Direct Electrochemistry and Electrocatalysis of Horseradish Peroxidase Immobilized in a DNA/Chitosan-Fe3O4 Magnetic Nanoparticle Bio-Complex Film
Abstract
: A DNA/chitosan-Fe3O4 magnetic nanoparticle bio-complex film was constructed for the immobilization of horseradish peroxidase (HRP) on a glassy carbon electrode. HRP was simply mixed with DNA, chitosan and Fe3O4 nanoparticles, and then applied to the electrode surface to form an enzyme-incorporated polyion complex film. Scanning electron microscopy (SEM) was used to study the surface features of DNA/chitosan/Fe3O4/HRP layer. The results of electrochemical impedance spectroscopy (EIS) show that Fe3O4 and enzyme were successfully immobilized on the electrode surface by the DNA/chitosan bio-polyion complex membrane. Direct electron transfer (DET) and bioelectrocatalysis of HRP in the DNA/chitosan/Fe3O4 film were investigated by cyclic voltammetry (CV) and constant potential amperometry. The HRP-immobilized electrode was found to undergo DET and exhibited a fast electron transfer rate constant of 3.7 s−1. The CV results showed that the modified electrode gave rise to well-defined peaks in phosphate buffer, corresponding to the electrochemical redox reaction between HRP(Fe(III)) and HRP(Fe(II)). The obtained electrode also displayed an electrocatalytic reduction behavior towards H2O2. The resulting DNA/chitosan/Fe3O4/HRP/glassy carbon electrode (GCE) shows a high sensitivity (20.8 A·cm−2·M−1) toward H2O2. A linear response to H2O2 measurement was obtained over the range from 2 μM to 100 μM (R2 = 0.99) and an amperometric detection limit of 1 μM (S/N = 3). The apparent Michaelis-Menten constant of HRP immobilized on the electrode was 0.28 mM. Furthermore, the electrode exhibits both good operational stability and storage stability.1. Introduction
The direct electron transfer (DET) between electrodes and redox proteins, particularly enzymes, has stimulated increasing interest because of its significance in both theoretical and practical applications in electrochemistry, such as fabricating biosensors, enzymatic bioreactors, and biomedical devices [1–3]. Nevertheless, DET between the enzyme and a conventional electrode is usually prohibited because enzymatic redox centers are deeply embedded in the structure of the enzyme. Therefore, new materials, such as carbon nanotube [4–6], quantum dot [7], nanoparticles [8], graphene [4], hybrid organic-inorganic film [9], room temperature ionic liquid matrix [10] and mesoporous matrix [11] are required to establish an electrical connection between these redox centers and the electrode for fabricating a third-generation enzyme biosensor or a mediatorless enzymatic biofuel cell.
In recent years, there has been an increasing trend in the design and development of magnetic nanoparticles for bioanalytical applications [12,13]. Magnetic nanoparticles have been considered as an interesting material for the immobilization of desired biomolecules because of some superior properties: electro-conductivity, bio-compatibility and ease of synthesis [14]. Enzyme-immobilized magnetic nanoparticles could potentially lead to unique properties such as large surface area, high bioactivity, and excellent stability [15,16]. However, pure iron oxide nanoparticles may not be very useful in biomedical and technological applications because they are very likely to aggregate, and they have limited functional groups for selective binding [17]. As a result, a number of biomaterial-functionalized magnetic nanoparticles have been used in bioelectronic applications [12–17], such as poly(ethyleneimine) layer-functionalized magnetic nanoparticles [18], Au-polydopamine-Fe3O4 magnetic nanoparticles [19], and magnetic core-shell Fe3O4@Al2O3 nanoparticles [20], which were used for capture of heme proteins for direct electrochemistry. Unfortunately, these methods may involve complicated electrode modification procedure or costly modification regents.
DNA/polycation based biocompatible films are formed by natural DNA molecules (negatively charged polyanions) and natural polycations molecules based on the electrostatic force of attraction. As novel electrochemical recognition layers, DNA/polycation based biocompatible films possess a number of unique properties [21–23]: (1) biocompatible microenvironment around the enzyme; (2) a host matrix of electrochemically active species (e.g., redox active intercalators) and metal ions which specifically binds to double-stranded DNA; (3) unique electron transfer property improving electron transfer characteristics between redox active species and the electrode surface; and (4) simplicity in procedure. In our previous reports, DNA/poly(allylamine) films have been successfully used as the support matrixes for co-immobilization of electron mediator-methylene blue and HRP [22–24], and immobilization of electrocatalytic element-copper ion [23,25] to fabricate novel amperomeric biosensors. More recently, glucose oxidase (GOD) was effectively immobilized on a DNA/chitsoan films modified glassy carbon electrode (GCE), and the direct electrochemistry of GOD and biosensing for glucose were performed successfully [26].
The DET of immobilized heme proteins such as horseradish peroxidase (HRP) [3], cytochrome C [27], and hemoglobin [28] is based on the Fe(III)/Fe(II) conversion in the active heme center of the proteins. Among them, HRP has been widely used for the fabrication of amperometric biosensors based on its direct electrochemistry to detect H2O2 due to its high purity, sensitivity, low cost and availability [29]. There are many ways to achieve the DET of immobilized HRP, for instance, using sol-gel matrices [30], biopolymers [31], and nanomaterials [32].
In this paper, a new type of DNA/chitosan/Fe3O4 magnetic nanoparticle bio-complex film was constructed for the immobilization of HRP. DNA/chitsoan film and Fe3O4 nanoparticles with the excellent biocompatibility and good conductivity were used to maintain the native structure of HRP and to facilitate the direct electrochemistry of HRP in the biofilm. Although a Fe3O4/chitosan/HRP-modified glassy carbon electrode has been reported for amperometric detection of H2O2 [33], methylene blue in solution was needed as an electron transfer mediator to transfer the electron between the HRP and electrode in the system. Here, the direct electron transfer and electrocatalysis of HRP based on the DNA/chitosan/Fe3O4 film was studied.
2. Results and Discussion
2.1. Morphologies of DNA/Chitosan/Fe3O4/HRP Film Surface
The surface morphologies of Fe3O4, DNA/chitosan, DNA/chitosan/Fe3O4 and DNA/chitosan/Fe3O4/HRP were examined by scanning electron microscopy (SEM). The micrograph of immobilized Fe3O4 shows aggregated particles with diameter of approximately 0.2 μm (Figure 1a). The micrograph of a DNA/chitosan film appears to be a smooth surface (Figure 1b), and that of a DNA/chitosan/Fe3O4 film shows uniformly distributed particles of approximately 0.4 μm diameter (Figure 1c). In contrast, following HRP immobilization, DNA/chitosan/Fe3O4/HRP film (Figure 1d) shows a rough surface with randomly distributed small spots, indicating that HRP was entrapped in the DNA/chitosan/Fe3O4 films. This is further supported by an increased electron transfer resistance of DNA/chitosan/Fe3O4/HRP-modified electrode reported in Section 2.2.
(a)
(b)
(c)
(d)
2.2. Electrochemical Impedance Spectroscopy (EIS) of DNA/Chitosan/Fe3O4/HRP Film
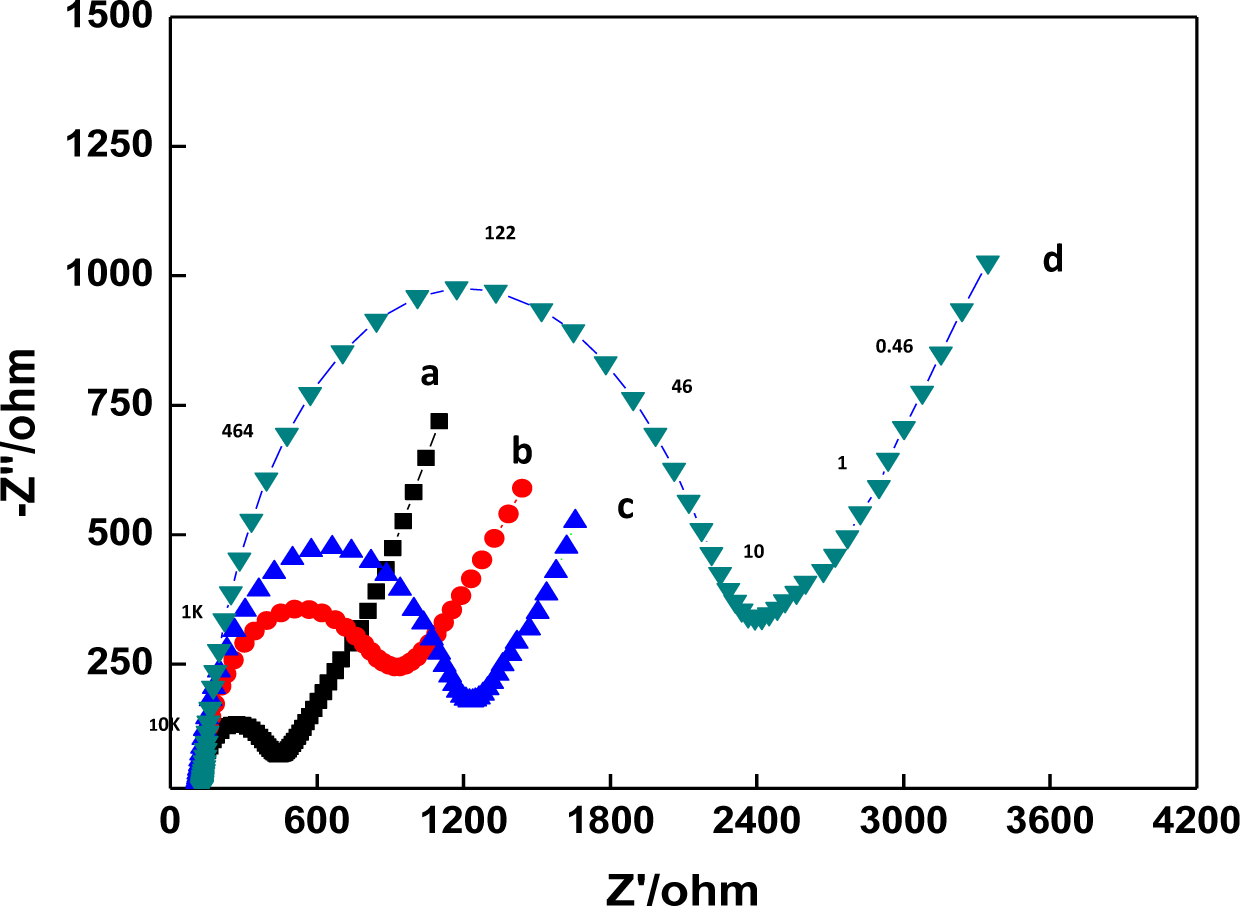
EIS can provide useful information on the impedance changes of the electrode surface during the fabrication process. In EIS, the diameter of a semicircle in the high frequency region corresponds to the electron transfer resistance, Ret. This resistance controls the electron transfer kinetics of redox probe at the electrode interface. Figure 2 displays the Nyquist plots of the EIS of GCE (a), a DNA/chitosan/GCE (b), a DNA/chitosan/Fe3O4/GCE (c) and a DNA/chitosan/Fe3O4/HRP/GCE (d) in 0.1 M KCl containing 5.0 mM K3Fe(CN)6/K4Fe(CN)6. The bare GCE reveals a very small semicircle domain (curve a, Ret = 446 Ω). After DNA/chitosan film was immobilized on the GCE, an increase of Ret to 1237 Ω was observed (curve b), which indicated that the DNA/chitostan polyion complex film hindered the electron transfer of K3Fe(CN)6/K4Fe(CN)6. For DNA/chitosan/Fe3O4/GCE, the value of Ret was found to be 940 Ω, implying that the incorporation of Fe3O4 facilitated electron transfer (curve c). There was further immobilization of enzymes: the Ret of DNA/chitosan/Fe3O4/HRP/GCE increased to 2392 Ω, which was caused by the hindrance of the macromolecular structure of HRP to the electron transfer. The above results clearly confirm that HRP was immobilized successfully onto the electrode.
2.3. Direct Electrochemistry of HRP at DNA/Chitosan/Fe3O4 Film
Figure 3 shows the cyclic voltammograms (CVs) of DNA/chitosan/GCE, DNA/chitosan/Fe3O4/GCE, DNA/chitosan/HRP/GCE and DNA/chitosan/Fe3O4/HRP/GCE in N2-saturated phosphate buffer. DNA/chitosan/GCE (curve a) and DNA/chitosan/Fe3O4/GCE (curve b) did not show any redox wave in the potential range studied. In contrast, a pair of well-defined quasi-reversible redox peaks was obtained at the −0.35 V and −0.30 V by DNA/chitosan/Fe3O4/HRP/GCE (Figure 3c). The formal potential (E0′) was estimated to be ~ −0.32 V (versus Ag/ACl in saturated KCl) by the average of the cathodic and anodic peak potentials. Therefore, it can be concluded that the redox waves should be ascribed only to HRP, which is characteristic of quasi-reversible DET process of HRP[Fe(III)] and HRP[Fe(II)] in the HRP previously reported in various films [30–32]. Thus, DET of HRP in the DNA/chitosan/Fe3O4 film has been achieved successfully. Similar to the case of DNA/chitosan/Fe3O4/HRP/GCE (curve c), the DNA/chitosan/HRP modified electrode (curve d) showed a pair of quasi-reversible redox peaks, but the oxidation and reduction peak currents were 87% and 53%, respectively, smaller than the corresponding peak currents obtained at DNA/chitosan/Fe3O4/GCE. This result clearly indicated that the electron transfer of HRP on the DNA/chitosan electrode was amplified by incorporation of Fe3O4 magnetic nanoparticles.
The cyclic voltammograms of DNA/chitosan/Fe3O4/HRP/GCE at various scan rates were investigated (Figure 4). The linear relationship between peak current and scan rate indicated that the redox process of HRP in the film was a surface-confined process. The electron transfer rate constant (ks) has been estimated from the peak potential separation value using the model of Laviron [34]. Taking a charge transfer coefficient α of 0.5, the electron transfer rate constant of HRP at the DNA/chitosan/Fe3O4/HRP/GCE was 3.7 s−1. The electron transfer rate is higher than the values reported for HRP immobilized in a polystyrene and multiwalled carbon nanotube composite film (1.15 s−1) [32], a colloidal gold modified screen-printed electrode (0.75 s−1) [35], and a dipalmitoyl phosphatidic acid film (1.13 s−1) [36]. Since DNA/chitsoan film and Fe3O4 nanoparticles with the excellent biocompatibility and good conductivity appeared to be capable of maintaining the native structure of HRP, it suggested that DNA/chitosan/Fe3O4 film was an excellent promoter for the direct electron transfer between HRP and GCE.
Based on the equation, Q = nFAΓ, where n denotes the charge of the redox reaction, Q denotes the quantity of electricity, F denotes the Faraday constant and A denotes the area of the electrode surface (in this case, A = 0.02 cm2), the surface coverage (ΓHRP) of HRP was estimated to be 3.5 × 10−10 mol·cm−2 at the DNA/chitosan/Fe3O4/HRP/GCE. The relative amount of electroactive HRP was 36.1% of the total amount of HRP deposited on the electrode surface. The results demonstrated that the composite film is efficiency for HRP immobilization.
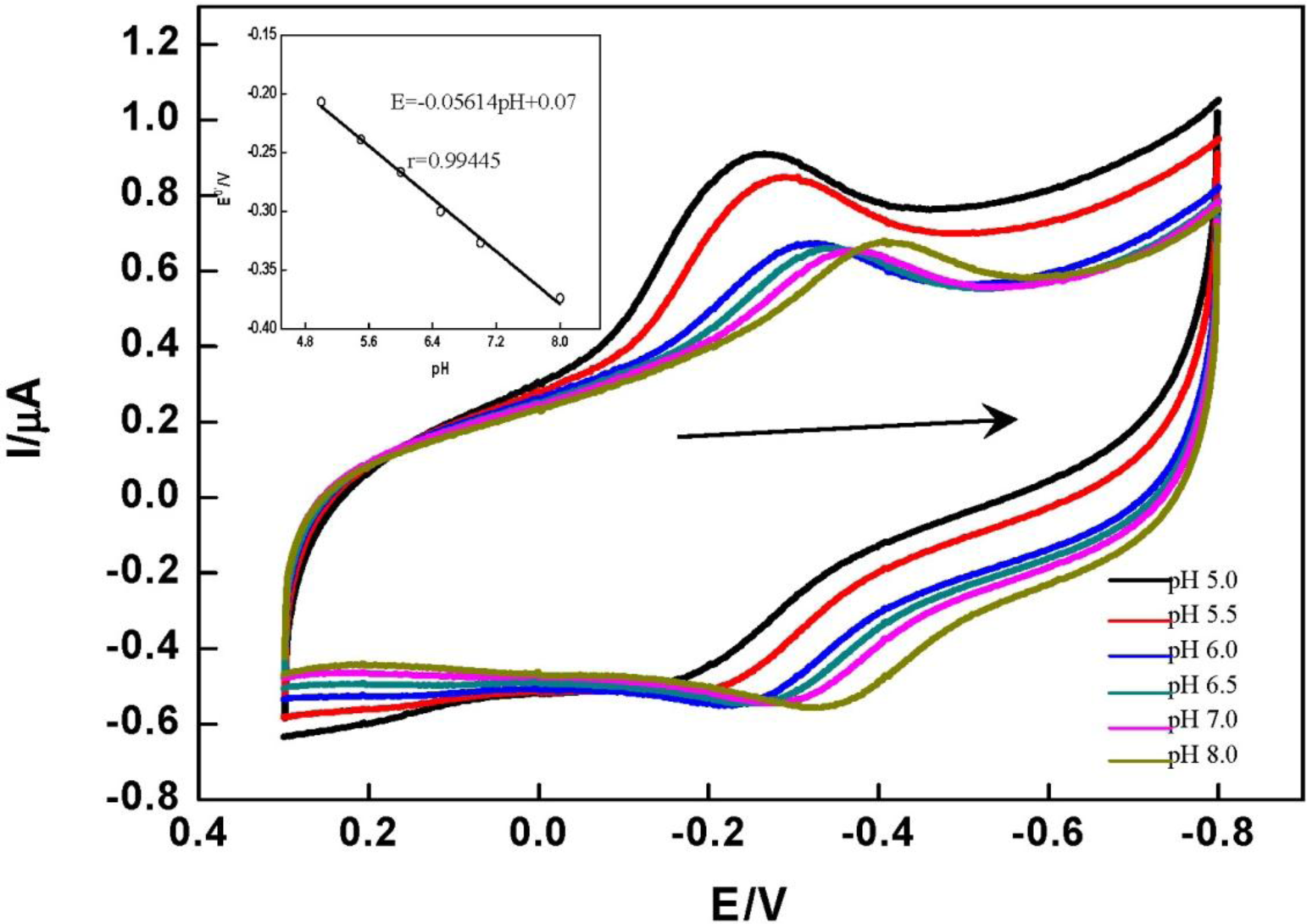
The DET of HRP immobilized on the DNA/chitosan/Fe3O4/HRP/GCE showed a strong dependence on solution pH. Figure 5 shows the CVs of the DNA/chitosan/Fe3O4/HRP/GCE in phosphate buffer at different pH values. CVs with stable and well defined peaks were observed in the pH range 5.0–8.0, but increasing pH caused a negative shift of both cathodic and anodic peak potentials. This is attributed to the involvement of proton transfer in the HRP[Fe(III)]/HRP[Fe(II)] redox couple. The E0′ value of HRP varied linearly in the range of pH from 5.0 to 8.0, with a slope of 56.14 mV pH−1 (inset graph in Figure 5). This value is very close to the theoretical value for the transfer of one proton and one electron in a reversible reduction (58 mV pH−1 at 25 °C) [37].
2.4. Electrocatalysis of DNA/Chitosan/Fe3O4/HRP/GCE
By using hydrogen peroxide as a probe, the electrocatalytic properties of the HRP in DNA/chitosan/Fe3O4/GCE were studied. Figure 6 shows the bioelectrocatalytic activity of the HRP in DNA/chitosan/Fe3O4/GCE toward the reduction of H2O2 at a scan rate of 0.1 V·s−1. A pair of quasi-reversible CV peaks appeared in the absence of H2O2 (curve a). Upon the addition of H2O2 to the pH 7.0 phosphate buffer, the reduction peak current of the immobilized HRP increased dramatically and the oxidation peak current decreased concomitantly (curve b, c and d). The reduction peak current increases with increasing concentration of H2O2 (curve b, c and d), indicating that an electrocatalytic reduction of H2O2 took place at electrode and the HRP entrapped in the DNA/chitosan/Fe3O4/HRP film maintained its bio-electrocatalytic activity. Similar electrocatalytic behavior has been reported at Nafion/HRP/graphene/GCE [3] and HRP-incorporated in dipalmitoyl phosphatidic acid film-modified pyrolytic graphite electrode [36].
The electrocatalytic mechanism of HRP immobilized in the DNA/chitosan/Fe3O4/HRP film to H2O2 reduction can be represented as follows [38]:
here, HRP contains heme as an active site; in the resting state, the heme-iron oxidation state is HRP[Fe(III)]. The basic catalytic mechanism of HRP occurs through the rapid reaction with H2O2 to give a two-equivalent oxidized form, called Compound I as shown in Equation (1); the rapid reaction of compound I with the substrate then regenerates the HRP[Fe(III)] ground state form via an intermediate called Compound II, as shown in Equations (2) and (3) [39].
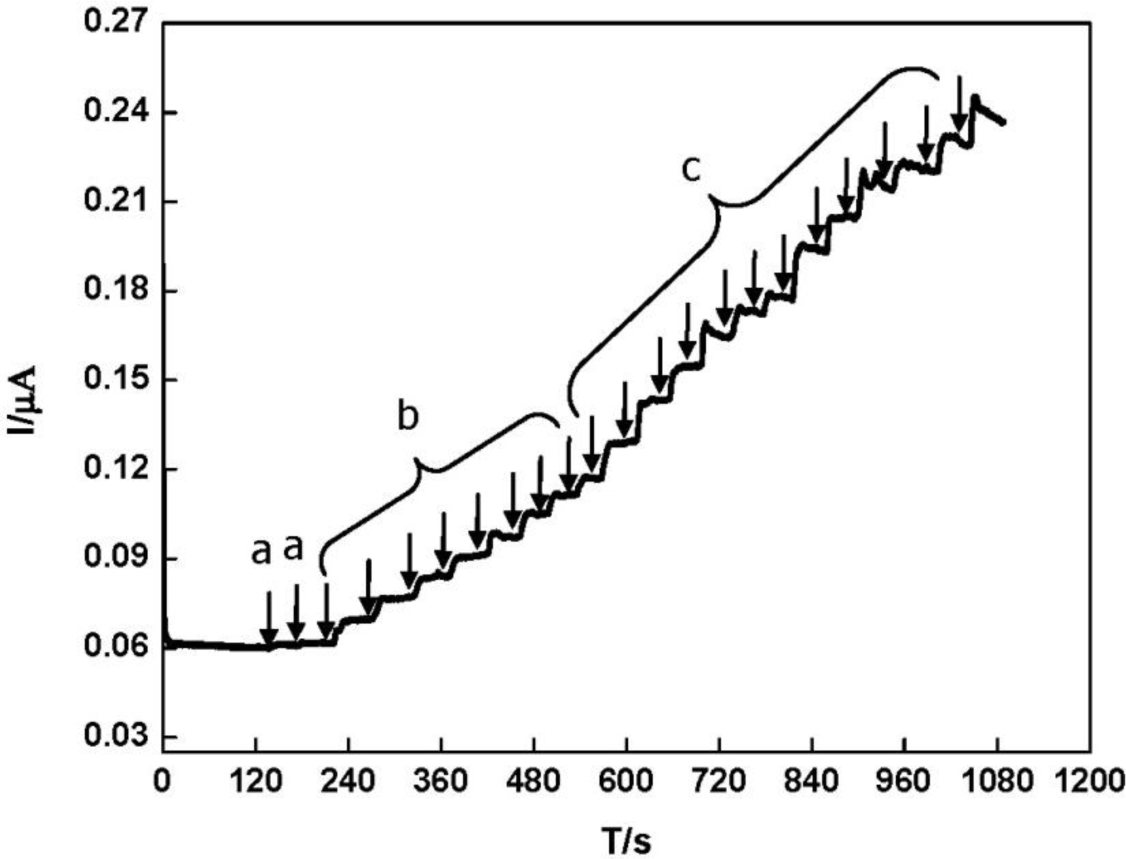
The electrocatalytic property of the DNA/chitosan/Fe3O4/HRP/GCE was also studied by the constant potential amperometry. To reduce the background current, we selected −0.25 V as an applied potential. Figure 7 illustrates a typical current-time curve of the DNA/chitosan/Fe3O4/HRP/GCE at −0.25 V with successive addition of H2O2. When H2O2 was added to the stirred phosphate buffer, the reduction current increased rapidly and 95% of the steady-state current was obtained within 6 s. The H2O2 reduction current at the DNA/chitosan/Fe3O4/HRP/GCE changes linearly from 2 μM to 100 μM with a correlation coefficient of 0.99 (n = 3). Using the linear calibration plot, we have estimated a detection limit of 1 μM, based on S/N = 3.
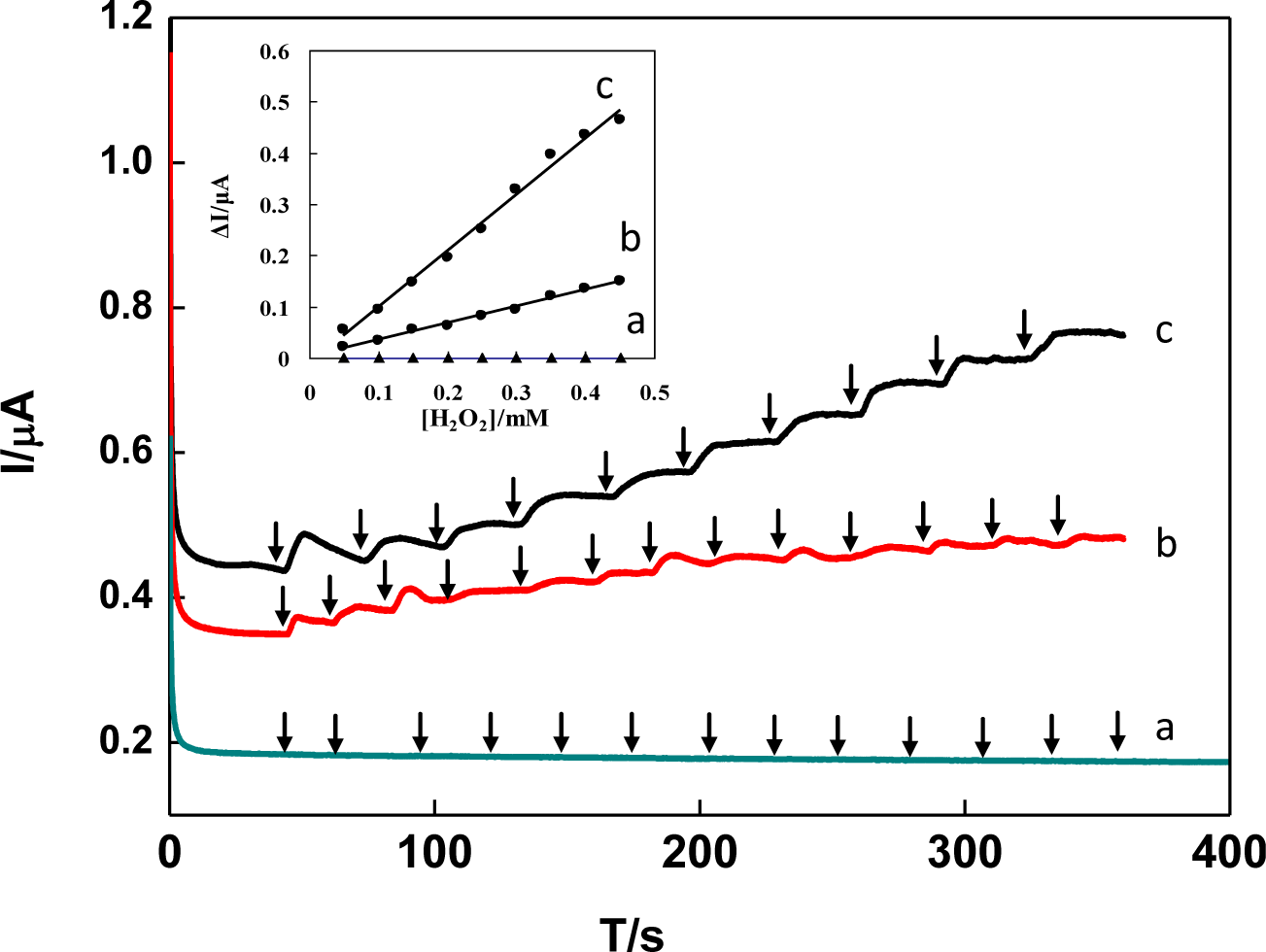
Figure 8 shows the comparison of amperometric current-time curves for successive addition of 0.1 mM H2O2 obtained by DNA/chitosan/GCE (a), DNA/chitosan/HRP/GCE (b) and DNA/chitosan/Fe3O4/HRP/GCE (c) at applied potential of −0.25 V (versus Ag/AgCl). The observed catalytic responses of electrodes were in the following order: DNA/chitosan/Fe3O4/HRP/GCE > DNA/chitosan/HRP/GCE > DNA/chitosan/GCE. No response was observed on the DNA/chitosan/GCE without HRP (Figure 8a), indicating that direct catalyzed reduction of H2O2 was negligible. The amperometric responses of DNA/chitosan/HRP/GCE (Figure 8b) were observed for 0.1 mM H2O2, which was contributed from the direct electron transfer between HRP embedded in the DNA/chitosan film and GCE. When Fe3O4 was present in the DNA/chitosan/HRP/GCE, the sensitivity of H2O2 responses obviously increased from 8.86 A·cm−2·M−1 to 20.8 A·cm−2·M−1. These results indicated that Fe3O4 nanoparticles in the film play an important role to enhance the DET and electrocatalytic properties of HRP molecules.
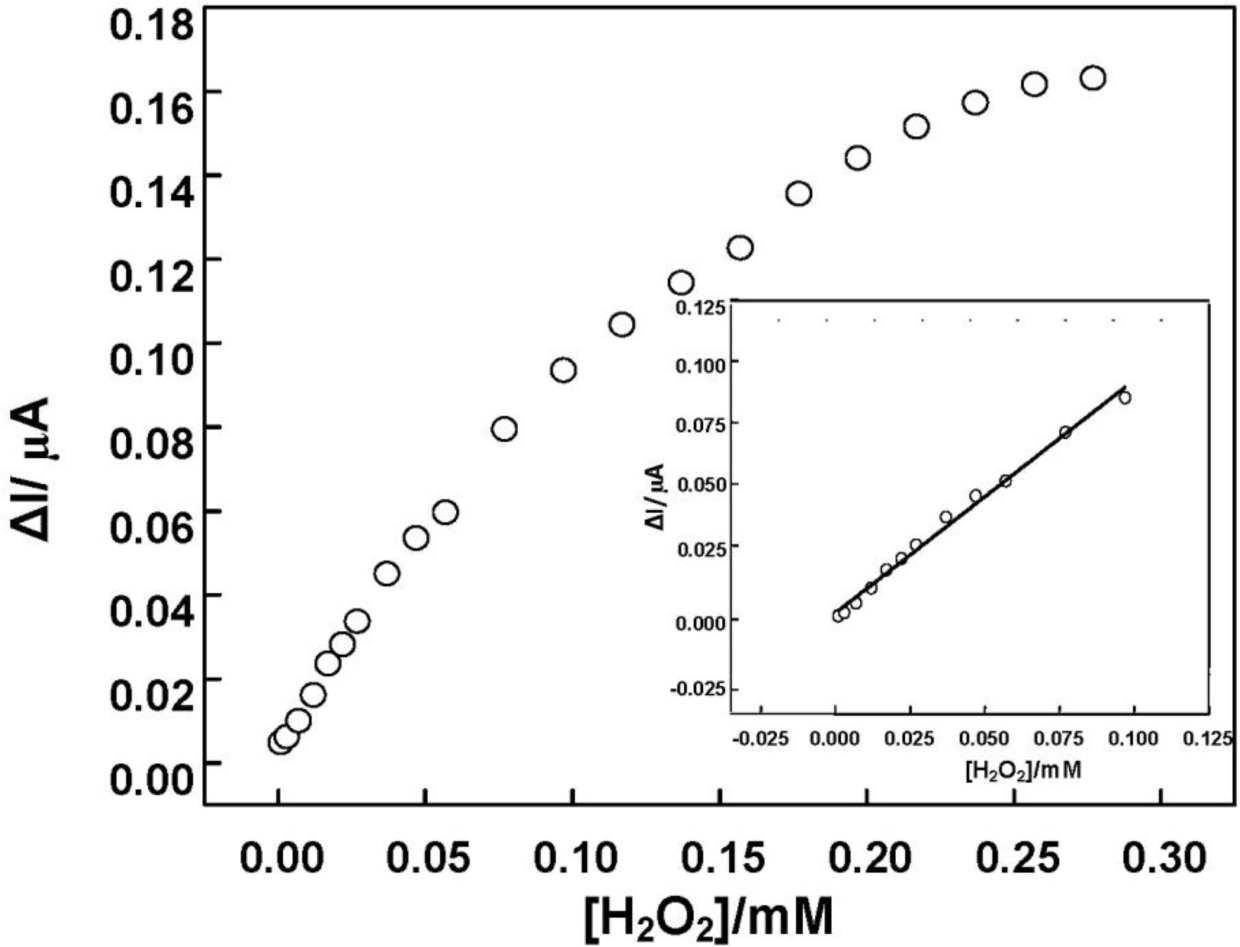
Figure 9 shows the calibration curve of amperometric response of DNA/chitosan/Fe3O4/HRP/GCE versus H2O2 concentration. The sensitivity of the DNA/chitosan/Fe3O4/HRP/GCE was calculated to be 20.8 A·cm−2·M−1. The relationship between the catalytic current and the concentration of H2O2 shows a Michaelis-Menten kinetic mechanism. The apparent Michaelis-Menten constant (Km) could be calculated from the electrochemical Lineweaver-Burk equation: 1/Iss = (Km/Iss) × (1/C) + 1/Imax, where Iss, Imax and C represent the steady current, maximum current and H2O2 concentration, respectively. According to the intercept and slope of above regression equation, Km was estimated to be 0.28 mM. The value is much smaller than 0.66 mM for HRP immobilized in a polystyrene and multiwalled carbon nanotube composite film [32], and 0.818 mM for HRP immobilized in γ-Al2O3 nanoparticles/chitosan film [40]. The small apparent Michaelis-Menten constant shows a high affinity to H2O2 and good bioactivity of DNA/chitosan/Fe3O4/HRP/GCE toward H2O2 reduction.
The long-term stability of electrode was investigated by examining its current response after storage in a refrigerator at 4 °C. The electrode exhibited no obvious decrease in current response in the first week and maintained about 95% of its initial value after three weeks. The relative standard deviation (R.S.D.) of the electrode response to 10 mM H2O2 for 5 successive measurements was 2.1%, indicating an acceptable electrode reproducibility. The selectivity of electrode was performed by comparing the amperometric response of 0.2 mM H2O2 before and after adding 2 mM of several known interfering species, respectively, in 0.1 M phosphate buffer (pH 7.0). The steady-state amperometric current ratio obtained in the presence to that in the absence of each of these interfering species is 0.98 for glucose, 0.93 for ascorbic acid and 0.96 for uric acid. Notably, there was minimal interference from glucose, ascorbic acid and uric acid in the determination of H2O2. The good selectivity of this electrode is largely attributed to the low working potential (−0.25 V).
3. Experimental Section
3.1. Reagents and Chemicals
Double-stranded DNA (dsDNA, from Calf Thymus DNA; mol wt 8.0–15 kb, 41.9% GC ), HRP (E.C. 1.11.1.7, ≥250 U·mg−1, from horseradish), d-(+)-glucose, ascorbic acid and uric acid were all obtained from Sigma-Aldrich (Shanghai) Trading Co., Ltd (Shanghai, China). Chitosan was purchased from Sinopharm Chemcial Reagent Co., (Shanghai, China). Fe3O4 magnetic nanoparticles (~20 nm, 99.9%) was purchased from Beijing Dk Nano technology Co., Ltd. (Beijing, China). All chemicals were of analytical grade and used without further purification. The supporting electrolyte phosphate buffer was prepared by mixing the stock solutions of KH2PO4 and K2HPO4. Stock solution of 1 mg·mL−1 HRP was prepared by directly dissolving HRP in pH 7.0 phosphate buffer and stored at 4 °C. The Fe3O4 magnetic nanoparticles were dispersed in water by ultra-sonication for about 30 min and stored at 4 °C for use. The supporting electrolyte solution used in electrochemical impedance spectroscopy (EIS) measurements was 0.1 M KCl containing 5 mM [Fe(CN)6]3−/4− (1:1). Double-distilled water was used throughout the experiments.
3.2. Preparation of DNA/Chitosan/Fe3O4/HRP/GCE
A GCE [3 mm diameter, Bioanalytical Systems (BAS), West Lafayette, IN, USA] was polished with 0.05 μm alumina slurry, rinsed with water, sonicated in water for 2 min, and dried. The DNA/chitosan/Fe3O4/HRP/GCE was prepared as follows: aqueous solutions of chitosan (0.5 mL, 1 mg/mL) and Fe3O4 (0.5 mL, 1 mg/mL) were mixed for 15 min. Then, 10 μL of dsDNA (1 mg/mL), 20 μL of the mixture of chitosan and Fe3O4, and 10 μL of HRP (1 mg/mL) aqueous solution were successively placed on the GCE surface to form a polyion complex layer. The electrode was allowed to dry for 24 h under a 1000 mL beaker at room temperature. After being rinsed with distilled water, the resulting DNA/chitosan/Fe3O4/HRP/GCE was stored in the refrigerator at 4 °C when not in use. Before electrochemical measurements, the electrodes were immersed in phosphate buffer for 15 min.
3.3. Electrochemical Measurements
All electrochemical experiments were performed with a conventional three-electrode system using a CHI 660D electrochemical workstation (Shanghai CH Instruments, Shanghai, China). An Ag/AgCl (sat. KCl) electrode, a platinum wire electrode (1 mm diameter) and a GCE were used as the reference electrode, the counter electrode, and the working electrode, respectively. CV and constant potential amperometry were carried out by using a deoxygenated (N2-saturated) 0.1 M phosphate buffer (10 mL). Deoxygenated electrolyte solutions were prepared by bubbling high purity grade nitrogen gas through the solution at least 20 min prior to the electrochemical measurements. EIS measurements were performed with 0.1 M KCl solution containing 5 mM [Fe(CN)6]3−/4− under applied potential of 170 mV with the frequency range from 0.01 to 100,000 Hz with the amplitude of 8 mV. All measurements were done at room temperature.
3.4. Scanning Electron Microscopic Analysis
The SEM analysis of DNA/chitosan/Fe3O4/HRP polyion complex membrane was performed using a JEOL-6480LV microscope (JEOL Ltd., Tokyo, Japan) operating at 15.0 kV. Prior to SEM analysis, ca. 10 nm of carbon film was sputtered on the samples.
4. Conclusions
As one kind of novel nanomaterial, magnetic nanomaterials not only have the common nano-characters, but also have some special properties. Acting as the carrier of biological molecules, DNA/chitosan/Fe3O4 film can provide a microenvironment which is similar to biological molecules of internal environment. In this paper, a simple method for constructing HRP modified electrode on a DNA/chitosan/Fe3O4 bio-magnetic polyion complex membrane to realize direct electron transfer and electrocatalysis was proposed. The results show that HRP was successfully immobilized on the electrode surface by DNA/chitosan/Fe3O4 bio-polyion complex membrane. The HRP on the electrode exhibited fast electron transfer rate, high affinity to H2O2 and good bioactivity toward H2O2 reduction.
Acknowledgments
The present work was supported by the Scientific Research Foundation for the Returned Overseas Chinese Scholars of State Education Ministry, Foundation of State Key Laboratory of Environmental Chemistry and Ecotoxicology, Research Center for Eco-Environmental Sciences, Chinese Academy of Sciences (KF2012-06), and Scientific Research Foundation of University of Science and Technology Liaoning.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Armstrong, F.A.; Hill, H.A.O.; Walton, N.J. Direct electrochemistry of redox proteins. Acc. Chem. Res 1988, 21, 407–413. [Google Scholar]
- Andreu, R.; Ferapontova, E.E.; Gorton, L.; Calvente, J.J. Direct electron transfer kinetics in horseradish peroxidase electrocatalysis. J. Phys. Chem. B 2007, 111, 469–477. [Google Scholar]
- Li, M.; Xu, S.; Tang, M.; Liu, L.; Gao, F.; Wang, Y. Direct electrochemistry of horseradish peroxidase on graphene-modified electrode for electrocatalytic reduction towards H2O2. Electrochim. Acta 2011, 56, 1144–1149. [Google Scholar]
- Zhang, J.D.; Feng, M.L.; Tachikawa, H. Layer-by-layer fabrication and direct electrochemistry of glucose oxidase on single wall carbon nanotubes. Bioelechemistry 2007, 22, 3036–3041. [Google Scholar]
- Deng, C.Y.; Chen, J.H.; Chen, X.L.; Mao, C.H.; Nie, L.H.; Yao, S.Z. Direct electrochemistry of glucose oxidase and biosensing for glucose based on boron-doped carbon nanotubes modified electrode. Biosens. Bioelectron 2008, 23, 1272–1277. [Google Scholar]
- Zebda, A.; Gondran, C.; Goff, A.L.; Holzinger, M.; Cinquin, P.; Cosnier, S. Mediatorless high-power glucose biofuel cells based on compressed carbon nanotube-enzyme electrodes. Nat. Commun 2011, 2, 370. [Google Scholar]
- Liu, Q.; Lu, X.B.; Li, J.; Li, Y.; Li, S.; Li, Y. Direct electrochemistry of glucose oxidase and electrochemical biosensing of glucose quantum dots/carbon nanotube electrodes. Biosens. Bioelectron 2007, 15, 3203–3209. [Google Scholar]
- Rahman, M.A.; Non, H.; Shim, Y. Direct electrochemistry of laccase immobilized on Au nanoparticles encapsulated-dendrimer bonded conducting polymer: Application for a catechin sensor. Anal. Chem 2008, 80, 8020–8027. [Google Scholar]
- Kang, X.H.; Wang, J.; Tang, Z.W.; Wu, H.; Lin, Y.H. Direct electrochemistry and electrocatalysis of horseradish peroxidase immobilized in hybrid organic-inorganic film of chitosan/sol-gel/carbon nanotubes. Talanta 2009, 78, 120–125. [Google Scholar]
- Lu, X.B.; Hu, J.Q.; Yao, X.; Wang, Z.; Li, J. Composite system based on chitosan and room-temperature ionicliquid direct electrochemistry and electrocatalysis of hemoglobin. Biomacromolecules 2006, 7, 975–980. [Google Scholar]
- Dai, Z.H.; Ni, J.; Huang, X.H.; Lu, G.F.; Bao, J.C. Direct electrochemistry of glucose oxidase immobilized on a hexagonal mesoporous silica-MCM-41 matrix. Bioelectrochemistry 2007, 70, 250–256. [Google Scholar]
- Yang, L.Q.; Ren, X.L.; Tang, F.Q.; Zhang, L. A practical glucose biosensor based on Fe3O4 nanoparticles and chitosan/nafion composite film. Biosens. Bioelectron 2009, 25, 889–895. [Google Scholar]
- Zhou, Y.; Yuan, P.X.; Yuan, R.; Chai, Y.Q.; Hong, C.L. Bienzyme functionalized threelayer composite magnetic nanoparticles for electrochemical immunosensors. Biomaterials 2009, 30, 2284–2290. [Google Scholar]
- Baby, T.T.; Ramaprabhu, S. SiO2 coated Fe3O4 magnetic nanoparticle dispersed multiwalled carbon nanotubes based amperometric glucose biosensor. Talanta 2010, 80, 2016–2022. [Google Scholar]
- Li, J.; Wei, X.; Yuan, Y. Synthesis of magnetic nanoparticles composed by Prussian blue and glucose oxidase for preparing highly sensitive and selective glucose biosensor. Sens. Actuators B 2009, 139, 400–406. [Google Scholar]
- Chen, X.; Zhu, J.; Chen, Z.; Xu, C.; Wang, Y.; Yao, C. A novel bienzyme glucose biosensor based on three-layer Au–Fe3O4@SiO2 magnetic nanocomposite. Sens. Actuators B 2011, 159, 220–228. [Google Scholar]
- Liu, S.Q.; Ju, H.X. Reagentless glucose biosensor based on direct electron transfer of glucose oxidase immobilized on colloidal gold modified carbon pasteelectrode. Biosens. Bioelectron 2003, 19, 177–183. [Google Scholar]
- Krishnan, S.; Walgama, C. Electrocatalytic features of a heme protein attached to polymer-functionalized magnetic nanoparticles. Anal. Chem 2013, 85, 11420–11426. [Google Scholar]
- Peng, H.P.; Liang, R.P.; Zhang, L.; Qiu, J.D. General preparation of novel core-shell heme protein-Au-polydopamine-Fe3O4 magnetic bionanoparticles for direct electrochemistry. J. Electroanal. Chem 2013, 700, 70–76. [Google Scholar]
- Peng, H.P.; Liang, R.P.; Zhang, L.; Qiu, J.D. Facile synthesis of Fe3O4@Al2O3 core-shell nanoparticles and their application to the highly specific capture of heme proteins for direct electrochemistry. Biosens. Bioelectron 2011, 26, 3005–3011. [Google Scholar]
- Kelley, S.O. Charge Migration through the DNA Double Helix. In Electroanalytical Methods for Biological Materials; Brajter-Toth, A., Chambers, J.Q., Eds.; Marcel Dekker: New York, NY, USA, 2002; pp. 1–27. [Google Scholar]
- Gu, T.; Hasebe, Y. Peroxidase and methylene blue-incorporated double stranded DNA-polyamine complex membrane for electrochemical sensing of hydrogen peroxide. Anal. Chim. Acta 2004, 525, 191–198. [Google Scholar]
- Gu, T.; Hasebe, Y. DNA–Cu(II) poly(amine) complex membrane as novel catalytic layer for highly sensitive amperometric determination of hydrogen peroxide. Biosens. Bioelectron 2006, 21, 2121–2128. [Google Scholar]
- Hasebe, Y.; Gu, T.; Fueki, T. Lactate biosensor based on coupled lactate oxidase/peroxidase system incorporated into the DNA/poly(allylamine) polyelectrolyte membrane. Sens. Lett 2005, 3, 304–308. [Google Scholar]
- Hasebe, Y.; Gu, T.; Kusakabe, H. Glutamate biosensor using a DNA-Cu(II)/polyamine membrane as a novel electrocataytic layer for cathodic determination of hydrogen peroxide. Electrochemistry 2006, 74, 179–182. [Google Scholar]
- Gu, T.; Zhang, Y.; Deng, F.; Zhang, J.; Hasebe, Y. Direct electrochemistry of glucose oxidase and biosensing for glucose based on DNA/chitosan film. J. Environ. Sci 2011, 23, S66–S70. [Google Scholar]
- Tian, Y.; Mao, L.; Okajima, T.; Ohsaka, T. Superoxide dismutase-based third-generation biosensor for superoxide anion. Anal. Chem 2002, 74, 2428–2434. [Google Scholar]
- Ma, X.; Liu, X.; Xiao, H.; Li, G. Direct electrochemistry and electrocatalysis of hemoglobin in poly-3-hydroxybutyrate membrane. Biosens. Bioelectron 2005, 20, 1836–1842. [Google Scholar]
- Upadhyay, A.K.; Ting, T.W.; Chen, S.M. Amperometric biosensor for hydrogen peroxide based on coimmobilized horseradish peroxidase and methylene green in ormosils matrix with multiwalled carbon nanotubes. Talanta 2009, 79, 38–45. [Google Scholar]
- Zhang, L.; Zhang, Q.; Li, J.H. Direct electrochemistry and electrocatalysis of hemoglobin immobilized in bimodal mesoporous silica and chitosan inorganic-organic hybrid film. Electrochem. Commun 2007, 9, 1530–1535. [Google Scholar]
- Huang, H.; Hu, N.; Zeng, Y.; Zhou, G. Electrochemistry and electrocatalysis with heme proteins in chitosan biopolymer films. Anal. Biochem 2002, 308, 141–151. [Google Scholar]
- Zhao, H.; Sheng, Q.; Zheng, J. Direct electrochemistry and electrocatalysis of horseradish peroxidase on a gold electrode modified with a polystyrene and multiwalled carbon nanotube composite film. Microchim. Acta 2012, 176, 177–184. [Google Scholar]
- Tan, X.; Zhang, J.; Tan, S.; Zhao, D.; Huang, Z.; Mi, Y.; Huang, Z. Amperometric hydrogen peroxide biosensor based on horseradish peroxidase immobilized on Fe3O4/chitosan modified glassy carbon electrode. Electroanalysis 2009, 21, 1514–1520. [Google Scholar]
- Laviron, E. General expression of the linear potential sweep voltammogram in the case of diffusionless electrochemical systems. J. Electroanal. Chem 1979, 101, 19–28. [Google Scholar]
- Xu, X.; Liu, S.; Ju, H. A novel hydrogen peroxide sensor via the direct electrochemistry of horseradish peroxidase immobilized on colloidal gold modified screen-printed electrode. Sensors 2003, 3, 350–360. [Google Scholar]
- Liu, X.; Huang, Y.S.; Wang, X.; Xiao, H.; Li, G. Electron transfer reactivity and the catalytic activity of horseradish peroxidase incorporated in dipalmitoyl phosphatidic acid films. Bioelectrochemistry 2006, 68, 98–104. [Google Scholar]
- Lan, Y.; Yuan, R.C.; Tang, D.; Dai, J.; Zhong, X. Direct electrochemistry of horseradish peroxidase immobilized on gold colloid/cysteine/nafion-modified platinum disk electrode. Sens. Actuators B 2006, 115, 105–109. [Google Scholar]
- Wang, Y.; Hasebe, Y. Carbon felt-based bioelectrocatalytic flow-through detectors: Highly sensitive amperometric determination of H2O2 based on a direct electrochemistry of covalently modified horseradish peroxidase using cyanuric chloride as a linking agent. Sens. Actuators B 2011, 155, 722–729. [Google Scholar]
- Lu, X.; Zhang, Q.; Zhang, L.; Li, J. Direct electron transfer of horseradish peroxidase and its biosensor based on chitosan and room temperature ionic liquid. Electrochem. Commun 2006, 8, 874–878. [Google Scholar]
- Liu, X.; Luo, L.; Ding, Y.; Xu, Y.; Li, F. Hydrogen peroxide biosensor based on the immobilization of horseradish peroxidase on γ-Al2O3 nanoparticles/chitosan film-modified electrode. J. Solid State Electrochem 2011, 15, 447–453. [Google Scholar]




© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gu, T.; Wang, J.; Xia, H.; Wang, S.; Yu, X. Direct Electrochemistry and Electrocatalysis of Horseradish Peroxidase Immobilized in a DNA/Chitosan-Fe3O4 Magnetic Nanoparticle Bio-Complex Film. Materials 2014, 7, 1069-1083. https://doi.org/10.3390/ma7021069
Gu T, Wang J, Xia H, Wang S, Yu X. Direct Electrochemistry and Electrocatalysis of Horseradish Peroxidase Immobilized in a DNA/Chitosan-Fe3O4 Magnetic Nanoparticle Bio-Complex Film. Materials. 2014; 7(2):1069-1083. https://doi.org/10.3390/ma7021069
Chicago/Turabian StyleGu, Tingting, Jianli Wang, Hongqi Xia, Si Wang, and Xiaoting Yu. 2014. "Direct Electrochemistry and Electrocatalysis of Horseradish Peroxidase Immobilized in a DNA/Chitosan-Fe3O4 Magnetic Nanoparticle Bio-Complex Film" Materials 7, no. 2: 1069-1083. https://doi.org/10.3390/ma7021069



