ZnO Nanostructure Templates as a Cost-Efficient Mass-Producible Route for the Development of Cellular Networks
Abstract
:1. Introduction
2. Results
2.1. HeLa Cultures on Patterned Si Wafers with ZnO Nanostructures
2.2. HeLa Cultures on Si3N4
2.3. HeLa Cultures on Emulated MEAs
2.4. HeLa Cultures on Conventional Cover Slips
3. Discussion
4. Materials and Methods
4.1. Short Description of Sol-Gel/Hydrothermal Methodology
- the deposition of a thin seeding (nucleation) layer that provides the necessary nucleation sites for the nanocrystal formation to commence. The deposition of the seeding layer is achieved via simple centrifugation of an appropriately selected sol-gel formed by the dissolution of zinc acetate dihydrate into an alcohol (ethanol or propanol), which may contain ethanolamine or triethylamine that promotes the formation of ZnO nanoparticles. It has been established that the seeding layer has a direct and critical effect on the structural properties of the ZnO nanostructures that are subsequently grown on top of it controlling the alignment, density, and morphological characteristics of the resulting structures. In other words, the seeding layer preparation conditions have a direct impact on the resulting structures and one can tailor them according to the targeted applications through several parameters like the number of spin-coatings, the annealing temperature and duration [29,72,73].
- the hydrothermal growth step per se, during which the substrates are either immersed or floating in the nutrient solution most commonly constituted of (as in our case) zinc nitrate hexahydrate (ZnNO3.6H2O) and hexamethylentetramine (C6H12N4, HMTA) [28]. The nutrient solution is heated and the formation of the ZnO nanostructures is achieved through the following reactions taking place over the nucleation sites of the seeding layer:C6H12N4 + 6H2O → 6HCHO + 4NH3NH3 + H2O → NH4+ + OH−Zn2+ + 4OH− → [Zn(OH)4]2+[Zn(OH)4]2+ → ZnO22− + 2H2OZn2+ surface + ZnO22− solution → 2ZnOO2−surface + ZnO22− solution + H2O → ZnO + 4OH−
4.2. Fabrication of Si and Si3N4 Templates with Patterns of ZnO Nanostructures
4.3. Fabrication of Templates with Metallic Patterns Modified with ZnO Nanostructures (Emulated MEAs)
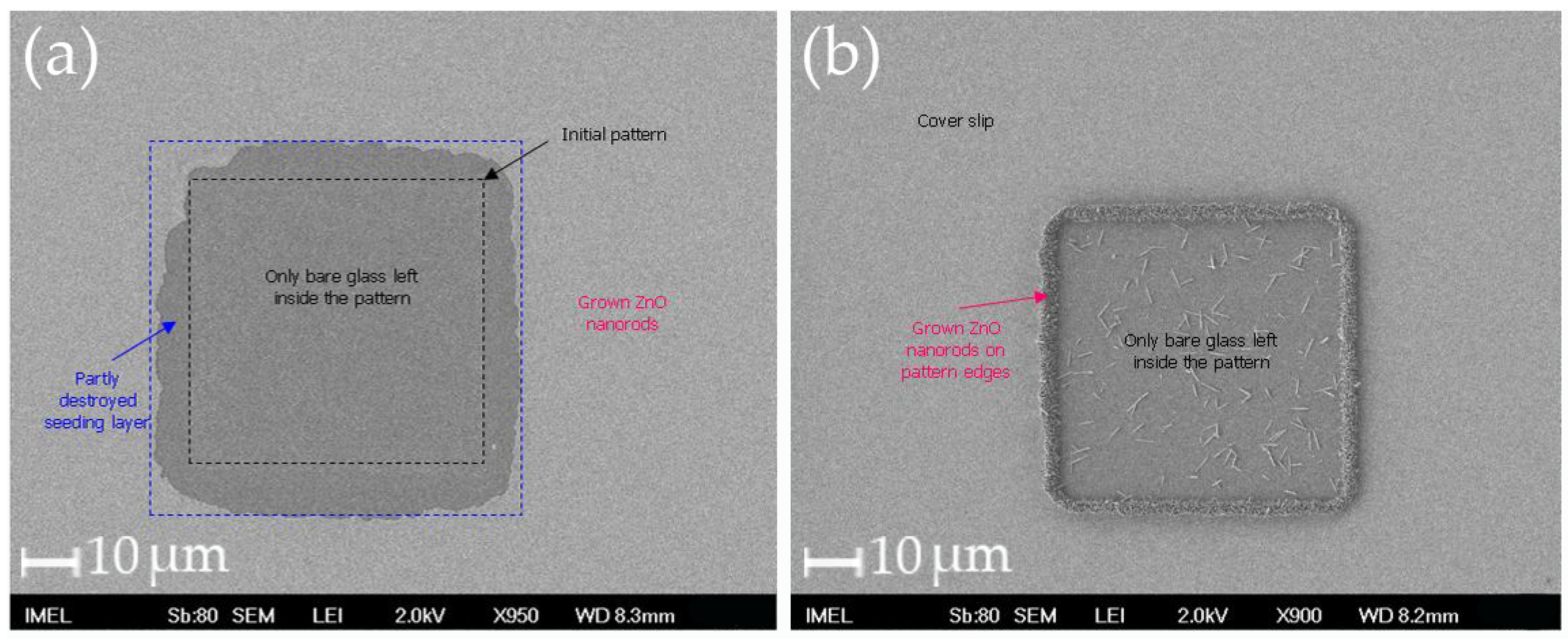
4.4. Fabrication of Cover Slip Templates with Patterns of ZnO Nanostructures
4.5. Solubility Studies on ZnO Nanostructures
4.6. HeLa Cell Cultures
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fang, Y.; Ferrie, A.M.; Fontaine, N.H.; Mauro, J.; Balakrishnan, J. Resonant waveguide grating biosensor for living cell sensing. Biophys. J. 2006, 91, 1925–1940. [Google Scholar] [CrossRef] [PubMed]
- Falconnet, D.; Csucs, G.; Michelle Grandin, H.; Textor, M. Surface engineering approaches to micropattern surfaces for cell-based assays. Biomaterials 2006, 27, 3044–3063. [Google Scholar] [CrossRef] [PubMed]
- Khademhosseini, A.; Langer, R.; Borenstein, J.T.; Vacanti, J.P. Microscale technologies for tissue engineering and biology. Proc. Natl. Acad. Sci. USA 2006, 103, 2480–2487. [Google Scholar] [CrossRef] [PubMed]
- Morin, F.; Nishimura, N.; Griscom, L.; LePioufle, B.; Fujita, H.; Takamura, Y.; Tamiya, E. Constraining the connectivity of neuronal networks cultured on microelectrode arrays with microfluidic techniques: A step towards neuron-based functional chips. Biosens. Bioelectron. 2006, 21, 1093–1100. [Google Scholar] [CrossRef] [PubMed]
- Merz, M.; Fromherz, P. Silicon chip interfaced with a geometrically defined net of snail neurons. Adv. Funct. Mater. 2005, 15, 739–743. [Google Scholar] [CrossRef]
- Krause, G.; Lehman, S.; Lehman, M.; Freund, I.; Schreiber, E.; Baumann, W. Measurement of electrical activity of long-term mammalian neuronal networks on semiconductor neurosensor chips and comparison with conventional microelectrode arrays. Biosens. Bioelectron. 2006, 21, 1272–1282. [Google Scholar] [CrossRef] [PubMed]
- Xiang, G.; Pan, L.; Huang, L.; Yu, Z.; Song, X.; Cheng, J.; Xing, W.; Zhou, Y. Microelectrode array-based system for neuropharmacological applications with cortical neurons cultured in vitro. Biosens. Bioelectron. 2007, 22, 2478–2484. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, S.N.; Yarmush, M.L.; Toner, M. Controlling cell interactions by micropatterning in co-cultures: Hepatocytes and 3T3 fibroblasts. J. Biomed. Mater. Res. 1997, 34, 189–199. [Google Scholar] [CrossRef]
- Preston, R.R.; McFadden, P.N. A two-cell biosensor that couples neuronal cells to optically monitored fish chromatophores. Biosens. Bioelectron. 2001, 16, 447–455. [Google Scholar] [CrossRef]
- Gilchrist, K.H.; Giovangrandi, L.; Whittington, R.H.; Kovacs, G.T.A. Sensitivity of cell-based biosensor to environmental variables. Biosens. Bioelectron. 2005, 20, 1397–1406. [Google Scholar] [CrossRef] [PubMed]
- Wrobel, G.; Seifert, R.; Ingebrandt, S.; Enderlein, J.; Ecken, H.; Baumann, A.; Kaupp, U.B.; Offenhausser, O. Cell-transistor coupling: Investigation of potassium currents recorded with p- and n-channel FETs. Biophys. J. 2005, 89, 3628–3638. [Google Scholar] [CrossRef] [PubMed]
- Lambacher, A.; Jenkner, M.; Merz, M.; Eversmann, B.; Kaul, R.A.; Hofmann, F.; Thewes, R.; Fromherz, P. Electrical imaging of neuronal activity by multi-transistor-arra (MTA) recording at 7.8 μm resolution. Appl. Phys. A 2004, 79, 1607–1611. [Google Scholar] [CrossRef]
- Stupp, S.I. Self-assembly and biomaterials. Nano Lett. 2010, 10, 4783–4786. [Google Scholar] [CrossRef] [PubMed]
- Kane, R.S.; Takayana, S.; Ostuni, E.; Ingber, D.E.; Whitesides, G.M. Patterning proteins and cells using soft lithography. Bioamterials 1999, 20, 2363–2376. [Google Scholar] [CrossRef]
- Wyart, C.; Ybert, C.; Bourdieu, L.; Herr, C.; Prinz, C.; Chatenay, D. Constrained synaptic connectivity in functional mammalian neuronal networks grown on patterned surfaces. J. Neurosci. Methods 2002, 117, 123–131. [Google Scholar] [CrossRef]
- Hynd, M.R.; Frampton, J.P.; Dowell-Mesfin, N.; Turner, J.N.; Shain, W. Directed cell growth on protein-functionalized hydrogel surfaces. J. Neurosci. Methods 2007, 162, 255–263. [Google Scholar] [CrossRef] [PubMed]
- James, C.D.; Davis, R.C.; Kam, L.; Craighead, H.G.; Isaacson, M.; Turner, J.N.; Shain, W. Patterned protein layers on solid substrates by thin stamp microcontact printing. Langmuir 1998, 14, 741–744. [Google Scholar] [CrossRef]
- Folch, A.; Byung-Ho, J.; Hurtado, O.; Beebe, D.J.; Toner, M. Microfabricated elastomeric stencils for micropatterning cell cultures. J. Biomed. Mater. Res. 2000, 52, 346–353. [Google Scholar] [CrossRef]
- Kenar, H.; Kocabas, A.; Aydinly, A.; Hasirci, V. Chemical and topographical modification of PHBV surface to promote osteoblast alignment and confinement. J. Biomed. Mater. Res. A 2008, 85, 1001–1010. [Google Scholar] [CrossRef] [PubMed]
- Zorlutuba, P.; Rong, Z.; Vadgama, P.; Sasirci, V. Influence of nanopatterns on endothelial cell adhesion: Enhanced cell retention under shear stress. Acta Biomater. 2009, 5, 2451–2459. [Google Scholar] [CrossRef] [PubMed]
- Khademhosseini, A.; Ferreira, L.; Blumling, J., III; Yeh, J.; Karp, J.M.; Fukuda, J.; Langer, R. Co-culture of human embryonic stem cells with murine embryonic fibroblasts on microwell-patterned substrates. Biomaterials 2006, 27, 5968–5977. [Google Scholar] [CrossRef] [PubMed]
- Kunzler, T.P.; Drobek, T.; Schuler, M.; Spencer, N.D. Systematic study of osteoblast and fibroblast response to roughness by means of surface-morphology gradients. Biomaterials 2007, 28, 2175–2182. [Google Scholar] [CrossRef] [PubMed]
- Arnold, M.; Cavalcanti-Adam, E.A.; Glass, R.; Blummel, J.; Eck, W.; Kantlehner, M.; Kessler, H.; Spatz, J.P. Activation of integrin function by nanopatterned adhesive interfaces. Chem. Phys. Chem. 2004, 5, 383–388. [Google Scholar] [CrossRef] [PubMed]
- Orgovan, N.; Peter, B.; Bősze, S.; Ramsden, J.J.; Szabó, B.; Horvath, R. Dependence of cancer cell adhesion kinetics on integrin ligand surface density measured by a high-throughput label-free resonant waveguide grating biosensor. Sci. Rep. 2014, 4, 4034. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nijdam, A.J.; Cheng, M.M.C.; Geho, D.H.; Fedele, R.; Herrmann, P.; Killian, K.; Espina, V.; Petricoin, E.F., III; Liotta, L.A.; Ferrari, M. Physicochemically modified silicon as a substrate for protein microarrays. Biomaterials 2007, 28, 550–558. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Thakar, R.G.; Wong, J.; McLeod, S.D.; Li, S. Control of cell adhesion on poly(methyl methacrylate). Biomaterials 2006, 27, 2890–2897. [Google Scholar] [CrossRef] [PubMed]
- Niarchos, G.; Makarona, E.; Tsamis, C. Growth of ZnO nanorods on patterned templates for efficient, large-area energy scavengers. Microsys. Technol. 2010, 16, 669–675. [Google Scholar] [CrossRef]
- Xu, S.; Wang, Z.L. One-dimensional ZnO nanostructures: Solution growth and functional properties. Nano Res. 2011, 4, 1013–1098. [Google Scholar] [CrossRef]
- Makarona, E.; Athanasiou, B.; Prionistis, C.; Tegou, E.; Tsamis, C. A cost-efficient solution-based process for the development of ZnO nanostructures: a comprehensive study of the role of the seeding layer formation conditions. Procedia Eng. 2015, 120, 447–450. [Google Scholar] [CrossRef]
- Gao, P.X.; Song, J.; Liu, J.; Wang, Z.L. Nanowire piezoelectric nanogenerators on plastic substrates as flexible power sources for nanodevices. Adv. Mater. 2007, 19, 67–72. [Google Scholar] [CrossRef]
- Chen, Y.; Chang, J. Growth of ZnO nanorod arrays on flexible PDMS substrates. posted on-line by Products Finishing. 2012. Available online: http://www.pfonline.com/articles/growth-of-zno-nanorod-arrays-on-flexible-pdms-substrates (accessed on 15 February 2012).
- Lee, C.Y.; Li, S.Y.; Lin, P.; Tseng, T.Y. ZnO nanowires hydrothermally grown on PET polymer substrates and their characteristics. J. Nanosci. Nanotechnol. 2005, 5, 1088–1094. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Jiang, Z.; Wang, G.; Tian, G.L.; Wang, H.K.; Song, Y. Surface functionalization of bamboo with nanostructured ZnO. Wood Sci. Technol. 2012, 46, 781–790. [Google Scholar] [CrossRef]
- Ko, H.U.; Mun, S.; Min, S.K.; Kim, G.W.; Kim, J. Fabrication of cellulose ZnO hybrid nanocomposite and its strain sensing behavior. Materials 2014, 7, 7000–7009. [Google Scholar] [CrossRef]
- Park, H.; Tong, F.; Sujan, A.; Chung, Y.; Park, M.; Tatarchuk, B.J.; Koo, H.; Ahn, H.; Yoon, Y.S.; Kim, D.J. Growth of nanostructured ZnO on wearable fabrics for functional garment. Mater. Letters 2014, 118, 47–50. [Google Scholar] [CrossRef]
- Morkoc, H.; Ozgur, U. Zinc Oxide: Fundamentals, Materials and Device Technologies; Wiley-VCH: Hoboken, NJ, USA, 2009. [Google Scholar]
- Kołodziejczak-Radzimska, A.; Jesionowski, T. Zinc Oxide—From Synthesis to Application: A Review. Materials 2014, 7, 2833–2881. [Google Scholar] [CrossRef]
- Mishra, Y.K.; Modi, G.; Cretu, V.; Postica, V.; Lupan, O.; Reimer, T.; Paulowicz, I.; Hrkac, V.; Benecke, W.; Kienle, L.; et al. Direct growth of freestanding ZnO tetrapod networks for multifunctional applications in photocatalysis, UV photodetection and gas sensing. ACS Appl. Mater. Interfaces 2015, 7, 14303–14316. [Google Scholar] [CrossRef] [PubMed]
- Mishra, Y.K.; Kaps, S.; Schuchardt, A.; Paulowicz, I.; Jin, X.; Gedamu, D.; Freitag, S.; Claus, M.; Wille, S.; Kovalev, A.; et al. Fabrication of Macroscopically Flexible and Highly Porous 3D Semiconductor Networks from Interpenetrating Nanostructures by a Simple Flame Transport Approach. Part. Part. Syst. Charact. 2013, 30, 775–783. [Google Scholar] [CrossRef]
- Lee, C.T. Fabrication methods and luminescent properties of ZnO materials for light-emitting diodes. Materials 2010, 3, 2218–2259. [Google Scholar] [CrossRef]
- Ibupoto, Z.H.; Khun, K.; Eriksson, M.; AlSalhi, M.; Atif, M.; Ansari, A.; Willander, M. Hydrothermal growth of vertically aligned ZnO nanorods using a biocomposite seed layer of ZnO nanoparticles. Materials 2013, 6, 3584–3597. [Google Scholar] [CrossRef]
- Li, Z.; Liu, X. Temperature-dependent photoluminescence property of self-assembly ZnO nanowires via chemical vapor deposition combined with hydrothermal pretreatment. Materials 2015, 8, 7598–7603. [Google Scholar] [CrossRef]
- Jung, S.H.; Oh, E.; Lee, K.H.; Yang, Y.; Park, C.G.; Park, W.; Jeong, S.H. Sonochemical preparation of shape-selective ZnO nanostructures. Cryst. Growth Des. 2008, 8, 265–269. [Google Scholar] [CrossRef]
- Gedamu, D.; Paulowicz, I.; Kaps, S.; Lupan, O.; Wille, S.; Haidarschin, G.; Mishra, Y.K.; Adelung, R. Nanonetworks: Rapid fabrication technique for interpenetrated ZnO nanotetrapod networks for fast UV sensors. Adv. Mater. 2014, 10, 1541–1550. [Google Scholar] [CrossRef] [PubMed]
- Shouli, B.; Liangyuan, C.; Dianqing, L.; Wensheng, Y.; Pengcheng, Y.; Zhiyong, L.; Aifan, C.; Liu, C.C. Different morphologies of ZnO nanorods and their sensing property. Sens. Act. B 2010, 146, 129–137. [Google Scholar] [CrossRef]
- Kim, K.H.; Utashiro, K.; Abe, Y.; Kawamura, M. Structural properties of zinc oxide nanorods grown on Al-doped zinc oxide seed layer and their applications in dye-sensitized solar cells. Materials 2014, 7, 2522–2533. [Google Scholar] [CrossRef]
- Lamberti, A.; Gazia, R.; Sacco, A.; Bianco, S.; Quaglio, M.; Chiodoni, A.; Tresso, E.; Pirri, C.F. Coral-shaped ZnO nanostructures for dye-sensitized solar cell photoanodes. Prog. Photovolt: Res. Appl. 2014, 22, 189–197. [Google Scholar] [CrossRef]
- Kyaw, A.K.K.; Sun, X.W.; Jiang, C.Y.; Lo, G.Q.; Zhao, D.W.; Kwong, D.L. An inverted organic solar cell employing a sol-gel derived ZnO electron selective layer and thermal evaporated MoO3 hole selective layer. Appl. Phys. Lett. 2008, 93, 221107. [Google Scholar]
- Polydorou, E.; Makarona, E.; Soultati, A.; Georgiadou, D.; Kyrasta, T.; Speliotis, T.; Tsamis, C.; Papanikolaou, N.; Argitis, P.; Kostis, I.; et al. Solution-processed nanostructured zinc oxide cathode interfacial layers for efficient inverted organic photovoltaics. Microel. Engin. 2014, 119, 100–104. [Google Scholar] [CrossRef]
- Ramanathan, K.; Contreras, M.A.; Perkins, C.L.; Asher, S.; Hasoon, F.S.; Keane, J.; Young, D.; Romero, M.; Metzger, W.; Noufi, R.; et al. Properties of 19.2% efficiency ZnO/CdS/CuInGaSe2 thin-film solar cells. Prog. Photovolt: Res. Appl. 2003, 11, 225–230. [Google Scholar] [CrossRef]
- Abbasi, M.A.; Ibupoto, Z.H.; Hussain, M.; Pozina, G.; Lu, J.; Hultman, L.; Nur, O.; Willander, M. Decoration of ZnO nanorods with coral reefs like NiO nanostructures by the hydrothermal growth method and their luminescence study. Materials 2014, 7, 430–440. [Google Scholar] [CrossRef]
- Wang, Z.L. From nanogenerators to piezotronics—A decade-long study of ZnO nanostructures. MRS Bulletin 2012, 37, 814–827. [Google Scholar] [CrossRef]
- Ko, Y.H.; Nagaraju, G.; Lee, S.H.; Yu, J.S. PDMS-based triboelectric and transparent nanogenerators with ZnO nanorod arrays. ACS Appl. Mater. Inter. 2014, 6, 6631–6637. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Xiao, X.; Li, Y.; Ding, Y.; Qiang, P.; Tan, X.; Mai, W.; Lin, Z.; Wu, W.; Li, T.; et al. Hydrogenated ZnO Core–Shell Nanocables for Flexible Supercapacitors and Self-Powered Systems. ACS Nano 2013, 7, 2617–2626. [Google Scholar] [CrossRef] [PubMed]
- Garinoa, N.; Lamberti, A.; Gazia, R.; Chiodonia, A.; Gerbaldia, C. Cycling behaviour of sponge-like nanostructured ZnO as thin-film Li-ion battery anodes. J. Alloys Compd. 2014, 615, S454–S458. [Google Scholar] [CrossRef]
- Chu, B.H.; Lee, J.; Chang, C.Y.; Jiang, P.; Tseng, Y.; Pearton, S.J.; Gupte, A.; Lele, T.; Ren, F. The study of low temperature hydrothermal growth of ZnO nanorods on stents and its applications of cell adhesion and viability. Appl. Surf. Sci. 2009, 255, 8309–8312. [Google Scholar] [CrossRef]
- Kritharidou, A.; Georgoussi, Z.; Tsamis, C.; Makarona, E. Zinc oxide nanostructures as low-cost templates for neuronal circuit. In Proceedings of SPIE 8765- Progress in Biomedical Optics and Imaging, Grenoble, France, 25–26 April 2013; pp. 87650G-1–87650G-7.
- Lee, J.; Kang, B.S.; Hicks, B.; Chancellor, T.F.J.; Chu, B.H.; Wang, H.T.; Keselowksy, B.G.; Ren, F.; Lele, T.P. The control of cell adhesion and viability by zinc oxide nanorods. Biomaterials 2008, 29, 3743–3749. [Google Scholar] [CrossRef] [PubMed]
- Mishra, Y.K.; Adelung, R.; Roehl, C.; Shukla, D.; Spors, F.; Tiwari, V. Virostatic potential of micro-nano filopodia-like ZnO structures against herpes simplex virus-1. Antiviral Res. 2011, 92, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Antoine, T.E.; Mishra, Y.K.; Trigilio, J.; Tiwari, V.; Adelung, R.; Shukla, D. Prophylactic, therapeutic and neutralizing effects of zinc oxide tetrapod structures against herpes simplex virus type-2 infection. Antiviral Res. 2012, 96, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Migliorini, E.; Ban, J.; Grenci, G.; Andolfi, L.; Pozzato, A.; Tormen, M.; Torre, V.; Lazzarino, M. Nanomechanics controls neuronal precursors adhesion and differentiation. Biotechnol. Bioeng. 2013, 110, 2301–2310. [Google Scholar] [CrossRef] [PubMed]
- Anselme, K.; Davidson, P.; Popa, A.M.; Giazzon, M.; Liley, M.; Ploux, L. The interaction of cells and bacteria with surfaces structured at the nanometer scale. Acta Biomater. 2010, 6, 3824–3846. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Lee, H.; Lee, Y.K.; Nam, J.M.; Levchenko, A. Biomimetic nanopatterns as enabling tools for analysis and control of live cell. Adv. Mater. 2010, 22, 4551–4566. [Google Scholar] [CrossRef] [PubMed]
- Seo, C.H.; Jeong, H.; Furukawa, K.S.; Suzuki, Y.; Ushida, T. The switching of focal adhesion maturation sites and actin filament activation for MSCs by topography of well-defined micropatterned surfaces. Biomaterials 2013, 34, 1764–1771. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, T.; Wolpwert, L. Cellular mechanisms in the morphogenesis of the sea urchin larva. The formation of arms. Exp. Cell Res. 1961, 22, 509–520. [Google Scholar] [CrossRef]
- Albrecht-Bueheler, G. Filopodia of spreading 3T3 cells. Do they have a substrate-exploring function? J. Cell Biol. 1976, 69, 275–286. [Google Scholar] [CrossRef]
- Davenport, R.W.; Dou, P.; Rehder, V.; Kater, S.B. A sensory role for neuronal growth cone filopodia. Nature 1993, 361, 721–724. [Google Scholar] [CrossRef] [PubMed]
- Bentley, D.; Toroian-Raymond, A. Disoriented pathfinding by pioneer neurone growth cones deprived of filopodia by cytochalasin treatment. Nature 1986, 323, 712–715. [Google Scholar] [CrossRef] [PubMed]
- Pula, G.; Schuh, K.; Nakayama, K.; Nakayama, K.I.; Walter, U.; Poole, A. PKCdelta regulates collagen-induced platelet aggregation through inhibition of VASP-mediated filopodia formation. Blood 2006, 108, 4035–4044. [Google Scholar] [CrossRef] [PubMed]
- Albuschies, J.; Vogel, V. The role of filopodia in the recognition of nanotopogrphie. Sci. Rep. 2013, 3, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Pitroff, A.; Park, J.; Bauer, S.; Schmuki, P. ECM spreading behavior on micropatterned TiO2 nanotube surfaces. Acta Biomater. 2012, 8, 2639–2647. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.H.; Utashiro, K.; Abe, Y.; Kawamura, M. Growth of Zinc oxide nanorods using various seed layer annealing temperatures and substrate materials. Int. J. Electrochem. Sci. 2014, 9, 2080–2089. [Google Scholar]
- Greene, L.E.; Law, M.; Tan, D.H.; Montano, M.; Goldberger, J.; Somorjai, G.; Yang, P. General route to vertical ZnO nanowire arrays using textured ZnO seeds. Nano Letters 2005, 5, 1231–1236. [Google Scholar] [CrossRef] [PubMed]
- Reddy, K.M.; Feris, K.; Bell, J.; Wingett, D.G.; Hanley, C.; Punnoose, A. Selective toxicity of zinc oxide nanoparticles to prokaryoitc and eukaryotic systems. Appl. Phys. Lett. 2007, 90, 2139021–2139023. [Google Scholar] [CrossRef] [PubMed]
- Papavlassopoulos, H.; Mishra, Y.K.; Kaps, S.; Paulowicz, I.; Abdelaziz, R.; Elbahri, M.; Maser, E.; Adelung, R.; Rohl, C. Toxicity of functional nano-micro zinc oxide tetrapods: Impact of cell culture conditions, cellular age and material properties. PLoS ONE 2014, 9, e84983. [Google Scholar] [CrossRef] [PubMed]





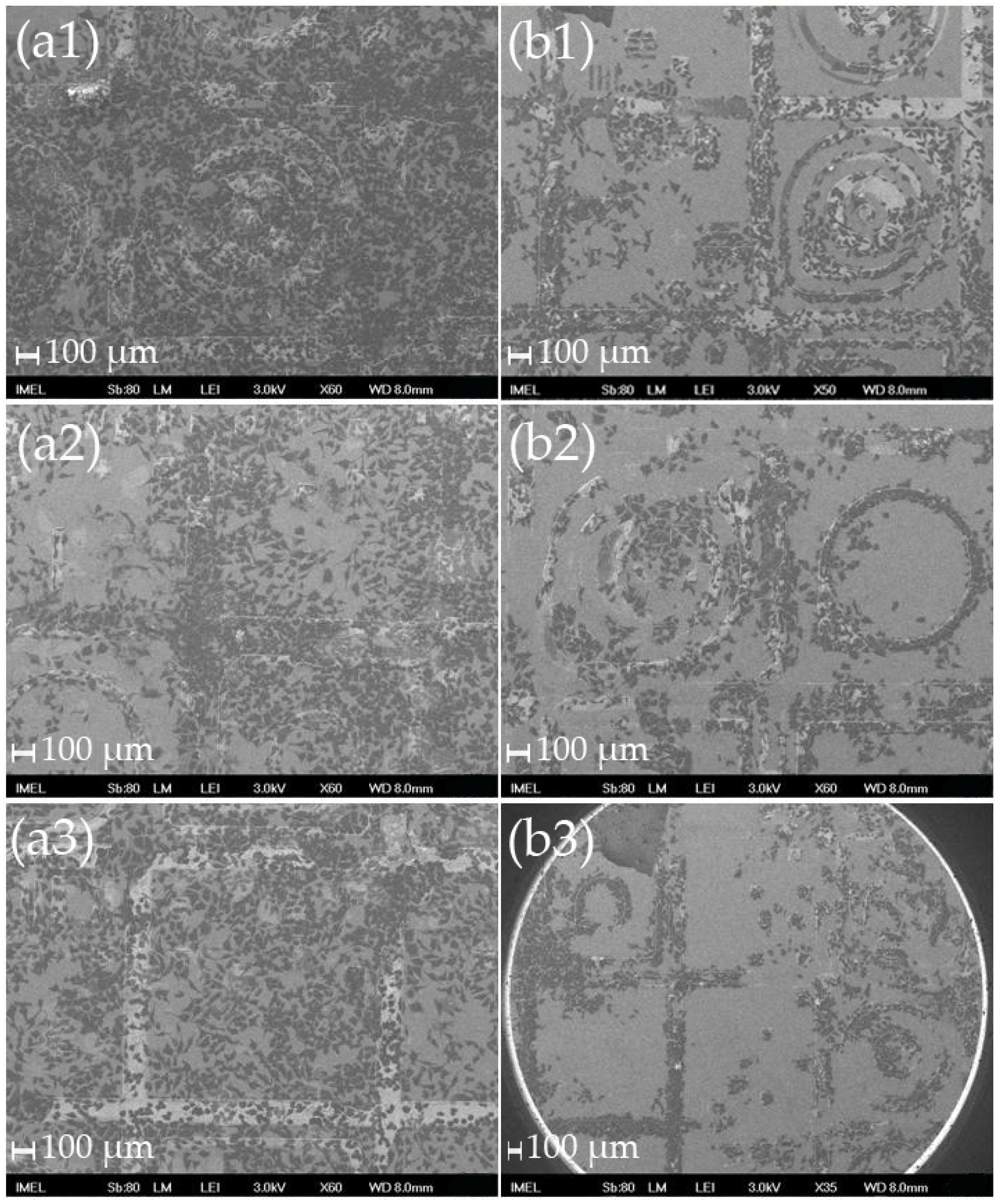







| ZnO Nanostructures on Patterned Si | ZnO Nanostructures on Patterned Si3N4 | ZnO Nanostructures on Patterned Al | ZnO Nanostructures on Patterned Pt | ZnO Nanostructures on Patterned Cover Slips |
|---|---|---|---|---|
| Substrates with small patterns: Opposite behavior to Neuro2A increased population onto nanorods at large angles decreased population onto vertical nanorods The flat areas too small to accommodate cell somata/not possible to determine selective adhesion | Substrates with small patterns: Cells tend to remain on flat areas by squeezing the somata inside the flat areas Flat areas too small to accommodate the cell somata/ not possible to determine selective adhesion | 2 days: No preferential adhesion | 2 days: Cells are more spherical and smaller compared to the Al-MEAs Conformation to the patterns at the borderlines- no clear preference | Cover slips uniformly covered with ZnO nanostructures: Necrosis since day 1 |
| Substrates with large patterns and large flat areas: Selective adhesion onto flat surfaces and avoidance of nanostructured areas Cell spreading and conformation to the patterns at the borderlines | Substrates with large patterns and large flat areas Selective adhesion onto the flat surfaces and avoidance of nanostructured areas Cell spreading and conformation to the patterns at the borderlines | 4 days: Clear migration and selective adhesion of cells onto nanopatterned metallic layers—almost all cells adhere onto the nanopatterns | 4 days: Clear migration and selective adhesion of the cells onto the nanopatterned metallic layers—almost all cells adhere onto the nanopatterns Cells obtain their “conventional” elongated shape | Cover slips with large ZnO nanostructure patterns: Cell necrosis after day 1 Cover slips with small ZnO nanostructure patterns: Clear migration and selective adhesion of the cells onto the nanopatterned areas after day 1 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Makarona, E.; Peter, B.; Szekacs, I.; Tsamis, C.; Horvath, R. ZnO Nanostructure Templates as a Cost-Efficient Mass-Producible Route for the Development of Cellular Networks. Materials 2016, 9, 256. https://doi.org/10.3390/ma9040256
Makarona E, Peter B, Szekacs I, Tsamis C, Horvath R. ZnO Nanostructure Templates as a Cost-Efficient Mass-Producible Route for the Development of Cellular Networks. Materials. 2016; 9(4):256. https://doi.org/10.3390/ma9040256
Chicago/Turabian StyleMakarona, Eleni, Beatrix Peter, Inna Szekacs, Christos Tsamis, and Robert Horvath. 2016. "ZnO Nanostructure Templates as a Cost-Efficient Mass-Producible Route for the Development of Cellular Networks" Materials 9, no. 4: 256. https://doi.org/10.3390/ma9040256









