Investigation of Iron Oxide Morphology in a Cyclic Redox Water Splitting Process for Hydrogen Generation
Abstract
:1. Introduction
2. Experimental Methods
2.1. Thermal Reactor Design
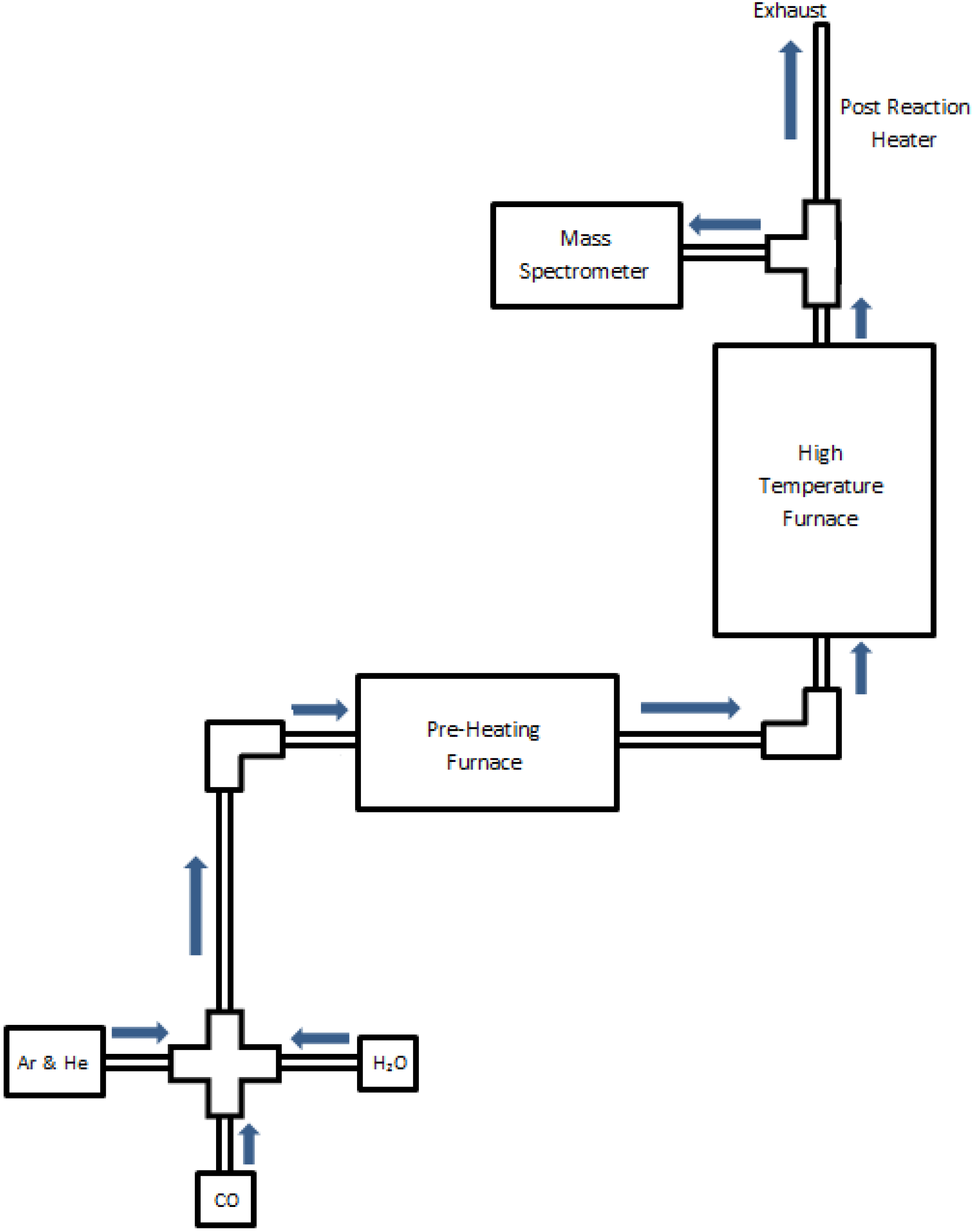
2.2. Data Acquisition
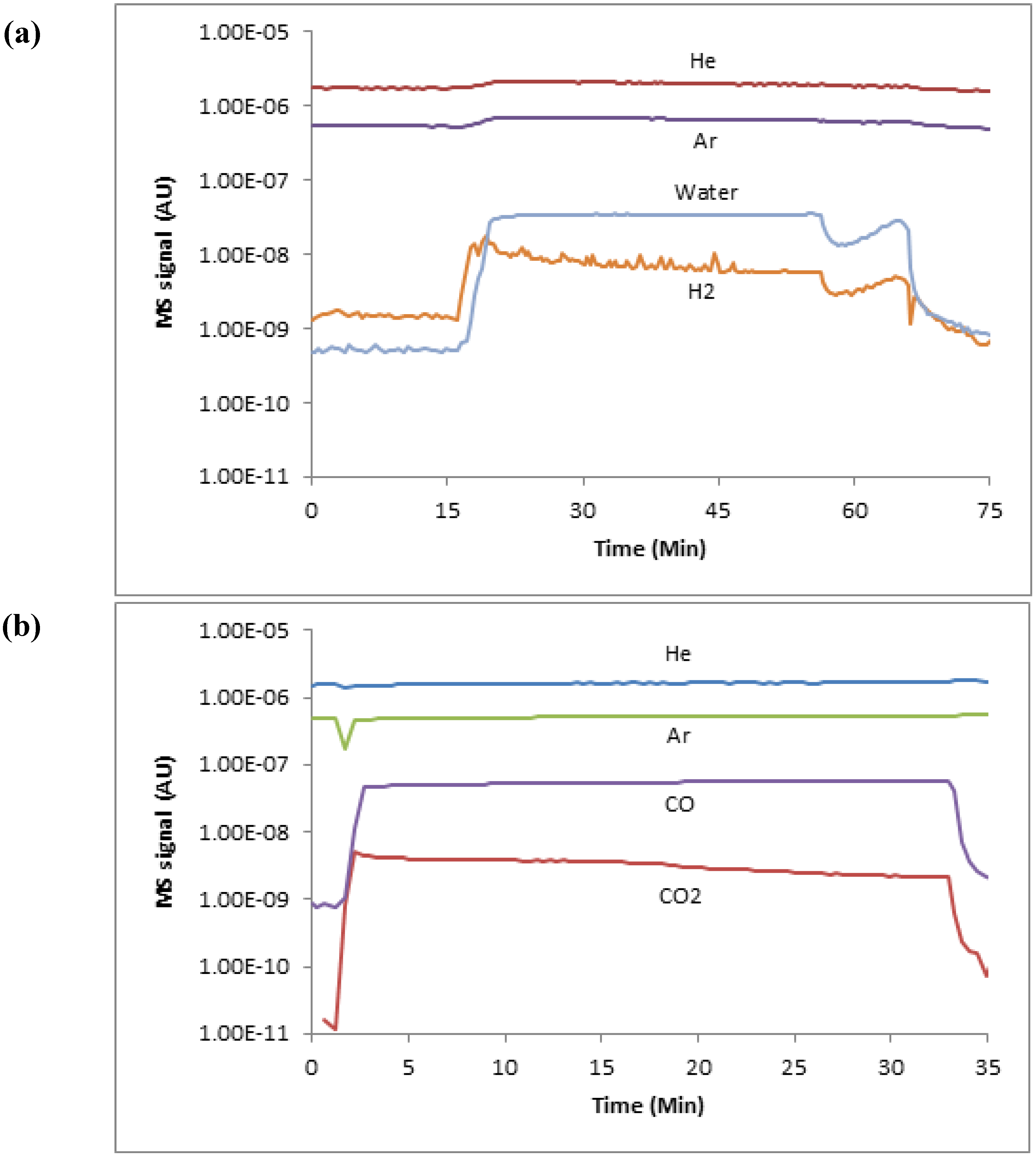
2.3. Experimental Procedure
3. Results
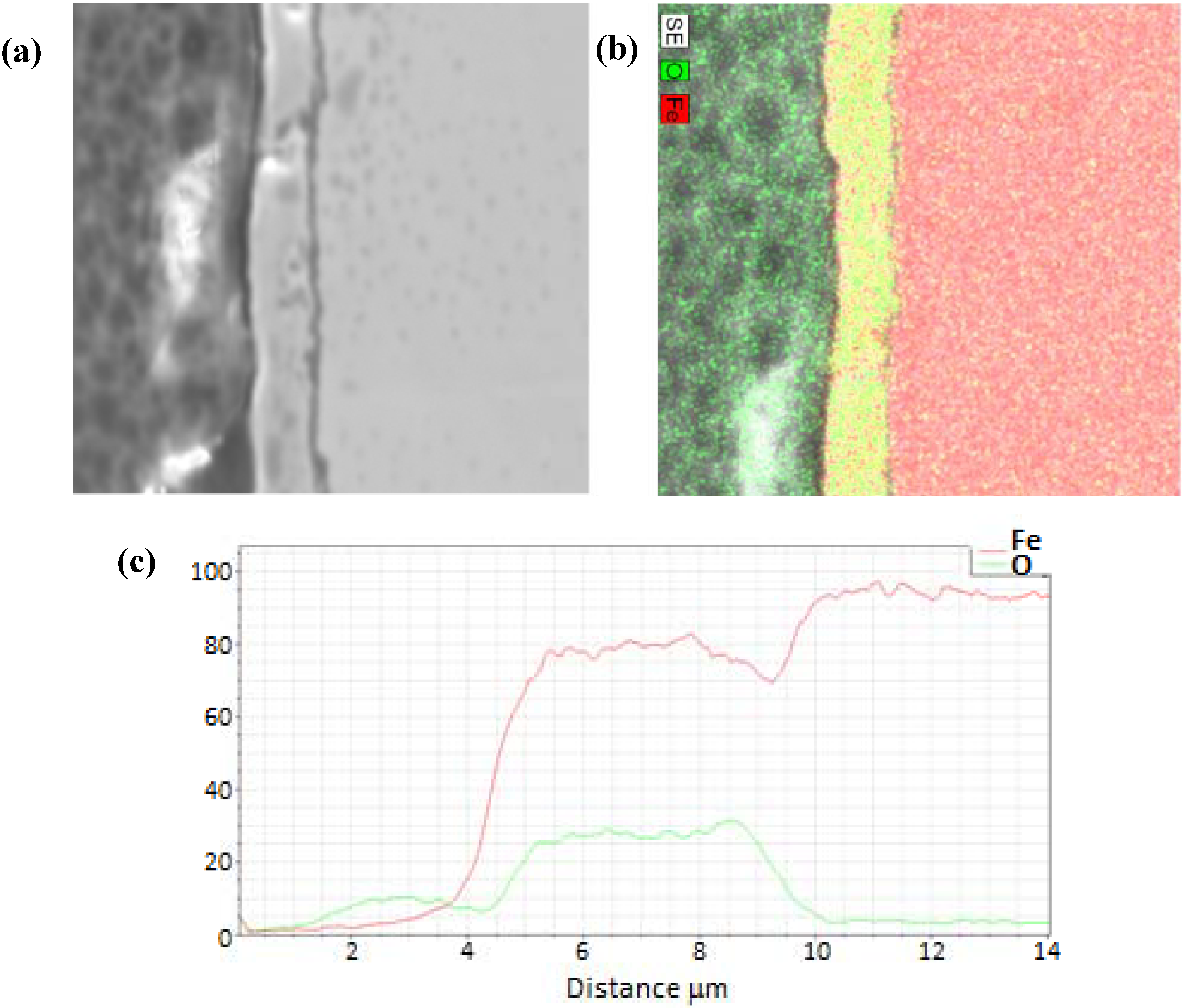
3.1. Termination Following Oxidation

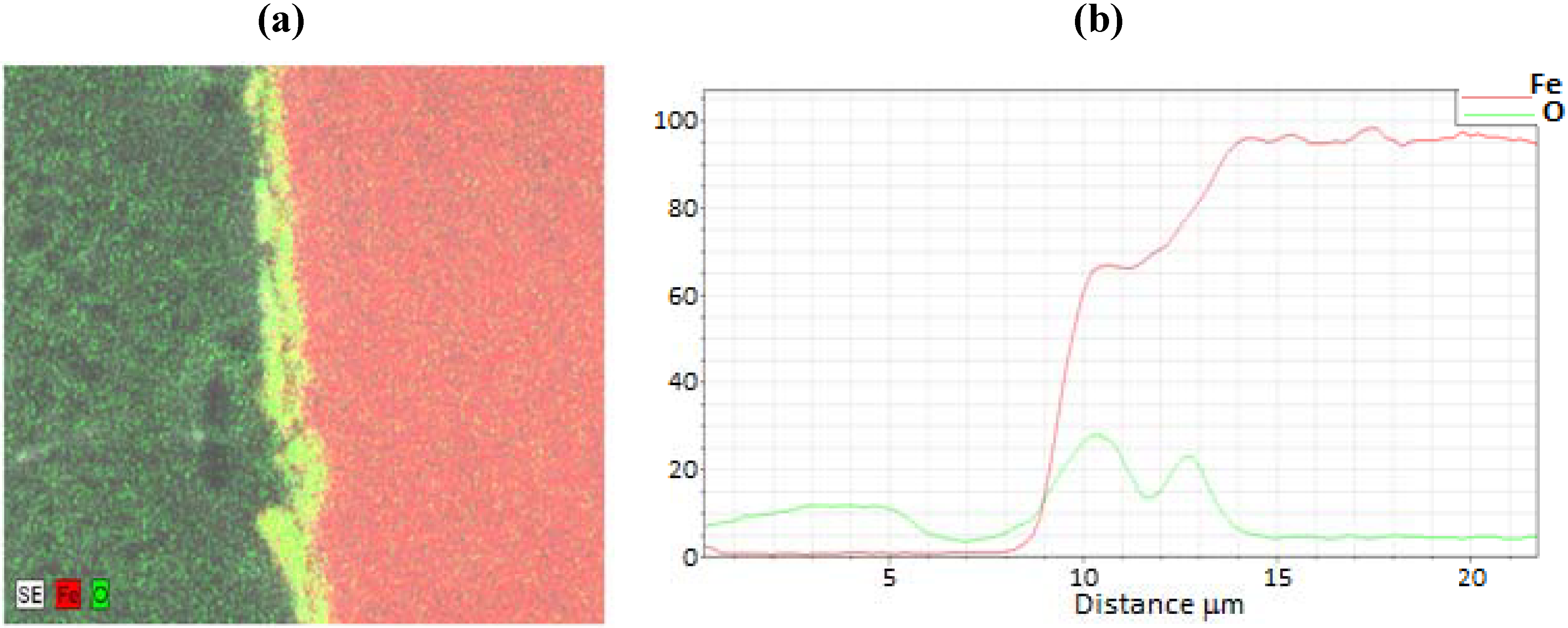
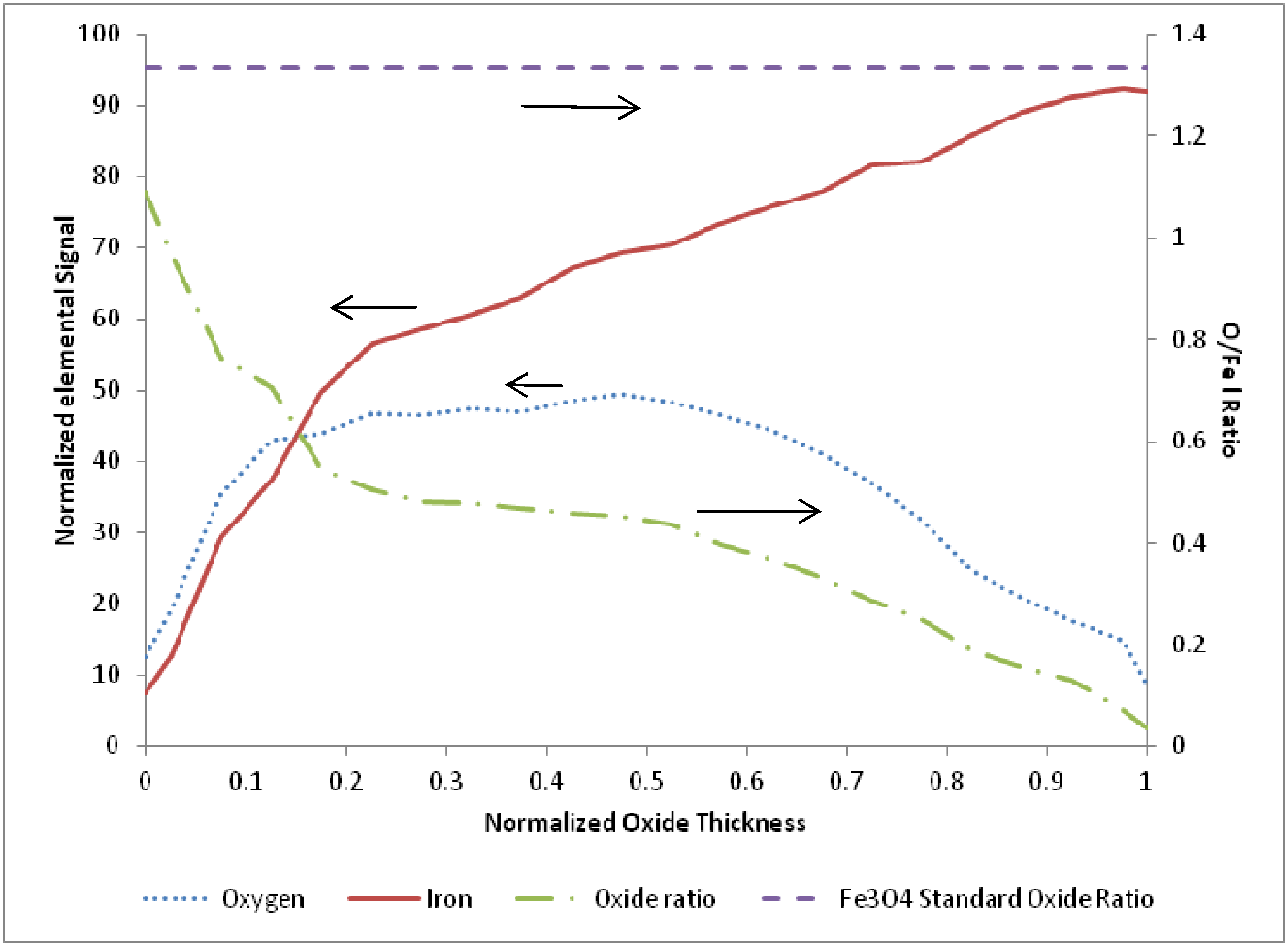
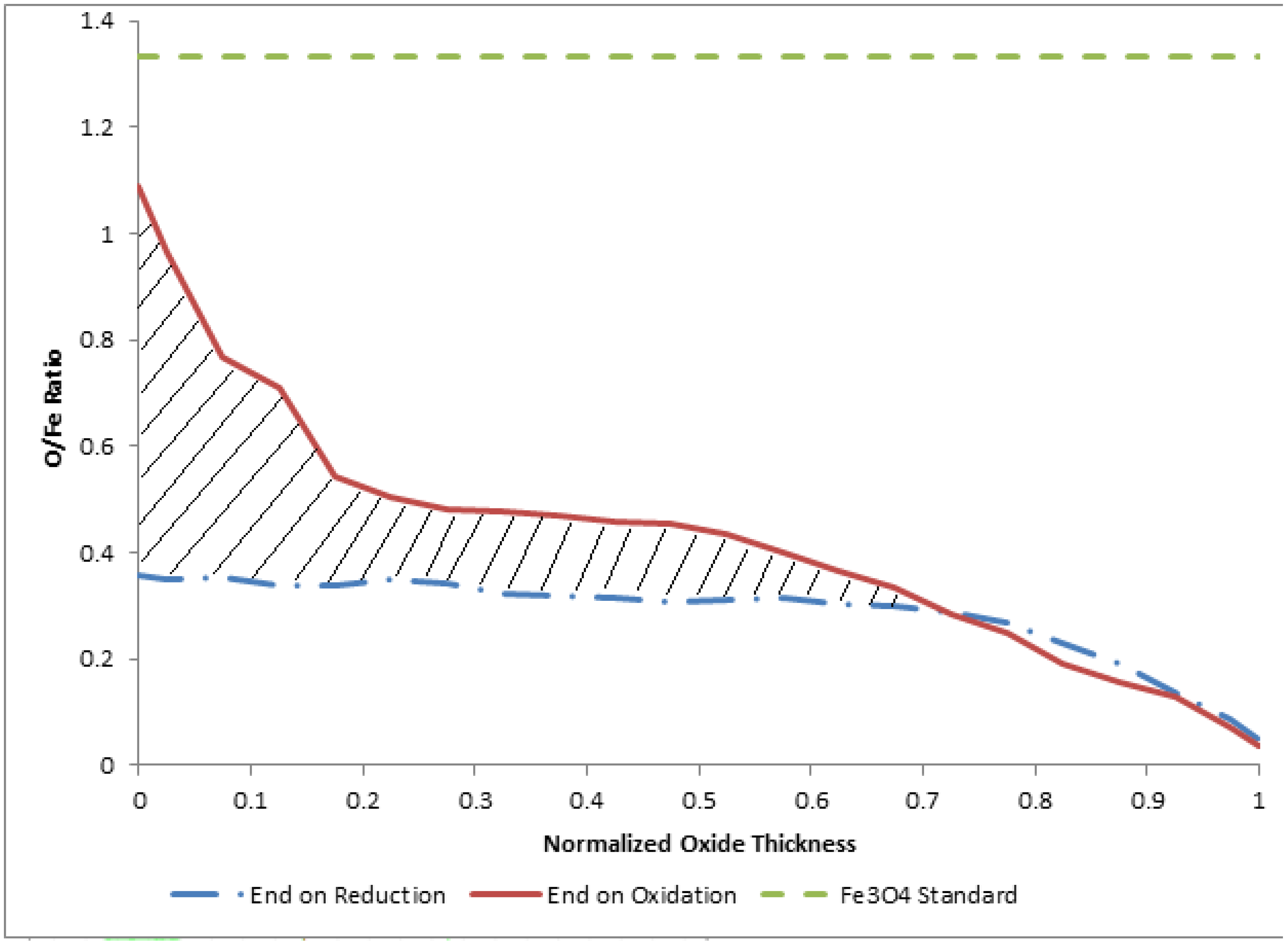
3.2. Termination Following Reduction
| Temperature (K) | Number of Cycles | Oxide Thickness (µm) |
|---|---|---|
| 873 | 1 | 5.9 |
| 873 | 1 | 3.5 |
| 796 | 21 | 4.8 |
| 796 | 21 | 9.8 |
| 796 | 21 | 3.1 |
| 673 | 22 | 8.8 |
| 673 | 22 | 5.6 |
| 761 | 26 | 4.9 |
| 761 | 26 | 5.9 |
| 761 | 26 | 12.4 |
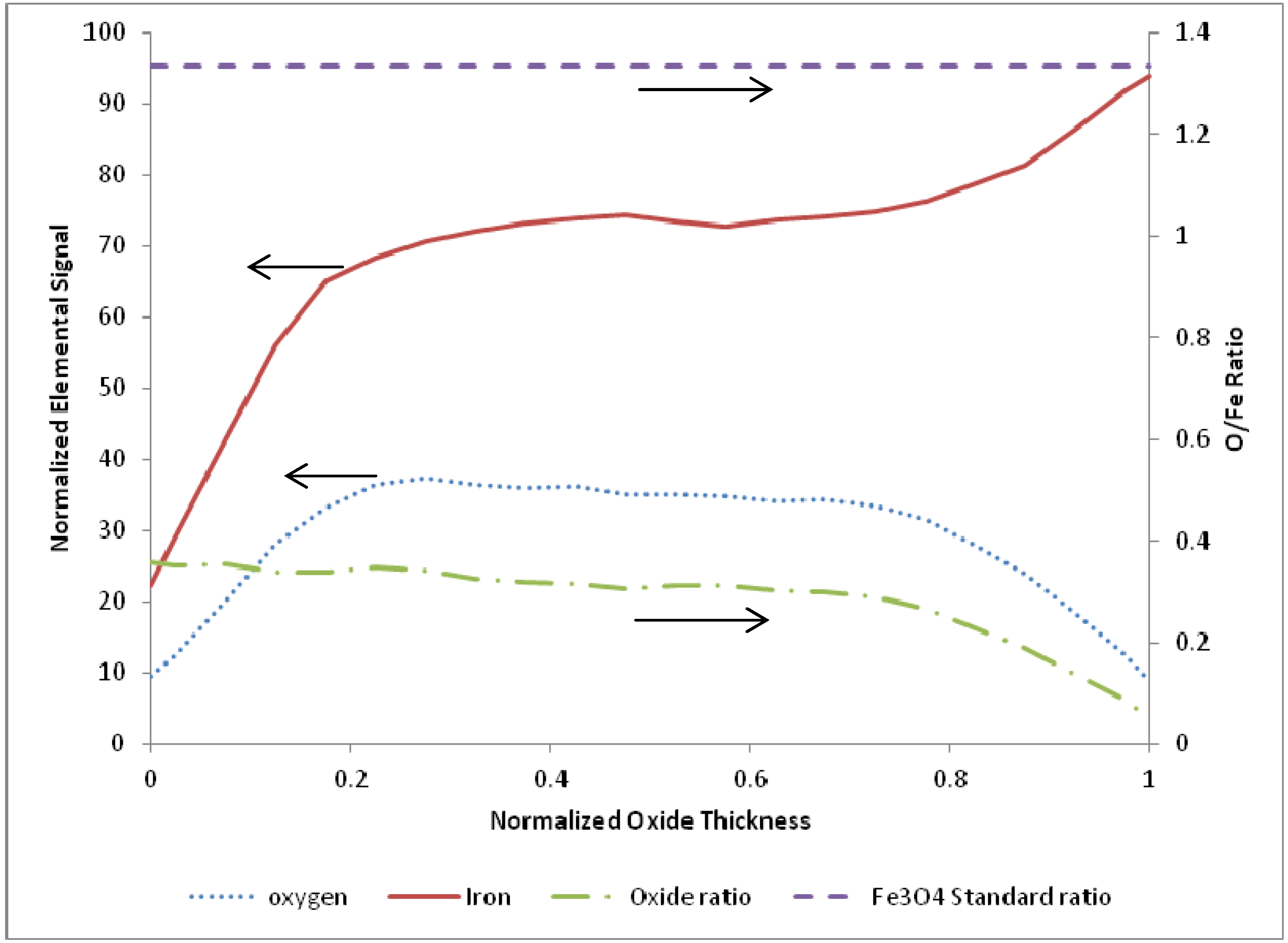
4. Discussion and Conclusions
Acknowledgements
References
- Steinfeld, A.; Weimer, A. Thermochemical production of fuels with concentrated solar energy. Opt. Express 2010, 18, A100–A111. [Google Scholar] [CrossRef] [PubMed]
- Kodoma, T. High-temperature solar chemistry for converting solar heat to chemical fuels. Prog. Energy Combust. Sci. 2003, 29, 567–597. [Google Scholar] [CrossRef]
- Perkins, C.; Weimer, A. Solar thermal production of renewable hydrogen. AIChE J. 2009, 55, 286–293. [Google Scholar] [CrossRef]
- Hacker, V.; Frankhauser, R.; Faleschini, G.; Fuchs, H.; Friedrich, K.; Muhr, M.; Kordesch, K. Hydrogen production by steam-iron process. J Power Sources 2000, 86, 531–535. [Google Scholar] [CrossRef]
- Lorente, E.; Pena, J.A.; Herguido, J. Separation and storage of hydrogen by steam-iron process: Effect of added metals upon hydrogen release and solid stability. J. Power Sources 2009, 192, 224–229. [Google Scholar] [CrossRef]
- Funk, J.E. Thermochemical hydrogen production: Past and present. Int. J. Hydrog. Energy 2001, 26, 185–190. [Google Scholar] [CrossRef]
- Steinfeld, A. Solar thermochemical production of hydrogen—A review. Solar Energy 2005, 78, 603–615. [Google Scholar] [CrossRef]
- Nakamura, T. Hydrogen-production from water utilizing solar heat at high-temperatures. Solar Energy 1976, 19, 467–475. [Google Scholar] [CrossRef]
- Stamatiou, A.; Loutzenhiser, P.G.; Steinfeld, A. Solar syngas production via H2O/CO2-splitting thermochemical cycles with Zn/ZnO and FeO/Fe3O4 redox reactions. Chem. Mater. 2010, 22, 851–859. [Google Scholar] [CrossRef]
- Go, K.S.; Son, S.R.; Kim, S.D.; Kang, K.S.; Park, C.S. Hydrogen production from two-step steam methane reforming in a fluidized bed reactor. Int. J. Hydrog. Energy 2009, 34, 1301–1309. [Google Scholar] [CrossRef]
- Otsuka, K.; Mito, A.; Takenaka, S.; Yamanaka, I. Production of hydrogen from methane without CO2 emissions mediated by indium oxide and iron oxide. Int. J. Hydrog. Energy 2001, 26, 191–194. [Google Scholar] [CrossRef]
- Gupta, P.; Velazquez-Vargas, L.G.; Fan, L.S. Syngas redox (SGR) process to produce hydrogen from coal derived syngas. Energy Fuels 2007, 21, 2900–2908. [Google Scholar] [CrossRef]
- Neises, M.; Roeb, M.; Schmücker, M.; Sattler, C.; Pitz-Paal, R. Kinetic investigations of the hydrogen step of a thermochemical cycle using mixed iron oxides coated on ceramic substrates. Int. J. Energy Res. 2010, 34, 651–661. [Google Scholar]
- Charvin, P.; Abanades, S.; Flamant, G.; Lemort, F. Two-step water splitting thermochemical cycle based on iron oxide redox pair for solar hydrogen production. Energy 2007, 32, 1124–1133. [Google Scholar] [CrossRef]
- Roeb, M.; Gathmann, N.; Neises, M.; Sattler, C.; Pitz-Paal, R. Thermodynamic analysis of two-step solar water splitting with mixed iron oxides. Int. J. Energy Res. 2009, 33, 893–902. [Google Scholar] [CrossRef]
- Go, K.S.; Son, S.R.; Kim, S.D. Reaction kinetics of reduction and oxidation of metal oxides for hydrogen production. Int. J. Hydrog. Energy 2008, 33, 5986–5995. [Google Scholar] [CrossRef]
- Mehdizadeh, A.M.; Klausner, J.F.; Barde, A.; Mei, R. Enhancement of thermochemical hydrogen production using an iron silica magnetically stabilized porous structure. Int. J. Hydrog. Energy 2012, 37, 8954–8963. [Google Scholar] [CrossRef]
- Schefe, J.; Li, J.; Weimer, A. A spinel ferrite/hercynite water-splitting redox cycle. Int. J. Hydrog. Energy 2010, 5, 3333–3340. [Google Scholar] [CrossRef]
- Gokon, N.; Murayama, H.; Umeda, J.; Hatamachi, T.; Kodama, T. Monoclinic zirconia-supported Fe2O3 for the two-step water-splitting thermochemical cycle at high thermal reduction temperatures of 1400–1600 °C. Int. J. Hydrog. Energy 2009, 34, 1208–1217. [Google Scholar] [CrossRef]
- Pineau, A.; Kanari, N.; Gabalah, I. Kinetics of reduction of iron oxides by H2. Part 1. Low temperature reduction of hematite. Thermochem. Acta 2006, 447, 89–100. [Google Scholar] [CrossRef]
- Stehle, R.C.; Bobek, M.M.; Hooper, R.; Hahn, D.W. Oxidation reaction kinetics for the steam-iron process in support of hydrogen production. Int. J. Hydrog. Energy 2011, 36, 15125–15135. [Google Scholar] [CrossRef]
- Svoboda, K.; Slowinski, G.; Rogut, J.; Baxter, D. Thermodynamic possibilities and constraints for pure hydrogen production by iron based chemical looping process at lower temperatures. Energy Convers. Manag. 2007, 48, 3063–3373. [Google Scholar] [CrossRef]
- Singh, A.; Al-Raqom, F.; Klausner, J.F.; Petrasch, J. Production of hydrogen via an Iron/Iron oxide looping cycle: Thermodynamic modeling and experimental validation. Int. J. Hydrog. Energy 2012, 37, 7442–7450. [Google Scholar] [CrossRef]
- Steinfeld, A.; Sanders, S.; Palumbo, R. Design aspects of solar thermochemical engineering—A case study: Two step water splitting cycle using the Fe3O4/FeO redox system. Solar Energy 1999, 65, 43–53. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bobek, M.M.; Stehle, R.C.; Hahn, D.W. Investigation of Iron Oxide Morphology in a Cyclic Redox Water Splitting Process for Hydrogen Generation. Materials 2012, 5, 2003-2014. https://doi.org/10.3390/ma5102003
Bobek MM, Stehle RC, Hahn DW. Investigation of Iron Oxide Morphology in a Cyclic Redox Water Splitting Process for Hydrogen Generation. Materials. 2012; 5(10):2003-2014. https://doi.org/10.3390/ma5102003
Chicago/Turabian StyleBobek, Michael M., Richard C. Stehle, and David W. Hahn. 2012. "Investigation of Iron Oxide Morphology in a Cyclic Redox Water Splitting Process for Hydrogen Generation" Materials 5, no. 10: 2003-2014. https://doi.org/10.3390/ma5102003
APA StyleBobek, M. M., Stehle, R. C., & Hahn, D. W. (2012). Investigation of Iron Oxide Morphology in a Cyclic Redox Water Splitting Process for Hydrogen Generation. Materials, 5(10), 2003-2014. https://doi.org/10.3390/ma5102003




