Micro- and Nanoscale Hydrogel Systems for Drug Delivery and Tissue Engineering
Abstract
:1. Introduction
2. Synthetic Polymer Based Hydrogels
2.1. Poly(N-isopropylacrylamide) in Drug Delivery and Biosensing
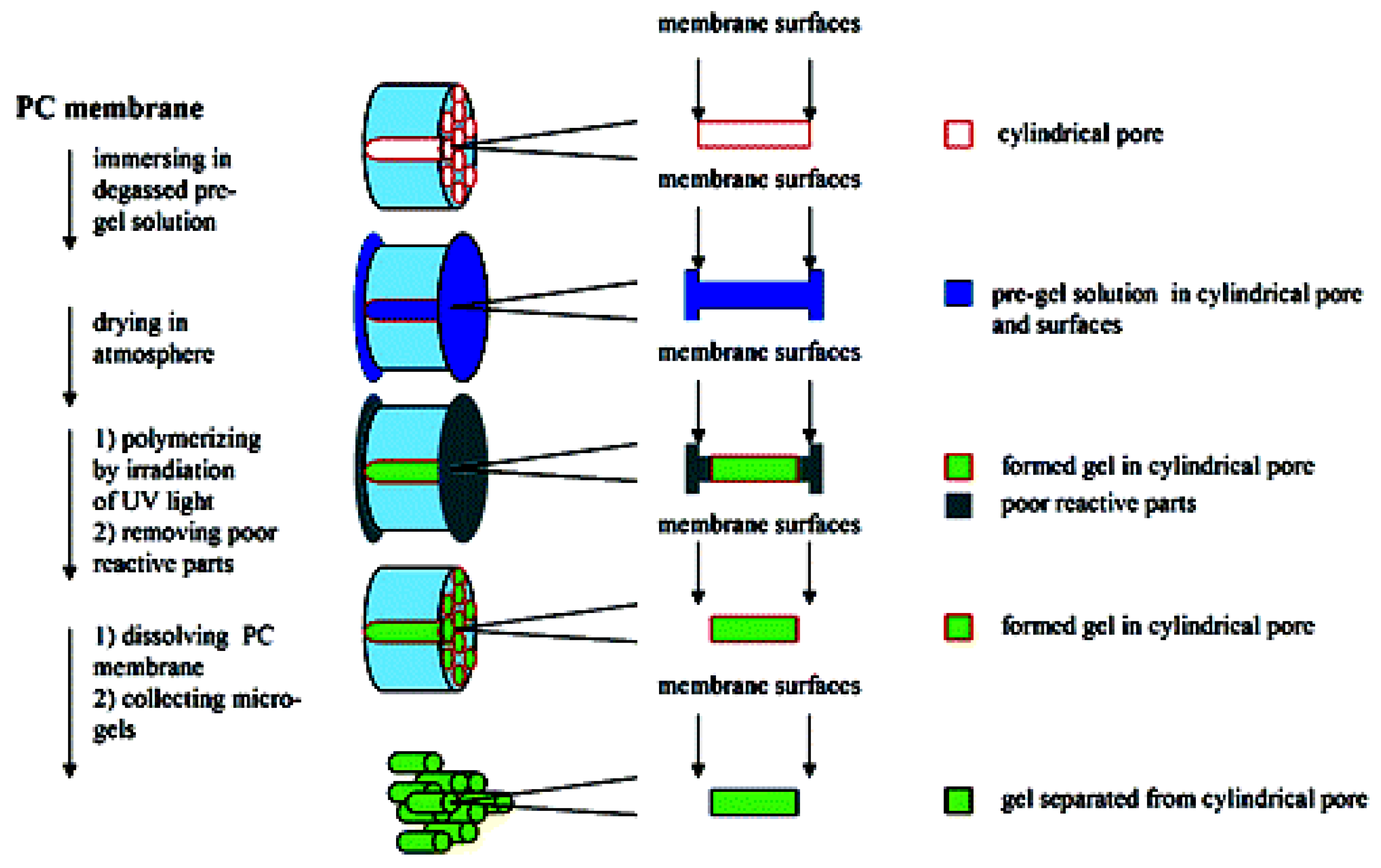
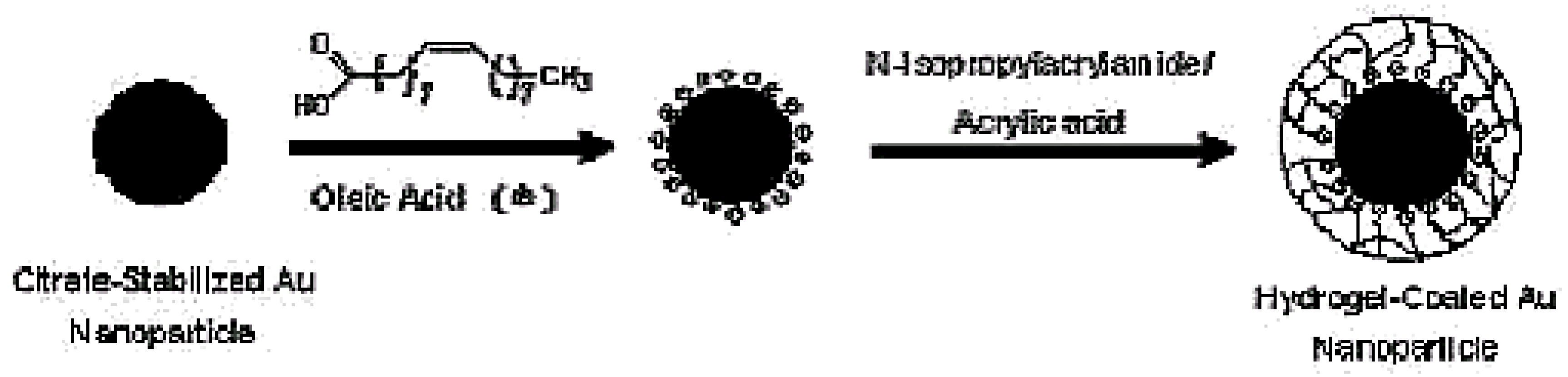

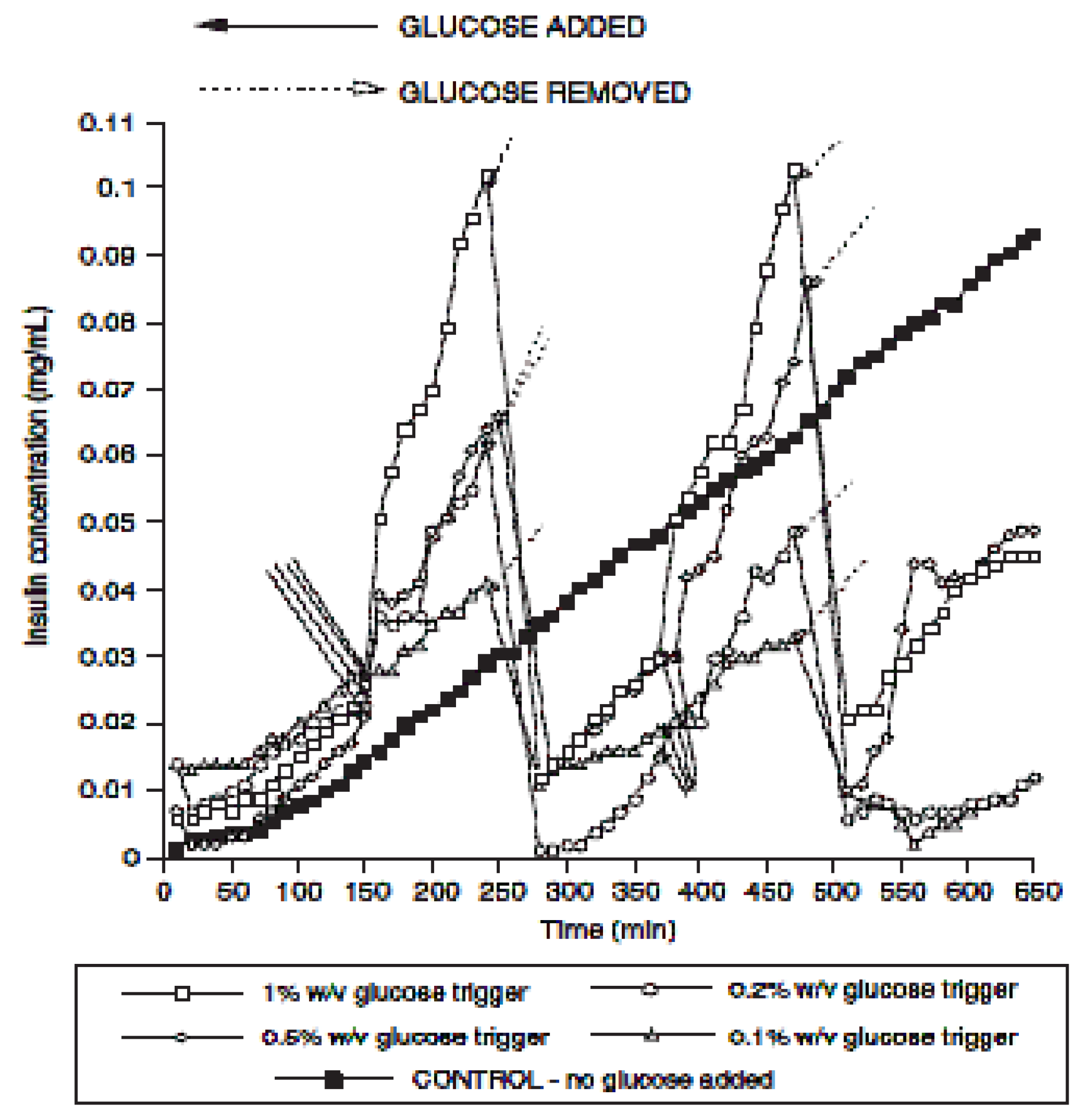
2.1.1. pNIPAm Hydrogels for Drug delivery in Treatment for Diabetes
2.1.2. pNIPAm Hydrogels as Actuators
2.1.3. pNIPAm Hydrogels in Ocular and Lens Applications
2.2. Polyethylene glycol (PEG) Based Hydrogels in Biosensing, and Drug Delivery
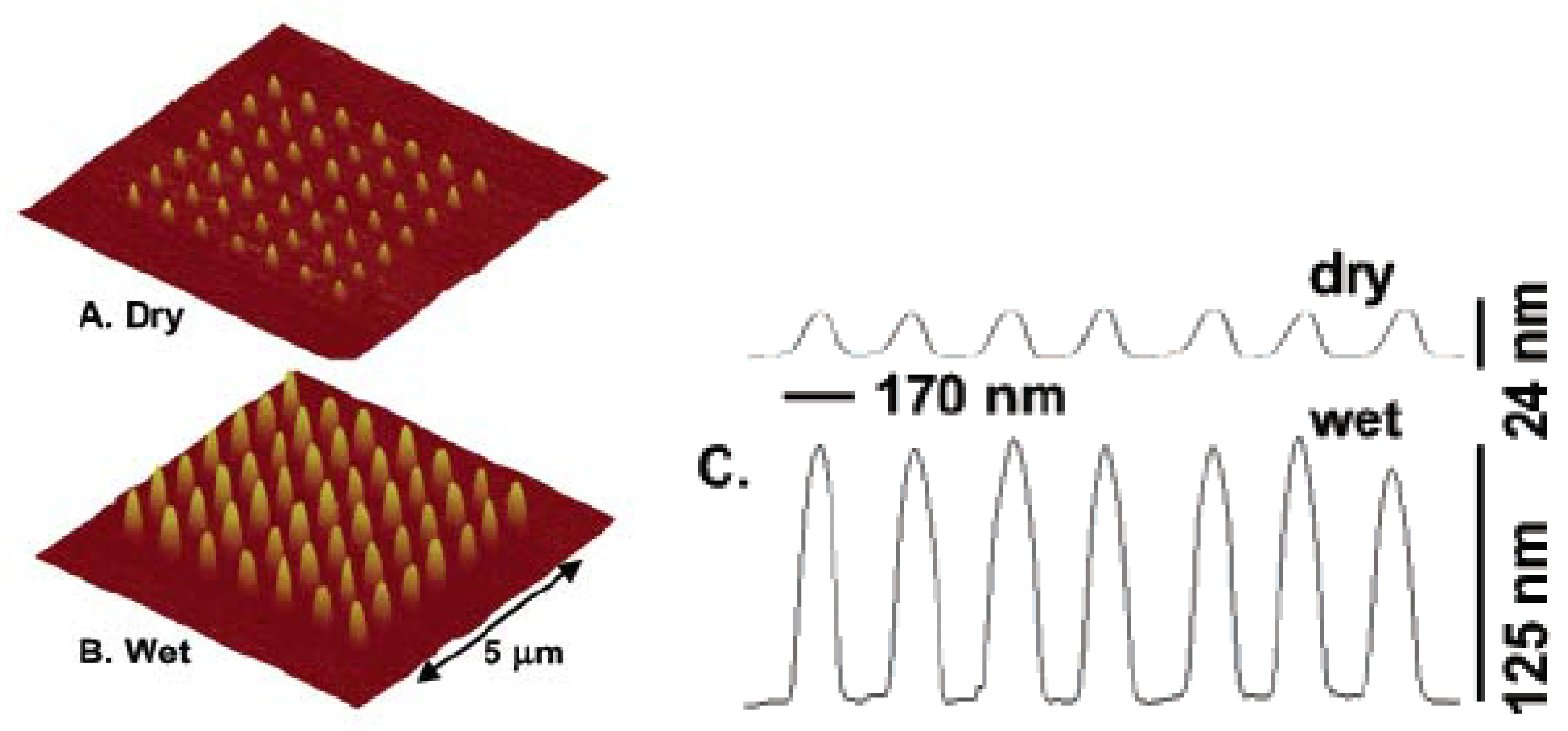
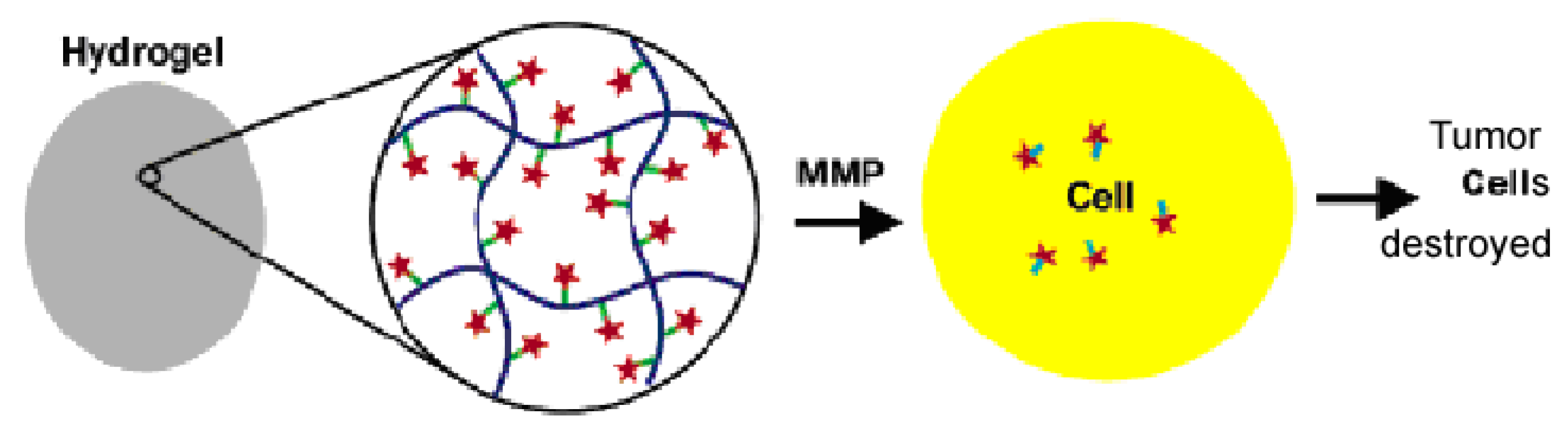
2.2.1. Polyethylene glycol (PEG) Based Hydrogels in Tissue Engineering

2.3. Polyphosphazenes Hydrogels in Drug Delivery
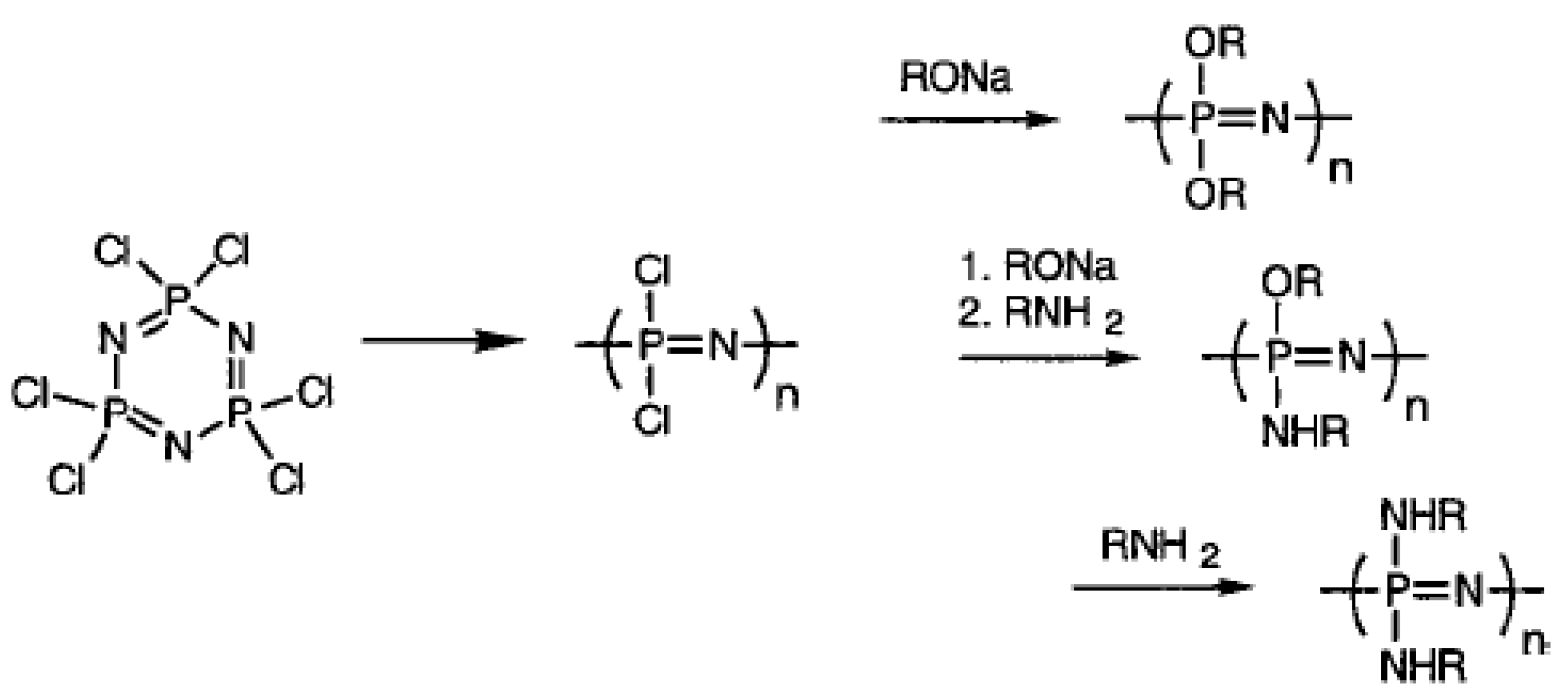
2.4. Acrylic Acid Hydrogels for Drug Delivery in Treatment of Diabetes
2.5. Poly(methacrylic acid) (PMAA ) in Cancer Treatment and Drug Delivery
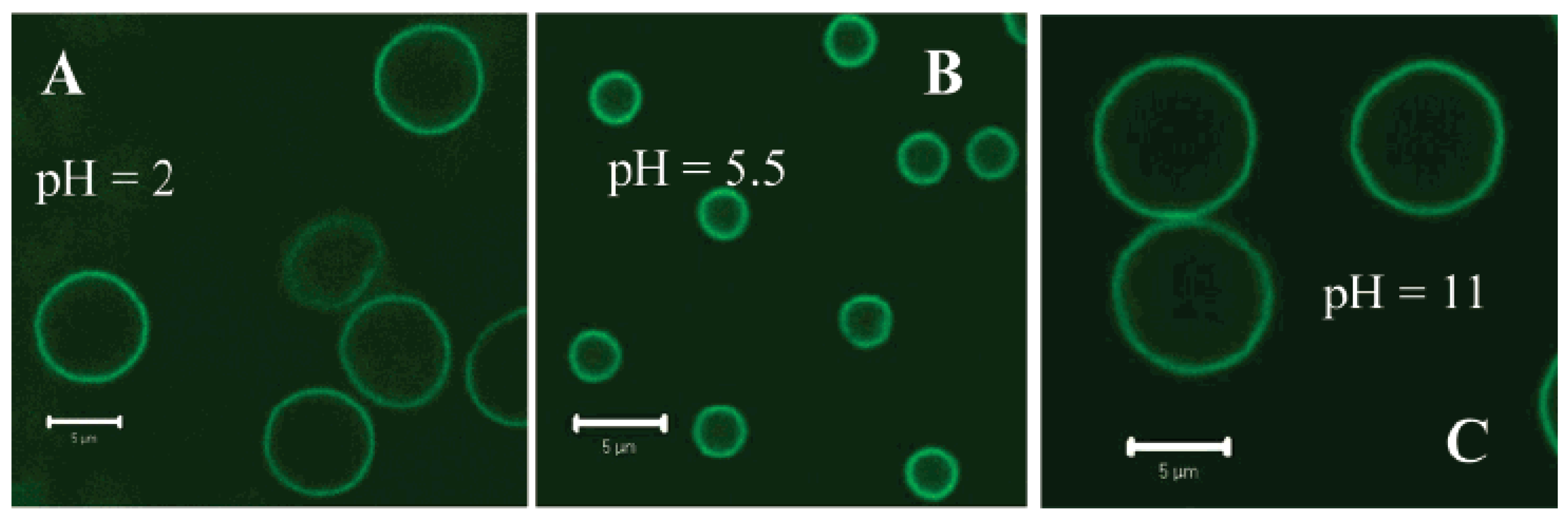
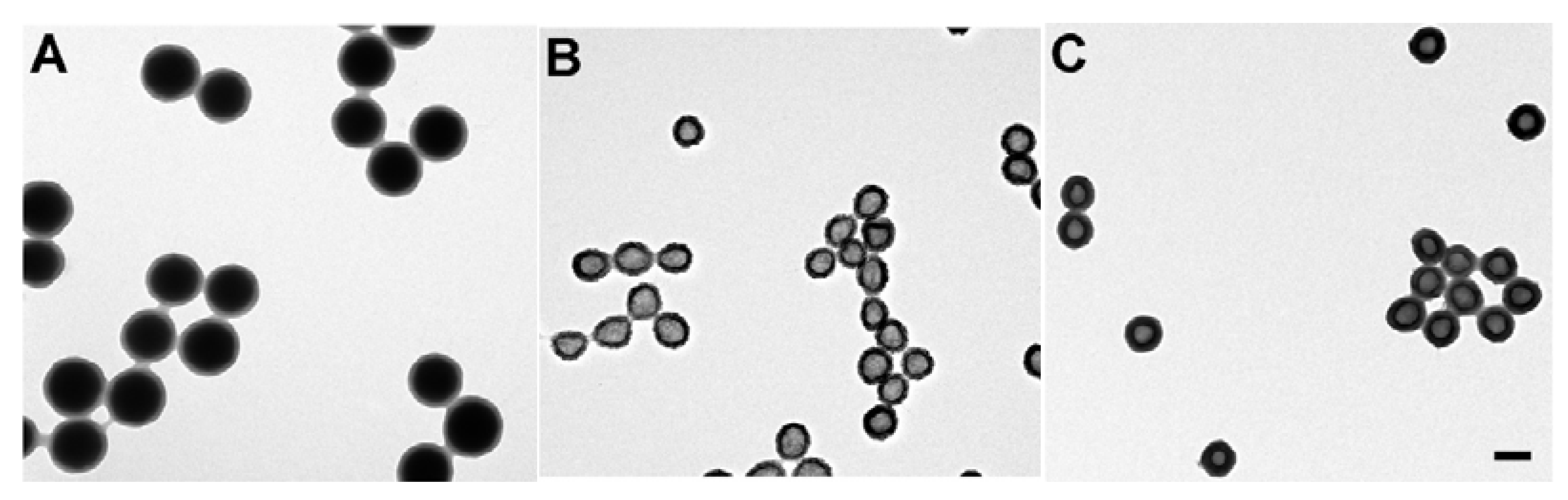
2.6. Polyvinylamine Hydrogels in Drug Delivery
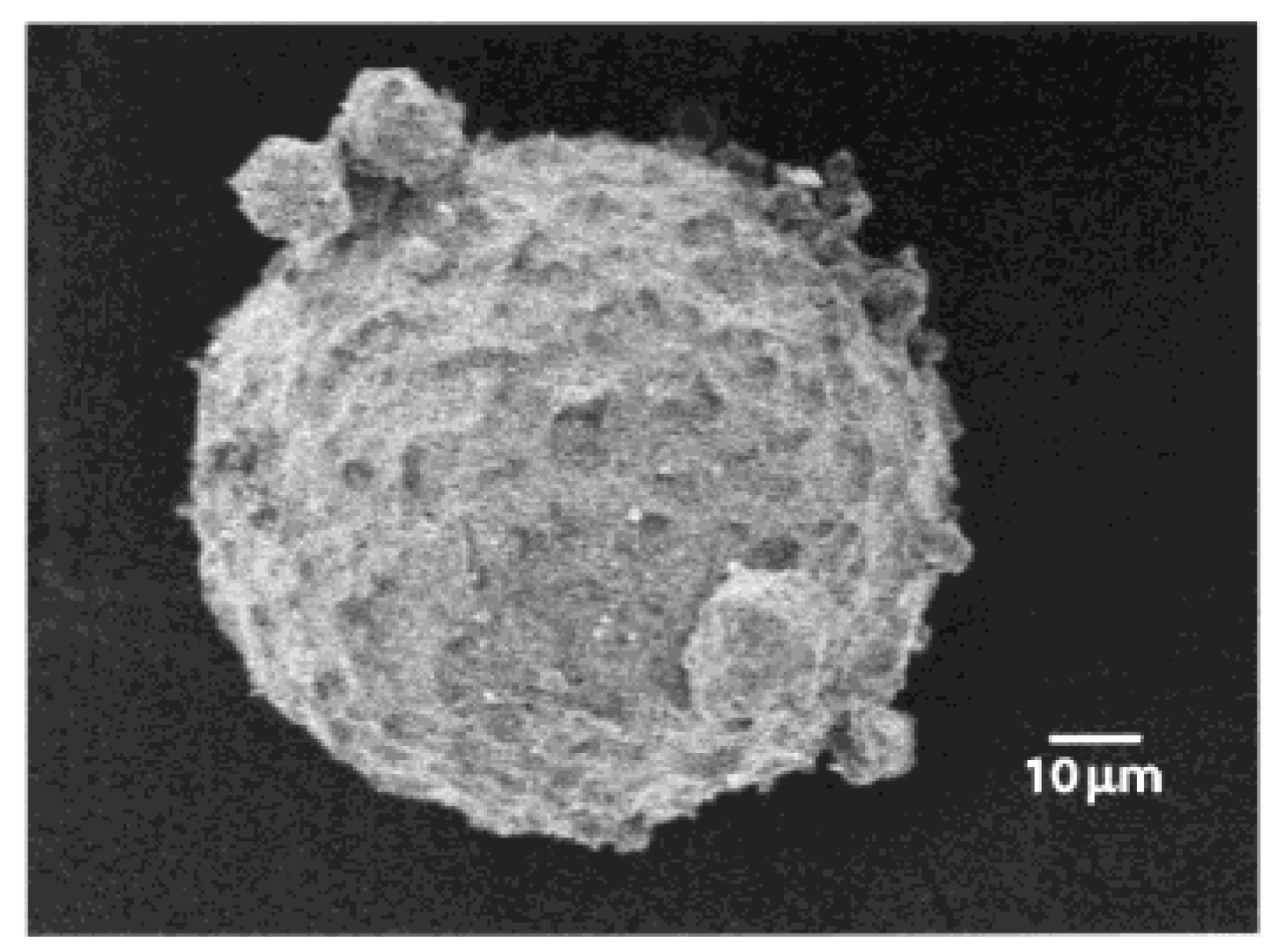
2.7. Poly (adipic anhydride) Microgels in Drug Delivery
2.8. Poly(N-vinyl formamide)in Enzyme Encapsulation and Drug Delivery
2.9. Pluronics in Drug Delivery
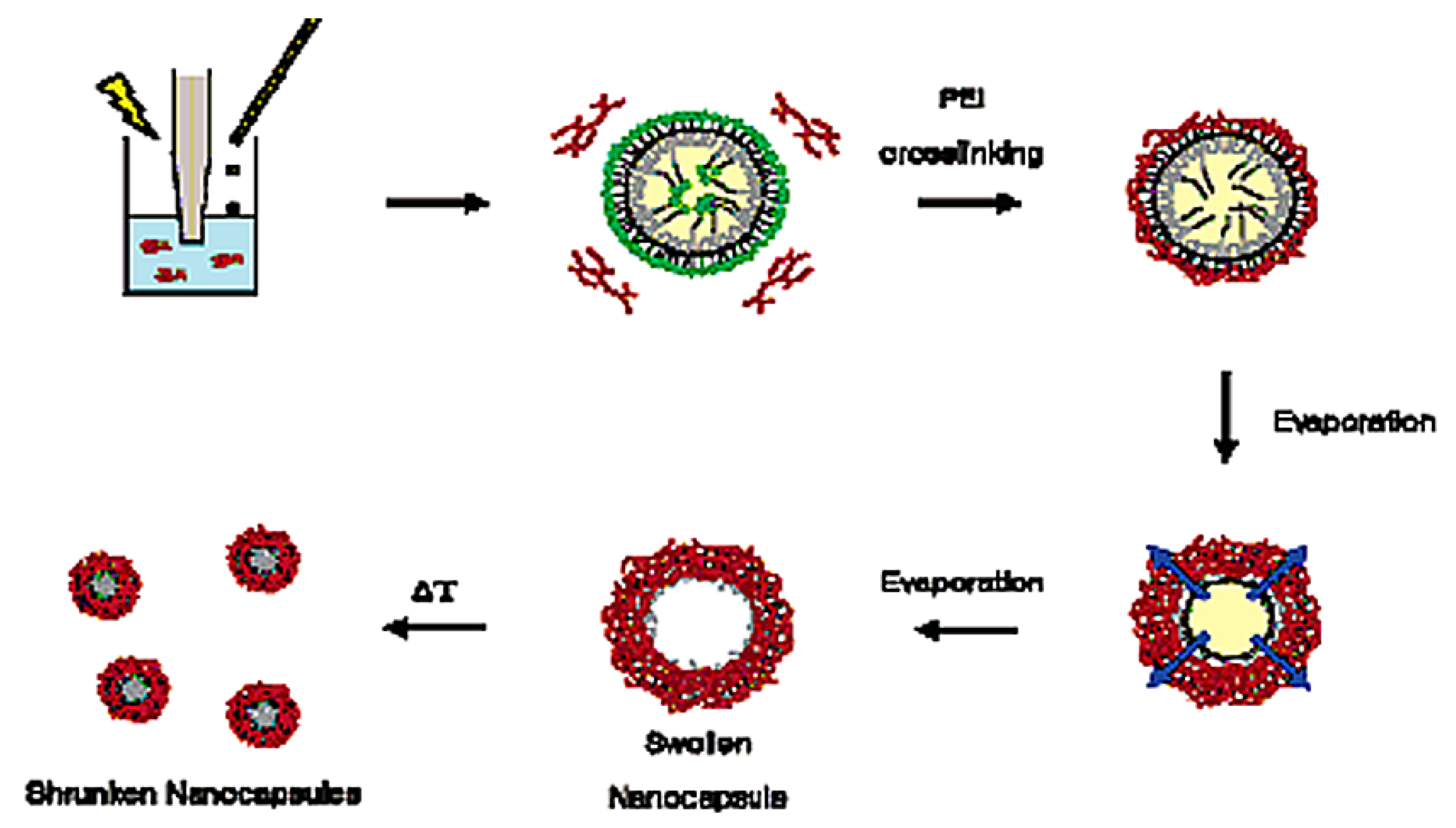
3. Supramolecular Hydrogels in Drug Delivery

4. Carbohydrate Hydrogels in Biomedicine
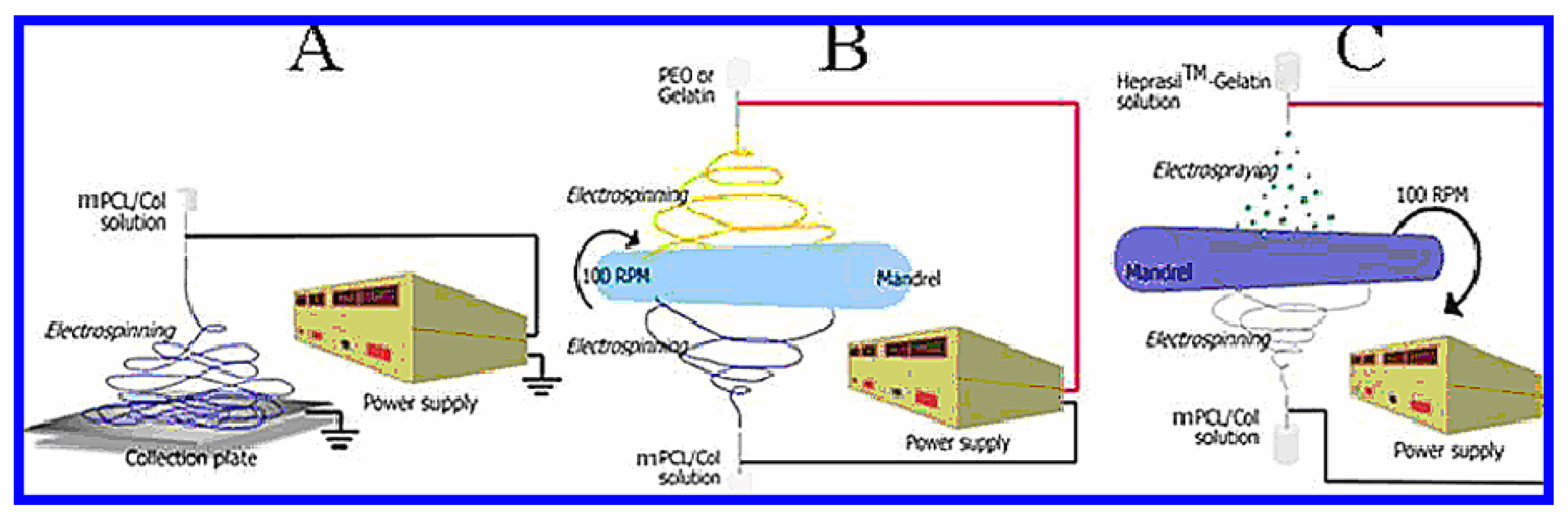
5. Peptide Hydrogels


6. Dendrimers in Drug Delivery
7. Toxicity Studies of Hydrogels
8. Future Perspectives and Concluding Remarks
References and Notes
- Wang, S. Ordered mesoporous materials for drug delivery. Microp. Mesop. Mater. 2009, 117, 1–9. [Google Scholar] [CrossRef]
- Zhang, H.; Mardyani, S.; Chan, W.C.W.; Kumacheva, E. Design of biocompatible chitosan microgels for targeted pH-mediated intracellular release of cancer therapeutics. Biomacromolecules 2006, 7, 1568–1572. [Google Scholar] [CrossRef] [PubMed]
- Ethirajan, A.; Schoeller, K.; Musyanovych, A.; Ziener, U.; Landfester, K. Synthesis and optimization of gelatin nanoparticles using the miniemulsion process. Biomacromolecules 2008, 9, 2383–2389. [Google Scholar] [CrossRef] [PubMed]
- Bertholon, I.; Hommel, H.; Labarre, D.; Vauthier, C. Properties of polysaccharides grafted on nanoparticles investigated by EPR. Langmuir 2006, 22, 5485–5490. [Google Scholar] [CrossRef] [PubMed]
- Wenk, E.; Wandrey, A.J.; Merkle, H.P.; Meinely, L. Silk fibroin spheres as a platform for controlled drug delivery. J. Contr. Rel. 2008, 132, 26–34. [Google Scholar] [CrossRef]
- Prato, M.; Kostarelos, K.; Bianco, A. Functionalized carbon nanotubes in drug design and discovery. Acc. Chem. Res. 2008, 41, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Lee, J.; Kang, J.; Lee, K.; Suh, J.; Yoon, H.; Huh, Y.; Haam, S. Hollow silica nanocontainers as drug delivery vehicles. Langmuir 2008, 24, 3417–3421. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, S.; Flynn, P.; Tseng, Y.; Yu, Y.B. Electrostatically controlled hydrogelation of oligopeptides and protein entrapment. Chem. Mater. 2005, 17, 6583–6588. [Google Scholar] [CrossRef]
- Li, C.; Tang, Y.; Armes, S.P. Synthesis and characterization of biocompatible thermo-responsive gelators based on ABA triblock copolymers. Biomacromolecules 2005, 6, 994–999. [Google Scholar] [CrossRef] [PubMed]
- Descamps, M.; Hornez, J.C.; Leriche, A. Manufacture of hydroxyapatite beads for medical applications. J. Eur. Ceram. Soc. 2009, 29, 369–375. [Google Scholar] [CrossRef]
- Zhao, Q.; Li, B. pH-controlled drug loading and release from biodegradable microcapsules. Nanomed. Nanotechnol. Biol. Med. 2008, 4, 302–310. [Google Scholar] [CrossRef]
- Bachelder, E.M.; Beaudette, T.T.; Broaders, K.E.; Dashe, J.; Freéchet, J.M.J. Acetal-derivatized dextran: An acid-responsive biodegradable material for therapeutic applications. J. Am. Chem. Soc. 2008, 130, 10494–10495. [Google Scholar] [CrossRef] [PubMed]
- Heffernan, M.J.; Murthy, N. Polyketal Nanoparticles: A new pH sensitive biodegradable Drug Delivery Vehicle. Bioconjug. Chem. 2005, 16, 1340–1342. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.C.; Bhide, M.; Crispe, I.N.; Pierce, R.H.; Murthy, N. Polyketal copolymers: A new acid-sensitive delivery vehicle for treating acute inflammatory disease. Bioconjug. Chem. 2008, 19, 1164–1169. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Maheshwari, R.; Kiick, K.L. Polymer-based therapeutics. Macromolecules 2009, 42, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Brookes, S.; Biessels, P.; Ng, N.F.L.; Woods, C.; Bell, D.N.; Adamson, G. Synthesis and characterization of a hemoglobin-ribavirin conjugate for targeted drug delivery. Bioconjug. Chem. 2006, 17, 530–537. [Google Scholar] [CrossRef] [PubMed]
- Temming, K.; Meyer, D.L.; Zabinski, R.; Dijkers, E.C.F.; Poelstra, K.; Molema, G.; Kok, R.J. Evaluation of RGD-targeted albumin carriers for specific delivery of auristatin E to tumor blood vessels. Bioconjug. Chem. 2006, 17, 1385–1394. [Google Scholar] [CrossRef] [PubMed]
- Temming, K.; Meyer, D.L.; Zabinski, R.; Senter, P.D.; Poelstra, K.; Molema, G.; Kok, R.J. Improved efficacy of αvβ3-targeted albumin conjugates by conjugation of a novel auristatin derivative. Mol. Pharm. 2007, 4, 686–694. [Google Scholar] [CrossRef] [PubMed]
- Xiong, X.; Mahmud, A.; Uludag, H.; Lavasanifar, A. Conjugation of arginine-glycine-aspartic acid peptides fo poly(ethylene oxide)-b-poly(ε-caprolactone) micelles for enhanced intracellular drug delivery to metastatic tumor cells. Biomacromolecules 2007, 8, 874–884. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Sun, X.; Chiu, J.; Sun, H.; He, Q. Transferrin-mediated gold nanoparticle cellular uptake. Bioconjug. Chem. 2005, 16, 494–496. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Wu, J.; Wu, J.; Gonit, M.; Yang, X.; Lee, A.; Xiang, G.; Li, H.; Liu, S.; Marcucci, G.; Ratnam, M.; Lee, R.J. Role of formulation composition in folate receptor-targeted liposomal doxorubicin delivery to Acute Myelogenous Leukemia cells. Mol. Pharm. 2007, 4, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, P.; Gupta, R.B. Supercritical CO2 based production of magnetically responsive micro- and nanoparticles for drug targeting. Ind. Eng. Chem. Res. 2002, 41, 6049–6058. [Google Scholar] [CrossRef]
- Backer, M.V.; Aloise, R.; Przekop, K.; Stoletov, K.; Backer, J.M. Molecular vehicles for targeted drug delivery. Bioconjug. Chem. 2002, 13, 462–467. [Google Scholar] [CrossRef] [PubMed]
- Ambade, A.V.; Savariar, E.N.; Thayumanavan, S. Dendrimeric micelles for controlled drug release and targeted delivery. Mol. Pharm. 2005, 2, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Wang, T.; Lu, B.; Xu, X.; Cheng, S.; Jiang, X.; Zhang, X.; Zhuo, R. Fabrication of supramolecular hydrogels for drug delivery and stem cell encapsulation. Langmuir 2008, 24, 10306–10312. [Google Scholar] [CrossRef] [PubMed]
- van de Manakker, F.; van der Pot, M.; Vermonden, T.; van Nostrum, C.F.; Hennink, W.E. Self-assembling hydrogels based on β-Cyclodextrin/cholesterol inclusion complexes. Macromolecules 2008, 41, 1766–1773. [Google Scholar]
- Wang, D.; Williams, C.G.; Li, Q.; Sharma, B.; Elisseeff, J.H. Synthesis and characterization of a novel degradable phosphate-containing hydrogel. Biomaterials 2003, 24, 3969–3980. [Google Scholar] [CrossRef] [PubMed]
- Jacchetti, E.; Emilitri, E.; Rodighiero, S.; Indrieri, M.; Gianfelice, A.; Lenardi, C.; Podestaà, A.; Ranucci, E.; Ferruti, P.; Milani, P. Biomimetic poly(amidoamine) hydrogels as synthetic materials for cell culture. J. Nanobiotechnol. 2008, 6. [Google Scholar] [CrossRef]
- Jansson, P.E.; Lindberg, B.; Sanford, P.A. Structural studies of gellan gum, an extracellular polysaccharide elaborated by Pseudomonas elodea. Carbohydr. Res. 1983, 124, 135–139. [Google Scholar] [CrossRef]
- Kurisawa, M.; Chung, J.E.; Yang, Y.Y.; Gao, S.J.; Uyama, H. Injectable biodegradable hydrogels composed of hyaluronic acid-tyramine conjugates for drug delivery and tissue engineering. Chem. Commun. 2005, 4312–4314. [Google Scholar] [CrossRef]
- Jia, X.; Burdick, J.A.; Kobler, J.; Clifton, R.J.; Rosowski, J.J.; Zeitels, S.M.; Langer, R. Synthesis and characterization of in situ cross-linkable hyaluronic acid-based hydrogels with potential application for vocal fold regeneration. Macromolecules 2004, 37, 3239–3248. [Google Scholar] [CrossRef]
- Jia, X.; Yeo, Y.; Clifton, R.J.; Jiao, T.; Kohane, D.S.; Kobler, J.B.; Zeitels, S.M.; Langer, R. Hyaluronic acid-based microgels and microgel networks for vocal fold regeneration. Biomacromolecules 2006, 7, 3336–3344. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, S.K.; Sanabria-DeLong, N.; Jemian, P.R.; Tew, G.N.; Bhatia, S.R. Micro- to nanoscale structure of biocompatible PLA-PEO-PLA hydrogels. Langmuir 2007, 23, 5039–5044. [Google Scholar] [CrossRef] [PubMed]
- Sanabria-DeLong, N.; Crosby, A.J.; Tew, G.N. Photo-cross-linked PLA-PEO-PLA hydrogels from self-assembled physical netoworks: Mechanical properties and influences of assumed constitutive relationships. Biomacromolecules 2008, 9, 2784–2791. [Google Scholar] [CrossRef]
- Blandino, A.; Maciías, M.; Cantero, D. Calcium alginate gel as encapsulation matrix for coimmobilized enzyme systems. Applied Biochemistry and Biotechnology 2003, 110, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Dai, H.; Chen, Q.; Qin, H.; Guan, Y.; Shen, D.; Hua, Y.; Tang, Y.; Xu, J. A temperature-responsive copolymer hydrogel in controlled drug delivery. Macromolecules 2006, 39, 6584–6589. [Google Scholar] [CrossRef]
- Wu, D.; Qiu, F.; Wang, T.; Jiang, X.; Zhang, X.; Zhuo, R. Toward the development of partially biodegradable and injectable thermoresponsive hydrogels for potential biomedical applications. Applied Materials and Interfaces. ACS Appl. Mater. Interfaces 2009, 1, 319–327. [Google Scholar] [CrossRef]
- Gong, Y.; Wang, C.; Lai, R.C.; Su, K.; Zhang, F.; Wang, D. An improved injectable polysaccharide hydrogel: modified gellan gum for long-term cartilage regeneration in vitro. J. Mater. Chem. 2009, 19, 1968–1977. [Google Scholar] [CrossRef]
- Matricardi, P.; Pontoriero, M.; Coviello, T.; Casadei, M.A.; Alhaique, F. In situ cross-linkable novel alginate-dextran methacrylate IPN hydrogels for biomedical applications: mechanical characterization and drug delivery properties. Biomacromolecules 2008, 9, 2014–2020. [Google Scholar] [CrossRef] [PubMed]
- Shim, W.S.; Yoo, J.S.; Bae, Y.H.; Lee, D.S. Novel injectable pH and temperature sensitive block copolymer hydrogel. Biomacromolecules 2005, 6, 2930–2934. [Google Scholar] [CrossRef]
- Langer, R.; Peppas, N.A. Advances in biomaterials, drug delivery, and bionanotechnology. AIChE J. 2003, 49, 2990–3006. [Google Scholar] [CrossRef]
- Sontjens, S.H.M.; Nettles, D.L.; Carnahan, M.A.; Setton, L.A.; Grinstaff, M.W. Biodendrimer-based hydrogel scaffolds for cartilage tissue repair. Biomacromolecules 2006, 7, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Saiani, A.; Gough, J.E.; Miller, A.F. Thermoreversible protein hydrogel as cell scaffold. Biomacromolecules 2006, 7, 2776–2782. [Google Scholar] [CrossRef] [PubMed]
- Shi, G.; Cai, Q.; Wang, C.; Lu, N.; Wang, S.; Bei, S. Fabrication and biocompatibility of cell scaffolds of poly (L-lactic acid) and poly (L-lactic-co-glycolic acid). Polym. Adv. Technol. 2002, 13, 227–232. [Google Scholar] [CrossRef]
- Xie, Y.; Yang, S.T.; Kniss, D.A. Three-dimensional cell-scaffold constructs promote efficient gene transfection: implications for cell-based gene therapy. Tissue Eng. 2001, 7, 585–598. [Google Scholar] [CrossRef] [PubMed]
- Halstenberg, S.; Panitch, A.; Rizzi, S.; Hall, H.; Hubbell, J.A. Biologically engineered protein-graft-poly(ethylene glycol) hydrogels: A cell adhesive and plasmin-degradable biosynthetic material for tissue repair. Biomacromolecules 2002, 3, 710–723. [Google Scholar] [CrossRef] [PubMed]
- Dadsetan, M.; Szatkowski, J.P.; Yaszemski, M.J.; Lu, L. Characterization of photo-cross-linked oligo[poly(ethylene glycol) fumarate] hydrogels for cartilage tissue engineering. Biomacromolecules 2007, 8, 1702–1709. [Google Scholar] [CrossRef] [PubMed]
- Franzesi, G.T.; Ni, B.; Ling, Y.; Khademhosseini, A. A controlled-release strategy for the generation of cross-linked hydrogel microstructures. J. Am. Chem. Soc. 2006, 128, 15064–15065. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Xu, K.; Wang, L.; Gu, H.; Wei, H.; Zhang, M.; Xu, B. Self-assembly of small molecules affords multifuntional supramolecular hydrogels for topically treating simulated uranium wounds. Chem. Commun. 2005, 4414–4416. [Google Scholar] [CrossRef]
- Raemdonck, K.; Demeester, J.; De Smedt, S. Advanced nanogel engineering for drug delivery. Soft Matter 2009, 5, 707–715. [Google Scholar] [CrossRef]
- Hamidi, M.; Azadi, A.; Rafiei, P. Hydrogel nanoparticles in drug delivery. Adv. Drug Deliv. Rev. 2008, 60, 1638–1649. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Gu, H.; Zhang, Y.; Wang, L.; Xu, B. Small molecule hydrogels based on a class of antiinflammatory agents. Chem. Commun. 2004, 2, 208–209. [Google Scholar] [CrossRef]
- Malmsten, M. Soft drug delivery systems. Soft Matter 2006, 2, 760–769. [Google Scholar] [CrossRef]
- Kumar, M.N.V.R. Nano and microparticles as controlled drug delivery devices. J. Pharm. Pharmaceut. Sci. 2000, 3, 234–258. [Google Scholar]
- Hoare, T.; Pelton, R. Impact of microgel morphology on functionalized microgel-drug interactions. Langmuir 2008, 24, 1005–1012. [Google Scholar] [CrossRef] [PubMed]
- Prevot, M.; Déjugnat, C.; Mohwald, H.; Sukhorukov, G.B. Behavior of temperature-sensitive pNIPAM confined in polyelectrolyte capsules. ChemPhysChem 2006, 7, 2497–2502. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.S.; Gupta, V.K. Photochromic cross-links in thermoresponsive hydrogels of poly(N-isopropylacrylamide): Enthalpic and entropic consequences on swelling behavior. J. Phys. Chem. B 2002, 106, 4127–4132. [Google Scholar] [CrossRef]
- Yan, H.; Nishino, M.; Tsuboi, Y.; Kitamura, N.; Tsujii, K. Template-guided synthesis and individual characterization of poly (N-isopropylacrylamide)-based microgels. Langmuir 2005, 21, 7076–7079. [Google Scholar] [CrossRef] [PubMed]
- Budhlall, B.M.; Marquez, M.; Velev, O.D. Microwave, photo- and thermally responsive pNIPAm-gold nanoparticles microgels. Langmuir 2008, 24, 11959–11966. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, T.R. Thermo- and pH-responsive hydrogel-coated gold nanoparticles. Chem. Mater. 2004, 16, 3647–3651. [Google Scholar] [CrossRef]
- Singh, N.; Lyon, A. Au nanoparticle templated synthesis of pNIPAm nanogels. Chem. Mater. 2007, 19, 719–726. [Google Scholar] [CrossRef]
- Kazakov, S.; Kaholek, M.; Gazaryan, I.; Krasnikov, B.; Miller, K.; Levon, K. Ion concentration of external solution as a characteristic of micro-and nanogel ionic reservoirs. J. Phys. Chem. B 2006, 110, 15107–15116. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Hu, Z. Phase behavior and stabilization of microgel arrays. Macromolecules 2007, 40, 3749–3756. [Google Scholar]
- Debord, J.D.; Lyon, L.A. Thermoresponsive photonic crystals. J. Phys. Chem. B 2000, 104, 6327–6331. [Google Scholar] [CrossRef]
- Kazakov, S.; Kaholek, M.; Teraoka, I.; Levon, K. UV-induced gelation on nanometer scale using liposome reactor. Macromolecules 2002, 35, 1911–1920. [Google Scholar] [CrossRef]
- Nolan, C.M.; Gelbaum, L.T.; Lyon, L.A. 1H NMR investigation of thermally triggered insulin release from poly(N-isopropylacrylamide) microgels. Biomacromolecules 2006, 7, 2918–2922. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Guan, Y.; Zhou, S. Synthesis and volume phase transitions of glucose-sensitive Microgels. Biomacromolecules 2006, 7, 3196–3201. [Google Scholar]
- Kato, N.; Sakai, Y.; Shibata, S. Wide-range control of deswelling time for thermosensitive poly(N-isopropylacrylamide) gel treated by freeze-drying. Macromolecules 2003, 36, 961–963. [Google Scholar] [CrossRef]
- Zhang, Y.; Guan, Y.; Zhou, S. Permeability control of glucose-sensitive nanoshells. Biomacromolecules 2007, 8, 3842–3847. [Google Scholar] [CrossRef] [PubMed]
- Hoare, T.; Pelton, R. Engineering glucose swelling responses in poly(N-isopropylacrlyamide)-based microgels. Macromolecules 2007, 40, 670–678. [Google Scholar] [CrossRef]
- Lawrence, D.B.; Cai, T.; Hu, Z.; Marquez, M.; Dinsmore, A.D. Temperature-responsive semipermeable capsules composed of colloidal microgel spheres. Langmuir 2007, 23, 395–398. [Google Scholar] [CrossRef] [PubMed]
- Sorrell, C.D.; Lyon, L.A. Bimodal swelling responses in microgel thin films. J. Phys. Chem. B 2007, 111, 4060–4066. [Google Scholar] [CrossRef] [PubMed]
- De Groot, J.H.; Spaans, C.J.; van Calck, R.V.; van Beijma, F.J.; Norrby, S.; Pennings, A.J. Hydrogels for an accommodating intraocular Lens: An explorative study. Biomacromolecules 2003, 4, 608–616. [Google Scholar] [CrossRef] [PubMed]
- Receveur, R.; Lindemans, F.W.; De Rooij, N.F. Microsystems technologies for implantable applications. J. Micromech. Microeng. 2007, 17, R50–R80. [Google Scholar] [CrossRef]
- Kim, J.; Nayak, S.; Lyon, A.L. Bioresponsive hydrogel microlenses. J. Am. Chem. Soc. 2005, 127, 9588–9592. [Google Scholar] [CrossRef]
- Allarakhia, L.; Lindstrom, R.L. Soft intraocular lenses. Int. J. Opthalmol. 1988, 12, 185–191. [Google Scholar]
- Dong, L.; Agarwal, A.K.; Beebe, D.J.; Jiang, H. Adaptive liquid microlenses activated by stimuli-responsive hydrogels. Nature 2006, 442, 551–554. [Google Scholar] [CrossRef] [PubMed]
- Verestiuc, L.; Nastasescu, O.; Barbu, E.; Sarvaiya, I.; Green, K.L.; Tsibouklis, J. Functionalized chitosan/NIPAM (HEMA) hybrid polymer networks as inserts for ocular drug delivery: Synthesis, in vitro assessment, and in vivo evaluation. J. Biomed Mater. Res. A 2006, 77A, 726–735. [Google Scholar] [CrossRef] [PubMed]
- DeLouise, L.A.; Fauchet, P.M.; Miller, B.L.; Pentland, A.A. Hydrogel-supported optical-microcavity sensors. Adv. Mater. 2005, 17, 2199–2203. [Google Scholar] [CrossRef]
- Mack, N.H.; Wackerly, J.M.; Malyarchuk, V.; Rogers, J.A.; Moore, J.S.; Nuzzo, R.G. Optical transduction of chemical forces. Nano Lett. 2007, 7, 733–737. [Google Scholar] [CrossRef]
- Yoshimura, I.; Miyahara, Y.; Kasagi, N.; Yamane, H.; Ojida, A.; Hamachi, I. Molecular recognition in a supramolecular hydrogel to afford a semi-wet sensor chip. J. Am. Chem. Soc. 2004, 126, 12204–12205. [Google Scholar]
- Frisk, M.L.; Tepp, W.H.; Lin, G.; Johnson, E.A.; Beebe, D.J. Substrate-modified hydrogels for autonomous sensing bolulinum neurotoxin Type A. Chem. Mater. 2007, 19, 5842–5844. [Google Scholar] [CrossRef]
- Hong, Y.; Krsko, P.; Libera, M. Protein surface patterning using nanoscale PEG hydrogels. Langmuir 2004, 20, 11123–11126. [Google Scholar] [CrossRef] [PubMed]
- An, S.Y.; Bui, M.N.; Nam, Y.J.; Han, K.N.; Li, C.A.; Choo, J.; Lee, E.K.; Katoh, S.; Kumada, Y.; Seong, G.H. Preparation of monodisperse and size-controlled poly(ethylene glycol) hydrogel nanoparticles using liposome templates. J. Colloid Interface Sci. 2009, 331, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Peppas, N.A.; Kavimandan, N.J. Nanoscale analysis of protein and peptide absorption: Insulin absorption using complexation and pH-sensitive hydrogels as delivery vehicles. Eur. J. Pharm. Sci. 2006, 29, 183–197. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, B.; Yadavalli, V.K.; Pishko, M.V. Encapsulation of enzymes within polymer spheres to create optical nanosensors for oxidative stress. Anal. Chem. 2005, 77, 6828–6833. [Google Scholar] [CrossRef] [PubMed]
- Tauro, J.R.; Gemeinhart, R.A. Extracellular protease activation of chemotherapeutics from hydrogel matrices: A new paradigm for local chemotherapy. Mol. Pharm. 2005, 2, 435–438. [Google Scholar] [CrossRef] [PubMed]
- Tauro, J.R.; Gemeinhart, R.A. Matrix metalloprotease triggered delivery of cancer chemotherapeutics from hydrogel matrixes. Bioconjug. Chem. 2005, 16, 1133–1139. [Google Scholar] [CrossRef] [PubMed]
- Koh, W.; Revzin, A.; Pishko, M.V. Poly(ethylene glycol) hydrogel microstructures encapsulating living cells. Langmuir 2002, 18, 2459–2462. [Google Scholar]
- Lensen, M.C.; Mela, P.; Mourran, A.; Groll, J.; Heuts, J.; Rong, H.; Möller. Micro- and nanopatterned star poly(ethylene glycol) (PEG) materials prepared by UV-based imprint lithography. Langmuir 2007, 23, 7841–7846. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Kim, P.; Song, I.; Cha, J.M.; Lee, S.H.; Kim, B.; Suh, K.Y. Guided three-dimensional growth of functional cardiomyocytes on polyethylene glycol nanostructures. Langmuir 2006, 22, 5419–5426. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Bae, Y.H.; Sohn, Y.S.; Jeong, B. Thermogelling aqueous solutions of alternating multiblock copolymers of poly(L-lactic acid) and poly(ethylene glycol). Biomacromolecules 2006, 7, 1729–1734. [Google Scholar] [CrossRef] [PubMed]
- Allcock, H. R. Biodegradable Polymers as Drug Delivery Systems; Chasin, M., Langer, R., Eds.; Marcel Dekker: New York, NY, USA, 1990. [Google Scholar]
- Andrianov, A.K.; Payner, L.G.; Visscher, K.B.; Allcock, H.R.; Langer, R. Hydrolytic degradation of ionically cross-linked polyphosphazene microspheres. J. Appl. Polym. Sci. 1994, 53, 1573–1578. [Google Scholar] [CrossRef]
- Cohen, S.; Bano, M.C.; Visscher, K.B.; Chow, M.; Allcock, H.; Langer, R. Ionically crosslinkable polyphosphazene: a novel polymer for microencapsulation. J. Am. Chem. Soc. 1990, 112, 7832–7833. [Google Scholar] [CrossRef]
- Greish, Y.E.; Bender, J.D.; Lakshmi, S.; Brown, P.W.; Allock, H.R.; Laurencin, C.T. Low temperature formation of hydroxyapatite-poly (alkyl oxybenzoate) phosphazene composites for biomedical applications. Biomaterials 2005, 26, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Vandorpe, J.; Schacht, E.; Dunn, S.; Hawley, A.; Stolnik, S.; Davis, S.S.; Garnett, M.C.; Davies, M.C.; Illum, L. Long circulating biodegradable poly(phosphazene) nanoparticles surface modified with poly(phosphazene)-poly(ethylene oxide) copolymer. Biomaterials. 1997, 18, 1147–1152. [Google Scholar] [CrossRef] [PubMed]
- Hoare, T.; Pelton, R. Charge-switching, amphoteric glucose-responsive microgels with physiological swelling activity. Biomacromolecules 2008, 9, 733–740. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.Y.; Shao, Y.H.; Lü, J. Preparation, properties and controlled release behaviors of pH- induced thermosensitive amphiphilic gels. Biomaterials. 2006, 27, 4016–4024. [Google Scholar] [CrossRef] [PubMed]
- Tartakovsky, A.; Drutis, D.M.; Carnali, J.O. The adsorption of cationic and amphoteric copolymers on glass surfaces: zeta potential measurements, adsorption isotherm determination, and FT Raman characterization. J. Colloid Interface Sci. 2003, 363, 408–419. [Google Scholar] [CrossRef]
- Cavalieri, F.; Chiessi, E.; Villa, R.; Vigano, L.; Zaffaroni, N.; Telling, M.F.; Paradossi, G. Novel PVA-based hydrogel microparticles for doxorubicin delivery. Biomacromolecules 2008, 9, 1967–1973. [Google Scholar] [CrossRef] [PubMed]
- Kiser, P.F.; Wilson, G.; Needham, D. A synthetic mimic of the secretory granule for drug delivery. Nature 1998, 394, 459–462. [Google Scholar] [CrossRef] [PubMed]
- Kozlovskaya, V.; Sukhishvili, S.A. Amphoteric hydrogel capsules: Multiple encapsulation and release routes. Macromolecules 2006, 39, 6191–6199. [Google Scholar] [CrossRef]
- Shi, L.; Berkland, C. Acid-labile polyvinylamine micro- and nanogel capsules. Macromolecules 2007, 40, 4635–4643. [Google Scholar] [CrossRef] [PubMed]
- Dinarvand, R.; Alimorad, M.M.; Amanlou, M.; Akbari, H. In vitro release of clomipramine HCl and buprenorphine HCl from poly adipic anhydride (PAA) and poly trimethylene carbonate (PTMC) blends. J. Biomed. Mater. Res. A 2005, 75A, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Khondee, S.; Linz, T.H.; Berkland, C. Poly (N-vinylformamide) nanogels capable of pH-sensitive protein release. Macromolecules 2008, 41, 6546–6554. [Google Scholar] [CrossRef]
- Maibaum, L.; Dinner, A.R.; Chandler, D. Micelle formation and the hydrophobic effect. J. Phys. Chem. B 2004, 108, 6778–6781. [Google Scholar]
- Bromberg, L.; Temchenko, M.; Hatton, T.A. Dually responsive microgels from polyether-modified poly(acrylic acid): Swelling and drug loading. Langmuir 2002, 18, 4944–4952. [Google Scholar] [CrossRef]
- Choi, S.H.; Lee, S.H.; Park, T.G. Temperature-sensitive pluronic/poly(ethylenimine) nanocapsules for thermally triggered disruption of intracellular endosomal compartment. Biomacromolecules 2006, 7, 1864–1870. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Xu, B. Supramolecular hydrogels based on biofunctional nanofibers of self-assembled small molecules. J. Mater. Chem. 2007, 17, 2385–2393. [Google Scholar] [CrossRef]
- Huang, H.; Remsen, E.E.; Wooley, K.L. Amphiphilic core-shell nanospheres obtained by intramicellar shell cross-linking of polymer micelles with poly(ethylene oxide) linkers. Chem. Commun. 1998, 1415–1416. [Google Scholar] [CrossRef]
- Ray, S.; Das, A.K.; Banerjee, A. pH-responsive, bolaamphiphile-based smart metallo-hydrogels as potential dye-absorbing agents, water purifier, and vitamin B12 carrier. Chem. Mater. 2007, 19, 1633–1639. [Google Scholar]
- Taylor, M.J.; Tanna, S.; Sahota, T.S.; Voermans, B. Rhelogical characterization of dextran-concanavalin A mixtures as a basis for a self-regulated drug delivery device. Eur. J. Pharm. Biopharm. 2006, 62, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Tanna, S.; Taylor, M.J.; Sahota, T.S.; Sawicka, K. Glucose-responsive UV polymerised dextran–Concanavalin A Acrylic Derivatized Mixtures for Closed-Loop Insulin Delivery. Biomaterials 2006, 27, 1586–1597. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Hu, J.; Pan, X.; Yao, P.; Jiang, M. Stable and pH-sensitive nanogels prepared by self-assembly of chitosan and ovalbumin. Langmuir 2006, 22, 2754–2759. [Google Scholar] [CrossRef] [PubMed]
- Muzzarelli, R.A.; Biagini, G.; Bellardini, M.; Simonelli, L.; Castaldini, C.; Fraatto, G. Osteoconduction exerted by methylpyrolidinone chitosan in dental surgery. Biomaterials 1993, 4, 39–43. [Google Scholar] [CrossRef]
- Feng, Z.Q.; Chu, X.; Huang, N.P.; Wang, T.; Wang, Y.; Shi, X.; Ding, Y.; Gu, Z.Z. The effect of nanofibrous galactosylated chitosan scaffolds on the formation of rat primary hepatocyte aggregates and the maintenance of liver function. Biomaterials 2009, 30, 2753–2763. [Google Scholar] [CrossRef]
- Illum, L. Chitosan and its use as a pharmaceutical excipient. Pharm. Res. 1998, 15, 1326–1331. [Google Scholar] [CrossRef] [PubMed]
- Henricus, M.M.; Fath, K.R.; Menzenski, M.Z.; Banerjee, I.A. Morphology controlled growth of chitosan-bound microtubes and a study of their biocompatibility and antibacterial activity. Macromol. Biosci. 2009, 9, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Azmi, N.E.; Ahmad, M.; Abdullah, J.; Sidek, H.; Heng, L.Y.; Karuppiah, N. Biosensor based on glutamate dehydrogenase immobilized in chitosan for the determination of ammonium in water samples. Anal. Biochem. 2009, 388, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.C.; Chen, S.Y.; Lee, C.A. Amperometric cholesterol biosensors on carbon nanotube- chitosan-platinum cholesterol oxidase nanobiocomposite. Sens. Actuat. B 2008, 135, 96–101. [Google Scholar] [CrossRef]
- Yi, H.; Wu, L.; Bently, W.E.; Ghodssi, R.; Rubloff, G.W.; Culver, J.N.; Payne, G.F. Biofabrication with Chitosan. Biomacromolecules 2005, 6, 2881–2894. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Mardyani, S.; Chan, W.C.W.; Kumacheva, E. Design of biocompatible chitosan microgels for targeted pH-mediated intracellular release of cancer therapeutics. Biomacromolecules 2006, 7, 1568–1572. [Google Scholar] [CrossRef] [PubMed]
- Brittain, H.G. Analytical Profiles of Drug Substances; Academic Press: California, USA, 1986; Vol. 5, pp. 284–306. [Google Scholar]
- Nishikawa, T.; Akiyoshi, K.; Sunamoto, J. Macromolecules complexation between bovine serum albumin and the self-assembled hydrogel nanoparticles of hydrophobicized polysaccharides. J. Am. Chem. Soc. 1996, 118, 6110–6115. [Google Scholar] [CrossRef]
- Akiyoshi, K.; Nishikawa, T.; Mitsui, Y.; Miyata, T.; Michiko, K.; Sunamato, J. Self-assembly of polymer amphiphiles: thermodynamics of complexation between BSA and self-aggregate of cholesterol bearing pullulan. Colloids Surf. A 2006, 112, 94–100. [Google Scholar]
- Nishikawa, T.; Akiyoshi, K.; Sunamoto, J. Supramolecular assembly between nanoparticles of hydrophobized polysaccharide and Soluble Protein complexation between the self-aggregate of cholesterol-bearing pullulan and α-chymotrypsin. Macromolecules 1994, 27, 7654–7659. [Google Scholar] [CrossRef]
- Akiyoshi, K.; Nishikawa, A.; Shichibe, S.; Sunamoto, J. Stabilization of insulin upon supramolecular complexation with hydrophobized polysaccharide nanoparticle. Chem. Lett. 1995, 24, 707–708. [Google Scholar] [CrossRef]
- Akiyoshi, K.; Sasaki, Y.; Sunamoto, J. Molecular chaperone-like activity of hydrogel nanoparticles of hydrophobicized pullulan: thermal stabilization with refolding of carbonic anhydrase B. Bioconjug. Chem. 1999, 10, 321–324. [Google Scholar] [CrossRef] [PubMed]
- Ayame, H.; Morimoto, N.; Akiyoshi, K. Self-assembled cationic nanogels for intracellular protein delivery. Bioconjug. Chem. 2008, 19, 882–890. [Google Scholar] [CrossRef] [PubMed]
- Na, K.; Lee, E.S.; Bae, Y.H. Self-organized nanogels responding to tumor extracellular pH: pH-dependent drug release and in vitro cytotoxicity against MCF-7 cells. Bioconj. Chem. 2007, 18, 1568–1574. [Google Scholar] [CrossRef]
- Sannino, A.; Madaghiele, M.; Conversano, F.; Mele, G.; Maffezzoli, A.; Netti, P.A.; Ambrosio, L.; Nicolais, L. Cellulose derivative-hyaluronic acid-based microporous hydrogels cross-linked through divinyl sulfone (DVS) to modulate equilibrium sorption capacity and network stability. Biomacromolecules 2004, 5, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Ekaputra, A.K.; Prestwich, G.D.; Cool, S.M.; Hutmacher, D.W. Combining electrospun scaffolds with electrosprayed hydrogels leads to three-dimensional cellularization of hybrid constructs. Biomacromolecules 2008, 9, 2097–2103. [Google Scholar] [CrossRef] [PubMed]
- Nukavarapu, S.P.; Kumbar, S.G.; Nair, L.S.; Laurencin, C.T. Biomedical Nanostructures; Gonsalves, K.E., Halberstadt, C.R., Laurencin, C.T., Nair, L.S., Eds.; Wiley & Sons, Inc.: Hoboken, NJ, USA, 2008; Chapter 15; p. 397. [Google Scholar]
- Shin, M.K.; Kim, S.I.; Kim, S.J. Controlled magnetic nanofiber hydrogels by clustering ferritin. Langmuir 2008, 24, 12107–12111. [Google Scholar] [CrossRef] [PubMed]
- Coradin, T.; Livage, J. Aqueous silicates in biological sol-gel applications: new perspectives for old precursors. Acc. Chem. Res. 2007, 40, 819–826. [Google Scholar] [CrossRef] [PubMed]
- Sidorenko, A.; Krupenkin, T.; Taylor, A.; Fratzl, P.; Aizenberg, J. Reversible switching of hydrogel-actuated nanostructures into complex micropatterns. Science 2007, 315, 487–490. [Google Scholar] [CrossRef] [PubMed]
- Tomalia, D.A.; Baker, H.; Dewald, J.R.; Hall, M.; Kallos, G.; Martin, S.; Roeck, J.; Ryder, J.; Smith, P. A new class of polymers: starbust-dendritic macromolecules. Polym. J. 1985, 17, 117–132. [Google Scholar] [CrossRef]
- Caminade, A.; Majoral, J. Nanomaterials based on phosphorous dendrimers. Acc. Chem. Res. 2004, 37, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Sigal, G.B.; Mammen, M.; Dahmann, G.; Whitesides, G.M. Polyacrylamides bearing pendant- sialoside groups strongly inhibit agglutination of erythrocytes by influenza virus: The strong inhibition reflects enhanced binding through cooperative polyvalent interactions. J. Am. Chem. Soc. 1996, 118, 3789–3800. [Google Scholar] [CrossRef]
- Roy, R.; Zanini, D.; Meunier, S.J.; Romaowska, A. Solid-Phase synthesis of dendritic sialoside inhibitors of influenza A Virus Haemagglutinin. Chem. Commun. 1993, 1869–1871. [Google Scholar] [CrossRef]
- Twyman, L.J.; Beezer, A.E.; Esfand, R.; Hardy, M.J.; Mitchell, J.C. The synthesis of water soluble dendrimers, and their application as possible drug delivery systems. Tetrahedron Lett. 1999, 40, 1743–1746. [Google Scholar] [CrossRef]
- Liu, M.; Kono, K.; Frechet, J.M.J. Water-soluble dendritic unimolecular micelles: Their potential as drug delivery agents. J. Contr. Rel. 2000, 65, 121–131. [Google Scholar] [CrossRef]
- Zhuo, R.X.; Du, B.; Lu, Z.R. In vitro release of 5-fluorouracil with cyclic core dendritic polymer. J. Contr. Rel. 1999, 57, 249–257. [Google Scholar] [CrossRef]
- Degoricija, L.; Bansal, P.N.; Sontjens, S.H.M.; Joshi, N.S.; Takahashi, M.; Snyder, B.; Grinstaff, M.W. Hydrogels for osteochondral repair based on photocrosslinkable carbamate dendrimers. Biomacromolecules 2008, 9, 2863–2872. [Google Scholar] [CrossRef] [PubMed]
- Haraguchi, K.; Takehisa, T.; Ebato, M. Control of Cell cultivation and Cell sheet detachment on the surface of polymer/clay nanocomposite hydrogels. Biomacromolecules 2006, 7, 3267–3765. [Google Scholar] [CrossRef] [PubMed]
- Grieshaber, S.E.; Farran, A.J.E.; Lin-Gibson, S.; Kiick, K.L.; Jia, X. Synthesis and characterization of elastin-mimetic hybrid polymers with multiblock, alternating molecular architecture and elastomeric properties. Macromolecules 2009, 42, 2532–2541. [Google Scholar] [CrossRef] [PubMed]
- Atzet, S.; Curtin, S.; Trinh, P.; Bryant, S.; Ratner, B. Degradable poly(2-hydroxyethyl methacrylate)-co-polycaprolactone hydrogels for tissue engineering scaffolds. Biomacromolecules 2008, 9, 3370–3373. [Google Scholar]
- Chao, G.T.; Huang, M.J.; Kan, B.; Gu, Y.C.; Gong, C.Y.; Yang, J.L.; Jia, W.J.; Liu, C.B.; Gou, M.L.; Tu, M.J.; Qian, Z.Y. Synthesis, characterization and hydrolytic degradation behavior of a novel biodegradable pH-sensitive hydrogel based on polycaprolactone, methacrylic acid and poly (ethylene glycol). J. Biomed. Mater. Res. A 2008, 85A, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.C.; Metters, A.T. Hydrogels in controlled release formulations: Network design and Mathematical Modeling. Adv. Drug Deliv. Rev. 2006, 58, 1379–1408. [Google Scholar] [CrossRef] [PubMed]
- Park, T.G.; Lu, W.Q.; Crotts, G. Importance of in-vitro experimental conditions on protein release kinetics, stability and polymer degradation in protein encapsulated poly (D,Llactic acid-co-glycolic acid) microspheres. J. Contr. Rel. 1995, 33, 211–222. [Google Scholar] [CrossRef]
- Jiang, W.L.; Gupta, R.K.; Deshpande, M.C.; Schwendeman, S.P. Biodegradable poly(lactic-co-glycolic acid) microparticles for injectable delivery of vaccine antigens. Adv. Drug Deliv. Rev. 2005, 57, 391–410. [Google Scholar] [CrossRef] [PubMed]
- Peppas, N.A.; Bures, P.; Leobandung, W.; Ichiwaka, H. Hydrogels in Pharmaceutical Formulations. Eur. J. Pharm. Biopharm. 2000, 50, 27–46. [Google Scholar] [CrossRef] [PubMed]
- TeBmar, J.; Brand, F.; Göpferich, A. Hydrogels for tissue engineering. In Fundamentals of Tissue Engineering and Regenerative Medicine; Springer: Berlin, Heidelberg, Germany, 2009; pp. 495–496. [Google Scholar]
- Ma, P.X.; Choi, J.W. Biodegradable Polymer Scaffolds with Well-Defined Interconnected Spherical Pore Network. Tissue Eng. 2001, 7, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Ferruti, P.; Bianchi, S.; Ranucci, E.; Chiellini, F.; Caruso, V. Novel Poly(amido-amine)-based hydrogels as scaffolds for tissue engineering. Macromol. Biosci. 2005, 5, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Leach, J.B; Bivens, K.A.; Patrick, C.W., Jr.; Schmidt, C.E. Photocrosslinked hyaluronic acid hydrogels: natural, biodegradable tissue engineering scaffolds. Biotechnol. Bioeng. 2003, 82, 578–589. [Google Scholar] [CrossRef] [PubMed]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Schwall, C.T.; Banerjee, I.A. Micro- and Nanoscale Hydrogel Systems for Drug Delivery and Tissue Engineering. Materials 2009, 2, 577-612. https://doi.org/10.3390/ma2020577
Schwall CT, Banerjee IA. Micro- and Nanoscale Hydrogel Systems for Drug Delivery and Tissue Engineering. Materials. 2009; 2(2):577-612. https://doi.org/10.3390/ma2020577
Chicago/Turabian StyleSchwall, Christine T., and Ipsita A. Banerjee. 2009. "Micro- and Nanoscale Hydrogel Systems for Drug Delivery and Tissue Engineering" Materials 2, no. 2: 577-612. https://doi.org/10.3390/ma2020577



