Methacrylate Polymer Monoliths for Separation Applications
Abstract
:1. Introduction
2. Methacrylate Polymer Monoliths
2.1. Morphology of Monoliths
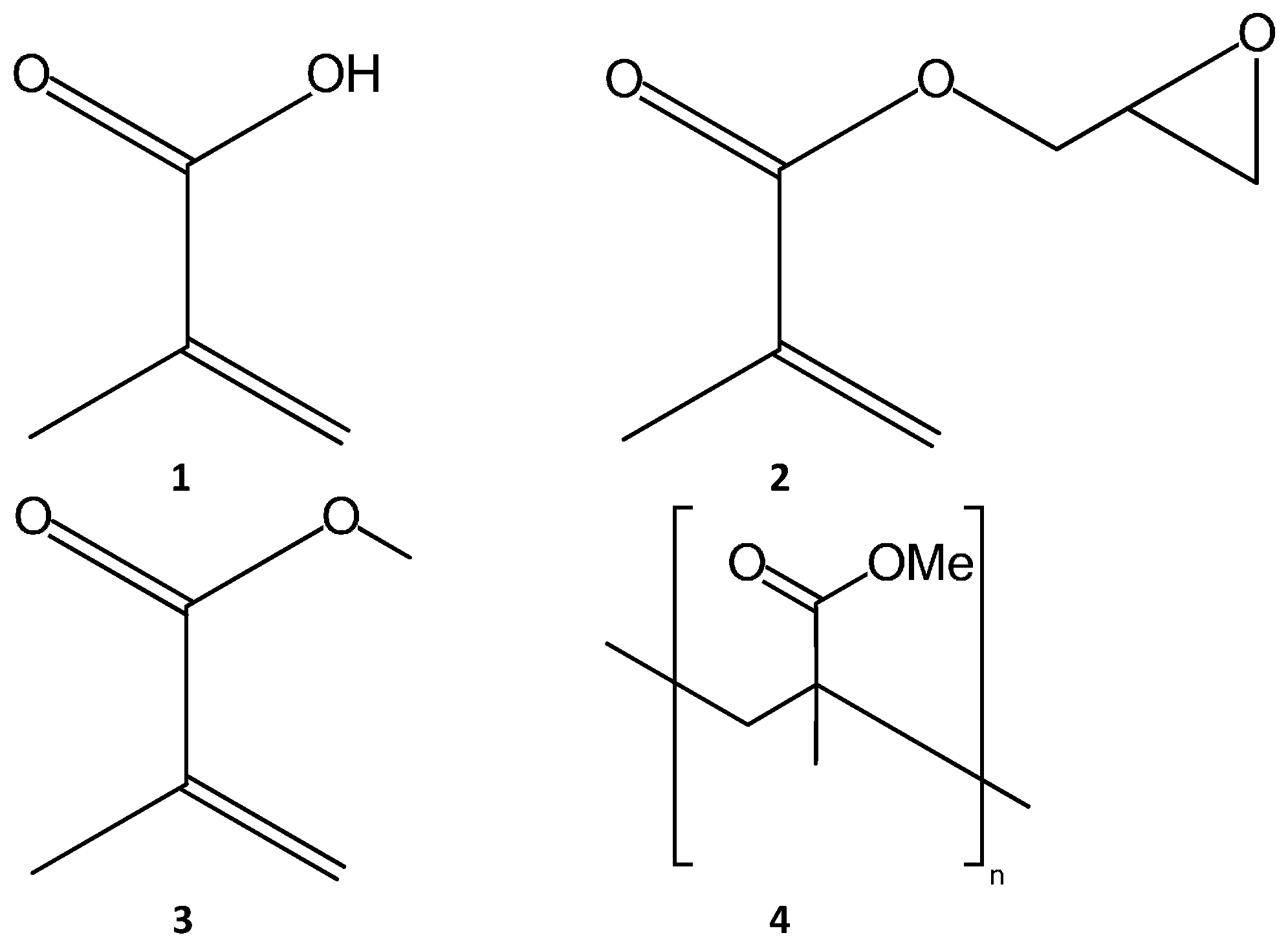
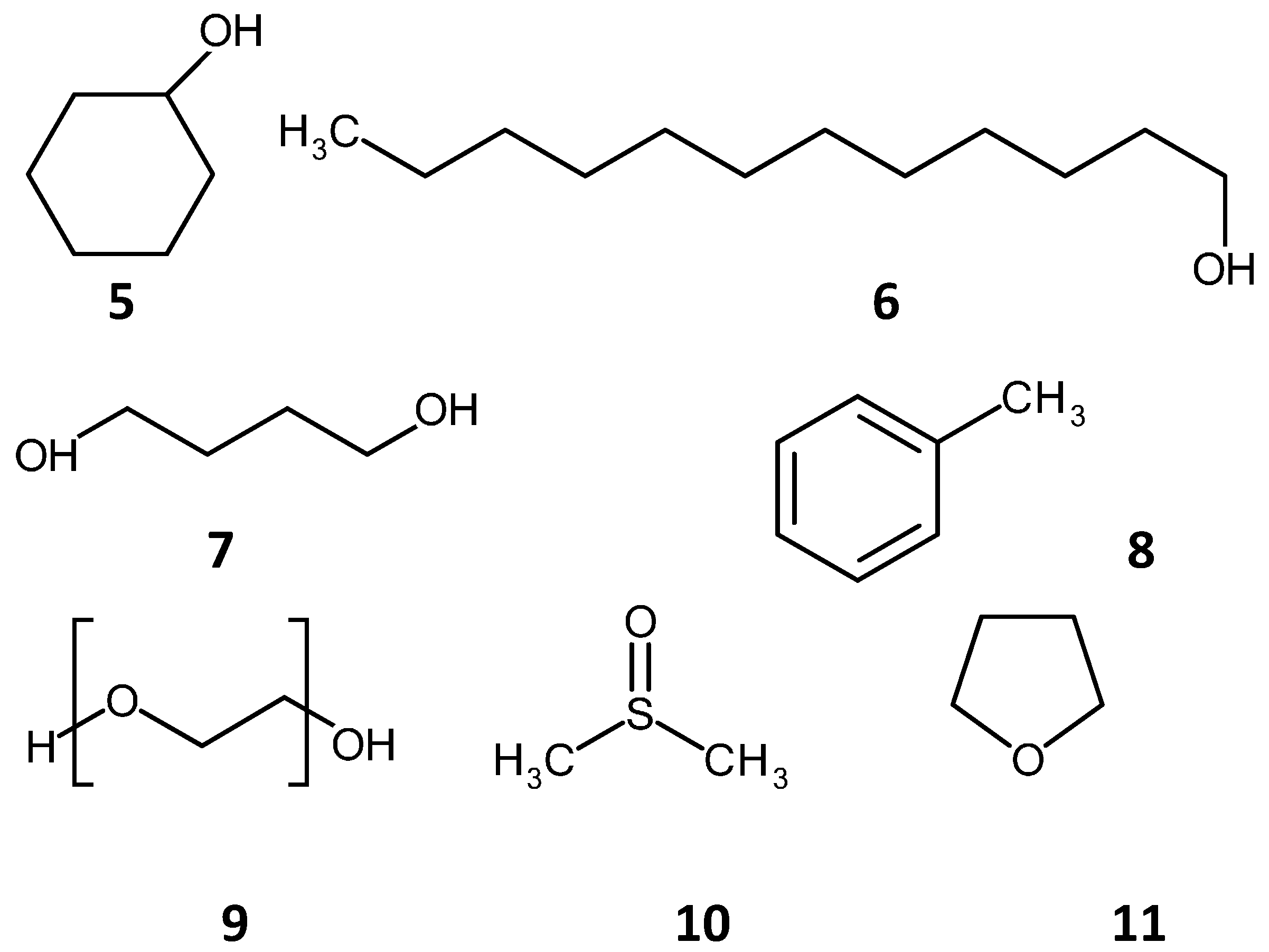
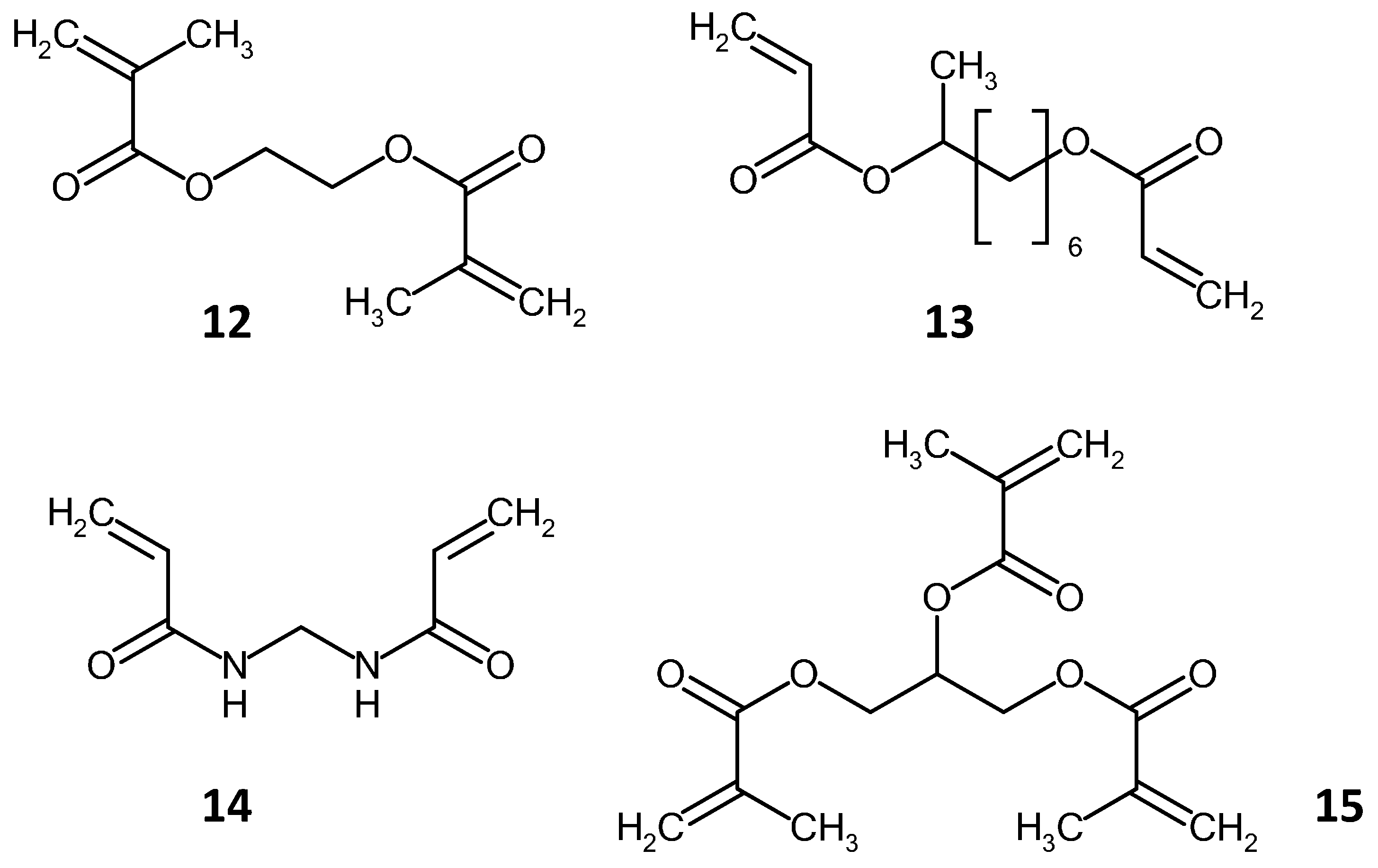
2.2. Methacrylate Monolith Preparation Methods and Compounds
2.3. Structural Properties
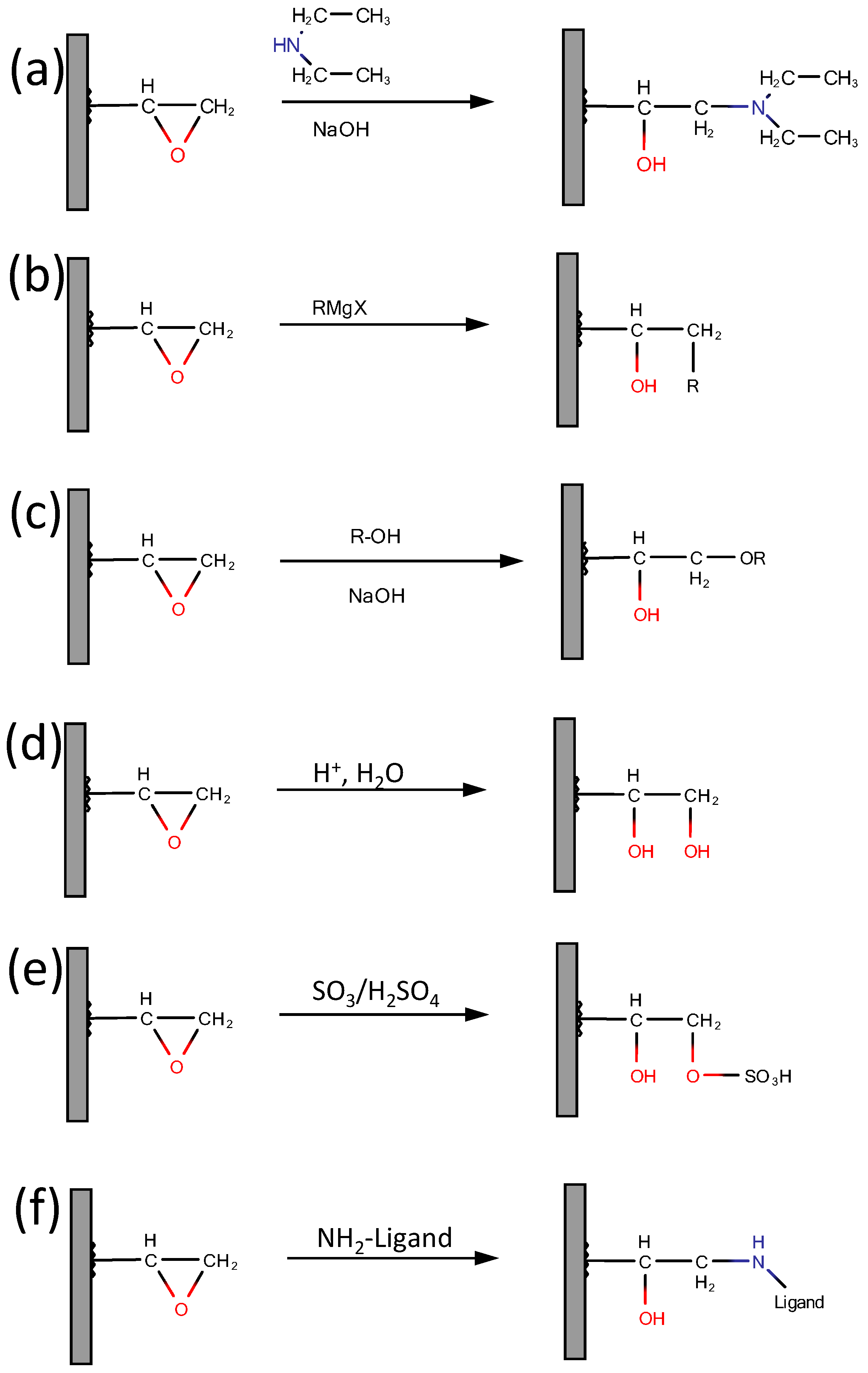
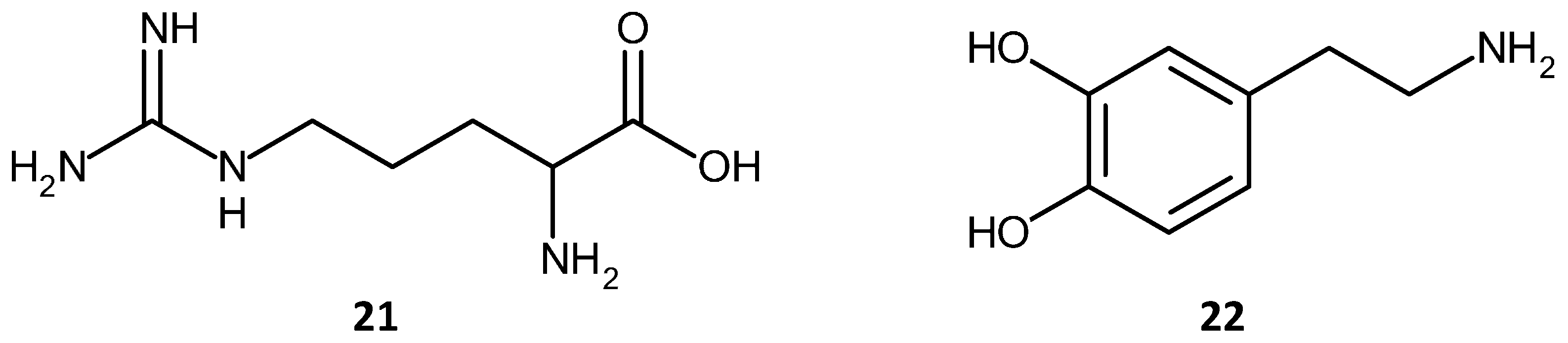
2.4. Functionalization Routes and Functional Groups
2.4.1. Epoxy Group Transformations
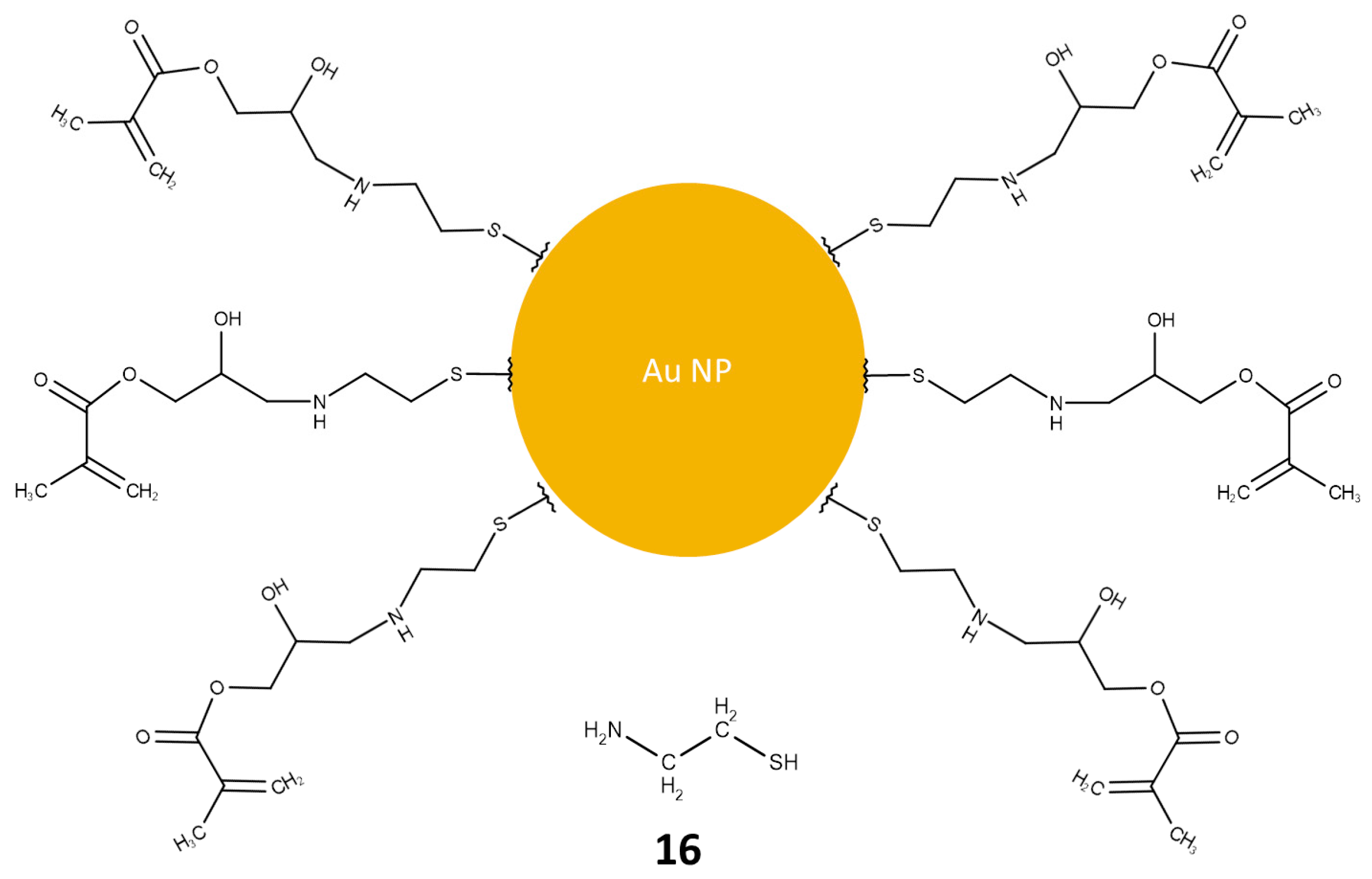
2.4.2. Nanoparticle Functionalization
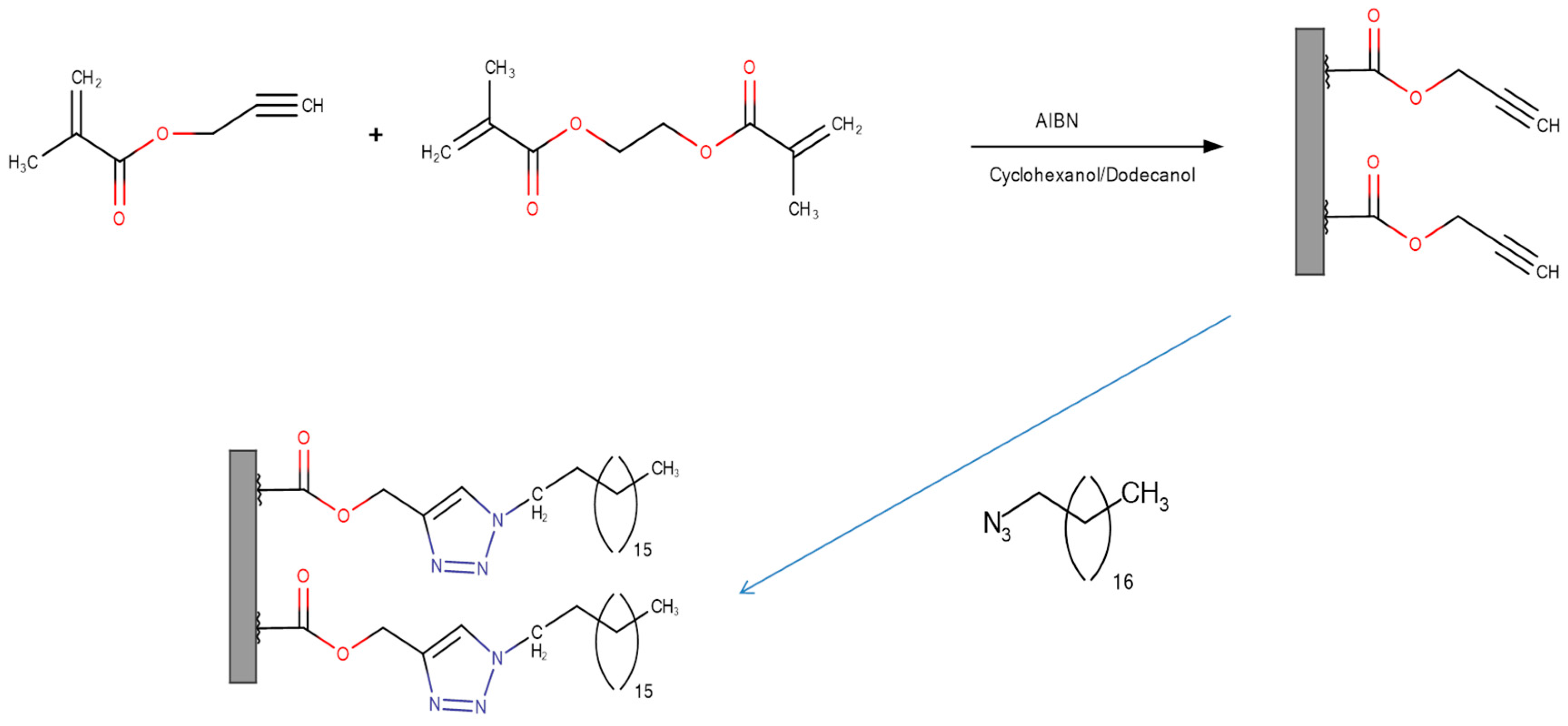
2.4.3. Click Chemistry
3. Applications of Methacrylate Polymer Monoliths in Separation Science
3.1. Separation and Pre-Concentration of Chemical Species
3.1.1. Solid Phase Extraction (SPE) and Pre-Concentration of Chemical Species
3.1.2. Chemical Separations
3.2. Biological Separations and DNA Purification
3.2.1. Amplification and Pre-Concentration of DNA, Plasmids, Viruses and Peptides
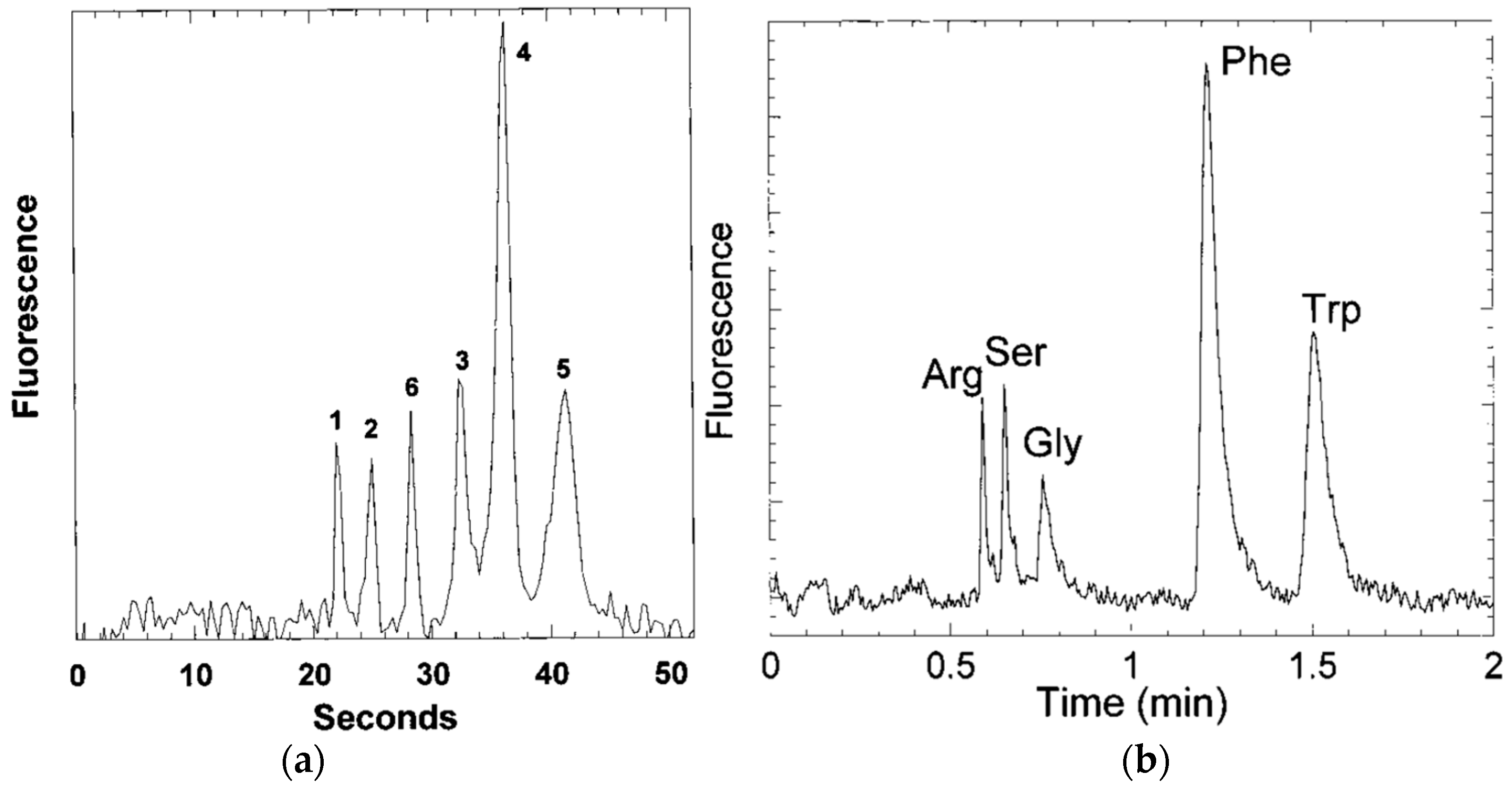
3.2.2. Chromatographic Separation of Proteins, Peptides and Amino Acids
3.3. Challenges for the Development of Methacrylate Monoliths
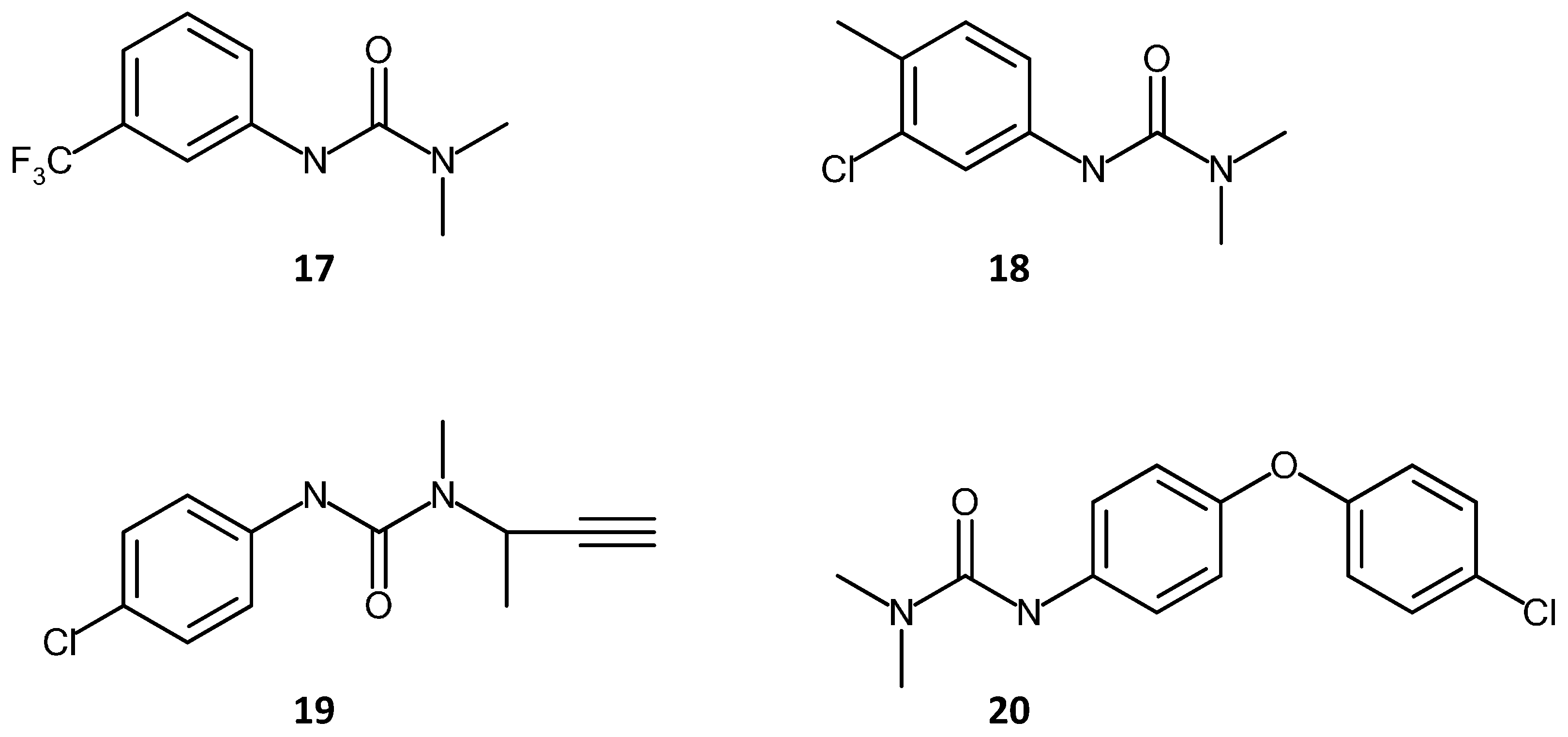
3.3.1. Poor Small Analyte Separation Efficiencies
3.3.2. Chemical Compositional Variations
3.3.3. Physical Architecture Variations
4. Conclusions and Outlook
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ABS | Acrylonitrile Butadiene Styrene |
| AHA | 6-Azido Hexanoic Acid |
| AIBN | Azo bis-isobutyronitrile |
| APTES | (3-Aminopropyl)triethoxysilane |
| ARTP | Atom Transfer Radical Polymerization |
| BET | Brunauer-Emmett-Teller |
| BSA | Bovine Serum Albumin |
| BuMA | Butyl Methacrylate |
| CA | Carbonic Anhydrase |
| CAD | Computer Aided Design |
| CD | Cyclodextrin |
| CEC | Capillary Electro Chromatography |
| CIM | Convective Interactive Media |
| COC | Cyclic Olefin Co-polymer |
| conA | conAlbumin |
| CuAAC | Cu(I) catalyzed 1,3 dipolar Azide-Alkyne cycloaddition |
| CVD | Chemical Vapour Deposition |
| DEAE | di-Ethyl Amino-Ethyl |
| DNA | Deoxyribonucleic acid |
| EDMA | Ethylene DiMethacrylate |
| EDX | Energy Dispersive X-ray Spectroscopy |
| FIA | Flow Injection Analysis |
| FIB | Focussed Ion Beam |
| FITC | Fluorescein Iso-Thiocyanate |
| GC | Gas Chromatography |
| GMA | Glycidyl Methacrylate |
| GMP | Good Manufacturing Practice |
| GO | Graphene Oxide |
| HEPES | 4-(2-Hydroxyethyl)piperazine-1-ethanesulfonic acid |
| HIC | Hydrophobic Interaction Chromatography |
| HILIC | Hydrophilic Interaction Liquid Chromatography |
| HIPE | High Internal Phase Emulsions |
| HPAA | Polyacrylic acid |
| HPLC | High Performance Liquid Chromatography |
| HRP | Horseradish Peroxidase |
| HSA | Human Serum Albumin |
| INS | Insulin |
| LSCM | Laser Scanning Confocal Microscopy |
| MALDI-TOF | Matrix Assisted Laser Desorption/Ionization—Time Of Flight |
| MB | Myoglobin |
| MIM | Molecular Imprinted Monolith |
| MMA | Methyl Methacrylate |
| OVA | Ovalbumin |
| PAH | Polycyclic Aromatic Hydrocarbons |
| PCR | Polymerase Chain Reaction |
| PDMS | Poly (di-Methyl Siloxane) |
| PEEK | Poly Ether Ether Ketone |
| PEG | Poly(Ethylene Glycol) |
| PEGDA | Poly Ethylene Glycol di-Acrylate |
| PLOT | Porous Layer Open Tubular Column |
| PMMA | Poly Methyl Methacrylate |
| PP2 | Phosphatase B, |
| PTFE | Poly Tetra Fluoro-Ethylene |
| PVA | Poly Vinyl Alcohol |
| RNA | Ribonucleic acid |
| RT-qPCR | Real Time quantitative Polymerase Chain Reaction |
| RP-LC | Reverse Phase Liquid Chromatography |
| SEM | Scanning Electron Microscopy |
| SPE | Solid Phase Extraction |
| TLC | Thin Layer Chromatography |
| UTLC | Ultra Thin Layer Chromatography |
References
- Rouquerol, J.; Avnir, D.; Fairbridge, C.W.; Everett, D.H.; Haynes, J.H.; Pernicone, N.; Ramsay, J.D.F.; Sing, K.S.W.; Unger, K.K. Recommendations for the characterization of porous solids. Pure Appl. Chem. 1994, 66, 1739–1758. [Google Scholar] [CrossRef]
- Synge, R.L.M.; Mould, D.L. Electrokinetic Ultrafiltration Analysis of Polysaccharides. Analyst 1952, 77, 964–969. [Google Scholar]
- Svec, F.; Fréchet, J.M.J. Continuous rods of macroporous polymer as high-performance liquid chromatography separation media. Anal. Chem. 1992, 64, 820–822. [Google Scholar] [CrossRef]
- Minakuchi, H.; Nakanishi, K.; Soga, N.; Ishizuka, N.; Tanaka, N. Octadecylsilylated porous silica rods as separation media for reversed-phase liquid chromatography. Anal. Chem. 1996, 68, 3498–3501. [Google Scholar] [CrossRef] [PubMed]
- Svec, F. Less common applications of monoliths: Preconcentration and solid-phase extraction. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2006, 841, 52–64. [Google Scholar] [CrossRef] [PubMed]
- Svec, F.; Kurganov, A.A. Less common applications of monoliths. III. Gas chromatography. J. Chromatogr. A 2008, 1184, 281–295. [Google Scholar] [CrossRef] [PubMed]
- Svec, F.; Lv, Y. Advances and Recent Trends in the Field of Monolithic Columns for Chromatography. Anal. Chem. 2015, 87, 250–273. [Google Scholar] [CrossRef] [PubMed]
- Svec, F. Porous polymer monoliths: Amazingly wide variety of techniques enabling their preparation. J. Chromatogr. A 2010, 1217, 902–924. [Google Scholar] [CrossRef] [PubMed]
- Vlakh, E.G.; Tennikova, T.B. Preparation of methacrylate monoliths. J. Sep. Sci. 2007, 30, 2801–2813. [Google Scholar] [CrossRef] [PubMed]
- Walsh, Z.; Levkin, P.A.; Abele, S.; Scarmagnani, S.; Heger, D.; Klán, P.; Diamond, D.; Paull, B.; Svec, F.; Macka, M. Polymerisation and surface modification of methacrylate monoliths in polyimide channels and polyimide coated capillaries using 660 nm light emitting diodes. J. Chromatogr. A 2011, 1218, 2954–2962. [Google Scholar] [CrossRef] [PubMed]
- Nischang, I. Porous polymer monoliths: Morphology, porous properties, polymer nanoscale gel structure and their impact on chromatographic performance. J. Chromatogr. A 2013, 1287, 39–58. [Google Scholar] [CrossRef] [PubMed]
- Urban, J.; Jandera, P. Recent advances in the design of organic polymer monoliths for reversed-phase and hydrophilic interaction chromatography separations of small molecules. Anal. Bioanal. Chem. 2013, 405, 2123–2131. [Google Scholar] [CrossRef] [PubMed]
- Barut, M.; Podgornik, A.; Urbas, L.; Gabor, B.; Brne, P.; Vidič, J.; Plevčak, S.; Štrancar, A. Methacrylate-based short monolithic columns: Enabling tools for rapid and efficient analyses of biomolecules and nanoparticles. J. Sep. Sci. 2008, 31, 1867–1880. [Google Scholar] [CrossRef] [PubMed]
- Ongkudon, C.M.; Kansil, T.; Wong, C. Challenges and strategies in the preparation of large-volume polymer-based monolithic chromatography adsorbents. J. Sep. Sci. 2014, 37, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Vlakh, E.G.; Tennikova, T.B. Applications of polymethacrylate-based monoliths in high-performance liquid chromatography. J. Chromatogr. A 2009, 1216, 2637–2650. [Google Scholar] [CrossRef] [PubMed]
- Podgornik, A.; Yamamoto, S.; Peterka, M.; Krajnc, N.L. Fast separation of large biomolecules using short monolithic columns. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2013, 927, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Švec, F.; Tennikova, T.B.; Deyl, Z. Monolithic Materials: Preparation, Properties, and Applications; Elsevier Ltd.: Amsterdam, The Netherlands, 2003. [Google Scholar]
- Nischang, I.; Teasdale, I.; Brüggemann, O. Porous polymer monoliths for small molecule separations: Advancements and limitations. Anal. Bioanal. Chem. 2011, 400, 2289–2304. [Google Scholar] [CrossRef] [PubMed]
- Svec, F. Quest for organic polymer-based monolithic columns affording enhanced efficiency in high performance liquid chromatography separations of small molecules in isocratic mode. J. Chromatogr. A 2012, 1228, 250–262. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.; Myers, P.; Zhang, H. Preparation of aligned porous silica monolithic capillary columns and their evaluation for HPLC. Anal. Methods 2012, 3942–3947. [Google Scholar] [CrossRef]
- Zheng, H.; Liu, Q.; Jia, Q. Preparation of poly(butyl methacrylate-co-ethyleneglyceldimethacrylate) monolithic column modified with B-cyclodextrin and nano-cuprous oxide and its application in polymer monolithic microextraction of polychlorinated biphenyls. J. Chromatogr. A 2014, 1343, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Mayadunne, E.; El Rassi, Z. Facile preparation of octadecyl monoliths with incorporated carbon nanotubes and neutral monoliths with coated carbon nanotubes stationary phases for HPLC of small and large molecules by hydrophobic and π-π interactions. Talanta 2014, 129, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Yan, C.; Bai, L.; Li, J.; Duan, Y. A simple and efficient method for preparation of a strong cation exchange monolith and its application to separation of HSA. Anal. Methods 2012, 4, 1098–1104. [Google Scholar] [CrossRef]
- Namera, A.; Miyazaki, S.; Saito, T.; Nakamoto, A. Monolithic silica with HPLC separation and solid phase extraction materials for determination of drugs in biological materials. Anal. Methods 2011, 3, 2189–2200. [Google Scholar] [CrossRef]
- Cao, Q.; Xu, Y.; Liu, F.; Svec, F.; Fréchet, J.M.J. Polymer monoliths with exchangeable chemistries: Use of gold nanoparticles as intermediate ligands for capillary columns with varying surface functionalities. Anal. Chem. 2010, 82, 7416–7421. [Google Scholar] [CrossRef] [PubMed]
- Shkolnikov, V.; Santiago, J.G. Coupling Isotachophoresis with Affinity Chromatography for Rapid and Selective Puri fi cation with High Column Utilization, Part 2: Experimental Study. Anal. Chem. 2014, 86, 6229–6236. [Google Scholar] [CrossRef] [PubMed]
- Nesterenko, E.; Yavorska, O.; Macka, M.; Yavorskyy, A.; Paull, B. Monolithic porous layer open tubular (monoPLOT) columns for low pressure liquid chromatography of proteins. Anal. Methods 2011, 3, 537–543. [Google Scholar] [CrossRef]
- Sabarudin, A.; Huang, J.; Shu, S.; Sakagawa, S.; Umemura, T. Preparation of methacrylate-based anion-exchange monolithic microbore column for chromatographic separation of DNA fragments and oligonucleotides. Anal. Chim. Acta 2012, 736, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Mallik, R.; Hage, D.S. Affinity monoliths for ultrafast immunoextraction. Anal. Chem. 2005, 77, 2362–2372. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Li, X.F.; Le, X.C. Aptamer-modified monolithic capillary chromatography for protein separation and detection. Anal. Chem. 2008, 80, 3915–3920. [Google Scholar] [CrossRef] [PubMed]
- Carrasco-Correa, E.J.; Ramis-Ramos, G.; Herrero-Martínez, J.M. Hybrid methacrylate monolithic columns containing magnetic nanoparticles for capillary electrochromatography. J. Chromatogr. A 2015, 1385, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Moyna, Á.; Connolly, D.; Nesterenko, E.; Nesterenko, P.N.; Paull, B. Iminodiacetic acid functionalised organopolymer monoliths: Application to the separation of metal cations by capillary high-performance chelation ion chromatography. Anal. Bioanal. Chem. 2013, 405, 2207–2217. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.; Wang, J.; Zhang, H.; Liu, S.; Qin, J.; Liu, H. Ionic liquid as porogen in the preparation of a polymer-based monolith for the separation of protein by high performance liquid chromatography. Anal. Methods 2015, 7, 607–613. [Google Scholar] [CrossRef]
- Bandari, R.; Elsner, C.; Knolle, W.; Kühnel, C.; Decker, U.; Buchmeiser, M.R. Separation behavior of electron-beam curing derived, acrylate-based monoliths. J. Sep. Sci. 2007, 30, 2821–2827. [Google Scholar] [CrossRef] [PubMed]
- Eeltink, S.; Herrero-Martinez, J.M.; Rozing, G.P.; Schoenmakers, P.J.; Kok, W.T. Tailoring the morphology of methacrylate ester-based monoliths for optimum efficiency in liquid chromatography. Anal. Chem. 2005, 77, 7342–7347. [Google Scholar] [CrossRef] [PubMed]
- Bakry, R.; Bonn, G.K.; Mair, D.; Svec, F. Monolithic porous polymer layer for the separation of peptides and proteins using thin-layer chromatography coupled with MALDI-TOF-MS. Anal. Chem. 2007, 79, 486–493. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Chen, C.F.; Tsao, C.W.; Chang, C.C.; Chu, C.C.; DeVoe, D.L. Polymer microchips integrating solid-phase extraction and high-performance liquid chromatography using reversed-phase polymethacrylate monoliths. Anal. Chem. 2009, 81, 2545–2554. [Google Scholar] [CrossRef] [PubMed]
- Eeltink, S.; Geiser, L.; Svec, F.; Fréchet, J.M.J. Optimization of the porous structure and polarity of polymethacrylate-based monolithc capillary columns for the LC-MS separation of enzymatic digests. J. Sep. Sci. 2007, 30, 2814–2820. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Wei, F.; Zhang, Y.F.; Nie, J.; Feng, Y.Q. Novel polymer monolith microextraction using a poly(methacrylic acid-ethylene glycol dimethacrylate) monolith and its application to simultaneous analysis of several angiotensin II receptor antagonists in human urine by capillary zone electrophoresis. J. Chromatogr. A 2006, 1102, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Fan, Y.; Li, G. Preparation and evaluation of a porous monolithic capillary column for microextraction of estrogens from urine and milk samples online coupled to high-performance liquid chromatography. J. Chromatogr. A 2012, 1228, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Zhai, H.; Li, J.; Chen, Z.; Su, Z.; Liu, Z.; Yu, X. A glass/PDMS electrophoresis microchip embedded with molecular imprinting SPE monolith for contactless conductivity detection. Microchem. J. 2014, 114, 223–228. [Google Scholar] [CrossRef]
- Krenkova, J.; Lacher, N.A.; Svec, F. Control of selectivity via nanochemistry: Monolithic capillary column containing hydroxyapatite nanoparticles for separation of proteins and enrichment of phosphopeptides. Anal. Chem. 2010, 82, 8335–8341. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Tolley, H.D.; Lee, M.L. Poly [hydroxyethyl acrylate-co-poly (ethylene glycol) diacrylate] Monolithic Column for Efficient Hydrophobic Interaction Chromatography of Proteins. Anal. Chem. 2009, 9416–9424. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Gu, B.; Dennis Tolley, H.; Lee, M.L. Preparation of polymeric monoliths by copolymerization of acrylate monomers with amine functionalities for anion-exchange capillary liquid chromatography of proteins. J. Chromatogr. A 2009, 1216, 5525–5532. [Google Scholar] [CrossRef] [PubMed]
- Holdšvendová, P.; Suchánková, J.; Bunček, M.; Bačkovská, V.; Coufal, P. Hydroxymethyl methacrylate-based monolithic columns designed for separation of oligonucleotides in hydrophilic-interaction capillary liquid chromatography. J. Biochem. Biophys. Methods 2007, 70, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Maksimova, E.F.; Vlakh, E.G.; Tennikova, T.B. Methacrylate-based monolithic layers for planar chromatography of polymers. J. Chromatogr. A 2011, 1218, 2425–2431. [Google Scholar] [CrossRef] [PubMed]
- Maksimova, E.; Vlakh, E.; Sinitsyna, E.; Tennikova, T. HPLC analysis of synthetic polymers on short monolithic columns. J. Sep. Sci. 2013, 36, 3741–3749. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Ou, J.; Wei, Y.; Wang, H.; Liu, Z.; Chen, L.; Zou, H. A novel polymeric monolith prepared with multi-acrylate crosslinker for retention-independent efficient separation of small molecules in capillary liquid chromatography. Anal. Chim. Acta 2015, 883, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Carrasco-Correa, E.J.; Ramis-Ramos, G.; Herrero-Martínez, J.M. Methacrylate monolithic columns functionalized with epinephrine for capillary electrochromatography applications. J. Chromatogr. A 2013, 1298, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.-L.; Wu, Y.-R.; Lin, T.-Y.; Fuh, M.-R. Preparation and evaluation of poly(alkyl methacrylate-co-methacrylic acid-co-ethylene dimethacrylate) monolithic columns for separating polar small molecules by capillary liquid chromatography. Anal. Chim. Acta 2015, 871, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Courtois, J.; Szumski, M.; Georgsson, F.; Irgum, K. Assessing the macroporous structure of monolithic columns by transmission electron microscopy. Anal. Chem. 2007, 79, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Dario Arrua, R.; Hilder, E.F. Highly ordered monolithic structures by directional freezing and UV-initiated cryopolymerisation. Evaluation as stationary phases in high performance liquid chromatography. RSC Adv. 2015, 5, 71131–71138. [Google Scholar] [CrossRef]
- Krajnc, P.; Leber, N.; Štefanec, D.; Kontrec, S.; Podgornik, A. Preparation and characterisation of poly(high internal phase emulsion) methacrylate monoliths and their application as separation media. J. Chromatogr. A 2005, 1065, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Su, C.-K.; Peng, P.-J.; Sun, Y.-C. Fully 3D-Printed Preconcentrator for Selective Extraction of Trace Elements in Seawater. Anal. Chem. 2015, 87, 6945–6950. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Pagaduan, J.V.; Yu, M.; Woolley, A.T. On chip preconcentration and fluorescence labeling of model proteins by use of monolithic columns: Device fabrication, optimization, and automation. Anal. Bioanal. Chem. 2015, 407, 737–747. [Google Scholar] [CrossRef] [PubMed]
- Nge, P.N.; Rogers, C.I.; Woolley, A.T. Advances in Microfluidic Materials, Functions, Integration, and Applications. Chem. Rev. 2013, 2550–2583. [Google Scholar] [CrossRef] [PubMed]
- Janasek, D.; Franzke, J.; Manz, A. Scaling and the design of miniaturized chemical-analysis systems. Nature 2006, 442, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.; Kwon, D.; Choi, W.; Jung, G.Y.; Jeon, S. 3D-Printed Microfluidic Device for the Detection of Pathogenic Bacteria Using Size-based Separation in Helical Channel with Trapezoid Cross-Section. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Araya-Farias, M.; Taverna, M.; Woytasik, M.; Bayle, F.; Guerrouache, M.; Ayed, I.; Cao, H.H.; Carbonnier, B.; Tran, N.T.T. A new strategy for simultaneous synthesis and efficient anchorage of polymer monoliths in native PDMS microchips. Polym. Guildf. 2015, 66, 249–258. [Google Scholar] [CrossRef]
- Thurmann, S.; Mauritz, L.; Heck, C.; Belder, D. High-performance liquid chromatography on glass chips using precisely defined porous polymer monoliths as particle retaining elements. J. Chromatogr. A 2014, 1370, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.H.; Yang, W.C.; Pan, T.; Woolley, A.T. Affinity monolith integrated poly(methyl methacrylate) microchips for on-line protein extraction and capillary electrophoresis. Anal. Chem. 2008, 80, 5126–5130. [Google Scholar] [CrossRef] [PubMed]
- Nazario, C.E.D.; Silva, M.R.; Franco, M.S.; Lanças, F.M. Evolution in miniaturized column liquid chromatography instrumentation and applications: An overview. J. Chromatogr. A 2015, 1421, 18–37. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.G.; Park, K.J.; Seok, S.; Shin, S.; Kim, D.H.; Park, J.Y.; Heo, Y.S.; Lee, S.J.; Lee, T.J. 3D printed modules for integrated microfluidic devices. RSC Adv. 2014, 4, 32876–32880. [Google Scholar] [CrossRef]
- Fee, C.; Nawada, S.; Dimartino, S. 3D printed porous media columns with fine control of column packing morphology. J. Chromatogr. A 2014, 1333, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Alzahrani, E.; Welham, K. Fabrication of an octadecylated silica monolith inside a glass microchip for protein enrichment. Analyst 2012, 137, 4751–4759. [Google Scholar] [CrossRef] [PubMed]
- Waldbaur, A.; Rapp, H.; Länge, K.; Rapp, B.E. Let there be chip—Towards rapid prototyping of microfluidic devices: One-step manufacturing processes. Anal. Methods 2011, 3, 2681–2716. [Google Scholar] [CrossRef]
- Kitson, P.J.; Rosnes, M.H.; Sans, V.; Dragone, V.; Cronin, L. Configurable 3D-Printed millifluidic and microfluidic “lab on a chip” reactionware devices. Lab Chip 2012, 12, 3267–3271. [Google Scholar] [CrossRef] [PubMed]
- Ladner, Y.; Bruchet, A.; Crétier, G.; Dugas, V.; Randon, J.; Faure, K. New “one-step” method for the simultaneous synthesis and anchoring of organic monolith inside COC microchip channels. Lab Chip 2012, 12, 1680–1685. [Google Scholar] [CrossRef] [PubMed]
- Nischang, I.; Svec, F.; Frechet, J.M.J. Downscaling limits and confinement effects in the miniaturization of porous polymer monoliths in narrow bore capillaries. Anal. Chem. 2009, 81, 7390–7396. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Zeng, Y.; Sun, X.; Harrison, D.J. Confinement effects on the morphology of photopatterned porous polymer monoliths for capillary and microchip electrophoresis of proteins. Electrophoresis 2008, 29, 2980–2986. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Xu, S.; Li, J. Recent advances in molecular imprinting technology: Current status, challenges and highlighted applications. Chem. Soc. Rev. 2011, 40, 2922–2942. [Google Scholar] [CrossRef] [PubMed]
- Derazshamshir, A.; Yılmaz, F.; Denizli, A. Molecularly imprinted hydrophobic polymers as a tool for separation in capillary electrochromatography. Anal. Methods 2015, 7, 2659–2669. [Google Scholar] [CrossRef]
- Zheng, Y.; Wang, X.; Ji, Y. Monoliths with proteins as chiral selectors for enantiomer separation. Talanta 2012, 91, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Svec, F.; Frechet, J.M.J. Kinetic Control of Pore Formation in Macroporous Polymers. Formation of “Molded” Porous Materials with High Flow Characteristics for Separations or Catalysis. Chem. Mater. 1995, 7, 707–715. [Google Scholar] [CrossRef]
- Viklund, C.; Ponten, E.; Glad, B.; Irgum, K.; Horstedt, P.; Svec, F. Molded macroporous poly(glycidyl methacrylate-co-trimethylolpropane trimethacrylate) materials with fine controlled porous properties: Preparation of monoliths using photoinitiated polymerization. Chem. Mater. 1997, 9, 463–471. [Google Scholar] [CrossRef]
- Walsh, Z.; Abele, S.; Lawless, B.; Heger, D.; Klán, P.; Breadmore, M.C.; Paull, B.; Macka, M. Photoinitiated polymerisation of monolithic stationary phases in polyimide coated capillaries using visible region LEDs. Chem. Commun. Camb. 2008, 7345, 6504–6506. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Q.; Adrus, N.; Tomicki, F.; Ulbricht, M. Composites of functional polymeric hydrogels and porous membranes. J. Mater. Chem. 2011, 21, 2783–2811. [Google Scholar] [CrossRef]
- Aggarwal, P.; Asthana, V.; Lawson, J.S.; Tolley, H.D.; Wheeler, D.R.; Mazzeo, B.A.; Lee, M.L. Correlation of chromatographic performance with morphological features of organic polymer monoliths. J. Chromatogr. A 2014, 1334, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Bai, L.; Lei, H.; Zhang, X.; Li, S.; Yang, G. Preparation of a novel skeleton weak anion-exchange monolith by single electron transfer-living radical polymerization for protein separation in high performance liquid chromatography. Anal. Methods 2013, 5, 939–945. [Google Scholar] [CrossRef]
- Vázquez, M.; Moore, D.; He, X.; Ben Azouz, A.; Nesterenko, E.; Nesterenko, P.; Paull, B.; Brabazon, D. Focussed ion beam serial sectioning and imaging of monolithic materials for 3D reconstruction and morphological parameter evaluation. Analyst 2014, 139, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Arrua, R.D.; Hitchcock, A.P.; Hon, W.B.; West, M.; Hilder, E.F. Characterization of polymer monoliths containing embedded nanoparticles by scanning transmission X-ray microscopy (STXM). Anal. Chem. 2014, 86, 2876–2881. [Google Scholar] [CrossRef] [PubMed]
- Müllner, T.; Zankel, A.; Mayrhofer, C.; Höltzel, A.; Lv, Y.; Svec, F.; Tallarek, U. Reconstruction and Characterization of Polymeric Monolithic Stationary Phases using Serial Block Face Scanning Electron Microscopy. Langmuir 2012, 28, 16733–16737. [Google Scholar] [CrossRef] [PubMed]
- Laher, M.; Causon, T.J.; Buchberger, W.; Hild, S.; Nischang, I. Assessing the nanoscale structure and mechanical properties of polymer monoliths used for chromatography. Anal. Chem. 2013, 85, 5645–5649. [Google Scholar] [CrossRef] [PubMed]
- Connolly, D.; Twamley, B.; Paull, B. High-capacity gold nanoparticle functionalised polymer monoliths. Chem. Commun. Camb. 2010, 46, 2109–2111. [Google Scholar] [CrossRef] [PubMed]
- Lv, Y.; Lin, Z.; Svec, F. “Thiol–ene” click chemistry: A facile and versatile route for the functionalization of porous polymer monoliths. Analyst 2012, 137, 4114–4118. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, J.P.; Hilder, E.F.; Shellie, R.; Smith, J.; Haddad, P.R. Towards high capacity latex-coated porous polymer monoliths as ion-exchange stationary phases. Analyst 2006, 131, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Ueki, Y.; Umemura, T.; Li, J.; Odake, T.; Tsunoda, K.I. Preparation and application of methacrylate-based cation-exchange monolithic columns for capillary ion chromatography. Anal. Chem. 2005, 76, 7007–7012. [Google Scholar] [CrossRef] [PubMed]
- Hjerten, S.; Nakazato, K.; Mohammad, J.; Eaker, D. Reversed-Phase Chromatography of Proteins and Peptides on Compressed Continuous Beds. Chromatographia 1993, 37, 287–294. [Google Scholar] [CrossRef]
- Sedlacek, O.; Kucka, J.; Svec, F.; Hruby, M. Silver-coated monolithic columns for separation in radiopharmaceutical applications. J. Sep. Sci. 2014, 37, 798–802. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Cao, Q.; Svec, F.; Fréchet, J.M.J. Porous polymer monolithic column with surface-bound gold nanoparticles for the capture and separation of cysteine-containing peptides. Anal. Chem. 2010, 82, 3352–3358. [Google Scholar] [CrossRef] [PubMed]
- Lv, Y.; Alejandro, F.M.; Fréchet, J.M.J.; Svec, F. Preparation of porous polymer monoliths featuring enhanced surface coverage with gold nanoparticles. J. Chromatogr. A 2012, 1261, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Alwael, H.; Connolly, D.; Clarke, P.; Thompson, R.; Twamley, B.; O’Connor, B.; Paull, B. Pipette-tip selective extraction of glycoproteins with lectin modified gold nano-particles on a polymer monolithic phase. Analyst 2011, 136, 2619–2628. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, M.; Tarawally, M.; Liu, X.; Liu, X.; Guo, L.; Yang, L.; Wang, G. Application of cyclodextrin-modified gold nanoparticles in enantioselective monolith capillary electrochromatography. Talanta 2013, 109, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Krenkova, J.; Foret, F. Nanoparticle-modified monolithic pipette tips for phosphopeptide enrichment. Anal. Bioanal. Chem. 2013, 405, 2175–2183. [Google Scholar] [CrossRef] [PubMed]
- Krenkova, J.; Foret, F. Iron oxide nanoparticle coating of organic polymer-based monolithic columns for phosphopeptide enrichment. J. Sep. Sci. 2011, 34, 2106–2112. [Google Scholar] [CrossRef] [PubMed]
- Pensa, E.; Cortés, E.; Corthey, G.; Carro, P.; Vericat, C.; Fonticelli, M.H.; Benítez, G.; Rubert, A.A.; Salvarezza, R.C. The chemistry of the sulfur-gold interface: In search of a unified model. Acc. Chem. Res. 2012, 45, 1183–1192. [Google Scholar] [CrossRef] [PubMed]
- Raubenheimer, H.G.; Schmidbaur, H. The Late Start and Amazing Upswing in Gold Chemistry. J. Chem. Educ. 2014, 91, 2024–2036. [Google Scholar] [CrossRef]
- Navarro-Pascual-Ahuir, M.; Lerma-García, M.J.; Ramis-Ramos, G.; Simó-Alfonso, E.F.; Herrero-Martínez, J.M. Preparation and evaluation of lauryl methacrylate monoliths with embedded silver nanoparticles for capillary electrochromatography. Electrophoresis 2013, 34, 925–934. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; White, I.; DeVoe, D.L. Nanoparticle-functionalized porous polymer monolith detection elements for surface-enhanced raman scattering. Anal. Chem. 2011, 83, 2119–2124. [Google Scholar] [CrossRef] [PubMed]
- Terborg, L.; Masini, J.C.; Lin, M.; Lipponen, K.; Riekolla, M.-L.; Svec, F. Porous polymer monolithic columns with gold nanoparticles as an intermediate ligand for the separation of proteins in reverse phase-ion exchange mixed mode. J. Adv. Res. 2014, 6, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Kolb, H.C.; Finn, M.G.; Sharpless, K.B. Click Chemistry: Diverse Chemical Function from a Few Good Reactions. Angew. Chem. Int. Ed. 2001, 40, 2004–2021. [Google Scholar] [CrossRef]
- Sun, X.; He, X.; Chen, L.; Zhang, Y. In-column “click” preparation of hydrophobic organic monolithic stationary phases for protein separation. Anal. Bioanal. Chem. 2011, 399, 3407–3413. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Yu, R.; Hu, W.; Zheng, J.; Tong, P.; Zhao, H.; Cai, Z. Preparation of a poly(3′-azido-3′-deoxythymidine-co-propargyl methacrylate-co-pentaerythritol triacrylate) monolithic column by in situ polymerization and a click reaction for capillary liquid chromatography of small molecules and proteins. Analyst 2015, 140, 4626–4635. [Google Scholar] [CrossRef] [PubMed]
- Le Droumaguet, B.; Nicolas, J. Recent advances in the design of bioconjugates from controlled/living radical polymerization. Polym. Chem. 2010, 1, 563–598. [Google Scholar] [CrossRef]
- Qin, A.; Lam, J.W.Y.; Tang, B.Z. Click Polymerization: Progresses, Challenges, and Opportunities. Macromolecules 2010, 43, 8693–8702. [Google Scholar] [CrossRef]
- Tang, W.; Becker, M.L. “Click” reactions: A versatile toolbox for the synthesis of peptide-conjugates. Chem. Soc. Rev. 2014, 43, 7013–7039. [Google Scholar] [CrossRef] [PubMed]
- Lowe, A.B. Thiol–ene “click” reactions and recent applications in polymer and materials synthesis: A first update. Polym. Chem. 2014, 5, 4820–4870. [Google Scholar] [CrossRef]
- Nair, D.P.; Podgórski, M.; Chatani, S.; Gong, T.; Xi, W.; Fenoli, C.R.; Bowman, C.N. The Thiol-Michael addition click reaction: A powerful and widely used tool in materials chemistry. Chem. Mater. 2014, 26, 724–744. [Google Scholar] [CrossRef]
- Espeel, P.; Du Prez, F.E. “Click”-Inspired Chemistry in Macromolecular Science: Matching Recent Progress and User Expectations. Macromolecules 2015, 48, 2–14. [Google Scholar] [CrossRef]
- Chu, C.; Liu, R. Application of click chemistry on preparation of separation materials for liquid chromatography. Chem. Soc. Rev. 2011, 40, 2177–2188. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Lin, D.; He, X.; Chen, L.; Zhang, Y. A facile and efficient strategy for one-step in situ preparation of hydrophobic organic monolithic stationary phases by click chemistry and its application on protein separation. Talanta 2010, 82, 404–408. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Zhang, Q.; Peng, Y.; Liu, Z.; Rao, L.; He, T.; Crommen, J.; Sun, P.; Jiang, Z. A facile and efficient one-step strategy for the preparation of β-cyclodextrin monoliths. J. Sep. Sci. 2013, 36, 2441–2449. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Zhang, Q.; Yao, Z.; Zhao, X.; Ran, D.; Crommen, J.; Jiang, Z. One-step strategy for the synthesis of a derivatized cyclodextrin-based monolithic column. J. Sep. Sci. 2014, 37, 1720–1727. [Google Scholar] [CrossRef] [PubMed]
- Rogeberg, M.; Malerod, H.; Roberg-Larsen, H.; Aass, C.; Wilson, S.R. On-line solid phase extraction-liquid chromatography, with emphasis on modern bioanalysis and miniaturized systems. J. Pharm. Biomed. Anal. 2014, 87, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Duffy, E.; He, X.; Nesterenko, P.N.; Paull, B. Hierarchical porous graphitic carbon monoliths with detonation nanodiamonds: Synthesis, characterisation and adsorptive properties. J. Mater. Sci. 2015, 50, 6245–6259. [Google Scholar] [CrossRef]
- Shi, Z.-G.; Chen, F.; Xing, J.; Feng, Y.-Q. Carbon monolith: Preparation, characterization and application as microextraction fiber. J. Chromatogr. A 2009, 1216, 5333–5339. [Google Scholar] [CrossRef] [PubMed]
- Goswami, S.; Bajwa, N.; Asuri, P.; Ci, L.; Ajayan, P.M.; Cramer, S.M. Aligned Carbon Nanotube Stationary Phases for Electrochromatographic Chip Separations. Chromatographia 2009, 69, 473–480. [Google Scholar] [CrossRef]
- He, X.; Nesterenko, E.P.; Nesterenko, P.N.; Brabazon, D.; Zhou, L.; Glennon, J.D.; Luong, J.H.T.; Paull, B. Fabrication and Characterization of Nanotemplated Carbon Monolithic Material. ACS Appl. Mater. Interfaces 2013, 5, 8572–8580. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Male, K.B.; Nesterenko, P.N.; Brabazon, D.; Paull, B.; Luong, J.H.T. Adsorption and desorption of methylene blue on porous carbon monoliths and nanocrystalline cellulose. ACS Appl. Mater. Interfaces 2013, 5, 8796–8804. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Wang, Y.; Feng, Y.Q. Extraction of clenbuterol from urine using hydroxylated poly(gycidyl methacrylate-co-ethylene dimethacrylate) monolith microextraction followed by high-performance liquid chromatography determination. J. Sep. Sci. 2007, 30, 2874–2880. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.-L.; Lirio, S.; Yang, Y.; Wu, L.-T.; Hsiao, S.-Y.; Huang, H.-Y. A poly(alkyl methacrylate-divinylbenzene-vinylbenzyl trimethylammonium chloride) monolithic column for solid-phase microextraction. J. Chromatogr. A 2015, 1395, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Augustin, V.; Proczek, G.; Dugay, J.; Descroix, S.; Hennion, M.C. Online preconcentration using monoliths in electrochromatography capillary format and microchips. J. Sep. Sci. 2007, 30, 2858–2865. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xie, X.; Lee, M.L.; Chen, J. Preparation and evaluation of hydrophilic C18 monolithic sorbents for enhanced polar compound retention in liquid chromatography and solid phase extraction. J. Chromatogr. A 2011, 1218, 8608–8616. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.-L.; Wu, Y.-R.; Fuh, M.-R. Polymer monolith microextraction using poly(butyl methacrylate-co-1,6-hexanediol ethoxylate diacrylate) monolithic sorbent for determination of phenylurea herbicides in water samples. Talanta 2016, 147, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhang, H.; Lv, Y.; Svec, F.; Tan, T. Polymer monoliths with chelating functionalities for solid phase extraction of metal ions from water. J. Chromatogr. A 2014, 1343, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Thabano, J.R.E.; Breadmore, M.C.; Hutchinson, J.P.; Johns, C.; Haddad, P.R. Silica nanoparticle-templated methacrylic acid monoliths for in-line solid-phase extraction-capillary electrophoresis of basic analytes. J. Chromatogr. A 2009, 1216, 4933–4940. [Google Scholar] [CrossRef] [PubMed]
- Nge, P.N.; Pagaduan, J.V.; Yu, M.; Woolley, A.T. Microfluidic chips with reversed-phase monoliths for solid phase extraction and on-chip labeling. J. Chromatogr. A 2012, 1261, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Nesterenko, E.P.; Nesterenko, P.N.; Connolly, D.; Lacroix, F.; Paull, B. Micro-bore titanium housed polymer monoliths for reversed-phase liquid chromatography of small molecules. J. Chromatogr. A 2010, 1217, 2138–2146. [Google Scholar] [CrossRef] [PubMed]
- Ghanem, A.; Ahmed, M.; Ishii, H.; Ikegami, T. Immobilized β-cyclodextrin-based silica vs polymer monoliths for chiral nano liquid chromatographic separation of racemates. Talanta 2015, 132, 301–314. [Google Scholar] [CrossRef] [PubMed]
- Yu, R.; Hu, W.; Lin, G.; Xiao, Q.; Zheng, J.; Lin, Z. One-pot synthesis of polymer monolithic column by combination of free radical polymerization and azide–alkyne cycloaddition “click” reaction and its application in capillary liquid chromatography. RSC Adv. 2015, 5, 9828–9836. [Google Scholar] [CrossRef]
- Bai, X.; Liu, H.; Wei, D.; Yang, G. Preparation of a novel porous poly (trimethylol propane triacrylate-co-ethylene dimethacrylate) monolithic column for highly efficient HPLC separations of small molecules. Talanta 2014, 119, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Smith, N.W.; Ferguson, P.D.; Taylor, M.T. Mixed-mode reversed-phase and ion-exchange monolithic columns for micro-HPLC. J. Sep. Sci. 2008, 31, 2774–2783. [Google Scholar] [CrossRef] [PubMed]
- Kip, Ç.; Erkakan, D.; Gökaltun, A.; Çelebi, B.; Tuncel, A. Synthesis of a reactive polymethacrylate capillary monolith and its use as a starting material for the preparation of a stationary phase for hydrophilic interaction chromatography. J. Chromatogr. A 2015, 1396, 86–97. [Google Scholar] [CrossRef] [PubMed]
- Alshitari, W.; Quigley, C.L.; Smith, N. Fabrication and evaluation of an organic monolithic column based upon the polymerisation of hexyl methacrylate with 1,6-hexanediol ethoxylate diacrylate for the separation of small molecules by capillary liquid chromatography. Talanta 2015, 141, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Gu, C.; Lin, L.; Chen, X.; Jia, J.; Wu, D.; Fang, N. Analysis of microcystins by capillary high performance liquid chromatography using a polymethacrylate-based monolithic column. J. Sep. Sci. 2007, 30, 2866–2873. [Google Scholar] [CrossRef] [PubMed]
- Gu, B.; Li, Y.; Lee, M.L. Polymer monoliths with low hydrophobicity for strong cation-exchange capillary liquid chromatography of peptides and proteins. Anal. Chem. 2007, 79, 5848–5855. [Google Scholar] [CrossRef] [PubMed]
- Andac, M.; Galaev, I.Y.; Denizli, A. Dye attached poly(hydroxyethyl methacrylate) cryogel for albumin depletion from human serum. J. Sep. Sci. 2012, 35, 1173–1182. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Tolley, H.D.; Lee, M.L. Monolithic capillary columns synthesized from a single phosphate-containing dimethacrylate monomer for cation-exchange chromatography of peptides and proteins. J. Chromatogr. A 2011, 1218, 4322–4331. [Google Scholar] [CrossRef] [PubMed]
- WHO Antimicrobial Resistance Fact Sheet. Available online: http://www.who.int/mediacentre/factsheets/fs194/en/ (accessed on 1 June 2016).
- Barut, M.; Podgornik, A.; Brne, P.; Strancar, A. Convective interaction media short monolithic columns: Enabling chromatographic supports for the separation and purification of large biomolecules. J. Sep. Sci. 2005, 28, 1876–1892. [Google Scholar] [CrossRef] [PubMed]
- Benčina, M.; Podgornik, A.; Štrancar, A. Characterization of methacrylate monoliths for purification of DNA molecules. J. Sep. Sci. 2004, 27, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Smrekar, F.; Podgornik, A.; Ciringer, M.; Kontrec, S.; Raspor, P.; Štrancar, A.; Peterka, M. Preparation of pharmaceutical-grade plasmid DNA using methacrylate monolithic columns. Vaccine 2010, 28, 2039–2045. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.J.; Tan, L.; Jeong, M.H.; Kim, J.-H.; Choe, W.-S. Monolith-based immobilized metal affinity chromatography increases production efficiency for plasmid DNA purification. J. Chromatogr. A 2011, 1218, 5273–5278. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, S.; Černigoj, U.; Lendero Krajnc, N.; Štrancar, A. Chromatographic purification of plasmid DNA on hydrophobic methacrylate monolithic supports. Sep. Purif. Technol. 2015, 147, 139–146. [Google Scholar] [CrossRef]
- Smrekar, F.; Ciringer, M.; Strancar, A.; Podgornik, A. Characterisation of methacrylate monoliths for bacteriophage purification. J. Chromatogr. A 2011, 1218, 2438–2444. [Google Scholar] [CrossRef] [PubMed]
- Urthaler, J.; Schlegl, R.; Podgornik, A.; Strancar, A.; Jungbauer, A.; Necina, R. Application of monoliths for plasmid DNA purification: Development and transfer to production. J. Chromatogr. A 2005, 1065, 93–106. [Google Scholar] [CrossRef] [PubMed]
- Jungbauer, A.; Hahn, R. Polymethacrylate monoliths for preparative and industrial separation of biomolecular assemblies. J. Chromatogr. A 2008, 1184, 62–79. [Google Scholar] [CrossRef] [PubMed]
- Podgornik, A.; Krajnc, N.L. Application of monoliths for bioparticle isolation. J. Sep. Sci. 2012, 35, 3059–3072. [Google Scholar] [CrossRef] [PubMed]
- Smrekar, V.; Smrekar, F.; Štrancar, A.; Podgornik, A. Single step plasmid DNA purification using methacrylate monolith bearing combination of ion-exchange and hydrophobic groups. J. Chromatogr. A 2013, 1276, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Krajnc, N.L.; Smrekar, F.; Černe, J.; Raspor, P.; Modic, M.; Krgovič, D.; Štrancar, A.; Podgornik, A. Purification of large plasmids with methacrylate monolithic columns. J. Sep. Sci. 2009, 32, 2682–2690. [Google Scholar] [CrossRef] [PubMed]
- Bicho, D.; Sousa, A.; Sousa, F.; Queiroz, J.; Tomaz, C.T. Effect of chromatographic conditions and plasmid DNA size on the dynamic binding capacity of a monolithic support. J. Sep. Sci. 2014, 37, 2284–2292. [Google Scholar] [CrossRef] [PubMed]
- Rački, N.; Kramberger, P.; Steyer, A.; Gašperšič, J.; Štrancar, A.; Ravnikar, M.; Gutierrez-Aguirre, I. Methacrylate monolith chromatography as a tool for waterborne virus removal. J. Chromatogr. A 2015, 1381, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Gerster, P.; Kopecky, E.M.; Hammerschmidt, N.; Klausberger, M.; Krammer, F.; Grabherr, R.; Mersich, C.; Urbas, L.; Kramberger, P.; Paril, T.; et al. Purification of infective baculoviruses by monoliths. J. Chromatogr. A 2013, 1290, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Jandera, P.; Urban, J.; Moravcová, D. Polymethacrylate and hybrid interparticle monolithic columns for fast separations of proteins by capillary liquid chromatography. J. Chromatogr. A 2006, 1109, 60–73. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Tolley, H.D.; Lee, M.L. Preparation of polymer monoliths that exhibit size exclusion properties for proteins and peptides. Anal. Chem. 2009, 81, 4406–4413. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Dong, J.; Ye, M.; Jiang, X.; Wu, R.; Zou, H. Online multidimensional separation with biphasic monolithic capillary column for shotgun proteome analysis. J. Proteome Res. 2008, 7, 306–310. [Google Scholar] [CrossRef] [PubMed]
- Albreht, A.; Vovk, I. Applicability of analytical and preparative monolithic columns to the separation and isolation of major whey proteins. J. Chromatogr. A 2012, 1227, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Blas, M.; Delaunay, N.; Rocca, J.L. Electrochromatographic separation on a poly(dimethylsiloxane)/glass chip by integration of a capillary containing an acrylate monolithic stationary phase. J. Sep. Sci. 2007, 30, 3043–3049. [Google Scholar] [CrossRef] [PubMed]
- Throckmorton, D.J.; Shepodd, T.J.; Singh, A.K. Electrochromatography in microchips: Reversed-phase separation of peptides and amino acids using photopatterned rigid polymer monoliths. Anal. Chem. 2002, 74, 784–789. [Google Scholar] [CrossRef] [PubMed]
- Stassen, C.; Desmet, G.; Broeckhoven, K.; Van Lokeren, L.; Eeltink, S. Characterization of polymer monolithic columns for small-molecule separations using total-pore-blocking conditions. J. Chromatogr. A 2014, 1325, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Nischang, I.; Brüggemann, O. Towards porous polymer monoliths for the efficient, retention-independent performance in the isocratic separation of small molecules by means of nano-liquid chromatography. J. Chromatogr. A 2010, 1217, 7514–7522. [Google Scholar] [CrossRef] [PubMed]
- Nischang, I.; Causon, T.J. Porous polymer monoliths: From their fundamental structure to analytical engineering applications. TrAC Trends Anal. Chem. 2016, 75, 108–117. [Google Scholar] [CrossRef]
- Moravcová, D.; Jandera, P.; Planeta, J. Characterization of polymer monolithic stationary phases for capillary HPLC. J. Sep. Sci. 2003, 26, 1005–1016. [Google Scholar] [CrossRef]
- Trilisky, E.I.; Koku, H.; Czymmek, K.J.; Lenhoff, A.M. Relation of structure to performance characteristics of monolithic and perfusive stationary phases. J. Chromatogr. A 2009, 1216, 6365–6376. [Google Scholar] [CrossRef] [PubMed]
- Mullner, T.; Zankel, A.; Svec, F.; Tallarek, U. Finite-size effects in the 3D reconstruction and morphological analysis of porous polymers. Mater. Today 2014, 17, 404–411. [Google Scholar] [CrossRef]
- Podgornik, A.; Jančar, J.; Merhar, M.; Kozamernik, S.; Glover, D.; Čuček, K.; Barut, M.; Štrancar, A. Large-scale methacrylate monolithic columns: Design and properties. J. Biochem. Biophys. Methods 2004, 60, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Photonic Professional GT; Nanoscribe GmbH Data Sheet; Nanoscribe GmbH: Eggenstein-Leopoldshafen, Germany, 2015.






© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Groarke, R.J.; Brabazon, D. Methacrylate Polymer Monoliths for Separation Applications. Materials 2016, 9, 446. https://doi.org/10.3390/ma9060446
Groarke RJ, Brabazon D. Methacrylate Polymer Monoliths for Separation Applications. Materials. 2016; 9(6):446. https://doi.org/10.3390/ma9060446
Chicago/Turabian StyleGroarke, Robert J., and Dermot Brabazon. 2016. "Methacrylate Polymer Monoliths for Separation Applications" Materials 9, no. 6: 446. https://doi.org/10.3390/ma9060446






