Synthesis and Enhanced Phosphate Recovery Property of Porous Calcium Silicate Hydrate Using Polyethyleneglycol as Pore-Generation Agent
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
| Chemical samples | Carbide residue | |||||||
|---|---|---|---|---|---|---|---|---|
| Chemical Components | CaO | SiO2 | Al2O3 | SO2 | MgO | Fe2O3 | SrO | H2O |
| Contents/% | 79.34 | 3.57 | 2.14 | 1.22 | 0.62 | 0.21 | 0.26 | 12.64 |
2.2. Preparation and Modification of CSH
2.2.1. Preparation of CSH
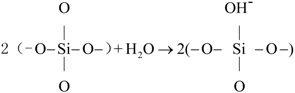
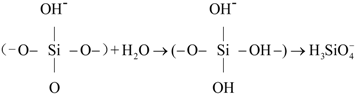
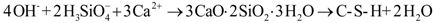
2.2.2. Modification Using PEG
2.3. Solubility Experiments
2.4. Experiments on Phosphate Recovery from Synthetic Solutions
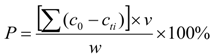
2.5. Characterization Instruments
3. Results and Discussion
3.1. Specific Surface Areas (SBET) and Pore Structure
| Samples | Total volume (cm3/g) | Peak pore diameter (nm) | SBET (m2/g) |
|---|---|---|---|
| CSH (PEG-0) | 0.28 | 12.97 | 85.92 |
| CSH (PEG-0.1%) | 0.20 | 10.28 | 77.95 |
| CSH (PEG-0.2%) | 0.28 | 9.34 | 118.77 |
| CSH (PEG-0.4%) | 0.40 | 10.79 | 148.11 |
| CSH (PEG-0.6%) | 0.87 | 9.83 | 352.96 |
| CSH (PEG-0.8%) | 0.31 | 10.17 | 119.94 |
3.2. Formation Mechanism of the Porous Structure
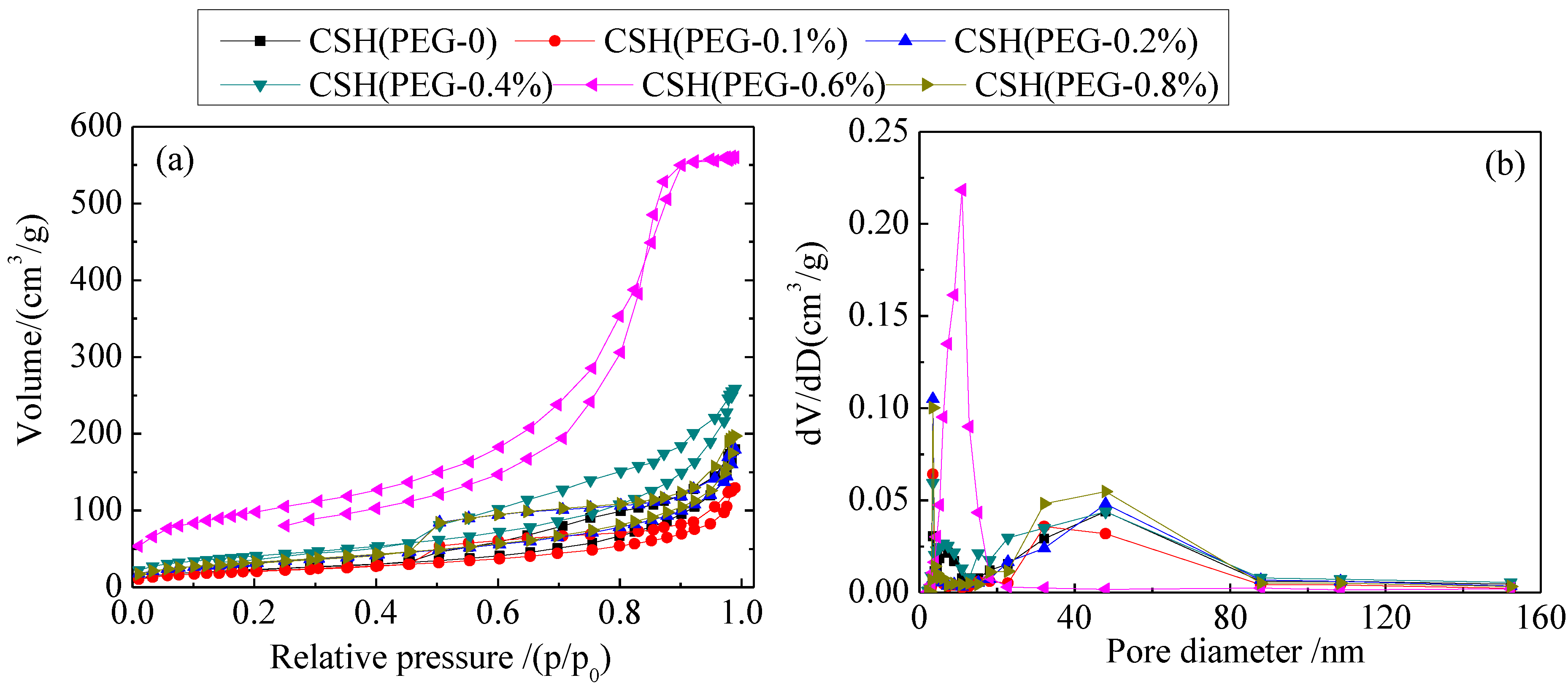
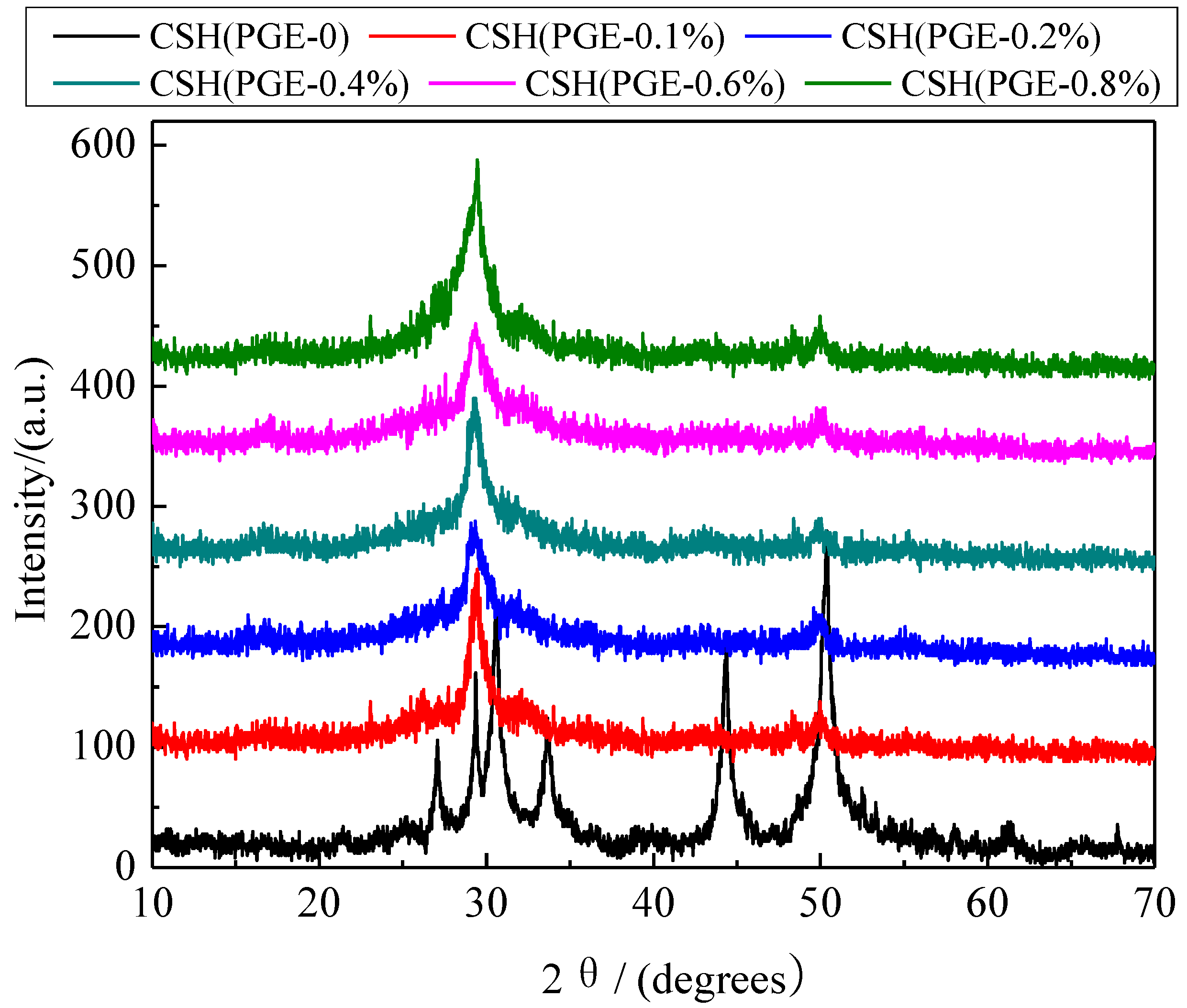
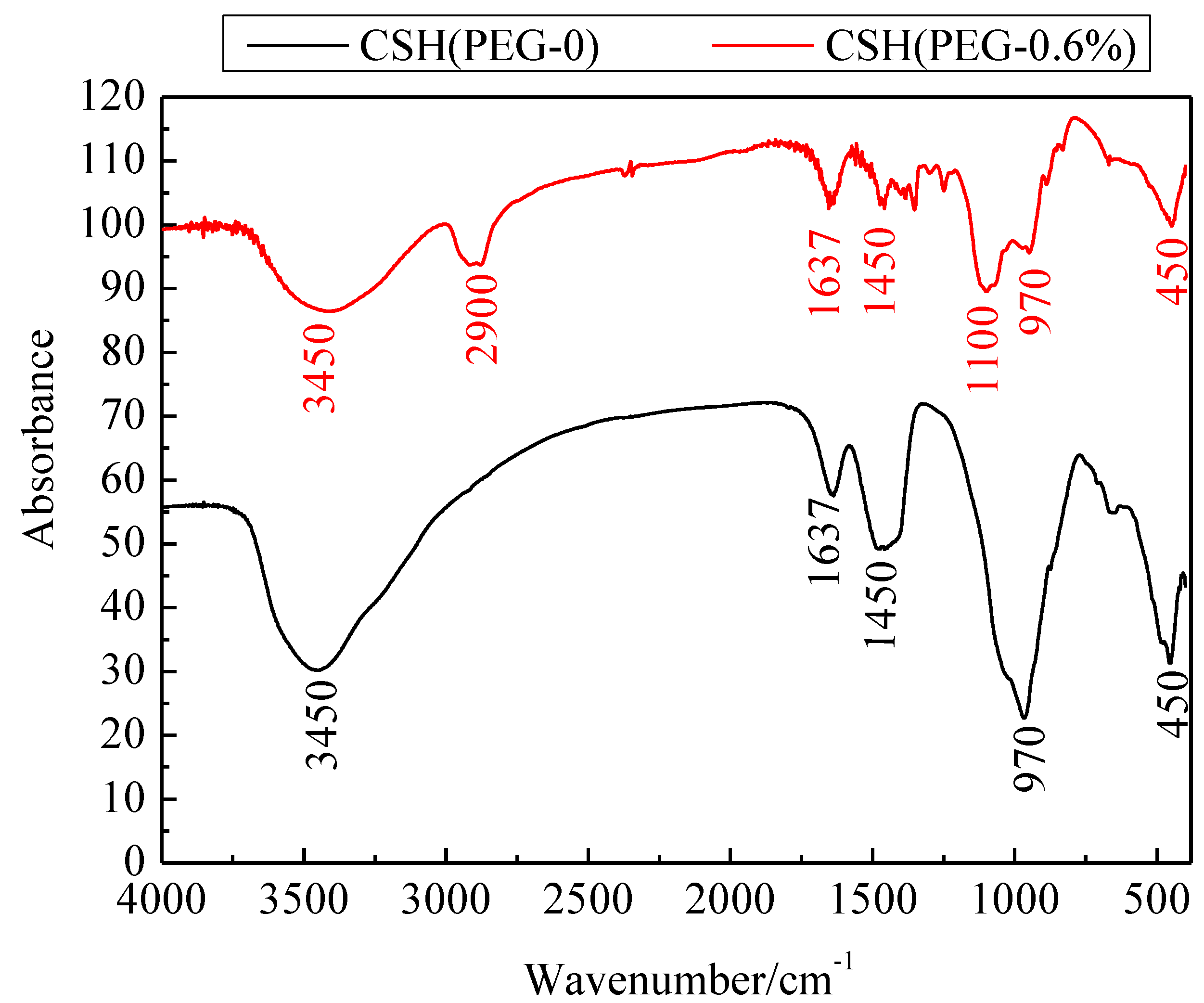

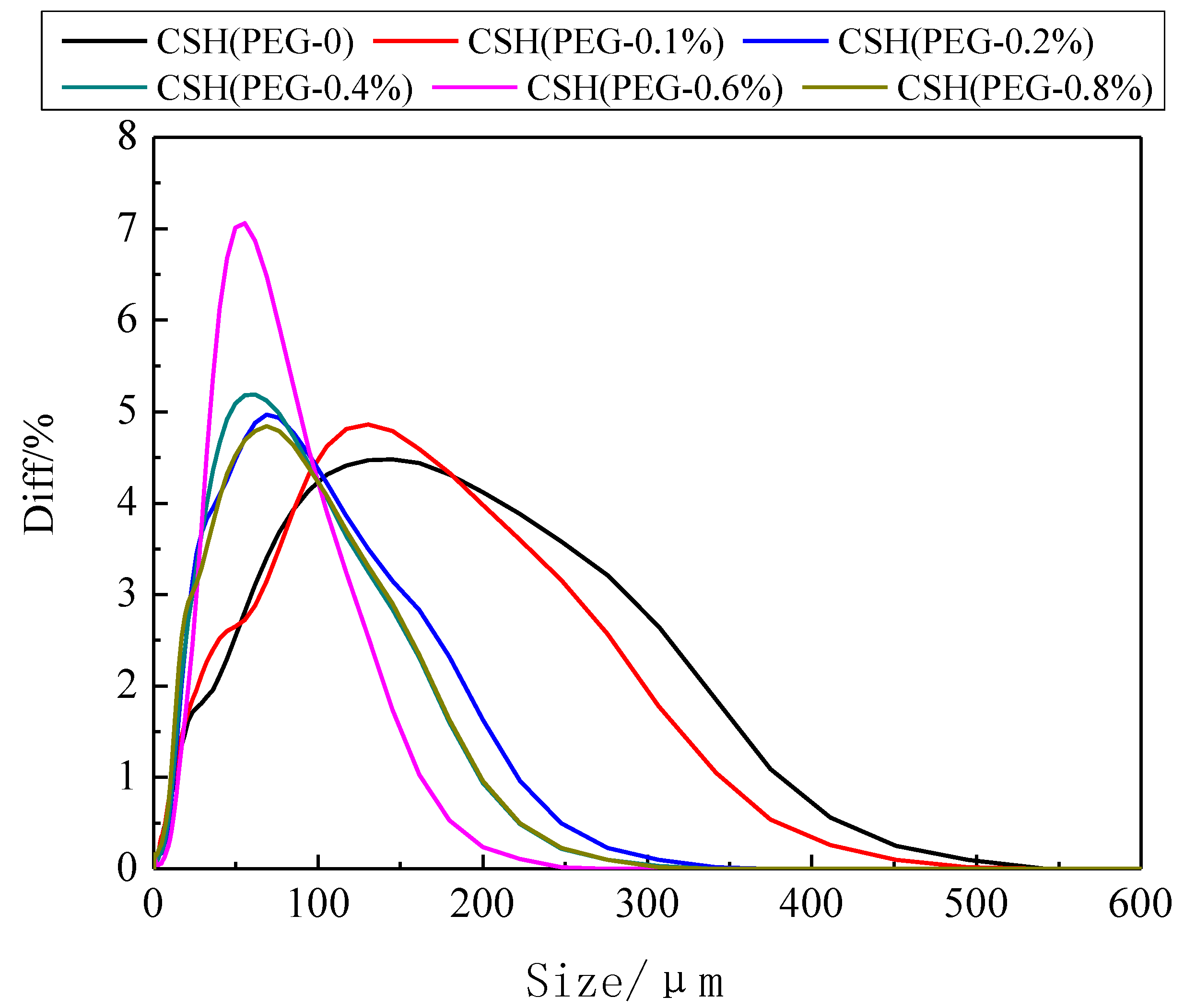
3.3. The Solubility and Dissolution Kinetics
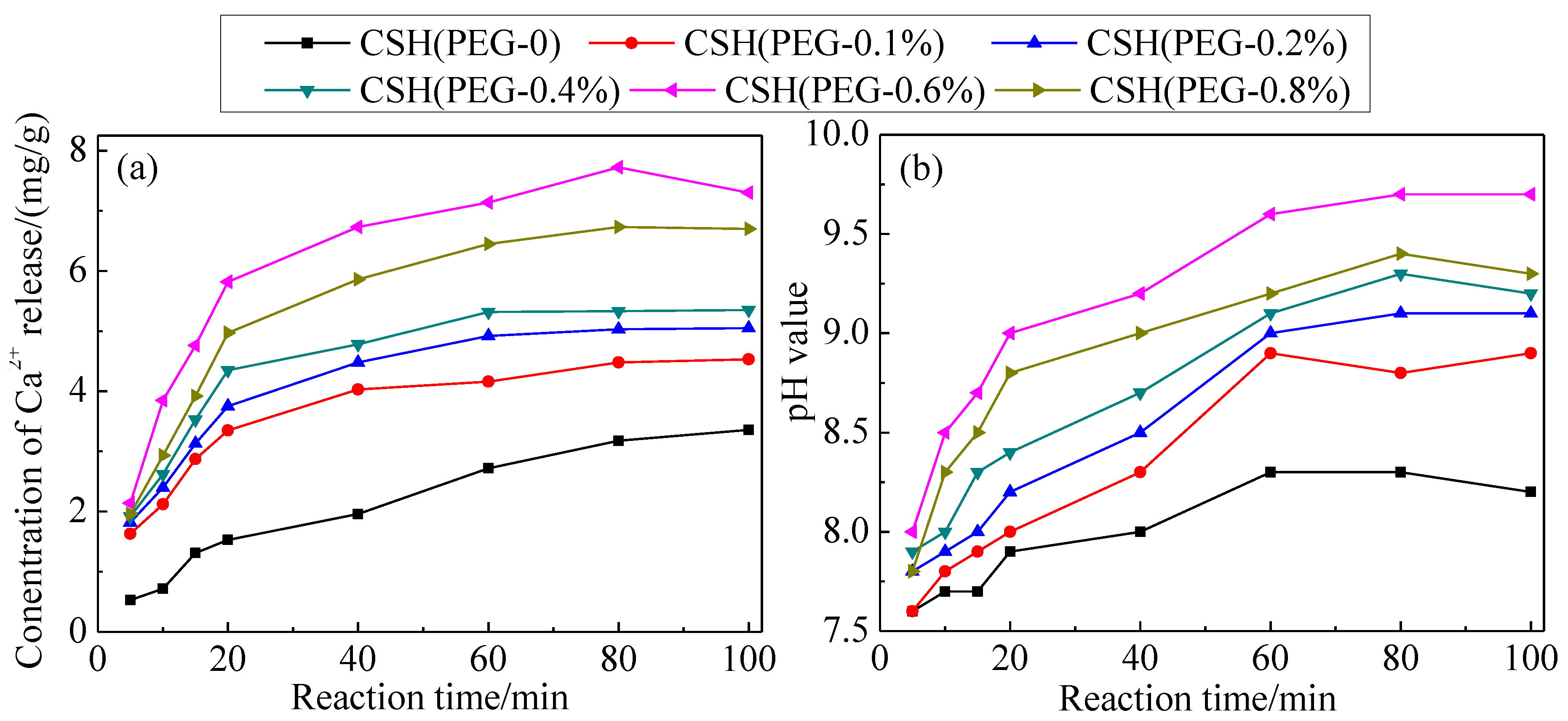
3.4. The Phosphate Recovery Property
| Samples | Quality (g) | Content of P (%) |
|---|---|---|
| CSH (PEG-0) | 1.02 | 10.53 |
| CSH (PEG-0.1%) | 1.02 | 9.81 |
| CSH (PEG-0.2%) | 1.03 | 11.75 |
| CSH (PEG-0.4%) | 1.04 | 12.95 |
| CSH (PEG-0.6%) | 1.07 | 15.40 |
| CSH (PEG-0.8%) | 1.05 | 13.24 |
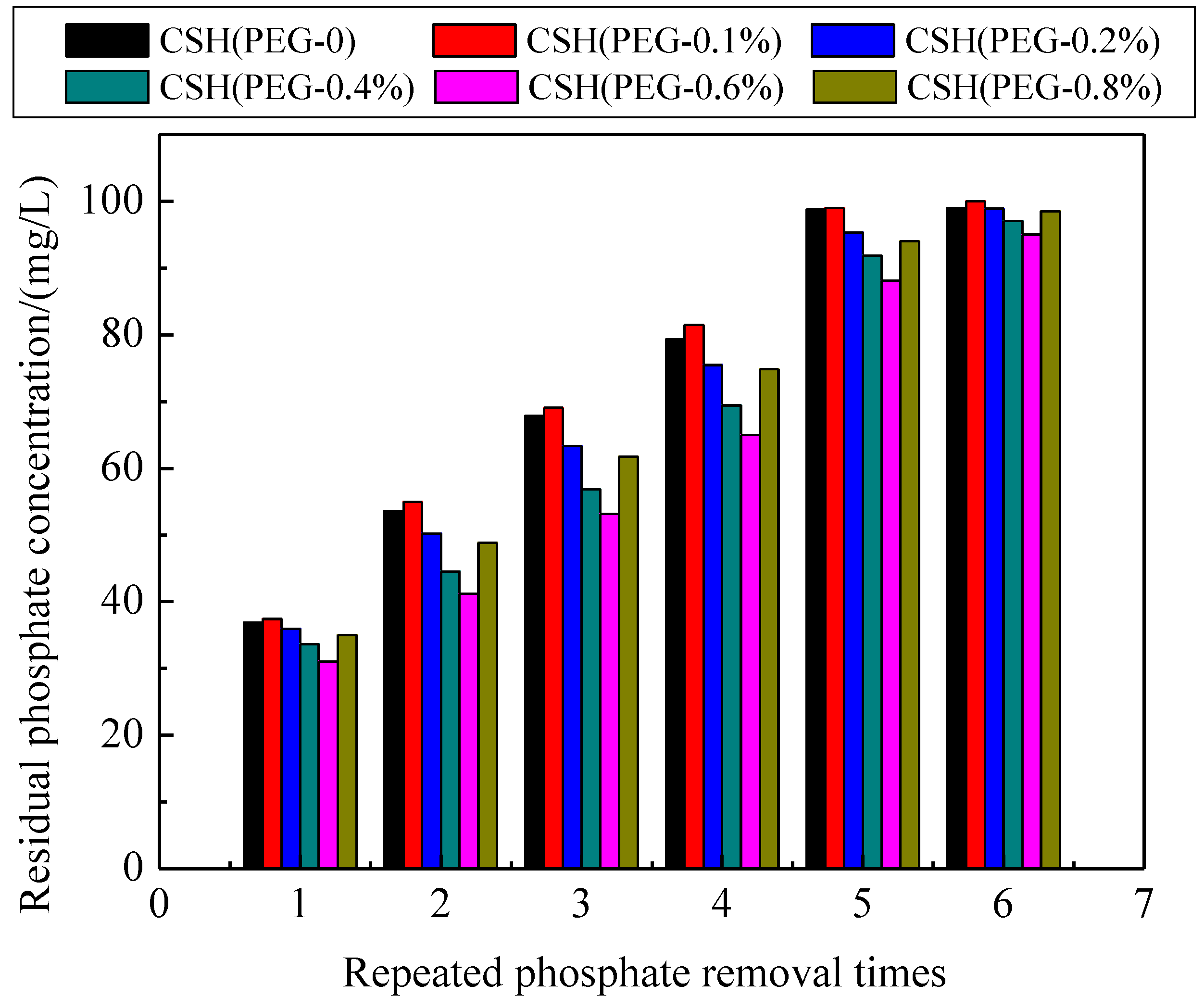
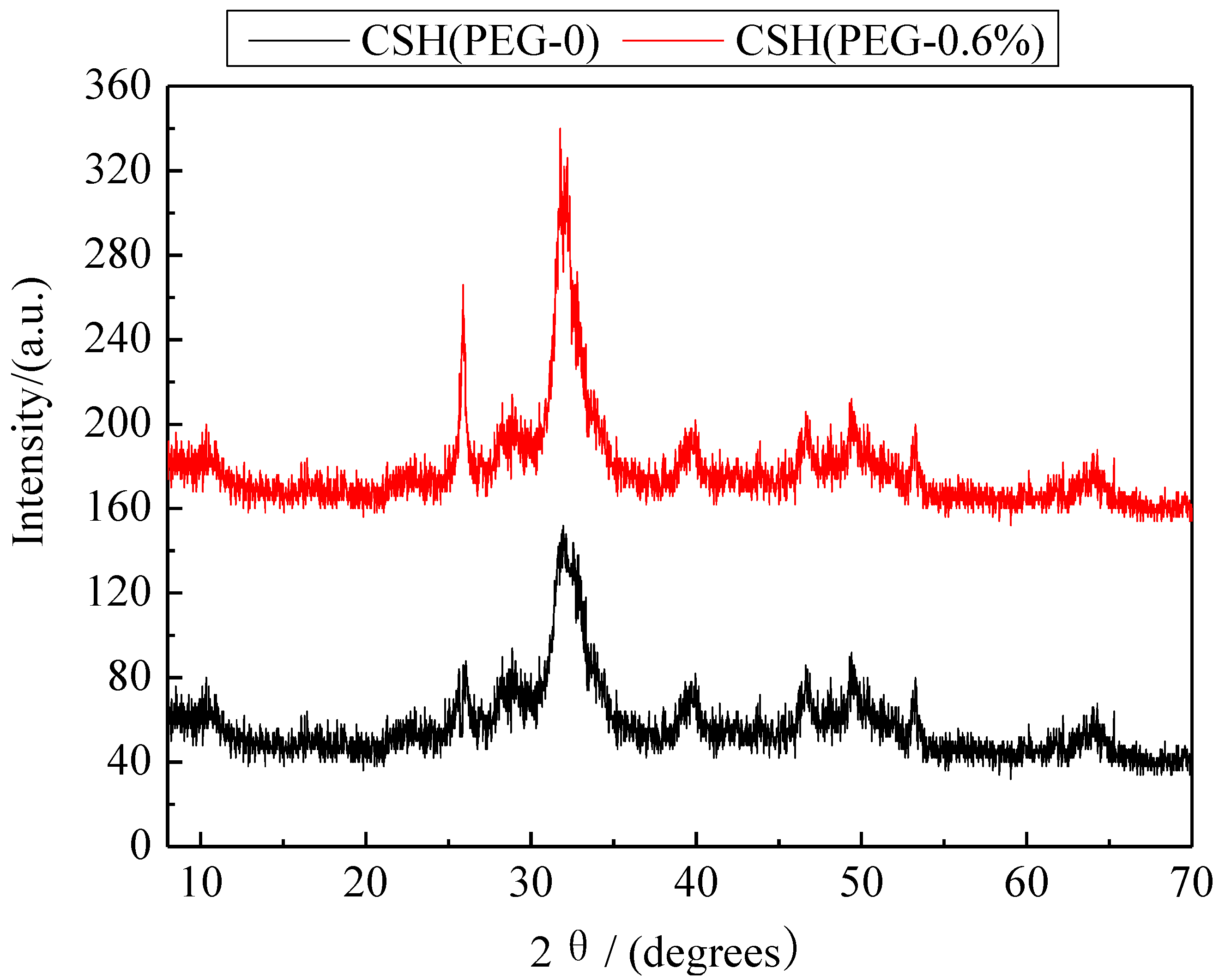
3.5. Mechanism of Phosphate Recovery Property Enhancement
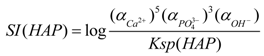
4. Conclusions
Conflict of Interest
References
- Cai, W.; Zhang, B.G.; Jin, Y.X.; Lei, Z.F.; Feng, C.P.; Ding, D.H.; Hu, W.W.; Chen, N.; Suemura, T. Behavior of total phosphorus removal in an intelligent controlled sequencing batch biofilm reactor for municipal wastewater treatment. Bioresour. Technol. 2013, 132, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.H.; Wang, Z.Y.; Lin, L.; Tian, B.H.; Pei, Y.S. Effect of low molecular weight organic acids on phosphorus adsorption by ferric-alum water treatment residuals. J. Hazard. Mater. 2012, 203–204, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Spears, B.M.; Meis, S.; Anderson, A.; Kellou, M. Comparison of phosphorus (P) removal properties of materials proposed for the control of sediment p release in UK lakes. Sci. Total Environ. 2013, 442, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Zhu, R.; Zhu, H.; Dai, X.H.; Yang, J. Phosphorus removal and simultaneous sludge reduction in humus soil sequencing batch reactor treating domestic wastewater. Chem. Eng. J. 2013, 215–216, 136–143. [Google Scholar] [CrossRef]
- Seyhan, D. Country-scale Phosphorus balancing as a base for resources conservation. Resour. Conserv. Recy. 2009, 53, 698–709. [Google Scholar] [CrossRef]
- Dawson, C.J.; Hilton, J. Fertiliser availability in a resource-limited world: Production and recycling of nitrogen and phosphorus. Food Policy 2011, 36, S14–S22. [Google Scholar] [CrossRef]
- Iizuka, A.; Sasaki, T.; Hongo, T.; Honma, M.; Hayakawa, Y.; Yamasaki, A.; Yanagisawa, Y. Phosphorus adsorbent derived from concrete sludge (PAdeCS) and its phosphorus recovery performance. Ind. Eng. Chem. Res. 2012, 51, 11266–11273. [Google Scholar] [CrossRef]
- Ye, Z.L.; Chen, S.H.; Wang, S.M.; Lin, L.F.; Yan, Y.J.; Zhang, Z.J.; Chen, J.S. Phosphorus recovery from synthetic swine wastewater by chemical precipitation using response surface methodology. J. Hazard. Mater. 2010, 176, 1083–1088. [Google Scholar] [CrossRef] [PubMed]
- Petzet, S.; Peplinski, B.; Cornel, P. On Wet chemical phosphorus recovery from sewage sludge ash by acidic or alkaline leaching and an optimized combination of both. Water Res. 2012, 46, 3769–3780. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.Y.; Zhou, Q.; Chen, J.H.; Zhang, L.; Chang, N. Phosphate adsorption on hydroxyl-iron-lanthanum doped activated carbon fiber. Chem. Eng. J. 2013, 215–216, 859–867. [Google Scholar] [CrossRef]
- Bashan, L.E.; Bashan, Y. Recent advances in removing phosphorus from wastewater and its future use as fertilizer (1997–2003). Water. Res. 2004, 38, 4222–4246. [Google Scholar] [CrossRef] [PubMed]
- Renman, G.; Renman, A. Sustainable Use of Crushed Autoclaved Aerated Concrete (CAAC) as a Filter Medium in Wastewater Purification. In Proceedings of the WASCON 2012 Conference, Gothenburg, Sweden, 1 June 2012.
- Renman, A.; Renman, G. Long-term phosphate removal by the calcium-silicate material polonite in wastewater filtration systems. Chemosphere 2010, 79, 659–664. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.C.; Kong, H.N.; Wu, D.Y.; Wang, X.Z.; Lin, Y.Y. Phosphate removal and recovery through crystallization of hydroxyapatite using xonotlite as seed crystal. J. Environ. Sci. 2009, 21, 575–580. [Google Scholar] [CrossRef]
- Baur, L.; Peter, K.; Mavrocordatos, D.; Wehrli, B.; Johnson, C.A. Dissolution-precipitation behaviour of ettringite, monosulfate, and calcium silicate hydrate. Cem. Concr. Res. 2004, 34, 341–348. [Google Scholar] [CrossRef]
- Wei, G.; Fang, Y.J.; Qing, K.C.; Peng, Y.; Qian, Z. Preparation and phosphorus recovery performance of porous calcium-silicate-hydrate. Ceram. Int. 2013, 39, 1385–1391. [Google Scholar] [CrossRef]
- Guan, W.; Ji, F.Y.; Chen, Q.K.; Yan, P.; Zhou, W.W. Influence of hydrothermal temperature on phosphorus recovery efficiency of porous calcium silicate hydrate. J. Nanomater. 2013, 9, 1–6. [Google Scholar]
- Song, Y.H.; Peter, G.W.; Berg, U.; Nueesch, R.; Donnert, D. Calcite-seed crystallization of calcium phosphate for phosphorus recovery. Chemosphere 2006, 63, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.H.; Lee, D.W.; Hwang, H.K.; Yim, S. Recovery of phosphates from wastewater using converter slag: Kinetics analysis of a completely mixed phosphorus crystallization process. Chemosphere 2006, 63, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Borrmann, T.; Johnston, J.H.; McFarlane, A.J.; MacKenzie, K.J.D.; Nukui, A. Structural elucidation of synthetic calcium silicates. Powder Diffr. 2008, 23, 204–212. [Google Scholar] [CrossRef]
- Sugiyama, D.; Fujita, T. A thermodynamic model of dissolution and precipitation of calcium silicate hydrates. Cem. Concr. Res. 2006, 36, 227–237. [Google Scholar] [CrossRef]
- Sezen, S.U.; Chae, S.R.; Benmore, C.J.; Wenk, H.R. Compositional evolution of calcium silicate hydrate(C–S–H) structures by total X-ray scattering. J. Am. Ceram. Soc. 2012, 2, 793–798. [Google Scholar]
- Maeda, H.; Ishida, E.H. Hydrothermal preparation of diatomaceous earth combined with calcium silicate hydrate gels. J. Hazard. Mater. 2011, 185, 858–861. [Google Scholar] [CrossRef] [PubMed]
- Southam, D.C.; Lewis, T.W.; McFarlane, A.J.; Borrmann, T.; Johnston, J.H. Calcium–phosphorus interactions at a nano-structured silicate surface. J. Colloid Interface Sci. 2008, 319, 489–497. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.X.; Ye, J.Z.; Zhang, G.W.; Gong, X.H.; Nie, L.H.; Liu, J.N. Polymorph and morphology control of CaCO3 via temperature and PEG during the decomposition of Ca(HCO3)2. J. Am. Ceram. Soc. 2012, 12, 3735–3738. [Google Scholar] [CrossRef]
- Knapen, E.; Gemert, D.V. Cement hydration and microstructure formation in the presence of water-soluble polymers. Cem. Concr. Res. 2009, 39, 6–13. [Google Scholar] [CrossRef]
- Wang, X.M.; Li, X.Y.; Shih, K.M. Spontaneous formation of nano-fibrillar boehmite and the enhancement effect of polyethylene glycol. J. Am. Ceram. Soc. 2011, 12, 4435–4443. [Google Scholar] [CrossRef]
- Li, S.; Qin, G.W.; Pei, T.W.; Ren, Y.P.; Zhang, Y.D.; Esling, C.; Zuo, L. Capping groups induced size and shage evolution of magnetite particles under hydrothermal condition and their magnetic properties. J. Am. Ceram. Soc. 2009, 3, 631–635. [Google Scholar] [CrossRef]
- Kim, J.G.; Tai, W.P.; Lee, K.J.; Cho, W.S. Effect of polyethylene glycol on the microstructure and PTCR characteristics of N-BaTiO3 ceramics. Ceram. Int. 2004, 30, 2223–2227. [Google Scholar] [CrossRef]
- Pourjavadi, A.; Fakoorpoor, S.M.; Hosseini, P.; Khaloo, A. Interactions between superabsorbent polymers and cement-based composites incorporating colloidal silica nanoparticles. Cem. Concr. Compos. 2013, 37, 196–204. [Google Scholar] [CrossRef]
- Urbanetz, N.A. Stabilization of solid dispersions of nimodipine and polyethylene glycol 2000. Eur. J. Pharm. Sci. 2006, 28, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Silva, D.A.; John, V.M.; Riberiro, J.L.D.; Roman, H.R. Pore size distribution of hydrated cement pastes modified with polymers. Cem. Concr. Res. 2001, 31, 1177–1184. [Google Scholar] [CrossRef]
- Cristian, B.; Claire, G.; Daniel, M.; Florent, C.; Yves, A. Phosphate removal from synthetic and real wastewater using steel slags produced in Europe. Water Res. 2012, 46, 2376–2384. [Google Scholar] [CrossRef] [PubMed]
- Dong, F.; Sun, Y.J.; Fu, M.; Wu, Z.B.; Lee, S.C. Room temperature synthesis and highly enhanced visible light photocatalytic activity of porous BiOI/BiOCl composites nanoplates microflowers. J. Hazard. Mater. 2012, 219–220, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Dong, F.; Sun, Y.J.; Fu, M.; Ho, W.K.; Lee, S.C.; Wu, Z.B. Novel in situ N-doped (BiO)2CO3 hierarchical microspheres self-assembled by nanosheets as efficient and durable visible light driven photocatalyst. Langmuir 2012, 28, 766–773. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Zhang, X.M. Metal-ion interactions with sugars. The crystal structure and FTIR study of an SrCl2-fructose complex. Carbohydr. Res. 2004, 339, 1421–1426. [Google Scholar] [CrossRef] [PubMed]
- Stark, J. Recent advances in the field of cement hydration and microstructure analysis. Cem. Concr. Res. 2011, 41, 666–678. [Google Scholar] [CrossRef]
- Richardson, I.G. Tobermorite/jennite-and tobermorite/calcium hydroxide-based models for the structure of C-S-H: Applicability to hardened pastes of tricalcium silicate, β-dicalcium silicate, portland cement, and blends of portland cement with blast-furnace slag, metakaolin, or silica fume. Cem. Concr. Res. 2004, 34, 1733–1777. [Google Scholar] [CrossRef]
- Roy, M.; Devoe, K.; Bandyopadhyay, A.; Bose, S. Mechanical property and in vitro biocompatibility of brushite cement modified by polyethylene glycol. Mater. Sci. Eng. C 2012, 32, 2145–2152. [Google Scholar] [CrossRef]
- Chen, F.D.; Liu, C.S.; Wei, J.; Chen, X.; Gao, Y.L. Preparation and characterization of injectable calcium phosphate cement paste modified by polyethylene glycol-6000. Mater. Chem. Phys. 2011, 125, 818–824. [Google Scholar] [CrossRef]
- Beaudoin, J.J.; Drame, H.; Raki, L. Formation and properties of C-S-H-PEG nanostructures. Mater. Struct. 2009, 42, 1003–1014. [Google Scholar] [CrossRef]
- Wyrwas, B.; Chraznowski, L.; Lawniczak, L.; Szulc, A.; Cyplik, P.; Bialas, W.; Szymanski, A.; Odachowska, A.H. Utilization of triton X-100 and polyethylene glycols during surfactant-mediated biodegradation of diesel fuel. J. Hazard. Mater. 2011, 197, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Johnston, C.T.; Parker, P.; Agnew, S.F. Infrared study of water sorption on Na-, Li-, Ca, and Mg-exchanged (SWy-1 and SAz-1) montmorillonite. Clays Clay Miner. 2000, 48, 120–131. [Google Scholar] [CrossRef]
- Ha, J.; Chae, S.; Chou, K.W.; Tyliszczak, T.; Monteiro, P.J.M. Effect of polymers on the nano structure and on the carbonation of calcium silicate hydrates: A scanning transmission X-ray microscopy study. J. Mater. Sci. 2012, 47, 976–989. [Google Scholar] [CrossRef]
- Celik, F.A.; Kazanc, S. Crystallization analysis and determination of Avrami exponents of CuAlNi alloy by molecular dynamics simulation. Phys. B 2013, 409, 63–70. [Google Scholar] [CrossRef]
- Berg, U.; Donnert, D.; Weidler, P.G.; Kaschka, E.; Knoll, G.; Nueesch, R. Phosphorus Removal and Recovery from Wastewater by Tobermorite-seeded Crystallization of Calcium Phosphate. In Proceedings of the 3rd IWA Leading-Edge Conference Water and Wastewater Treatment Technologies, Sapporo, Japan, 6–8 June 2005.
- Sinirkaya, M.; Ozer, A.K.; Gulaboglu, M.S. Investigation of the changes of P2O5 content of phosphate rock during simultaneous calcinations/salvation. Powder Technol. 2011, 211, 72–76. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Guan, W.; Ji, F.; Chen, Q.; Yan, P.; Pei, L. Synthesis and Enhanced Phosphate Recovery Property of Porous Calcium Silicate Hydrate Using Polyethyleneglycol as Pore-Generation Agent. Materials 2013, 6, 2846-2861. https://doi.org/10.3390/ma6072846
Guan W, Ji F, Chen Q, Yan P, Pei L. Synthesis and Enhanced Phosphate Recovery Property of Porous Calcium Silicate Hydrate Using Polyethyleneglycol as Pore-Generation Agent. Materials. 2013; 6(7):2846-2861. https://doi.org/10.3390/ma6072846
Chicago/Turabian StyleGuan, Wei, Fangying Ji, Qingkong Chen, Peng Yan, and Ling Pei. 2013. "Synthesis and Enhanced Phosphate Recovery Property of Porous Calcium Silicate Hydrate Using Polyethyleneglycol as Pore-Generation Agent" Materials 6, no. 7: 2846-2861. https://doi.org/10.3390/ma6072846




