Nanostructural Engineering of Nanoporous Anodic Alumina for Biosensing Applications
Abstract
:1. Introduction
2. Nanoporous Anodic Alumina Pore Engineering
2.1. Ex Situ Strategies: Hierarchical Nanopore Structures

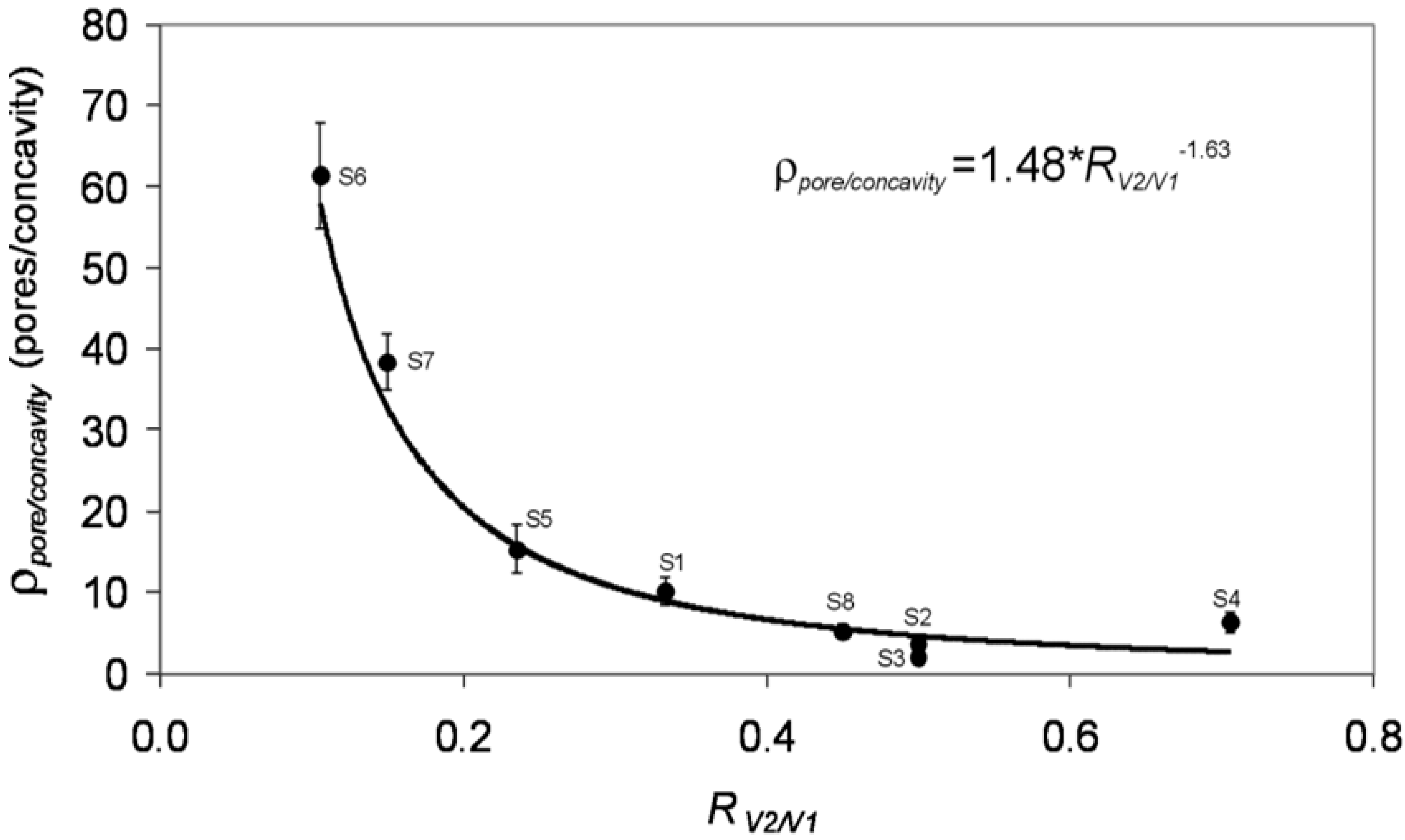
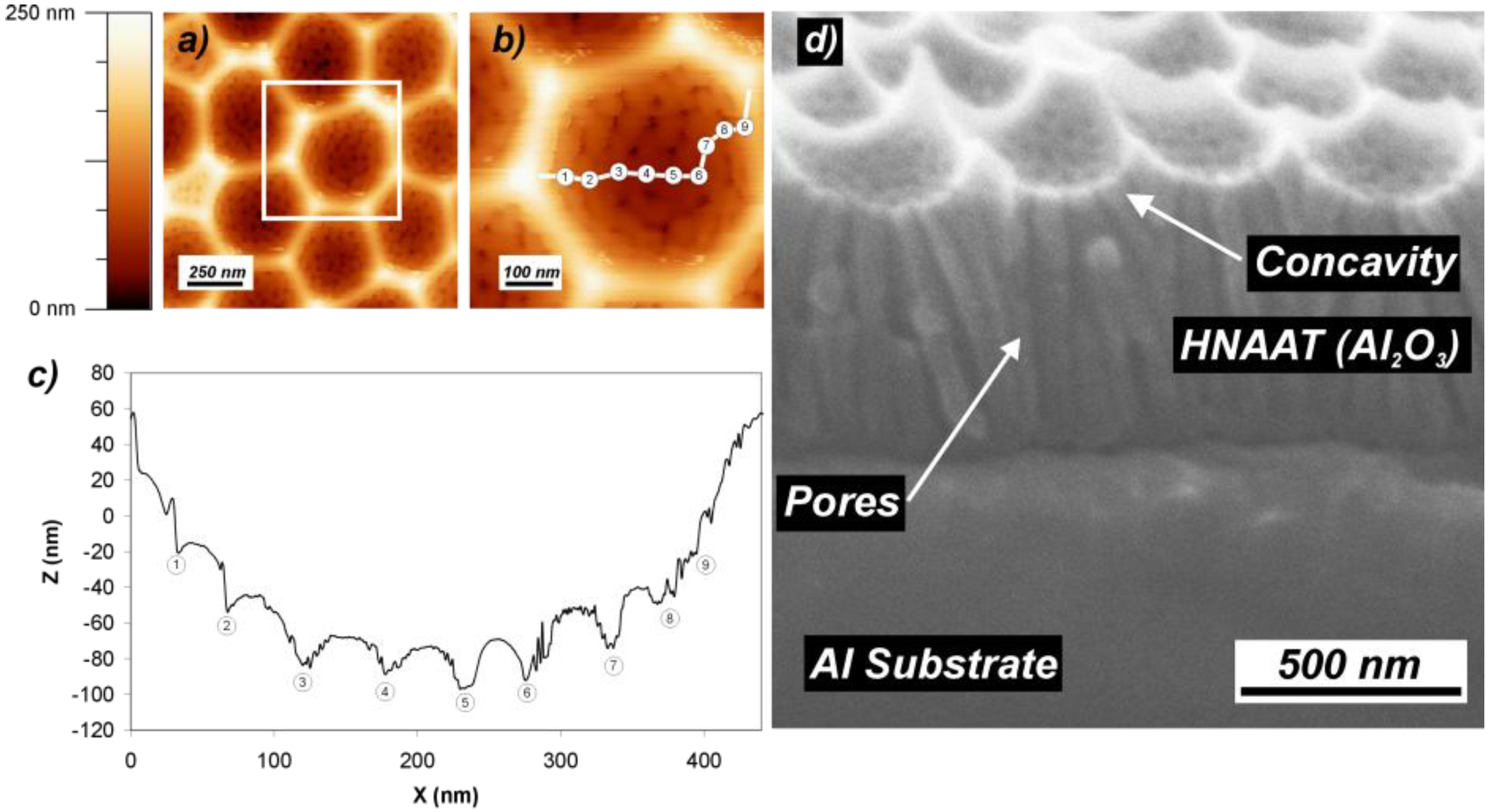
2.2. In Situ Strategies: Pore Modulation by Continuous Variation of Anodization Parameters
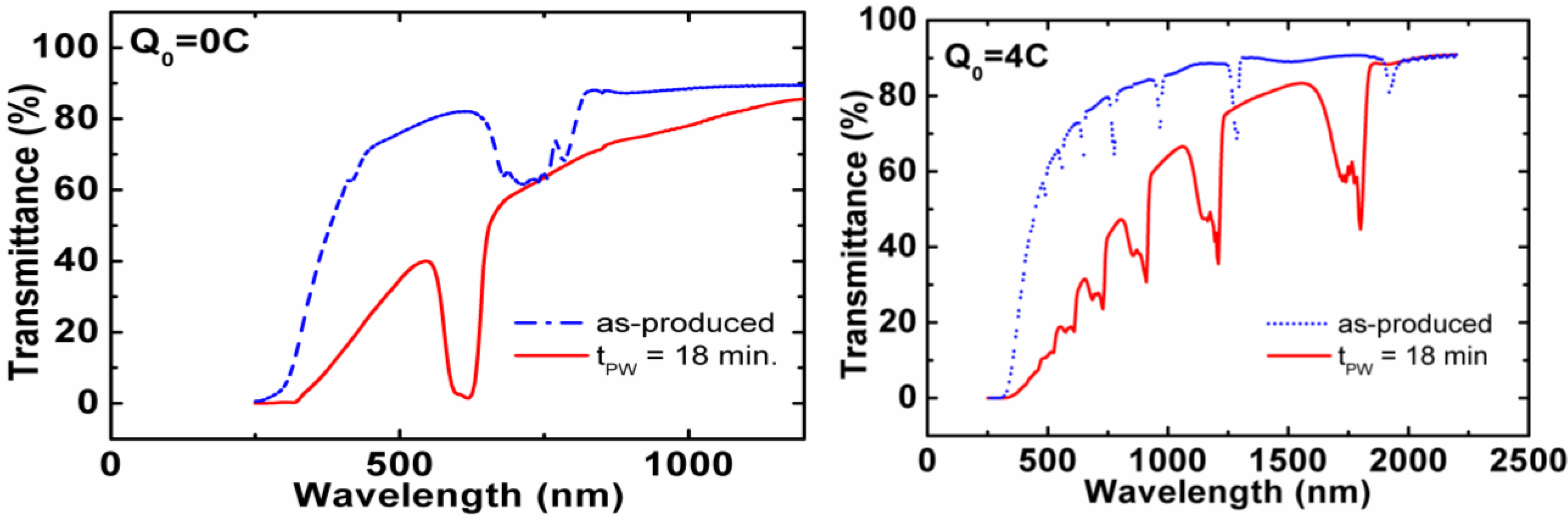
2.2.1. In Situ Strategies: Pore Modulation by Anodization Voltage Variation, NAA-Based DBR



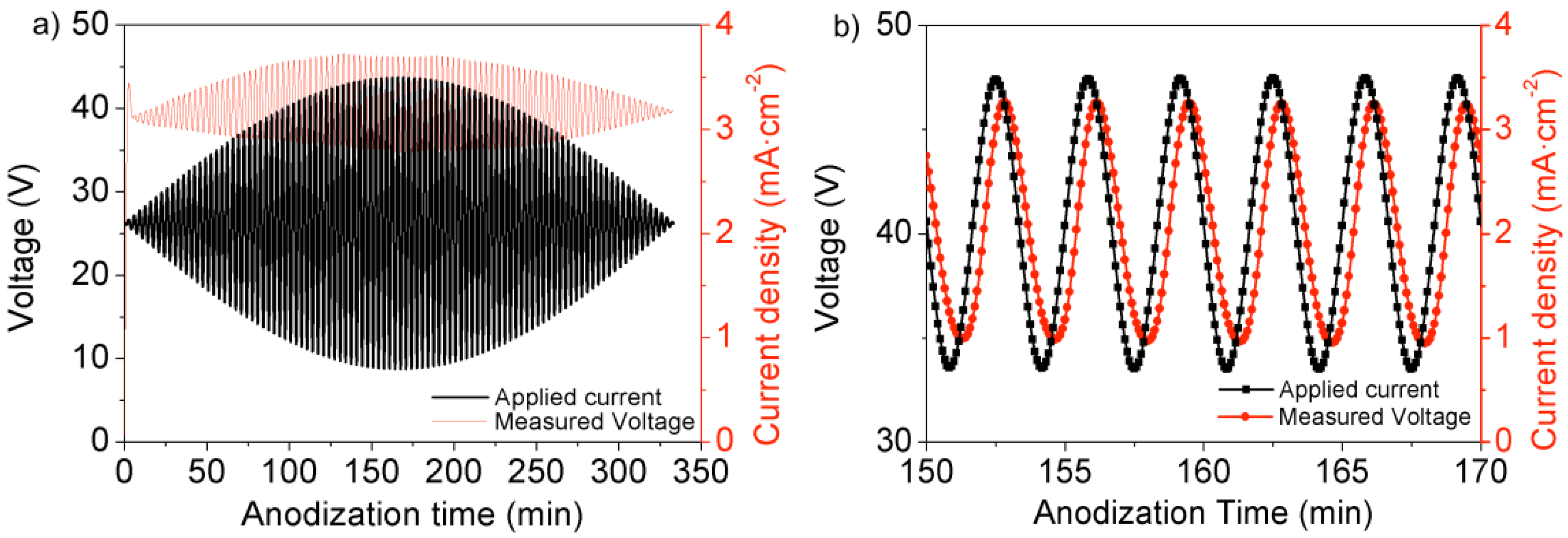
2.2.2. In Situ Strategies: Pore Modulation by Anodization Current Variation: NAA-Based Rugate Filters
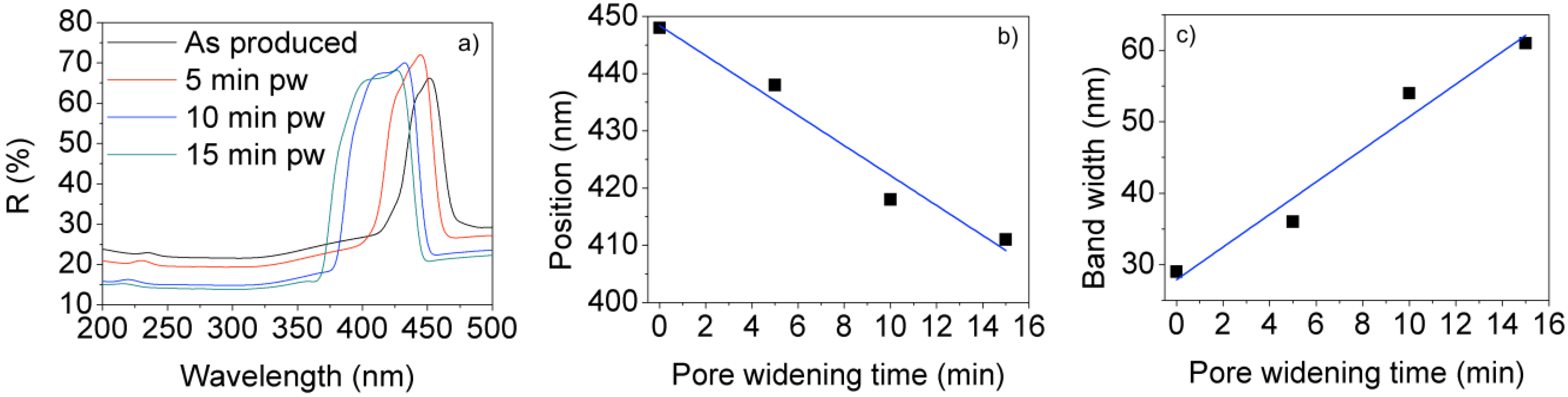
3. Applications of Nanoengineered NAA to Biosensing
3.1. Single Layer Structures: NAA-Based Barcodes


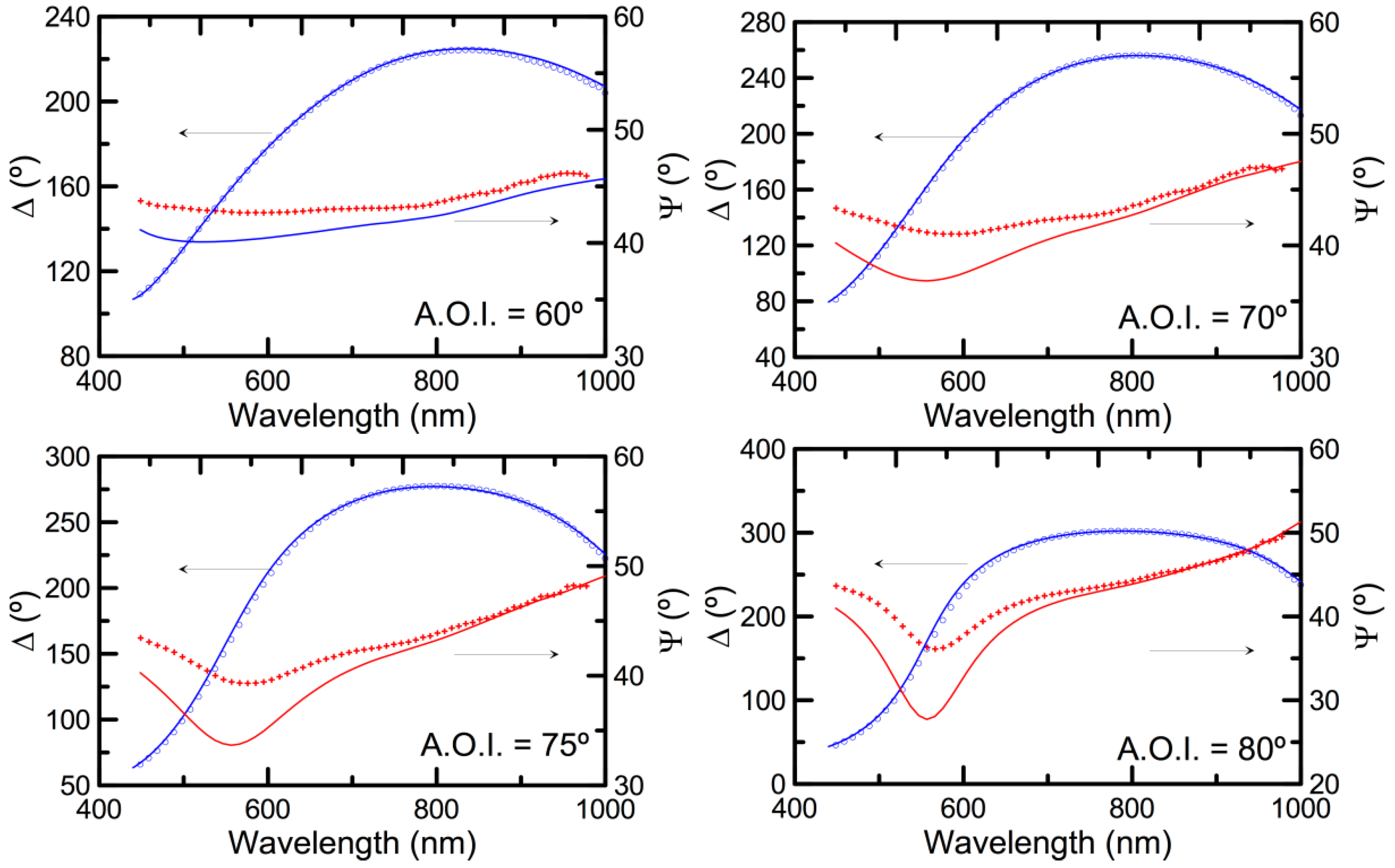
3.2. Double-Layer Gold-Coated NAA Sensors
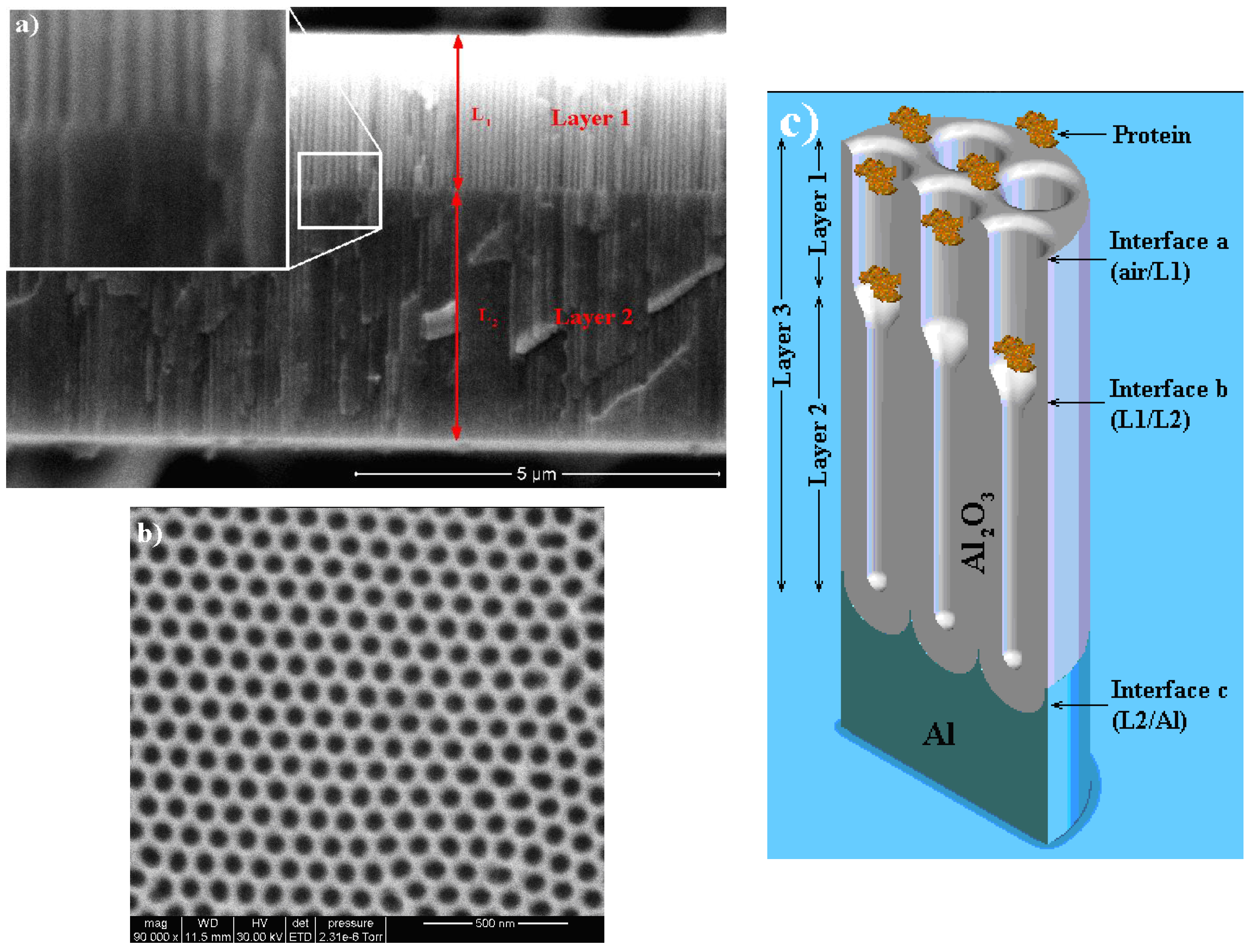

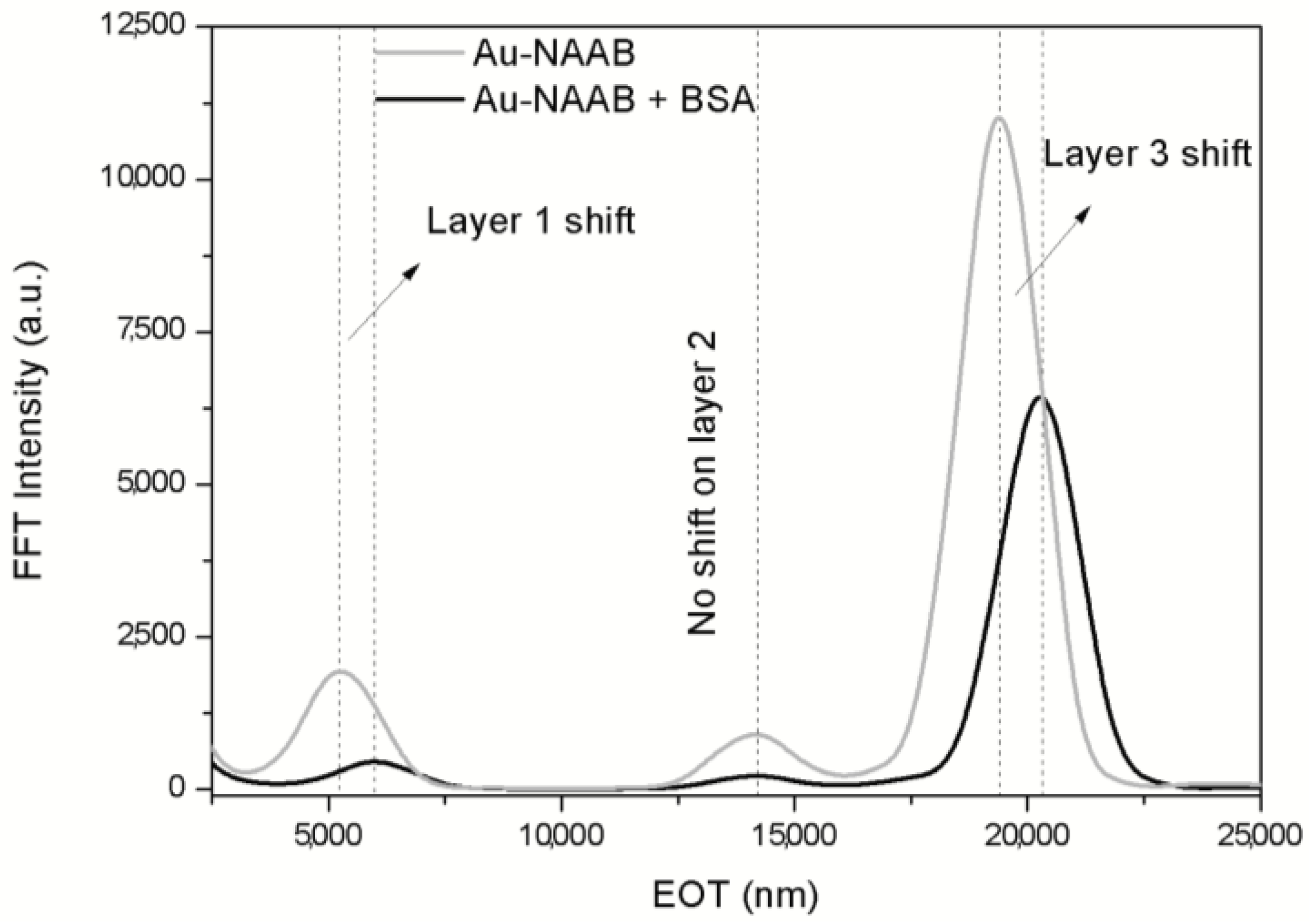
3.3. Optical Microcavity Rugate Filters
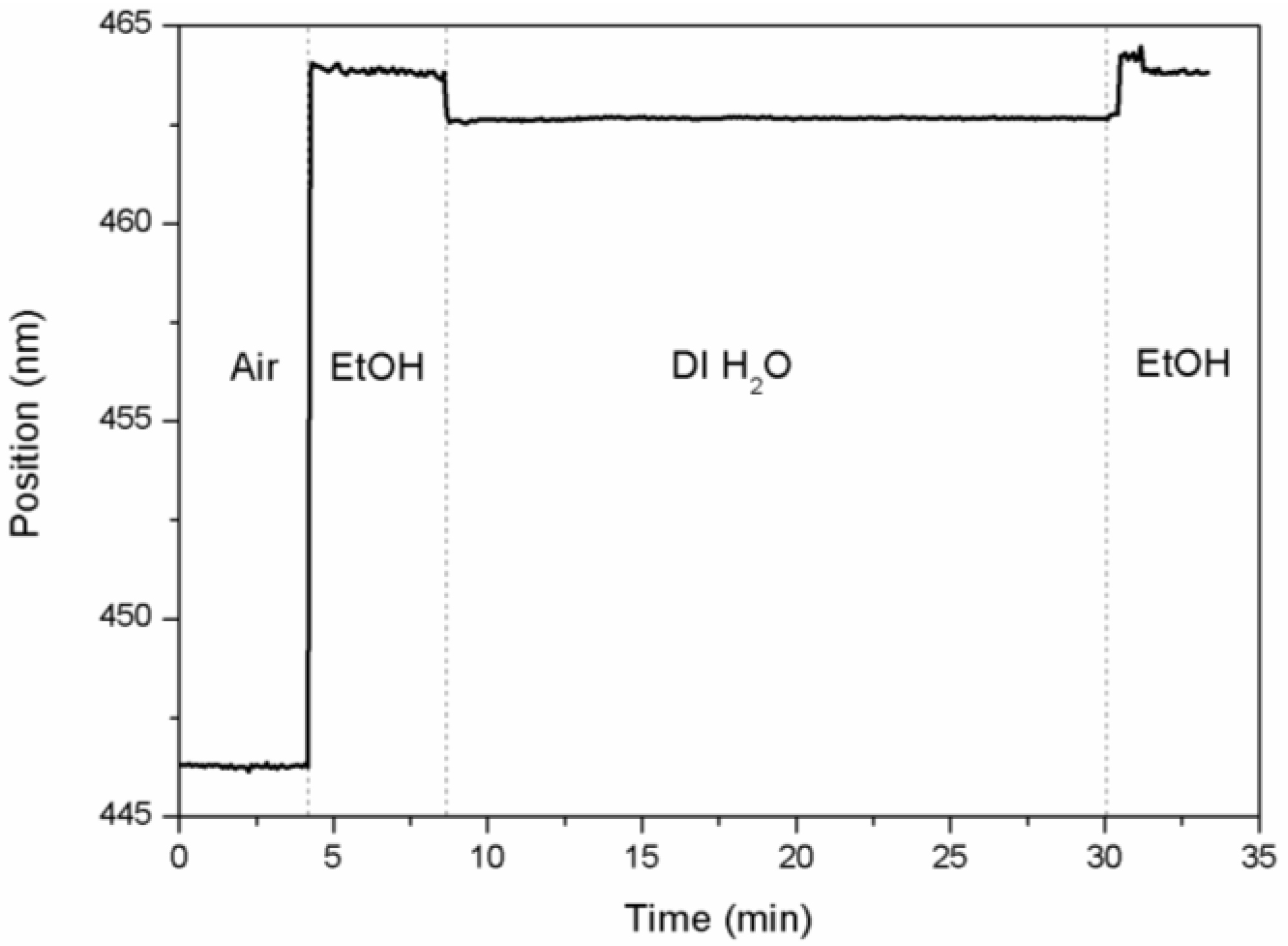
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sulka, G.D. Nanostructured Materials in Electrochemistry; Eftekhari, A., Ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2008; pp. 1–116. [Google Scholar]
- Lee, W. The anodization of aluminum for nanotechnology applications. JOM 2010, 62, 57–63. [Google Scholar] [CrossRef]
- Warkiani, M.E.; Bhagat, A.A.S.; Khoo, B.L.; Han, J.; Lim, C.T.; Gong, H.Q.; Fane, A.G. Isoporous micro/nanoengineered membranes. ACS Nano 2013, 7, 1882–1904. [Google Scholar] [CrossRef]
- Banerjee, P.; Perez, I.; Henn-Lecordier, L.; Lee, S.B.; Rubloff, G.W. Nanotubular metal-insulator-metal capacitor arrays for energy storage. Nat. Nanotechnol. 2009, 4, 292–296. [Google Scholar] [CrossRef]
- Santos, A.; Ferré-Borrull, J.; Marsal, L.F.; Pallarés, J.; Formentín, P. Fabrication and characterization of high-density arrays of P3HT nanopillars on ITO/glass substrates. Sol. Energy Mater. Sol. Cells 2010, 94, 1247–1253. [Google Scholar] [CrossRef]
- Kato, T.; Hayase, S. Quasi-solid dye sensitized solar cell with straight ion paths. J. Electrochem. Soc. 2007, 154, B117–B121. [Google Scholar] [CrossRef]
- Foong, T.R.B.; Shen, Y.; Hu, X.; Sellinger, A. Template-directed liquid ald growth of TiO2 nanotube arrays: Properties and potential in photovoltaic devices. Adv. Funct. Mater. 2010, 20, 1390–1396. [Google Scholar] [CrossRef]
- Santos, A.; Vojkuvka, L.; Pallarés, J.; Ferré-Borrull, J.; Marsal, L.F. Cobalt and nickel nanopillars on aluminium substrates by direct current electrodeposition process. Nanoscale Res. Lett. 2009, 4, 1021–1028. [Google Scholar] [CrossRef]
- González-Díaz, J.B.; García-Martín, A.; Armelles, G.; Navas, D.; Vázquez, M.; Nielsch, K.; Wehrspohn, R.B.; Gösele, U. Enhanced magneto-optics and size effects in ferromagnetic nanowire arrays. Adv. Mater. 2007, 19, 2643–2647. [Google Scholar] [CrossRef]
- Pallarés, J.; Marsal, L.F.; Ferré-Borrull, J.; Santos, A.; Formentin, P. Quasi-ordered P3HT nanopillar-nanocap structures with controlled size. Mater. Lett. 2010, 64, 371–374. [Google Scholar] [CrossRef]
- Platschek, B.; Keilbach, A.; Bein, T. Mesoporous structures confined in anodic alumina membranes. Adv. Mater. 2011, 23, 2395–2412. [Google Scholar] [CrossRef]
- Ingham, C.J.; ter Maat, J.; de Vos, W.M. Where bio meets nano: The many uses for nanoporous aluminum oxide in biotechnology. Biotechnol. Adv. 2012, 30, 1089–1099. [Google Scholar] [CrossRef]
- Gultepe, E.; Nagesha, D.; Sridhar, S.; Amiji, M. Nanoporous inorganic membranes or coatings for sustained drug delivery in implantable devices. Adv. Drug Deliv. Rev. 2010, 62, 305–315. [Google Scholar] [CrossRef]
- De la Escosura-Muñiz, A.; Merkoçi, A. Nanochannels preparation and application in biosensing. ACS Nano 2012, 6, 7556–7583. [Google Scholar] [CrossRef]
- Santos, A.; Kumeria, T.; Losic, D. Nanoporous anodic aluminum oxide for chemical sensing and biosensors. TrAC Trends Anal. Chem. 2013, 44, 25–38. [Google Scholar] [CrossRef]
- Adiga, S.P.; Jin, C.; Curtiss, L.A.; Monteiro-Riviere, N.A.; Narayan, R.J. Nanoporous membranes for medical and biological applications. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2005, 1, 568–581. [Google Scholar]
- Santos, A.; Pallarès, J.; Formentín, P.; Alba, M.; Ferré-Borrull, J.; Marsal, L.F.; Balderrama, V.S. Tunable Fabry-Pérot interferometer based on nanoporous anodic alumina for optical biosensing purposes. Nanoscale Res. Lett. 2012, 7. [Google Scholar] [CrossRef]
- Li, Y.B.; Zheng, M.J.; Ma, L. High-speed growth and photoluminescence of porous anodic alumina films with controllable interpore distances over a large range. Appl. Phys. Lett. 2007, 91. [Google Scholar] [CrossRef]
- Maksymov, I.; Ferré-Borrull, J.; Pallarès, J.; Marsal, L.F. Photonic stop bands in quasi-random nanoporous anodic alumina structures. Photonics Nanostruct. Fundam. Appl. 2012, 10, 459–462. [Google Scholar] [CrossRef]
- Masuda, H.; Fukuda, K. Ordered metal nanohole arrays made by a two-step replication of honeycomb structures of anodic alumina. Science 1995, 268, 1466–1468. [Google Scholar]
- Masuda, H.; Satoh, M. Fabrication of gold nanodot array using anodic porous alumina as an evaporation mask. Jpn. J. Appl. Phys. 1996, 35, L126–L129. [Google Scholar]
- Li, A.P.; Müller, F.; Birner, A.; Nielsch, K.; Gösele, U. Hexagonal pore arrays with a 50–420 nm interpore distance formed by self-organization in anodic alumina. J. Appl. Phys. 1998, 84. [Google Scholar] [CrossRef]
- Sulka, G.D.; Parkoła, K.G. Temperature influence on well-ordered nanopore structures grown by anodization of aluminium in sulphuric acid. Electrochim. Acta 2007, 52, 1880–1888. [Google Scholar] [CrossRef]
- Ono, S.; Saito, M.; Asoh, H. Self-ordering of anodic porous alumina formed in organic acid electrolytes. Electrochim. Acta 2005, 51, 827–833. [Google Scholar] [CrossRef]
- Chu, S.Z.; Wada, K.; Inoue, S.; Todoroki, S. Formation and microstructures of anodic alumina films from aluminum sputtered on glass substrate. J. Electrochem. Soc. 2002, 149, B321–B327. [Google Scholar] [CrossRef]
- Ono, S.; Saito, M.; Ishiguro, M.; Asoh, H. Controlling factor of self-ordering of anodic porous alumina. J. Electrochem. Soc. 2004, 151, B473–B478. [Google Scholar] [CrossRef]
- Lee, W.; Ji, R.; Gösele, U.; Nielsch, K. Fast fabrication of long-range ordered porous alumina membranes by hard anodization. Nat. Mater. 2006, 5, 741–747. [Google Scholar] [CrossRef]
- Lee, W.; Schwirn, K.; Steinhart, M.; Pippel, E.; Scholz, R.; Gösele, U. Structural engineering of nanoporous anodic aluminium oxide by pulse anodization of aluminium. Nat. Nanotechnol. 2008, 3, 234–239. [Google Scholar] [CrossRef]
- Schwirn, K.; Lee, W.; Hillebrand, R.; Steinhart, M.; Nielsch, K.; Gösele, U. Self-ordered anodic aluminum oxide formed by H2SO4 hard anodization. ACS Nano 2008, 2, 302–310. [Google Scholar] [CrossRef]
- Santos, A.; Formentín, P.; Ferré-Borrull, J.; Pallarès, J.; Marsal, L.F. Nanoporous anodic alumina obtained without protective oxide layer by hard anodization. Mater. Lett. 2012, 67, 296–299. [Google Scholar] [CrossRef]
- Hotta, K.; Yamaguchi, A.; Teramae, N. Deposition of polyelectrolyte multilayer film on a nanoporous alumina membrane for stable label-free optical biosensing. J. Phys. Chem. C 2012, 116, 23533–23539. [Google Scholar] [CrossRef]
- Hotta, K.; Yamaguchi, A.; Teramae, N. Nanoporous waveguide sensor with optimized nanoarchitectures for highly sensitive label-free biosensing. ACS Nano 2012, 6, 1541–1547. [Google Scholar] [CrossRef]
- Lau, K.H.A.; Tan, L.-S.; Tamada, K.; Sander, M.S.; Knoll, W. Highly sensitive detection of processes occurring inside nanoporous anodic alumina templates: A waveguide optical study. J. Phys. Chem. B 2004, 108, 10812–10818. [Google Scholar]
- Yeom, S.-H.; Kim, O.-G.; Kang, B.-H.; Kim, K.-J.; Yuan, H.; Kwon, D.-H.; Kim, H.-R.; Kang, S.-W. Highly sensitive nano-porous lattice biosensor based on localized surface plasmon resonance and interference. Opt. Express 2011, 19, 22882–22891. [Google Scholar]
- Alvarez, S.D.; Li, C.-P.; Chiang, C.E.; Schuller, I.K.; Sailor, M.J. A label-free porous alumina interferometric immunosensor. ACS Nano 2009, 3, 3301–3307. [Google Scholar] [CrossRef]
- Santos, A.; MacÍas, G.; Ferré-Borrull, J.; Pallarès, J.; Marsal, L.F. Photoluminescent enzymatic sensor based on nanoporous anodic alumina. ACS Appl. Mater. Interfaces 2012, 4, 3584–3588. [Google Scholar] [CrossRef]
- Wang, B.; Fei, G.T.; Wang, M.; Kong, M.G.; Zhang, D.L. Preparation of photonic crystals made of air pores in anodic alumina. Nanotechnology 2007, 18. [Google Scholar] [CrossRef]
- Rahman, M.M.; Garcia-Caurel, E.; Santos, A.; Marsal, L.F.; Pallarès, J.; Ferré-Borrull, J. Effect of the anodization voltage on the pore-widening rate of nanoporous anodic alumina. Nanoscale Res. Lett. 2012, 7. [Google Scholar] [CrossRef] [Green Version]
- Rahman, M.M.; Marsal, L.F.; Pallarès, J.; Ferré-Borrull, J. Tuning the photonic stop bands of nanoporous anodic alumina-based distributed bragg reflectors by pore widening. ACS Appl. Mater. Interfaces 2013, 5, 13375–13381. [Google Scholar] [CrossRef]
- Losic, D.; Lillo, M. Porous alumina with shaped pore geometries and complex pore architectures fabricated by cyclic anodization. Small 2009, 5, 1392–1397. [Google Scholar] [CrossRef]
- Santos, A.; Ferré-Borrull, J.; Pallarès, J.; Marsal, L.F. Hierarchical nanoporous anodic alumina templates by asymmetric two-step anodization. Phys. Status Solidi 2011, 208, 668–674. [Google Scholar] [CrossRef]
- Santos, A.; Formentín, P.; Pallarès, J.; Ferré-Borrull, J.; Marsal, L.F. Structural engineering of nanoporous anodic alumina funnels with high aspect ratio. J. Electroanal. Chem. 2011, 655, 73–78. [Google Scholar] [CrossRef]
- Oh, J.; Thompson, C.V. The role of electric field in pore formation during aluminum anodization. Electrochim. Acta 2011, 56, 4044–4051. [Google Scholar] [CrossRef]
- Lee, W.; Kim, J.-C. Highly ordered porous alumina with tailor-made pore structures fabricated by pulse anodization. Nanotechnology 2010, 21. [Google Scholar] [CrossRef]
- Lee, S.; Kim, D.; Gillette, E.; Oh, J.; Han, S.W.; Lee, S.B. Anodized pore structural evolution of focused ion beam patterned Al: Direct analysis of branched nanopores and nanosacks. Phys. Chem. Chem. Phys. 2013, 15, 10659–10665. [Google Scholar] [CrossRef]
- Yin, A.; Guico, R.S.; Xu, J. Fabrication of anodic aluminium oxide templates on curved surfaces. Nanotechnology 2007, 18. [Google Scholar] [CrossRef]
- He, B.; Son, S.J.; Lee, S.B. Shape-coded silica nanotubes for biosensing. Langmuir 2006, 22, 8263–8265. [Google Scholar] [CrossRef]
- He, B.; Son, S.J.; Lee, S.B. Suspension array with shape-coded silica nanotubes for multiplexed immunoassays. Anal. Chem. 2007, 79, 5257–5263. [Google Scholar] [CrossRef]
- He, B.; Kim, S.K.; Son, S.J.; Lee, S.B. Shape-coded silica nanotubes for multiplexed bioassay: Rapid and reliable magnetic decoding protocols. Nanomed. Lond. 2010, 5, 77–88. [Google Scholar] [CrossRef]
- Nagaura, T.; Takeuchi, F.; Inoue, S. Fabrication and structural control of anodic alumina films with inverted cone porous structure using multi-step anodizing. Electrochim. Acta 2008, 53, 2109–2114. [Google Scholar] [CrossRef]
- Yamauchi, Y.; Nagaura, T.; Ishikawa, A.; Chikyow, T.; Inoue, S. Evolution of standing mesochannels on porous anodic alumina substrates with designed conical holes. J. Am. Chem. Soc. 2008, 130, 10165–10170. [Google Scholar] [CrossRef]
- Ho, A.Y.Y.; Gao, H.; Lam, Y.C.; Rodríguez, I. Controlled fabrication of multitiered three-dimensional nanostructures in porous alumina. Adv. Funct. Mater. 2008, 18, 2057–2063. [Google Scholar] [CrossRef]
- Lee, K.H.; Lim, X.Y.; Wai, K.W.; Romanato, F.; Wong, C.C. Variation of nanopore diameter along porous anodic alumina channels by multi-step anodization. J. Nanosci. Nanotechnol. 2011, 11, 1147–1153. [Google Scholar] [CrossRef]
- Eason, R.W.; Nistor, M.; Perriere, J.; Solis, J.; Zergioti, I.; Sanza, F.J.; Laguna, M.F.; Casquel, R.; Holgado, M.; Barrios, C.A.; et al. Cost-effective SU-8 micro-structures by DUV excimer laser lithography for label-free biosensing. Appl. Surf. Sci. 2011, 257, 5403–5407. [Google Scholar] [CrossRef]
- Li, J.; Li, C.; Chen, C.; Hao, Q.; Wang, Z.; Zhu, J.; Gao, X. Facile method for modulating the profiles and periods of self-ordered three-dimensional alumina taper-nanopores. ACS Appl. Mater. Interfaces 2012, 4, 5678–5683. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, L.; Lee, W.; Knez, M.; Liu, L. Novel three-dimensional nanoporous alumina as a template for hierarchical TiO2 nanotube arrays. Small 2013, 9, 1025–1029. [Google Scholar] [CrossRef]
- Lukatskaya, M.R.; Gogotsi, Y. Three-dimensional nanostructures from porous anodic alumina. MRS Commun. 2012, 2, 51–54. [Google Scholar] [CrossRef]
- Vojkuvka, L.; Marsal, L.F.; Ferré-Borrull, J.; Formentin, P.; Pallarés, J. Self-ordered porous alumina membranes with large lattice constant fabricated by hard anodization. Superlattices Microstruct. 2008, 44, 577–582. [Google Scholar] [CrossRef]
- Choi, J.; Wehrspohn, R.B.; Gösele, U. Mechanism of guided self-organization producing quasi-monodomain porous alumina. Electrochim. Acta 2005, 50, 2591–2595. [Google Scholar] [CrossRef]
- Cheng, W.; Steinhart, M.; Gösele, U.; Wehrspohn, R.B. Tree-like alumina nanopores generated in a non-steady-state anodization. J. Mater. Chem. 2007, 17, 3493–3495. [Google Scholar] [CrossRef]
- Li, D.; Jiang, C.; Jiang, J.; Lu, J.G. Self-Assembly of Periodic Serrated Nanostructures. Chem. Mater. 2009, 21, 253–258. [Google Scholar] [CrossRef]
- Li, D.; Zhao, L.; Jiang, C.; Lu, J.G. Formation of anodic aluminum oxide with serrated nanochannels. Nano Lett. 2010, 10, 2766–2771. [Google Scholar] [CrossRef]
- Lee, W.; Scholz, R.; Gösele, U. A continuous process for structurally well-defined Al2O3 nanotubes based on pulse anodization of aluminum. Nano Lett. 2008, 8, 2155–2160. [Google Scholar] [CrossRef]
- Zhang, J.; Day, C.S.; Carroll, D.L. Controlled growth of novel hyper-branched nanostructures in nanoporous alumina membrane. Chem. Commun. Camb. 2009, 7, 6937–6939. [Google Scholar] [CrossRef]
- Losic, D. Preparation of porous anodic alumina with periodically perforated pores. Langmuir 2009, 25, 5426–5431. [Google Scholar] [CrossRef]
- Su, Y.; Fei, G.T.; Zhang, Y.; Li, H.; Yan, P.; Shang, G.L.; de Zhang, L. Anodic alumina photonic crystal heterostructures. J. Opt. Soc. Am. B 2011, 28, 2931–2933. [Google Scholar] [CrossRef]
- Sulka, G.D.; Hnida, K. Distributed Bragg reflector based on porous anodic alumina fabricated by pulse anodization. Nanotechnology 2012, 23. [Google Scholar] [CrossRef]
- Biswas, K.G.; el Matbouly, H.; Rawat, V.; Schroeder, J.L.; Sands, T.D. Self-supporting nanowire arrays templated in sacrificial branched porous anodic alumina for thermoelectric devices. Appl. Phys. Lett. 2009, 95. [Google Scholar] [CrossRef]
- Chung, C.K.; Chang, W.T.; Liao, M.W.; Chang, H.C.; Lee, C.T. Fabrication of enhanced anodic aluminum oxide performance at room temperatures using hybrid pulse anodization with effective cooling. Electrochim. Acta 2011, 56, 6489–6497. [Google Scholar] [CrossRef]
- Noormohammadi, M.; Moradi, M. Structural engineering of nanoporous alumina by direct cooling the barrier layer during the aluminum hard anodization. Mater. Chem. Phys. 2012, 135, 1089–1095. [Google Scholar] [CrossRef]
- Moradi, M.; Noormohammadi, M.; Behzadi, F. Three-dimensional structural engineering of nanoporous alumina by controlled sprinkling of an electrolyte on a porous anodic alumina (PAA) template. J. Phys. D. Appl. Phys. 2011, 44. [Google Scholar] [CrossRef]
- Lee, W.; Kim, J.-C.; Gösele, U. Spontaneous current oscillations during hard anodization of aluminum under potentiostatic conditions. Adv. Funct. Mater. 2010, 20, 21–27. [Google Scholar] [CrossRef]
- Shang, G.L.; Fei, G.T.; Xu, S.H.; Yan, P.; Zhang, D.L. Preparation of the very uniform pore diameter of anodic alumina oxidation by voltage compensation mode. Mater. Lett. 2013, 110, 156–159. [Google Scholar] [CrossRef]
- Urteaga, R.; Acquaroli, L.N.; Koropecki, R.R.; Santos, A.; Alba, M.; Pallarès, J.; Marsal, L.F.; Berli, C.L.A. Optofluidic characterization of nanoporous membranes. Langmuir 2013, 29, 2784–2789. [Google Scholar]
- Nielsch, K.; Choi, J.; Schwirn, K.; Wehrspohn, R.B.; Gösele, U. Self-ordering regimes of porous alumina: The 10 porosity rule. Nano Lett. 2002, 2, 677–680. [Google Scholar] [CrossRef]
- Zaraska, L.; Kurowska, E.; Sulka, G.D.; Jaskuła, M. Porous alumina membranes with branched nanopores as templates for fabrication of Y-shaped nanowire arrays. J. Solid State Electrochem. 2012, 16, 3611–3619. [Google Scholar] [CrossRef]
- Nagaura, T.; Takeuchi, F.; Yamauchi, Y.; Wada, K.; Inoue, S. Fabrication of ordered Ni nanocones using a porous anodic alumina template. Electrochem. Commun. 2008, 10, 681–685. [Google Scholar] [CrossRef]
- Meng, G.; Jung, Y.J.; Cao, A.; Vajtai, R.; Ajayan, P.M. Controlled fabrication of hierarchically branched nanopores, nanotubes, and nanowires. Proc. Natl. Acad. Sci. USA 2005, 102, 7074–7078. [Google Scholar] [CrossRef]
- Li, J.; Papadopoulos, C.; Xu, J. Nanoelectronics: Growing Y-junction carbon nanotubes. Nature 1999, 402, 253–254. [Google Scholar]
- Papadopoulos, C.; Rakitin, A.; Li, J.; Vedeneev, A.; Xu, J. Electronic Transport in Y-Junction Carbon Nanotubes. Phys. Rev. Lett. 2000, 85, 3476–3479. [Google Scholar] [CrossRef]
- Pitzschel, K.; Moreno, J.M.M.; Escrig, J.; Albrecht, O.; Nielsch, K.; Bachmann, J. Controlled introduction of diameter modulations in arrayed magnetic iron oxide nanotubes. ACS Nano 2009, 3, 3463–3468. [Google Scholar] [CrossRef]
- Minguez-Bacho, I.; Rodriguez-López, S.; Vázquez, M.; Hernández-Vélez, M.; Nielsch, K. Electrochemical synthesis and magnetic characterization of periodically modulated Co nanowires. Nanotechnology 2014, 25. [Google Scholar] [CrossRef]
- Yamaguchi, A.; Hotta, K.; Teramae, N. Optical waveguide sensor based on a porous anodic alumina/aluminum multilayer film. Anal. Chem. 2009, 81, 105–111. [Google Scholar] [CrossRef]
- Gitsas, A.; Yameen, B.; Lazzara, T.D.; Steinhart, M.; Duran, H.; Knoll, W. Polycyanurate nanorod arrays for optical-waveguide-based biosensing. Nano Lett. 2010, 10, 2173–2177. [Google Scholar]
- Kim, D.-K.; Kerman, K.; Hiep, H.M.; Saito, M.; Yamamura, S.; Takamura, Y.; Kwon, Y.-S.; Tamiya, E. Label-free optical detection of aptamer-protein interactions using gold-capped oxide nanostructures. Anal. Biochem. 2008, 379, 1–7. [Google Scholar] [CrossRef]
- Shaban, M.; Hady, A.G.A.; Serry, M. A new sensor for heavy metals detection in aqueous media. IEEE Sens. J. 2014, 14, 436–441. [Google Scholar] [CrossRef]
- Fu, C.; Gu, Y.; Wu, Z.; Wang, Y.; Xu, S.; Xu, W. Surface-enhanced Raman scattering (SERS) biosensing based on nanoporous dielectric waveguide resonance. Sens. Actuators B Chem. 2014, 201, 173–176. [Google Scholar]
- Markovics, Á.; Nagy, G.; Kovács, B. Reflection-based sensor for gaseous ammonia. Sens. Actuators B Chem. 2009, 139, 252–257. [Google Scholar] [CrossRef]
- Kumeria, T.; Losic, D. Reflective interferometric gas sensing using nanoporous anodic aluminium oxide (AAO). Phys. Status Solidi Rapid Res. Lett. 2011, 5, 406–408. [Google Scholar] [CrossRef]
- Kumeria, T.; Parkinson, L.; Losic, D. A nanoporous interferometric micro-sensor for biomedical detection of volatile sulphur compounds. Nanoscale Res. Lett. 2011, 6. [Google Scholar] [CrossRef]
- Kumeria, T.; Kurkuri, M.D.; Diener, K.R.; Parkinson, L.; Losic, D. Label-free reflectometric interference microchip biosensor based on nanoporous alumina for detection of circulating tumour cells. Biosens. Bioelectron. 2012, 35, 167–173. [Google Scholar]
- Toccafondi, C.; Thorat, S.; la Rocca, R.; Scarpellini, A.; Salerno, M.; Dante, S.; Das, G. Multifunctional substrates of thin porous alumina for cell biosensors. J. Mater. Sci. Mater. Med. 2014. [Google Scholar] [CrossRef]
- Kumeria, T.; Santos, A.; Losic, D. Ultrasensitive nanoporous interferometric sensor for label-free detection of gold(III) ions. ACS Appl. Mater. Interfaces 2013, 5, 11783–11790. [Google Scholar] [CrossRef]
- Santos, A.; Kumeria, T.; Losic, D. Optically optimized photoluminescent and interferometric biosensors based on nanoporous anodic alumina: A comparison. Anal. Chem. 2013, 85, 7904–7911. [Google Scholar] [CrossRef]
- Kumeria, T.; Rahman, M.M.; Santos, A.; Ferré-Borrull, J.; Marsal, L.F.; Losic, D. Structural and optical nanoengineering of nanoporous anodic alumina rugate filters for real-time and label-free biosensing applications. Anal. Chem. 2014, 86, 1837–1844. [Google Scholar] [CrossRef]
- Trivinho-Strixino, F.; Guerreiro, H.A.; Gomes, C.S.; Pereira, E.C.; Guimarães, F.E.G. Active waveguide effects from porous anodic alumina: An optical sensor proposition. Appl. Phys. Lett. 2010, 97. [Google Scholar] [CrossRef]
- Santos, A.; Alba, M.; Rahman, M.M.; Formentín, P.; Ferré-Borrull, J.; Pallarès, J.; Marsal, L.F. Structural tuning of photoluminescence in nanoporous anodic alumina by hard anodization in oxalic and malonic acids. Nanoscale Res. Lett. 2012, 7. [Google Scholar] [CrossRef]
- Nguyen, B.T.T.; Koh, G.; Lim, H.S.; Chua, A.J.S.; Ng, M.M.L.; Toh, C.-S. Membrane-based electrochemical nanobiosensor for the detection of virus. Anal. Chem. 2009, 81, 7226–7234. [Google Scholar]
- Heilmann, A.; Teuscher, N.; Kiesow, A.; Janasek, D.; Spohn, U. Nanoporous aluminum oxide as a novel support material for enzyme biosensors. J. Nanosci. Nanotechnol. 2003, 3, 375–379. [Google Scholar]
- Santos, A.; Balderrama, V.S.; Alba, M.; Formentín, P.; Ferré-Borrull, J.; Pallarès, J.; Marsal, L.F. Nanoporous anodic alumina barcodes: Toward smart optical biosensors. Adv. Mater. 2012, 24, 1050–1054. [Google Scholar]
- Ramiro-Manzano, F.; Fenollosa, R.; Xifré-Pérez, E.; Garín, M.; Meseguer, F. Porous silicon microcavities based photonic barcodes. Adv. Mater. 2011, 23, 3022–3025. [Google Scholar]
- Meade, S.O.; Chen, M.Y.; Sailor, M.J.; Miskelly, G.M. Multiplexed DNA detection using spectrally encoded porous SiO2 photonic crystal particles. Anal. Chem. 2009, 81, 2618–2625. [Google Scholar]
- Orosco, M.M.; Pacholski, C.; Sailor, M.J. Real-time monitoring of enzyme activity in a mesoporous silicon double layer. Nat. Nanotechnol. 2009, 4, 255–258. [Google Scholar]
- Kumeria, T.; Losic, D. Controlling interferometric properties of nanoporous anodic aluminium oxide. Nanoscale Res. Lett. 2012, 7. [Google Scholar] [CrossRef]
- Macias, G.; Hernández-Eguía, L.P.; Ferré-Borrull, J.; Pallares, J.; Marsal, L.F. Gold-coated ordered nanoporous anodic alumina bilayers for future label-free interferometric biosensors. ACS Appl. Mater. Interfaces 2013, 5, 8093–8098. [Google Scholar]
Appendix
| Changing parameter | Sample | First step | Second step | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Acid | V (V) | C (M) | T (°C) | Acid | V (V20) | C (M) | T (°C) | ||
| Anodization Voltage | S1 | H2C2O4 | 120 | 0.3 | 0 | H2C2O4 | 40 | 0.3 | 5 |
| S2 | H2C2O4 | 40 | 0.3 | 5 | H2C2O4 | 20 | 0.3 | 5 | |
| S3 | H3PO4 | 170 | 0.3 | 5 | H3PO4 | 85 | 0.3 | 5 | |
| Kind of Acid | S4 | H3PO4 | 170 | 0.3 | 5 | H2C2O4 | 120 | 0.3 | 0 |
| S5 | H3PO4 | 170 | 0.3 | 5 | H2C2O4 | 40 | 0.3 | 5 | |
| S6 | H3PO4 | 170 | 0.3 | 5 | H3PO4 | 18 | 0.3 | 5 | |
| S7 | H2C2O4 | 120 | 0.3 | 5 | H2SO4 | 18 | 0.3 | 5 | |
| S8 | H2C2O4 | 40 | 0.3 | 5 | H2SO4 | 18 | 0.3 | 5 | |
| Concentration | S9 | H3PO4 | 170 | 0.3 | 5 | H3PO4 | 170 | 0.2 | 5 |
| S10 | H2C2O4 | 120 | 0.3 | 0 | H2C2O4 | 120 | 0.1 | 0 | |
| Sample | dinter-concavity (nm) | φconcavity (nm) | dinterpore (nm) | φpore (nm) | ρpore/ρconcavity Pores per concavity |
|---|---|---|---|---|---|
| S1 | 249.3 ± 32.9 | 219.0 ± 18.7 | 105.0 ± 9.2 | 45.9 ± 10.4 | 10.1 ± 1.7 |
| S2 | 104.0 ± 9.7 | 64.5 ± 7.8 | 74.4 ± 8.7 | 22.1 ± 3.6 | 1.9 ± 0.8 |
| S3 | 393.1 ± 32.2 | 326.2 ± 31.5 | 126.2 ± 16.0 | 108.0 ± 23.3 | 3.6 ± 0.7 |
| S4 | 405.6 ± 46.7 | 303.5 ± 37.0 | 282.8 ± 28.1 | 64.0 ± 16.4 | 6.3 ± 1.2 |
| S5 | 413.6 ± 37.1 | 367.6 ± 30.0 | 97.6 ± 9.7 | 36.5 ± 7.0 | 15.3 ± 3.0 |
| S6 | 397.0 ± 45.4 | 369.2 ± 53.3 | 48.1 ± 8.6 | 23.1 ± 3.7 | 61.4 ± 6.5 |
| S7 | 292.0 ± 35.2 | 264.4 ± 41.0 | 47.5 ± 7.3 | 23.4 ± 4.5 | 38.4 ± 3.5 |
| S8 | 104.4 ± 10.5 | 83.2 ± 11.5 | 55.5 ± 9.3 | 21.0 ± 3.0 | 5.2 ± 0.8 |
| Sample | Anodization voltage (V) | Anodization time (min) | Total charge (C) | Temperature (ºC) | Pore widening time (min) |
|---|---|---|---|---|---|
| S1 | 20 | 10 | 0.8 | 5 | 0 |
| S2 | 15 | 1.1 | 6 | 3 | |
| S3 | 20 | 1.6 | 6 | 6 | |
| S4 | 25 | 1.8 | 5 | 9 | |
| S5 | 30 | 10 | 1.2 | 6 | 0 |
| S6 | 15 | 1.9 | 6 | 3 | |
| S7 | 20 | 2.8 | 7 | 6 | |
| S8 | 25 | 3.5 | 5 | 9 | |
| S9 | 40 | 10 | 2.6 | 5 | 0 |
| S10 | 15 | 4.2 | 5 | 3 | |
| S11 | 20 | 5.2 | 6 | 6 | |
| S12 | 25 | 8.4 | 5 | 9 | |
| S13 | 50 | 10 | 4.9 | 5 | 0 |
| S14 | 15 | 7.3 | 5 | 3 | |
| S15 | 20 | 9.6 | 5 | 6 | |
| S16 | 25 | 11.2 | 5 | 9 |

© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ferré-Borrull, J.; Pallarès, J.; Macías, G.; Marsal, L.F. Nanostructural Engineering of Nanoporous Anodic Alumina for Biosensing Applications. Materials 2014, 7, 5225-5253. https://doi.org/10.3390/ma7075225
Ferré-Borrull J, Pallarès J, Macías G, Marsal LF. Nanostructural Engineering of Nanoporous Anodic Alumina for Biosensing Applications. Materials. 2014; 7(7):5225-5253. https://doi.org/10.3390/ma7075225
Chicago/Turabian StyleFerré-Borrull, Josep, Josep Pallarès, Gerard Macías, and Lluis F. Marsal. 2014. "Nanostructural Engineering of Nanoporous Anodic Alumina for Biosensing Applications" Materials 7, no. 7: 5225-5253. https://doi.org/10.3390/ma7075225
APA StyleFerré-Borrull, J., Pallarès, J., Macías, G., & Marsal, L. F. (2014). Nanostructural Engineering of Nanoporous Anodic Alumina for Biosensing Applications. Materials, 7(7), 5225-5253. https://doi.org/10.3390/ma7075225







