Modification of 13X Molecular Sieve by Chitosan for Adsorptive Removal of Cadmium from Simulated Wastewater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of CMS
2.3. Cd2+ Adsorption Experiments
2.4. Material Characterization
3. Results and Discussion
3.1. Effects of Different Modification Conditions on Cd2+ Removal
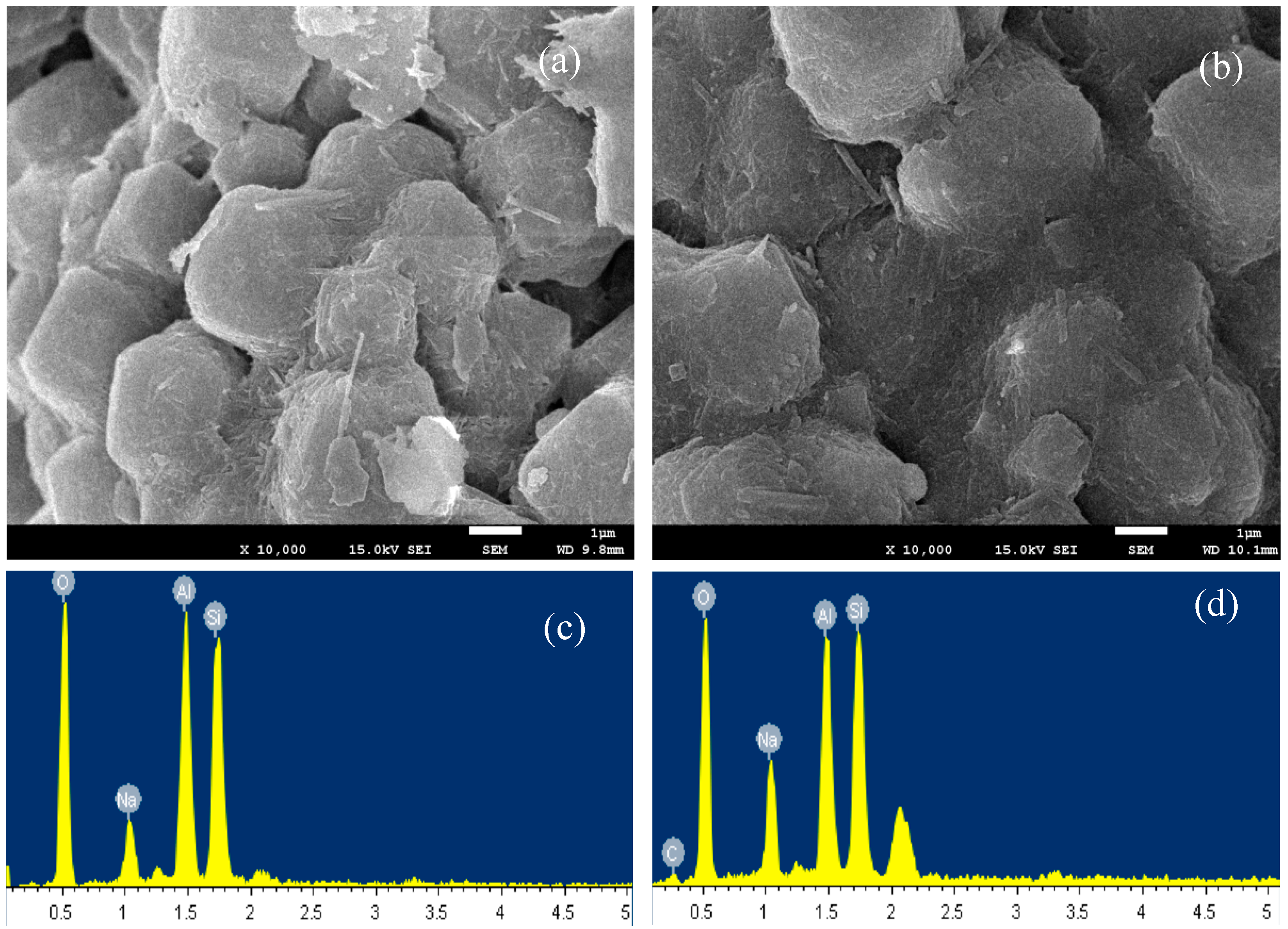
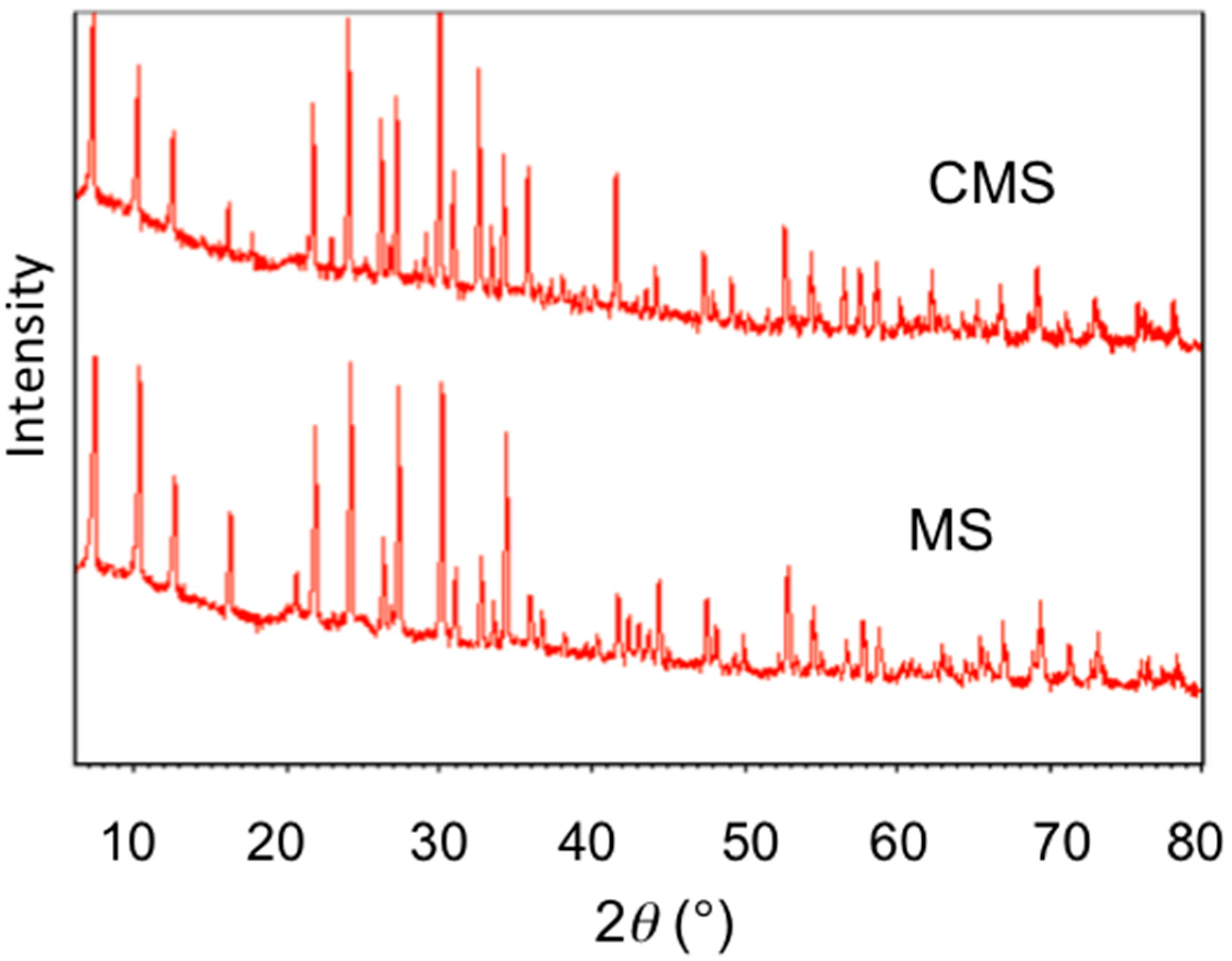
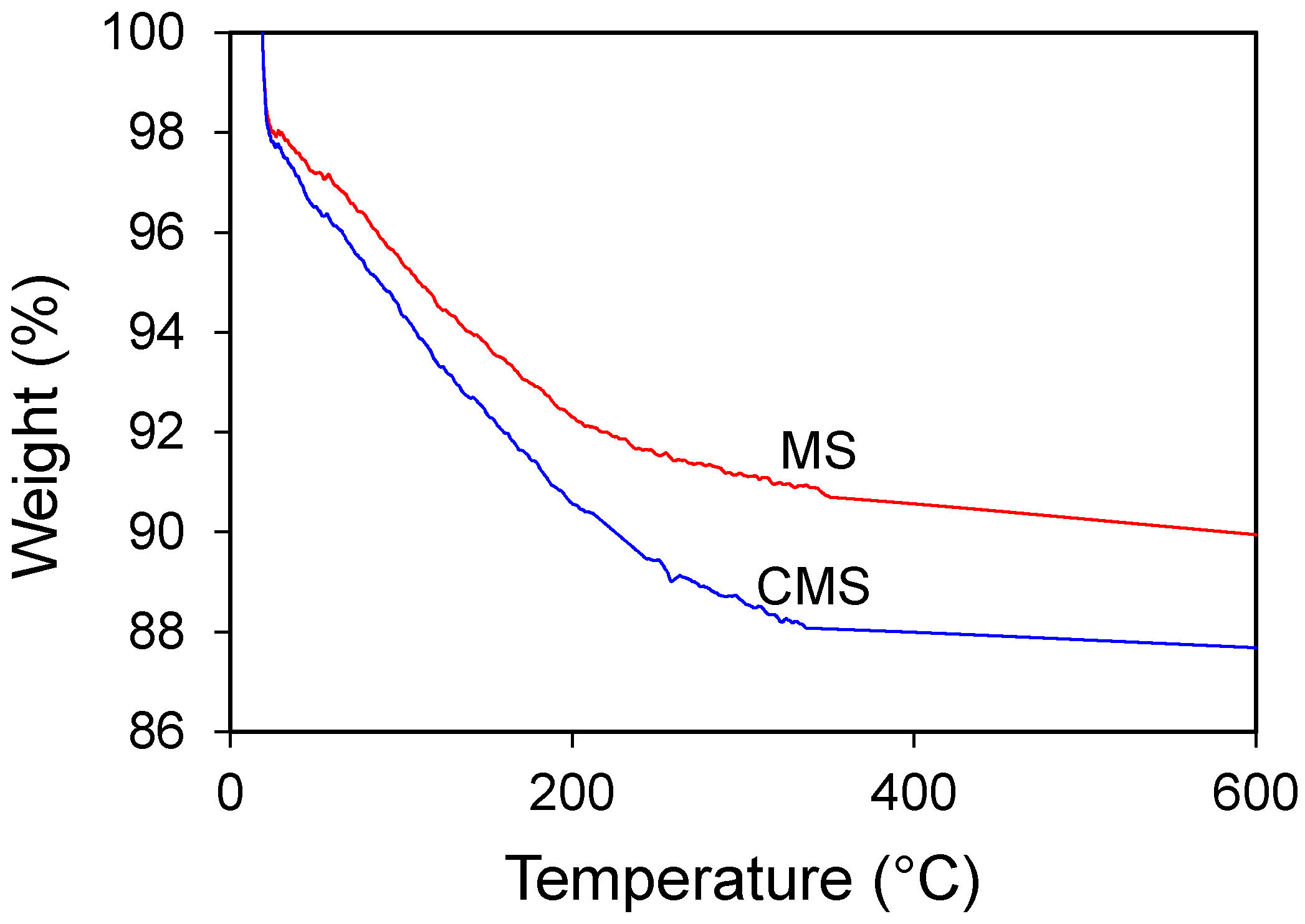
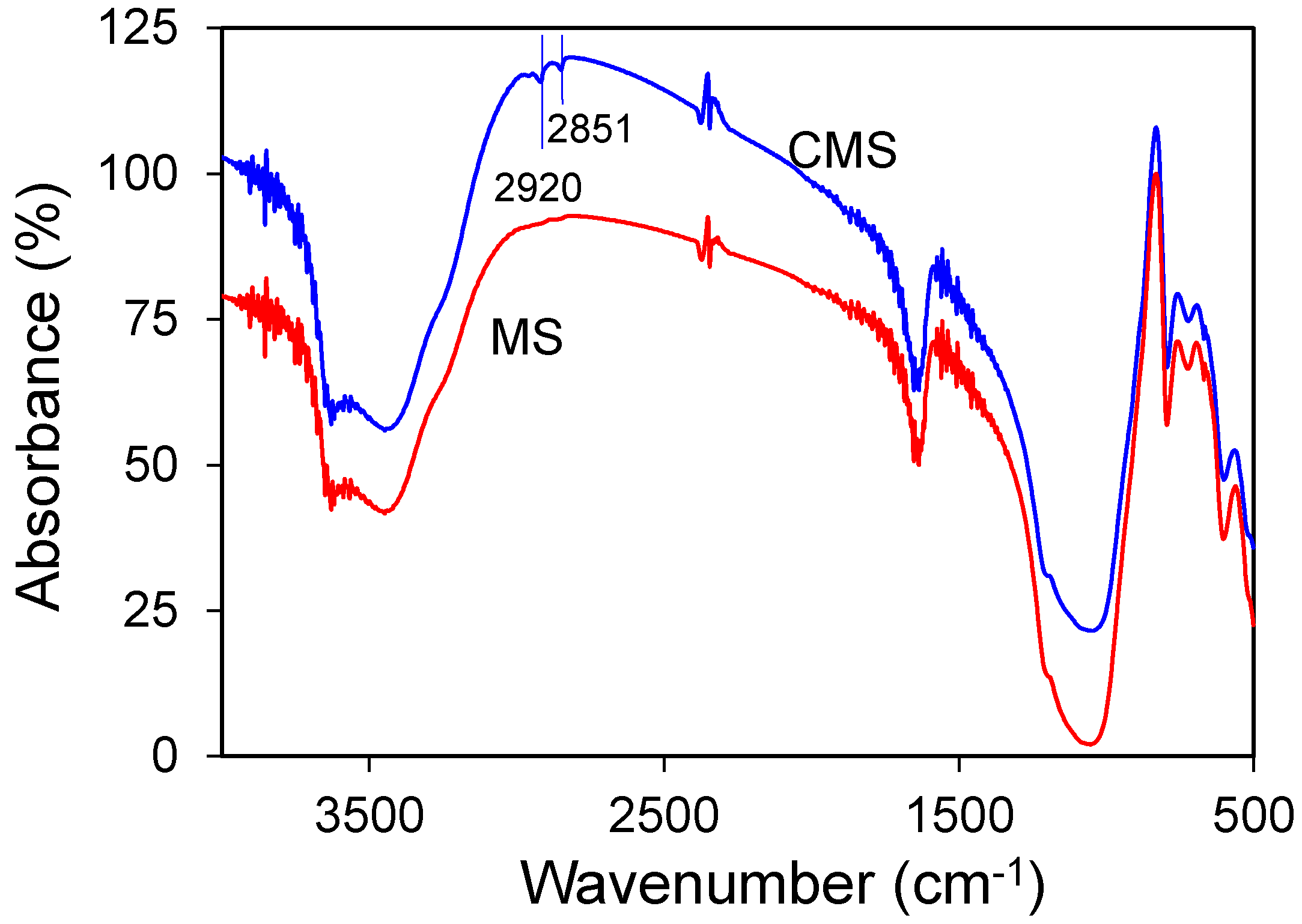
3.2. Material Characterization of MS and CMS
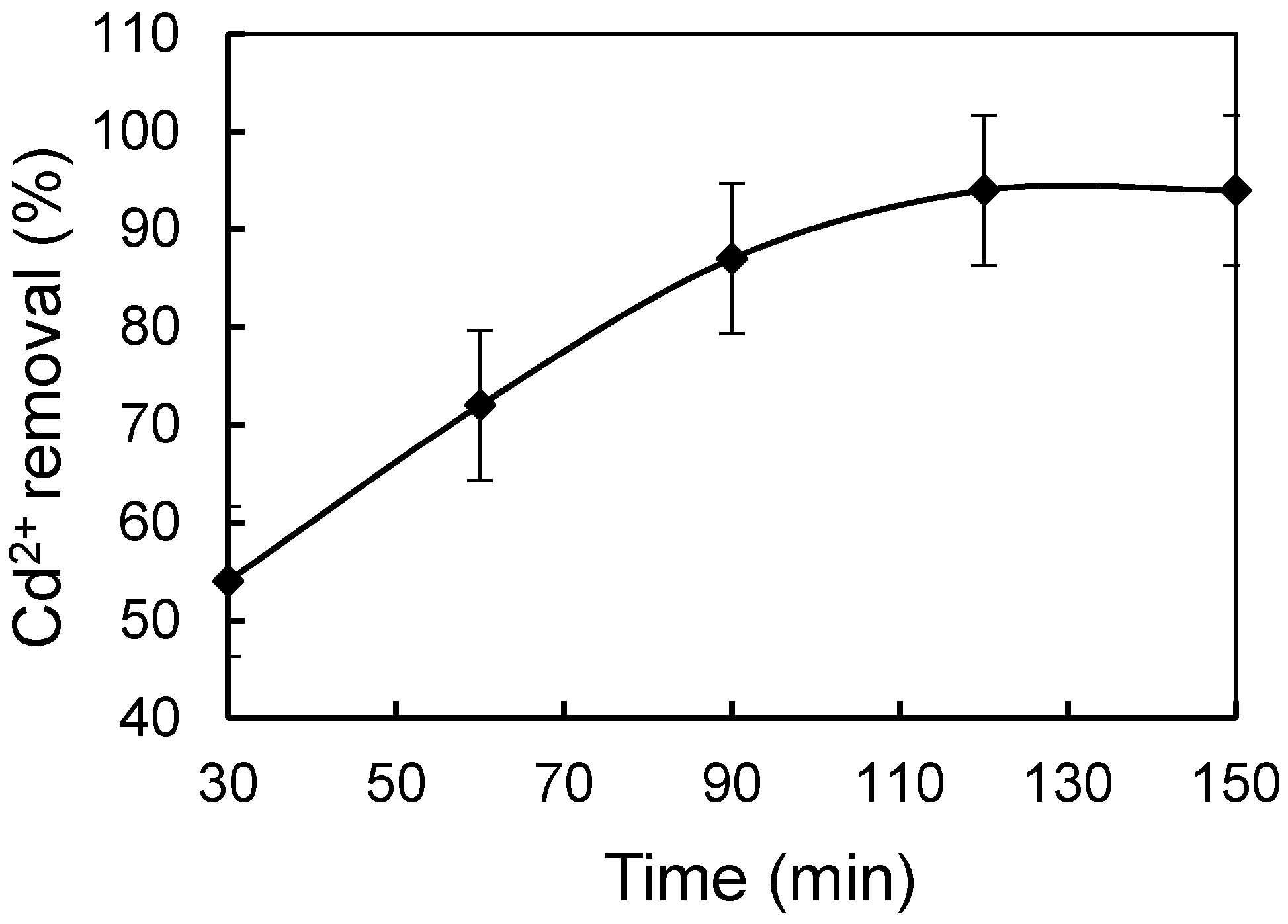
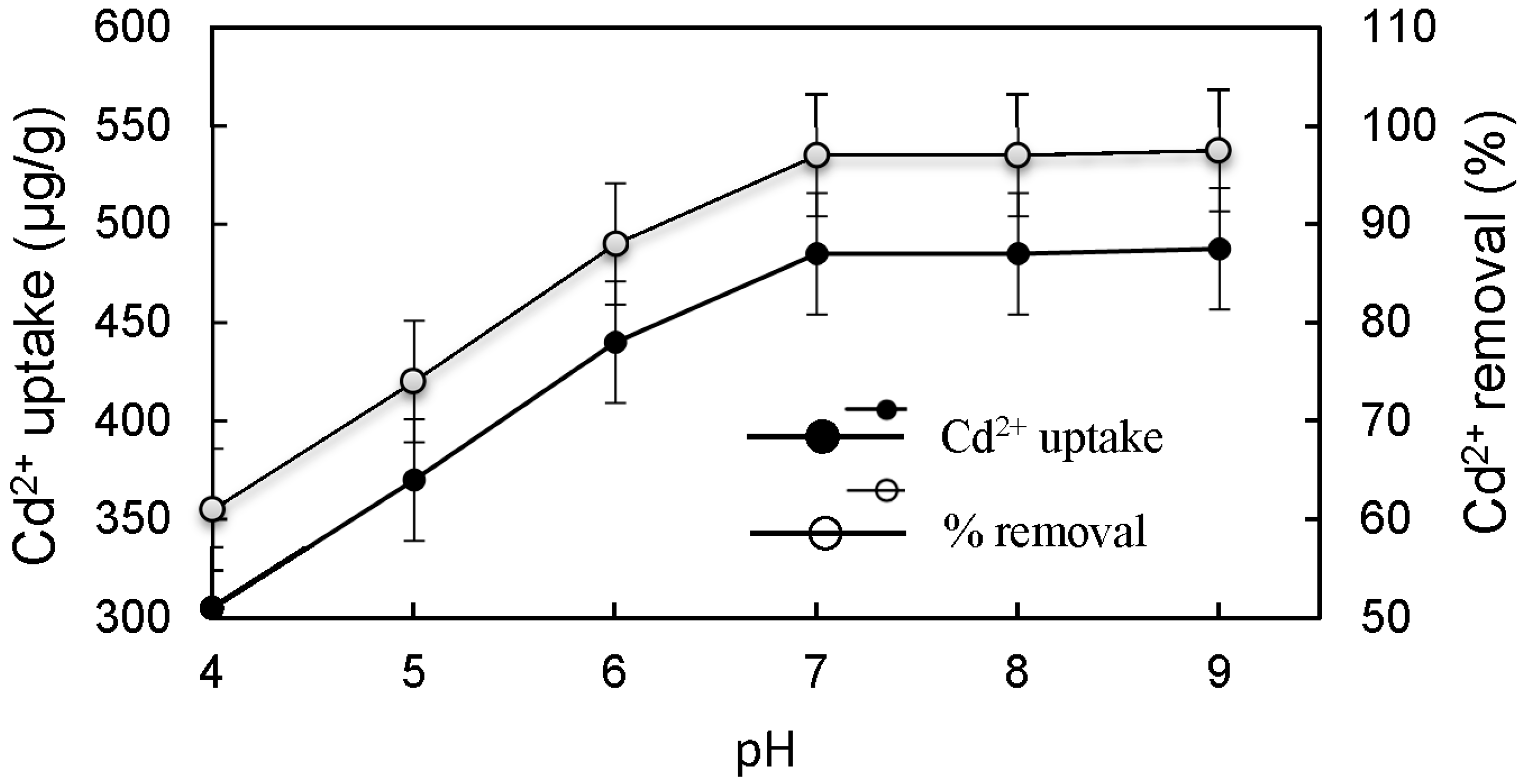
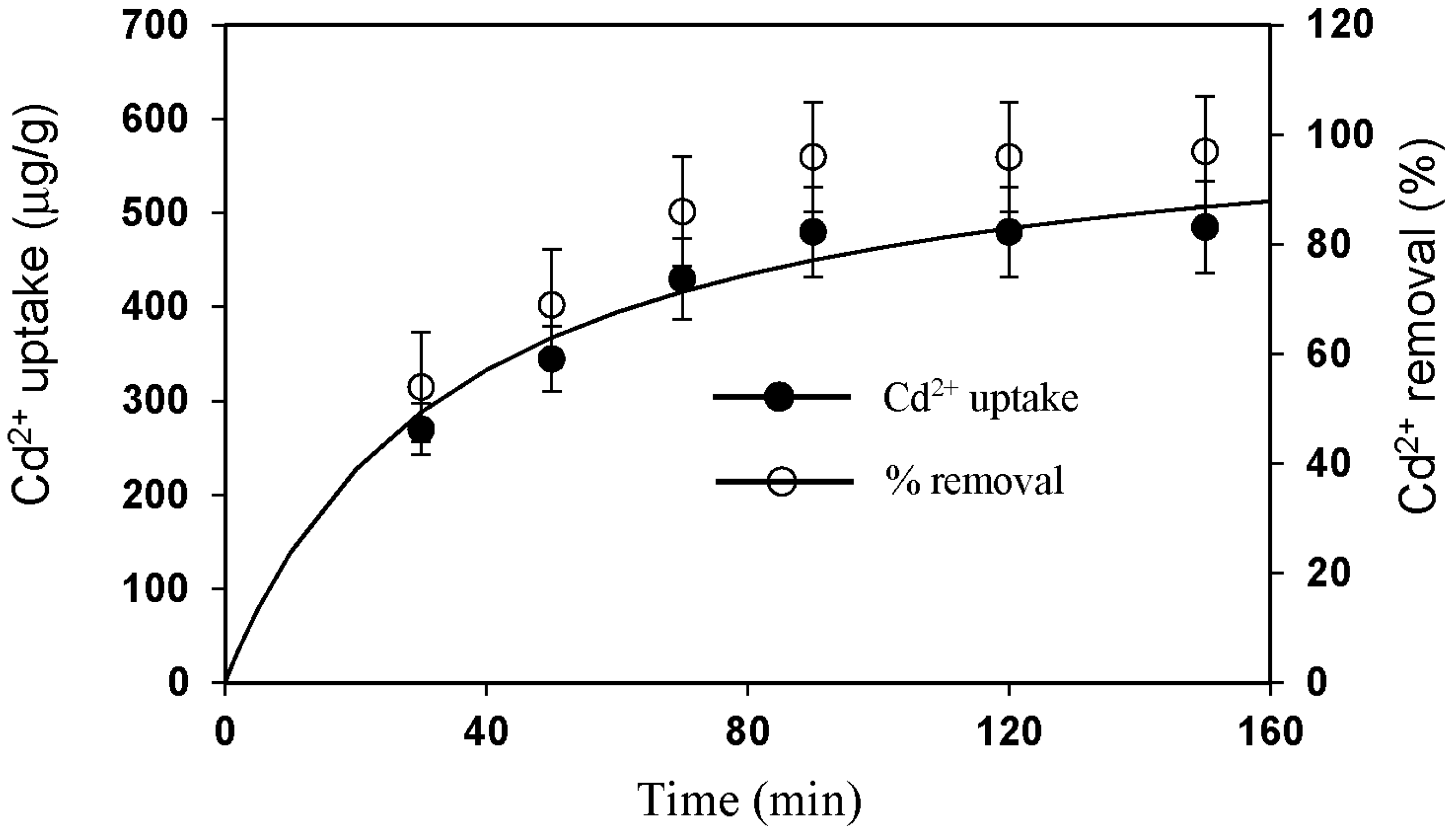
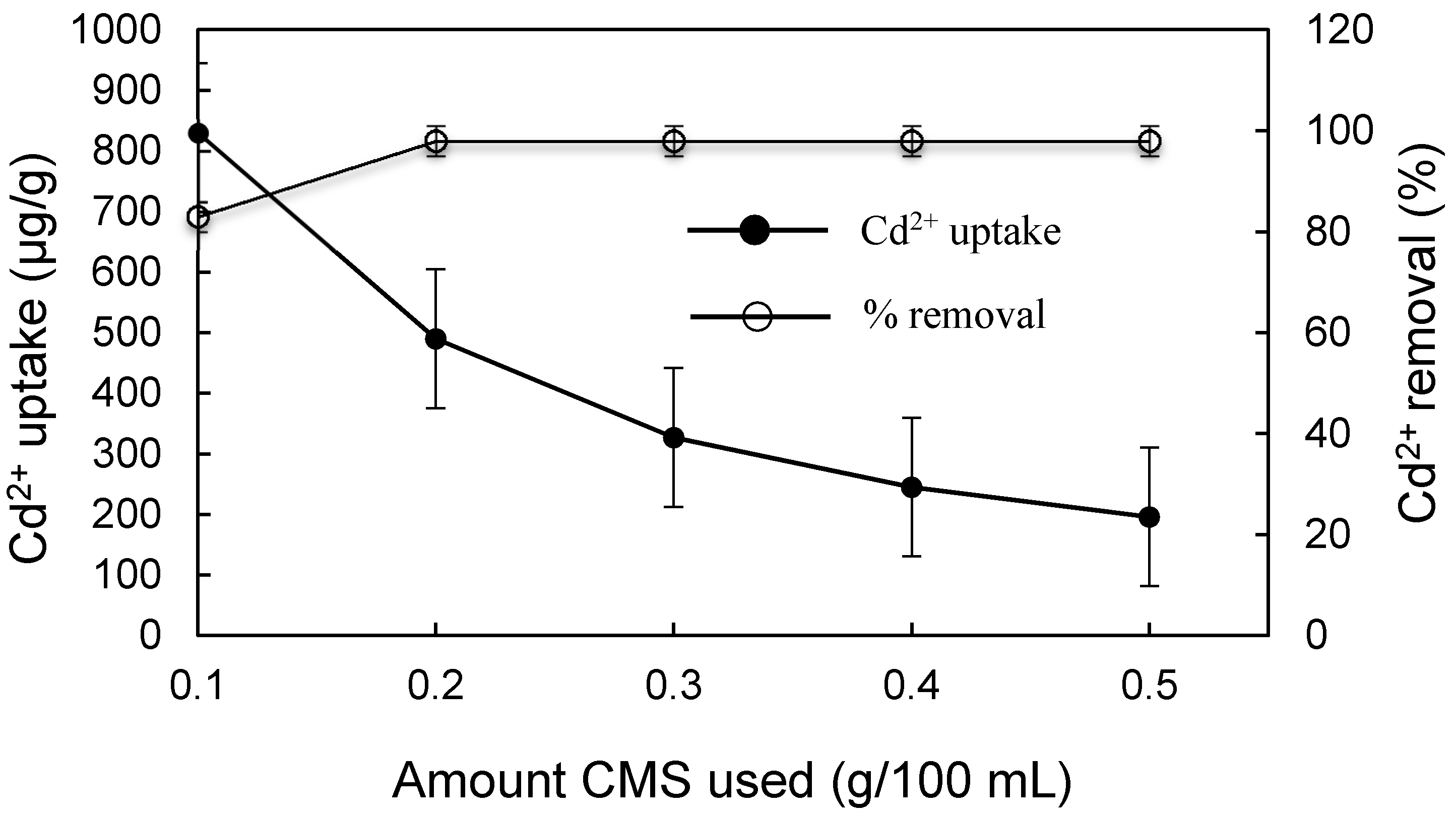
3.3. Effect of Different Physicochemical Conditions on Cd2+ Removal by CMS
4. Conclusions
- The optimal condition for the preparation of CMS was mixing 2% chitosan solution with MS for 2 h.
- SEM and XRD results showed no change in crystal morphology of MS after modification. However, the increase in C and N contents in EDS spectra, the increase in 2% of mass loss in TGA analysis, and the presence of 2850 and 2920 cm−1 bands in FTIR analyses confirmed chitosan uptake on MS after modification.
- The static single factor experiment results showed that under the condition of room temperature, pH of 7, vibration adsorption time of 90 min, and adsorbent dosage of 2 g/L, the Cd2+ removal from simulated wastewater with an initial concentration of 100 μg/L was over 96% when using CMS as the adsorbent, which meets the standard Cd2+ discharge prescribed in the Standards for Irrigation Water Quality (GB5084-2005) and the MCL and MCLG for groundwater and drinking water standards set by USEPA.
- The adsorption process of lower concentration of Cd2+ in water by CMS fitted with the Langmuir adsorption isotherm model well, with a saturated adsorption capacity of 1 mg/g.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, H.D.; Fang, F.M.; Xie, H.F. Research situation and outlook on heavy metal pollution in water environment of China. Guangdong Trace Elem. Sci. 2010, 17, 14–18. [Google Scholar]
- Zhan, J.; Wei, S.H. Methods of inhibiting cadmium toxicity and its mechanism: A review. Asian J. Ecotoxicol. 2012, 4, 354–359. [Google Scholar]
- Yue, X.Y.; Yi, J. Cadmium poisoning and preventing. Sichuan Anim. Vet. Sci. 2000, 7, 28–29. [Google Scholar]
- Du, L.N.; Yu, R.Z.; Wang, H.Y.; Lu, Y.; Liu, Z.T. Pollution and toxicity of cadmium: A review of recent studies. J. Environ. Health 2013, 2, 167–174. [Google Scholar]
- USEPA. National Primary Drinking Water Regulations. Available online: https://www.epa.gov/ground-water-and-drinking-water/national-primary-drinking-water-regulations (accessed on 15 September 2017).
- Friberg, L.; Nordberg, G.F.; Vouk, V.B. Handbook on the Toxicology of Metals, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2007; pp. 130–184. [Google Scholar]
- World Health Organization; United Nations Environment Programme; Global Environment Monitoring System. Global Fresh Water Quality; Published on behalf of the World Health Organization/United Nations Environment Programme; Blackwell Reference: Oxford, UK, 1989. [Google Scholar]
- Mustafa, H.T. Cadmium and zinc concentrations in the potable water of the eastern provinces of Saudi Arabia. Bull. Environ. Contam. Toxicol. 1988, 40, 462–467. [Google Scholar] [CrossRef] [PubMed]
- Dai, S.M.; Lv, X.W. Advances on cadmium pollution water treatment technology. Saf. Environ. Eng. 2006, 13, 63–65. [Google Scholar]
- Hu, K.W.; Jia, D.Y.; Zha, C.M. Effect of zeolite on competitive sdsorption of heavy metals ions. Soil Fertil. Sci. China 2008, 3, 66–69. [Google Scholar]
- He, H.P.; Guo, J.G.; Zhu, J.X. An experimental study of adsorption capacity of montmorillonite, kaolinite and illite for heavy metals. Acta Petrol. Mineral. 2001, 20, 573–578. [Google Scholar]
- Zou, Z.H.; He, S.F.; Han, C.Y. Progress of heavy metals liquid waste processing technique. Technol. Water Treat. 2010, 6, 19–20. [Google Scholar]
- Sun, X. Zeolite NaA and NaX Synthesis from Fly Ash and Their Adsorption of Heavy Metal Ions in Waste Water; Nan Jing University of Science and Technology: Nanjing, China, 2007. [Google Scholar]
- Van Mao, R.L.; Xiao, S.; Ramsaran, A.; Yao, J. Selective removal of silicon from zeolite frameworks using sodium carbonate. J. Mater. Chem. 1994, 4, 605–610. [Google Scholar] [CrossRef]
- Xu, R.R.; Pang, W.Q.; Yu, J.H. Chemistry-Zeolite and Oorous Materials; Science Press: Beijing, China, 2004; pp. 356–409. [Google Scholar]
- Qin, C.; Li, H.; Xiao, Q.; Liu, Y.; Zhu, J.; Du, Y. Water-solubility of chitosan and its antimicrobial activity. Carbohydr. Polym. 2006, 63, 367–374. [Google Scholar] [CrossRef]
- Hasan, S.; Ghosh, T.; Viswanath, D. Dispersion of chitosan on perlite for enhancement of copper adsorption capacity. J. Hazard. Mater. 2008, 152, 826–837. [Google Scholar] [CrossRef] [PubMed]
- Ngah, W.S.W.; Teong, L.C.; Toh, R.H.; Hanafiah, M.A.K.M. Utilization of chitosan-zeolite composite in the removal of Cu(II) from aqueous solution: Adsorption, desorption and fixed bed column studies. Chem. Eng. J. 2012, 209, 46–53. [Google Scholar] [CrossRef]
- Ngah, W.S.W.; Teong, L.C.; Toh, R.H.; Hanafiah, M.A.K.M. Comparative study on adsorption and desorption of Cu (II) ions by three types of chitosan–zeolite composites. Chem. Eng. J. 2013, 223, 231–238. [Google Scholar] [CrossRef]
- Xie, J.; Li, C.; Chi, L.; Wu, D. Chitosan modified zeolite as a versatile adsorbent for the removal of different pollutants from water. Fuel 2013, 103, 480–485. [Google Scholar] [CrossRef]
- Lu, C.; Yu, S.; Yao, T.; Zeng, C.; Wang, C.; Zhang, L. Zeolite X/chitosan hybrid microspheres and their adsorption properties for Cu (II) ions in aqueous solutions. J. Porous Mater. 2015, 22, 1255–1263. [Google Scholar] [CrossRef]
- Djelad, A.; Morsli, A.; Robitzer, M.; Bengueddach, A.; Di Renzo, F.; Quignard, F. Sorption of Cu (II) ions on chitosan-zeolite X composites: Impact of gelling and drying conditions. Molecules 2016, 21, 109. [Google Scholar] [CrossRef] [PubMed]
- Kołodyńska, D.; Hałas, P.; Franus, M.; Hubicki, Z. Zeolite properties improvement by chitosan modification—Sorption studies. J. Ind. Eng. Chem. 2017, 52, 187–196. [Google Scholar] [CrossRef]
- Hałas, P.; Kołodyńska, D.; Płaza, A.; Gęca, M.; Hubicki, Z. Modified fly ash and zeolites as an effective adsorbent for metal ions from aqueous solution. Adsorpt. Sci. Technol. 2017, 35, 519–533. [Google Scholar] [CrossRef]
- Płaza, A.; Kołodyńska, D.; Hałas, P.; Gęca, M.; Franus, M.; Hubicki, Z. The zeolite modified by chitosan as an adsorbent for environmental applications. Adsorpt. Sci. Technol. 2017. [Google Scholar] [CrossRef]
- Hamza, M.F.; Aly, M.M.; Abdel-Rahman, A.A.H.; Ramadan, S.; Raslan, H.; Wang, S.; Vincent, T.; Guibal, E. Functionalization of magnetic chitosan particles for the sorption of U (VI), Cu (II) and Zn (II)—Hydrazide derivative of glycine-grafted chitosan. Materials 2017, 10, 539. [Google Scholar] [CrossRef] [PubMed]

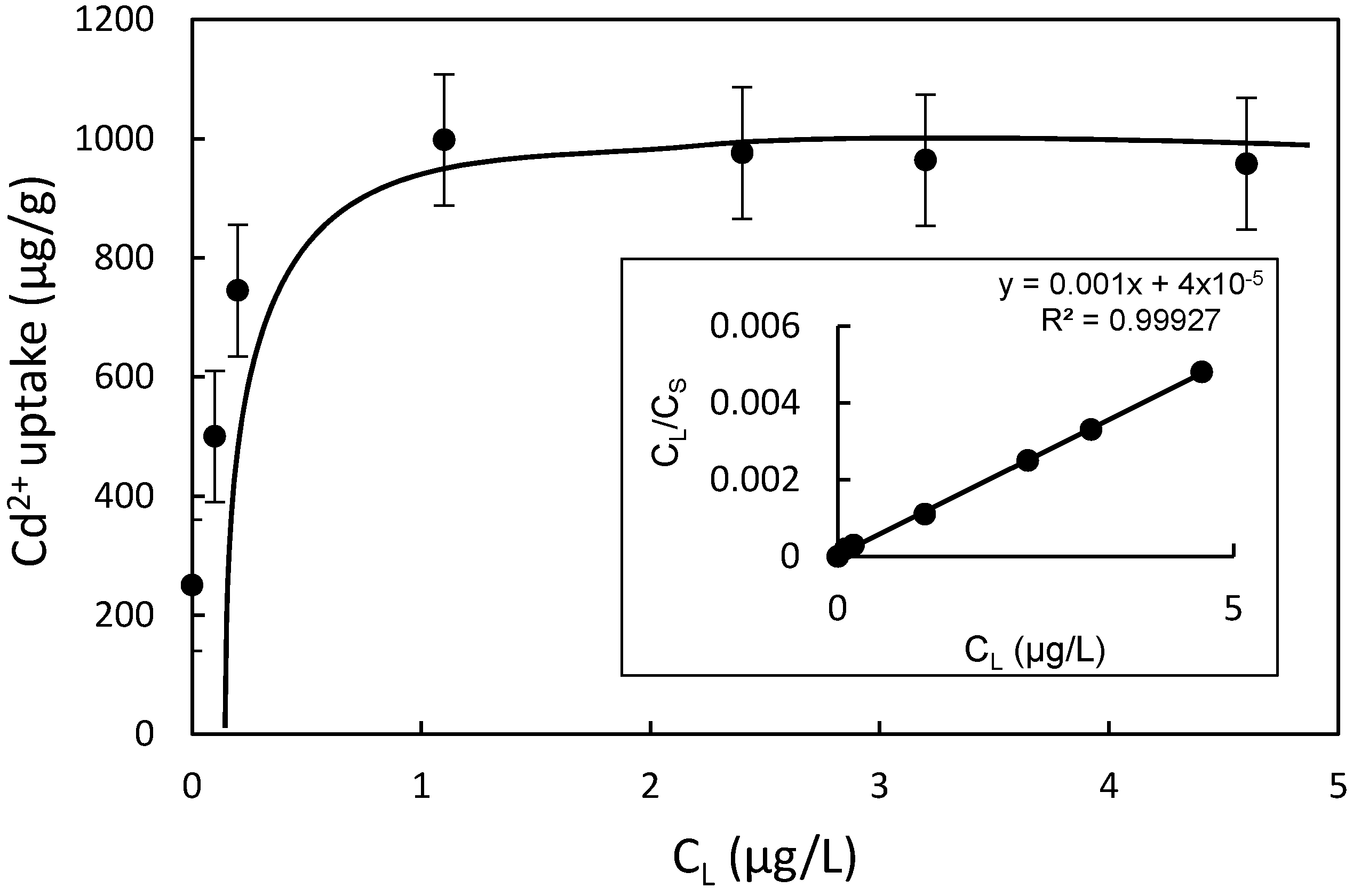
| Langmuir Constant | Freundlich Constant | ||||
|---|---|---|---|---|---|
| Sm (μg/g) | KL (L/μg) | R2 | logk | 1/n | R2 |
| 1000 | 25 | 0.9993 | 2.70 | 0.534 | 0.47 |
| Sample | CL (μg/L) | Removal (%) | CS (μg/g) |
|---|---|---|---|
| MS | 82.1 | 17.9 | 89.5 |
| CMS | 1.1 | 98.7 | 985 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, Y.; Sun, K.; Huo, L.; Li, X.; Qi, X.; Li, Z. Modification of 13X Molecular Sieve by Chitosan for Adsorptive Removal of Cadmium from Simulated Wastewater. Materials 2017, 10, 1101. https://doi.org/10.3390/ma10091101
Shi Y, Sun K, Huo L, Li X, Qi X, Li Z. Modification of 13X Molecular Sieve by Chitosan for Adsorptive Removal of Cadmium from Simulated Wastewater. Materials. 2017; 10(9):1101. https://doi.org/10.3390/ma10091101
Chicago/Turabian StyleShi, Yan, Ken Sun, Lixin Huo, Xiuxiu Li, Xuebin Qi, and Zhaohui Li. 2017. "Modification of 13X Molecular Sieve by Chitosan for Adsorptive Removal of Cadmium from Simulated Wastewater" Materials 10, no. 9: 1101. https://doi.org/10.3390/ma10091101
APA StyleShi, Y., Sun, K., Huo, L., Li, X., Qi, X., & Li, Z. (2017). Modification of 13X Molecular Sieve by Chitosan for Adsorptive Removal of Cadmium from Simulated Wastewater. Materials, 10(9), 1101. https://doi.org/10.3390/ma10091101







