003, 46, 329–337. [Google Scholar] [CrossRef]
- Haywood, J.D.; Tiarks, A.E.; Snow, G.A. Combinations of fungicide and cultural practices influence the incidence and impact of fusiform rust in slash pine plantations. South. J. Appl. For. 1994, 18, 53–59. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Scott, D.A.; Bliss, C.M. Phosphorus Fertilizer Rate, Soil P Availability, and Long-Term Growth Response in a Loblolly Pine Plantation on a Weathered Ultisol. Forests 2012, 3, 1071-1085. https://doi.org/10.3390/f3041071
Scott DA, Bliss CM. Phosphorus Fertilizer Rate, Soil P Availability, and Long-Term Growth Response in a Loblolly Pine Plantation on a Weathered Ultisol. Forests. 2012; 3(4):1071-1085. https://doi.org/10.3390/f3041071
Chicago/Turabian StyleScott, D. Andrew, and Christine M. Bliss. 2012. "Phosphorus Fertilizer Rate, Soil P Availability, and Long-Term Growth Response in a Loblolly Pine Plantation on a Weathered Ultisol" Forests 3, no. 4: 1071-1085. https://doi.org/10.3390/f3041071
APA StyleScott, D. A., & Bliss, C. M. (2012). Phosphorus Fertilizer Rate, Soil P Availability, and Long-Term Growth Response in a Loblolly Pine Plantation on a Weathered Ultisol. Forests, 3(4), 1071-1085. https://doi.org/10.3390/f3041071




