Characterization and In Vitro and In Vivo Assessment of a Novel Cellulose Acetate-Coated Mg-Based Alloy for Orthopedic Applications
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials Synthesis and Characterization
2.1.1. Alloy Synthesis, Thermo-Mechanical Processing, Microstructural and Mechanical Characterization
2.1.2. Coating Formulation
2.1.3. Biomaterials Characterization
2.2. In Vitro Cellular Response
2.2.1. Preparation of Extracts of the Biomaterials
2.2.2. Cell Culture
2.2.3. Quantitative and Qualitative Assessment of Cellular Survival and Cell Proliferation
2.2.4. Fluorescence Microscopic Evaluation of Cell Adhesion and Morphology
2.2.5. Determination of Intracellular Alkaline Phosphatase Activity
2.2.6. Quantification of Osteopontin Secretion
2.2.7. Quantitative Assessment of Extracellular Matrix Mineralization
2.3. In Vivo Animal Studies
2.3.1. Implant Preparation and Surgery
2.3.2. Histological Examination
2.4. Statistical Analysis
3. Results
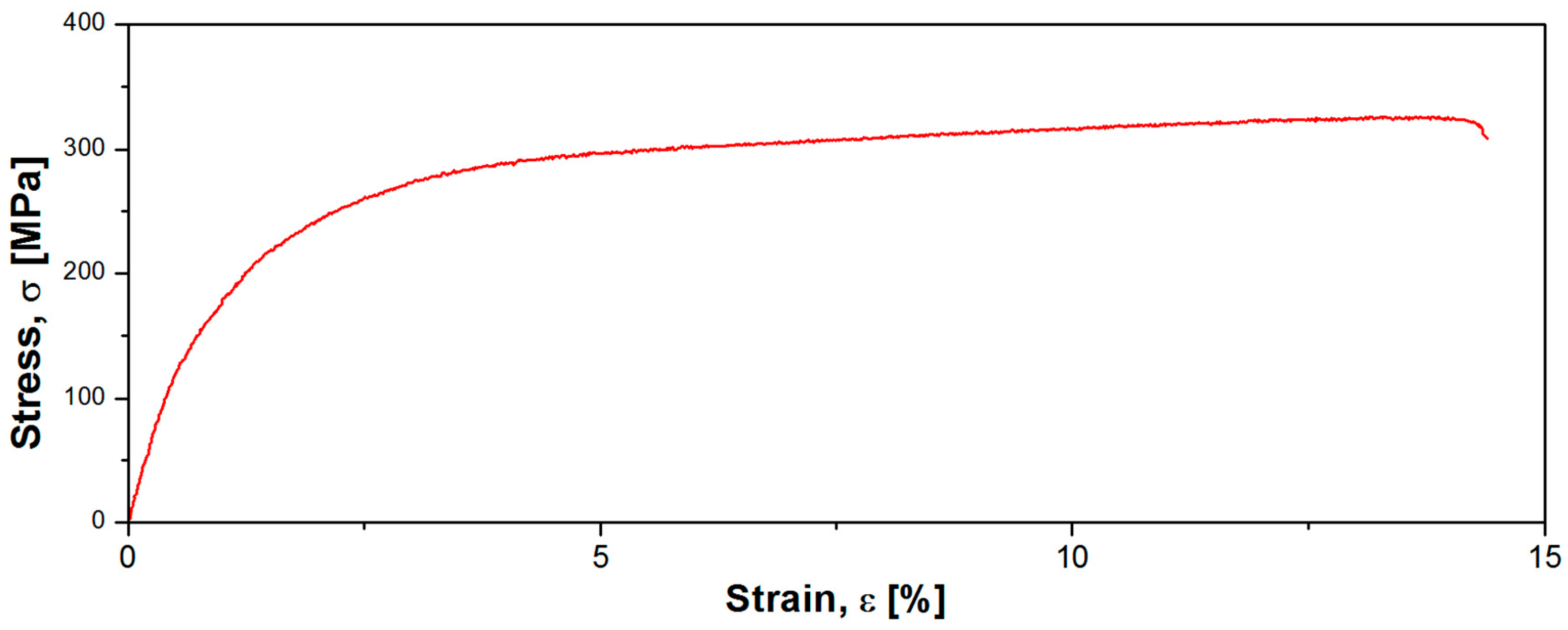
3.1. Alloy’s Microstructural and Mechanical Characterization
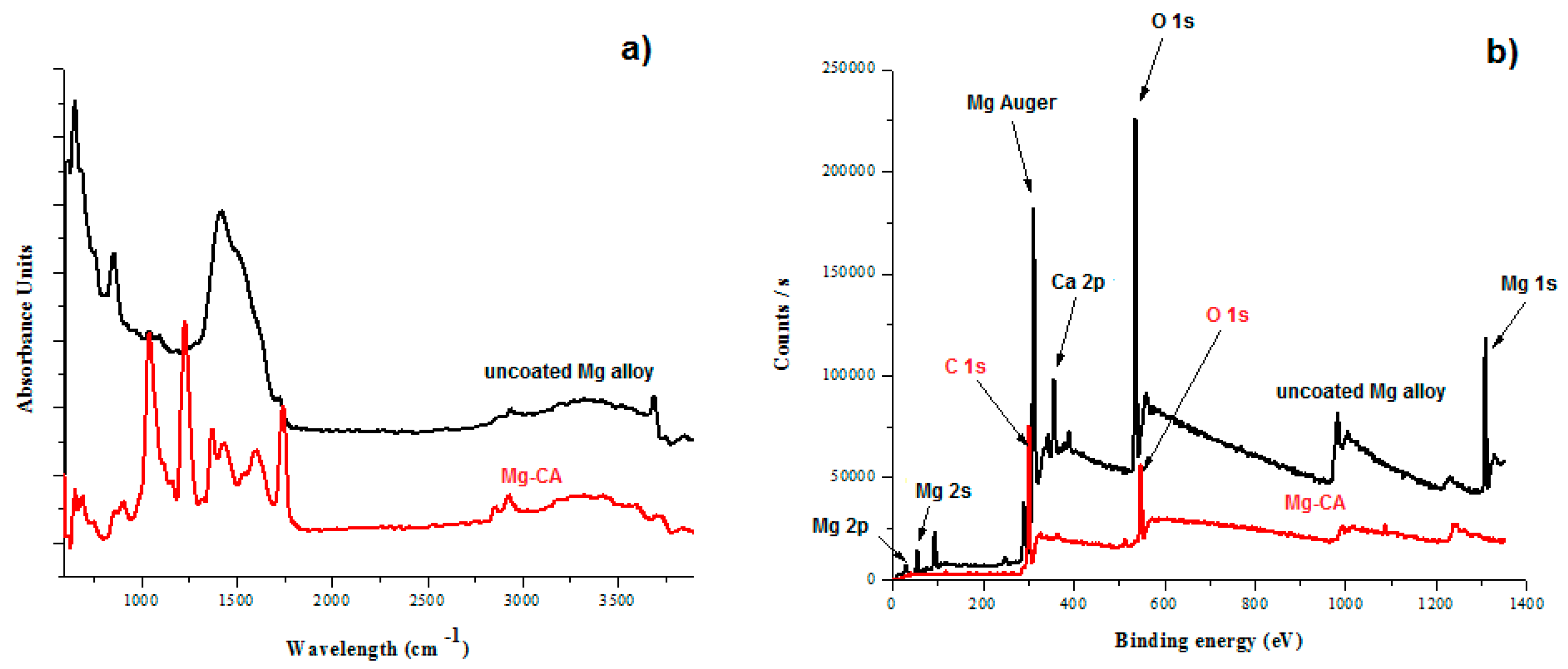
3.2. Characterization of Uncoated and Coated Mg-Based Alloy Samples
3.3. In Vitro Behavior of Osteoblast-Like Cells
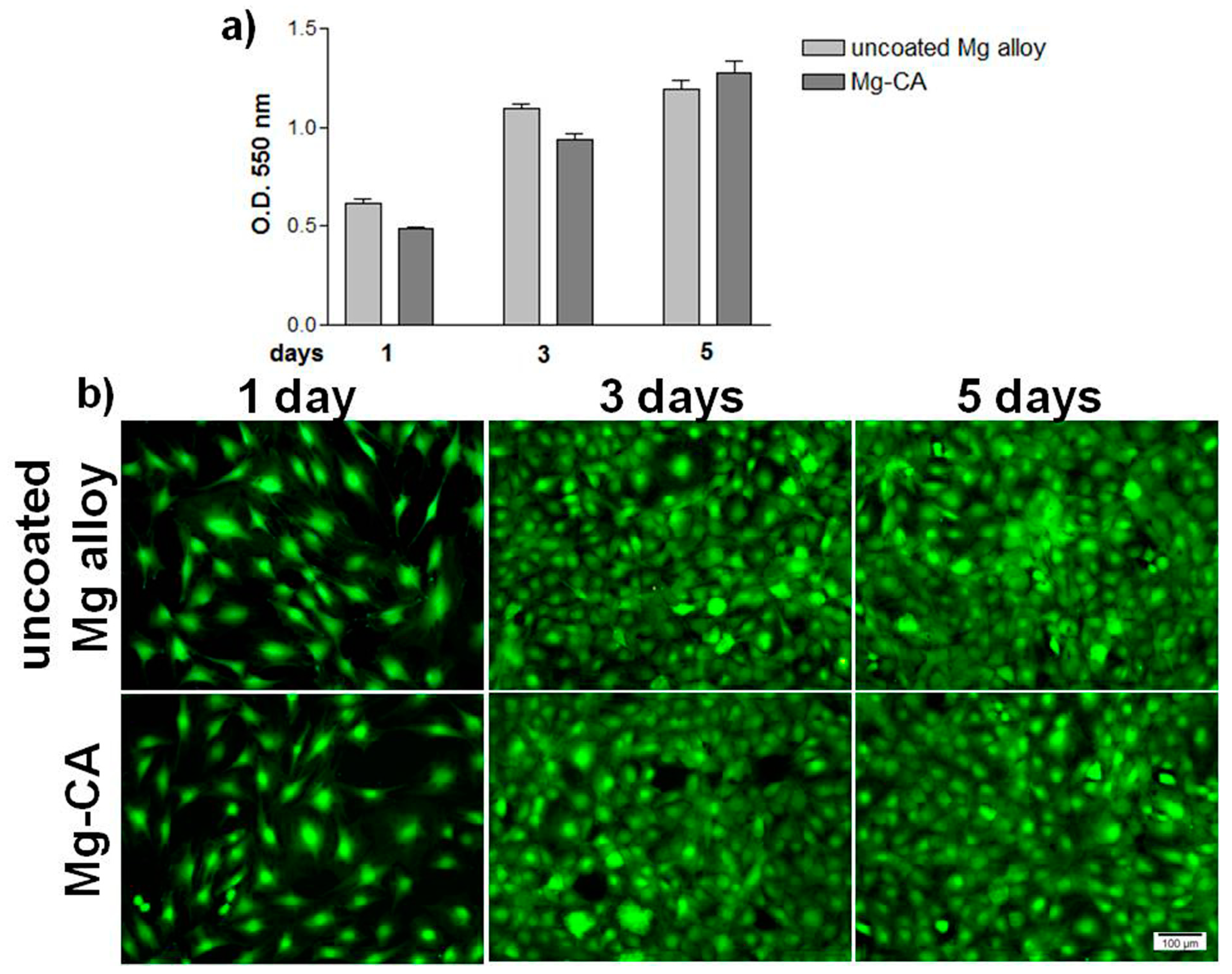
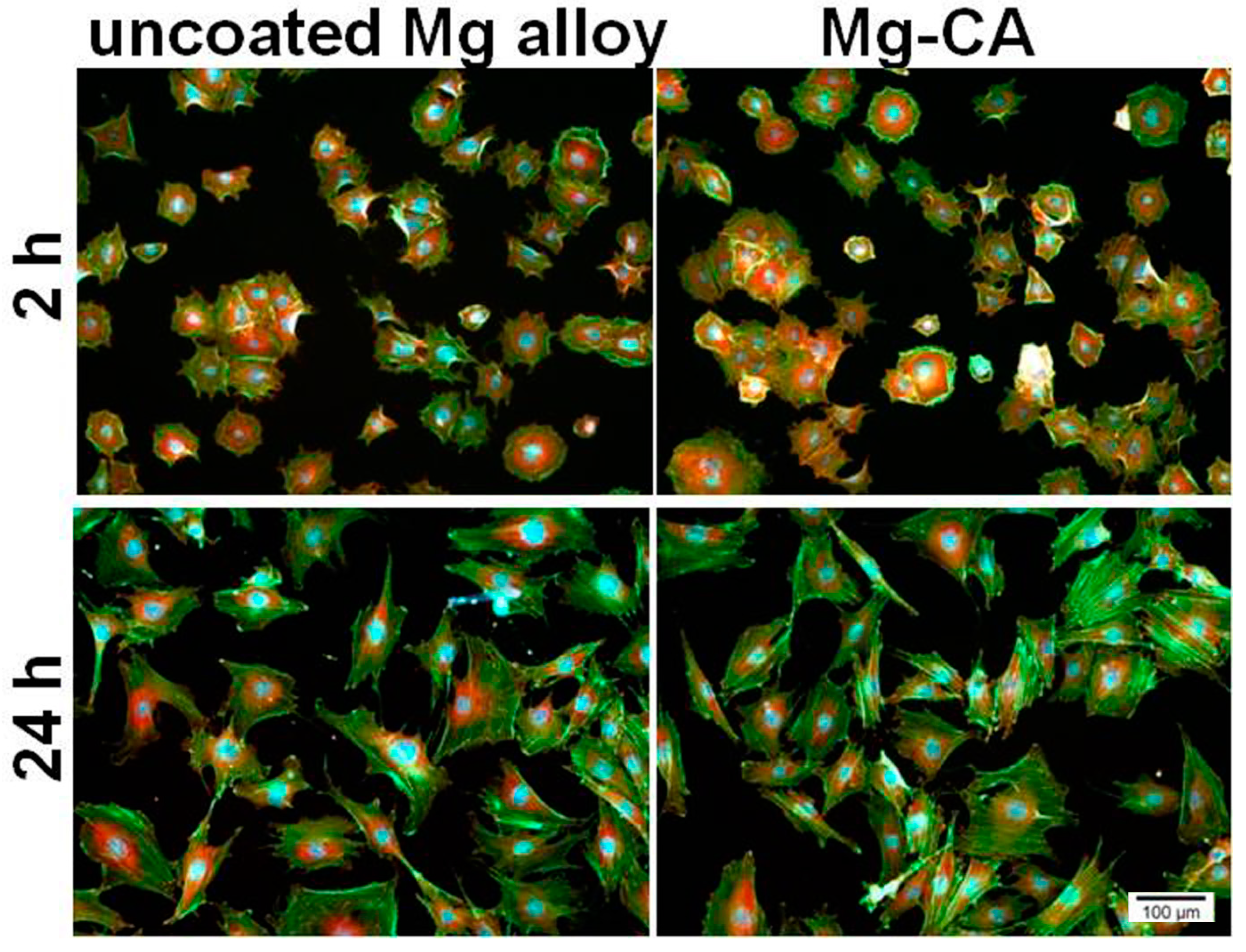
3.3.1. Cell Viability/Proliferation and Morphological Features
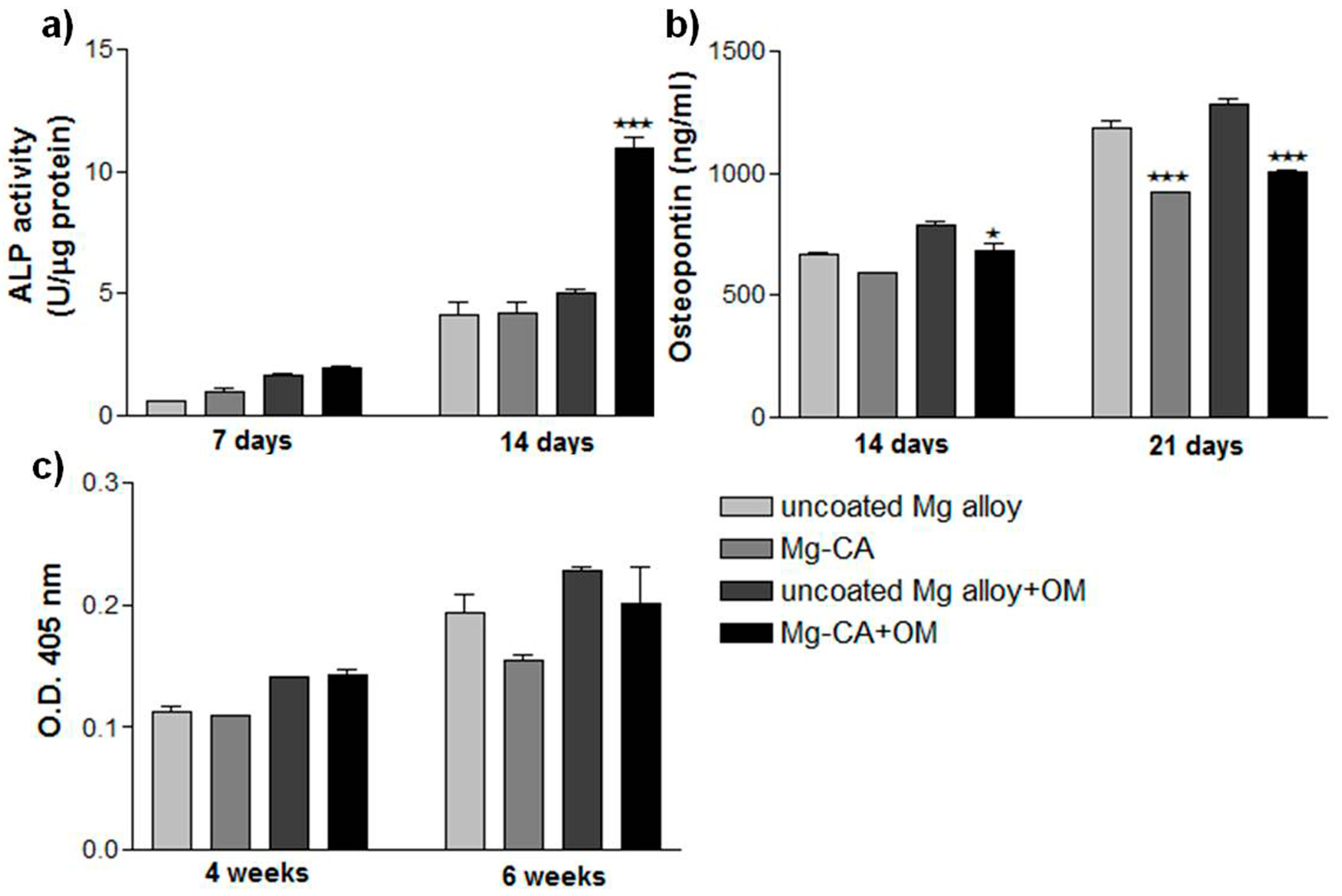
3.3.2. The Function of MC3T3-E1 Pre-Osteoblasts
3.4. In Vivo Biocompatibility of the Mg-Based Biomaterials
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Troitskskii, V.V.; Tsitrin, D.N. The resorbing metallic alloy ‘OSteosinthezit’ as material for fastening broken bone. Khirurgiia 1944, 8, 60–63. [Google Scholar]
- Znamenskii, M.S. Metallic osteosynthesis by means of an apparatus made of resorbing metal. Khirurgiia 1945, 12, 60–63. [Google Scholar]
- Poinern, G.E.; Brundavanam, S.; Fawcett, D. Biomedical Magnesium Alloys: A Review of Material Properties, Surface Modifications and Potential as a Biodegradable Orthopaedic Implant. Am. J. Biomed. Eng. 2012, 2, 218–240. [Google Scholar] [CrossRef]
- Babkin, V.M. Effect of zirconium on the grain size of magnesium containing 4.5% Zn. Met. Sci. Heat Treat. 2004, 5, 543–544. [Google Scholar] [CrossRef]
- Xu, L.; Pan, F.; Yu, G.; Yang, L.; Zhang, E.; Yang, K. In vitro and in vivo evaluation of the surface bioactivity of a calcium phosphate coated magnesium alloy. Biomaterials 2009, 30, 1512–1523. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Yu, G.; Zhang, E.; Pan, F.; Yang, K. In vivo corrosion behavior of Mg-Mn-Zn alloy for bone implant application. J. Biomed. Mater. Res. A. 2007, 83, 703–711. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Li, J.; Song, Y.; Zhao, C.; Zhang, X.; Xie, C.; Zhang, Y.; Tao, H.; He, Y.; Jiang, Y.; et al. In vitro degradation, hemolysis and MC3T3-E1 cell adhesion of biodegradable Mg-Zn alloy. Mater. Sci. Eng. C 2009, 29, 1907–1912. [Google Scholar] [CrossRef]
- Wang, H.X.; Guan, S.K.; Wang, X.; Ren, C.X.; Wang, L.G. In vitro degradation and mechanical integrity of Mg-Zn-Ca alloy coated with Ca-deficient hydroxyapatite by the pulse electrodeposition process. Acta Biomater. 2009, 6, 1749–1755. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Cao, L.; Song, Y.; Zhang, S.; Zhao, C.; Zhang, F.; Zhang, X. The Bioactivated Interfacial Behavior of the Fluoridated Hydroxyapatite-Coated Mg-Zn Alloy in Cell Culture Environments. Bioinorg. Chem. Appl. 2011, 2011, 192671. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.N.; Zheng, Y.F.; Lan, Q.X.; Cheng, Y.; Zhang, Z.X.; Xi, T.F.; Zhang, D.Y. Surface modification of Mg-1Ca alloy to slow down its biocorrosion by chitosan. Biomed. Mater. 2009, 4, 044109. [Google Scholar] [CrossRef] [PubMed]
- Hahn, B.D.; Park, D.S.; Choi, J.J.; Ryu, J.; Yoon, W.H.; Choi, J.H.; Kim, H.E.; Kim, S.G. Aerosol deposition of hydroxyapatite–chitosan composite coatings on biodegradable magnesium alloy. Surf. Coat. Technol. 2011, 205, 3112–3118. [Google Scholar] [CrossRef]
- Zberg, B.; Uggowitzer, P.J.; Löffler, J.F. MgZnCa glasses without clinically observable hydrogen evolution for biodegradable implants. Nat. Mater. 2009, 8, 887–891. [Google Scholar] [CrossRef] [PubMed]
- Chai, H.; Guo, L.; Wang, X.; Gao, X.; Liu, K.; Fu, Y.; Guan, J.; Tan, L.; Yang, K. In vitro and in vivo evaluations on osteogenesis and biodegradability of a b-tricalcium phosphate coated magnesium alloy. J. Biomed. Mater. Res. A 2012, 100, 293–304. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.L.; Wang, Q.; Geng, F.; Xi, X.S.; Qiu, J.H.; Yang, K. Preparation and characterization of Ca-P coating on AZ31 magnesium alloy. Trans. Nonferr. Met. Soc. China 2010, 20, 648–654. [Google Scholar] [CrossRef]
- Puleo, A.C.; Paul, D.R.; Kelley, S.S. The Effect of Degree of Acetylation on Gas Sorption and Transport Behavior in Cellulose Acetate. J. Membr. Sci. 1989, 47, 301–332. [Google Scholar] [CrossRef]
- Basu, S.; Khan, A.L.; Cano-Odena, A.; Liu, C.; Vankelecom, I.F.J. Membrane-based Technologies for Biogas Separations. Chem. Soc. Rev. 2010, 39, 750–768. [Google Scholar] [CrossRef] [PubMed]
- Reid, C.E.; Breton, E.J. Water and Ion Flow Across Cellulosic Membranes. J. Appl. Polym. Sci. 1959, 1, 133–143. [Google Scholar] [CrossRef]
- De Oliveira, C.R.; Carvalho, J.L.; Novikoff, S.; Berti, F.V.; Porto, L.M.; Gomes, D.A.; de Goes, A.M. Bacterial cellulose membranes constitute biocompatible biomaterials for mesenchymal and induced pluripotent stem cell culture and tissue engineering. J. Tissue Sci. Eng. 2012, 11, 005. [Google Scholar] [CrossRef]
- Qiu, X.; Hu, S. “Smart” Materials Based on Cellulose: A Review of the Preparations, Properties, and Applications. Materials 2013, 6, 738–781. [Google Scholar] [CrossRef]
- Mo, Y.; Guo, R.; Liu, J.; Lan, Y.; Zhang, Y.; Xue, W.; Zhang, Y. Preparation and properties of PLGA nanofiber membranes reinforced with cellulose nanocrystals. Colloids Surf. B 2015, 132, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Yliniemi, K.; Wilson, B.P.; Singer, F.; Höhn, S.; Kontturi, E.; Virtanen, S. Dissolution control of Mg by cellulose acetate-polyelectrolyte membranes. ACS Appl. Mater. Interfaces 2014, 6, 22393–22399. [Google Scholar] [CrossRef] [PubMed]
- Staiger, M.P.; Pietak, A.M.; Huadmai, J.; Dias, G. Magnesium and its alloys as orthopedic biomaterials: A review. Biomaterials 2008, 27, 1728–1734. [Google Scholar] [CrossRef] [PubMed]
- Zhang, E.; Xu, L.; Yang, K. Investigation on new biomedical degradable magnesium alloys. In Proceedings of the Beijing International Materials Week, Beijing, China, 25–30 June 2006. [Google Scholar]
- Zhang, S.; Zhang, X.; Zhao, C.; Li, J.; Xie, C.; Tao, H.; Yang, Y.; He, Y.; Jiang, Y.; Bian, Y. Research on an Mg-Zn alloy as a degradable biomaterial. Acta Biomater. 2010, 6, 626–640. [Google Scholar] [CrossRef] [PubMed]
- Kokubo, T.; Takadama, H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2007, 27, 2907–2915. [Google Scholar] [CrossRef] [PubMed]
- ASTM G59-97, Standard Test Method for Conducting Potentiodynamic Polarization Resistance Measurements; ASTM International: West Conshohocken, PA, USA, 2014.
- A Practical Guide to ISO 10993-12: Sample Preparation and Reference Materials; MDDI: Los Angeles, CA, USA, 1998.
- Ion, R.; Gordin, D.M.; Mitran, V.; Osiceanu, P.; Dinescu, S.; Gloriant, T.; Cimpean, A. In vitro bio-functional performances of the novel superelastic beta-type Ti-23Nb-0.7Ta-2Zr-0.5N alloy. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 35, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Neacsu, P.; Mazare, A.; Cimpean, A.; Park, J.; Costache, M.; Schmuki, P.; Demetrescu, I. Reduced inflammatory activity of RAW 264.7 macrophages on titania nanotube modified Ti surface. Int. J. Biochem. Cell Biol. 2014, 55, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Niinomi, M. Recent research and development in titanium alloys for biomedical applications and healthcare goods. Sci. Technol. Adv. Mater. 2003, 4, 445–454. [Google Scholar] [CrossRef]
- Niinomi, M. Biologically and mechanically biocompatible titanium alloys. Mater. Trans. 2008, 49, 2170–2178. [Google Scholar] [CrossRef]
- Voicu, S.I.; Condruz, R.M.; Mitran, V.; Cimpean, A.; Miculescu, F.; Andronescu, C.; Miculescu, M.; Thakur, V.K. Sericin Covalent Immobilization onto Cellulose Acetate Membranes. ACS Sustain. Chem. Eng. 2016, 4, 1765–1774. [Google Scholar] [CrossRef]
- Ionita, M.; Crica, L.E.; Voicu, S.I.; Pandele, A.M.; Iovu, H. Fabrication of Cellulose Triacetate/Graphene Oxide Porous Membrane. Polym. Adv. Technol. 2016, 27, 350–357. [Google Scholar] [CrossRef]
- Corobea, M.C.; Muhulet, O.; Miculescu, F.; Antoniac, I.V.; Vuluga, Z.; Florea, D.; Vuluga, D.M.; Butnaru, M.; Ivanov, D.; Voicu, S.I.; et al. Novel Nanocomposite Membranes from Cellulose Acetate and Clay-Silica Nanowires. Polym. Adv. Technol. 2016, 27, 1586–1595. [Google Scholar] [CrossRef]
- Yoo, Y.H.; Le, D.P.; Kim, J.G.; Kim, S.K.; Vinh, P.V. Corrosion behavior of TiN, TiAlN, TiAlSiN thin films deposited on tool steel in the 3.5 wt.% NaCl solution. Thin Solid Films 2008, 516, 3544–3548. [Google Scholar] [CrossRef]
- Voicu, I.; Ninciuleanu, C.M.; Muhulet, O.; Miculescu, M. Cellulose acetate membranes with controlled porosity and their use for the separation of aminoacids and proteins. J. Optoelectron. Adv. Mater. 2014, 16, 903–908. [Google Scholar]
- Thakur, V.K.; Voicu, S.I. Recent Advances in Cellulose and Chitosan Membranes for Water Purification: A Concise Review. Carbohydr. Polym. 2016, 146, 148–165. [Google Scholar] [CrossRef] [PubMed]
- Voicu, S.I.; Dobrica, A.; Sava, S.; Ivan, A.; Naftanaila, L. Cationic surfactants-controlled geometry and dimensions of polymeric membrane pores. J. Optoelectron. Adv. Mater. 2012, 14, 923–928. [Google Scholar]
- Miculescu, M.; Thakur, V.K.; Miculescu, F.; Voicu, S.I. Graphene based polymer nanocomposite membranes: A Review. Polym. Adv. Technol. 2016, 27, 844–859. [Google Scholar] [CrossRef]
- Liao, Y.; Xu, Q.; Zhang, J.; Niu, J.; Yuan, G.; Jiang, Y.; He, Y.; Wang, X. Cellular response of chondrocytes to magnesium alloys for orthopedic applications. Int. J. Mol. Med. 2015, 36, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Witte, F.; Feyerabend, F.; Maier, P.; Fischer, J.; Störmer, M.; Blawert, C.; Dietzel, W.; Hort, N. Biodegradable magnesium–hydroxyapatite metal matrix composites. Biomaterials 2007, 28, 2163–2174. [Google Scholar] [CrossRef] [PubMed]
- Fischer, J.; Pröfrock, D.; Hort, N.; Willumeit, R.; Feyerabend, F. Improved cytotoxicity testing of magnesium materials. Mater. Sci. Eng. B 2011, 176, 830–834. [Google Scholar] [CrossRef]
- Paul, W.; Sharma, C.P. Nanoceramic matrices: Biomedical applications. Am. J. Biochem. Biotechnol. 2006, 2, 41–48. [Google Scholar] [CrossRef]
- Zreiqat, H.; Howlett, C.R.; Zannettino, A.; Evans, P.; Schulze-Tanzil, G.; Knabe, C.; Shakibaei, M. Mechanisms of magnesium-stimulated adhesion of osteoblastic cells to commonly used orthopaedic implants. J. Biomed. Mater. Res. 2002, 62, 175–184. [Google Scholar] [CrossRef] [PubMed]
- LeGeros, R.Z.; Gatti, A.M.; Kijkowska, R.; Mijares, D.Q.; LeGeros, J.P. Magnesium tricalcium phosphate: Formation and properties. Key Eng. Mater. 2004, 254–256, 127–130. [Google Scholar] [CrossRef]
- Park, J.W.; Kim, Y.J.; Jang, J.H.; Song, H. Osteoblast response to magnesium ion-incorporated nanoporous titanium oxide surfaces. Clin. Oral Implants Res. 2010, 21, 1278–1287. [Google Scholar] [CrossRef] [PubMed]
- Takebe, J.; Itoh, S.; Okada, J.; Ishibashi, K. Anodic oxidation and hydrothermal treatment of titanium results in a surface that causes increased attachment and altered cytoskeletal morphology of rat bone marrow stromal cells in vitro. J. Biomed. Mater. Res. 2000, 51, 398–407. [Google Scholar] [CrossRef]
- Cifuentes, S.C.; Bensiamar, F.; Gallardo-Moreno, A.M.; Osswald, T.A.; González-Carrasco, J.L.; Benavente, R.; González-Martín, M.L.; García-Rey, E.; Vilaboa, N.; Saldaña, L. Incorporation of Mg particles into PDLLA regulates mesenchymal stem cell and macrophage responses. J. Biomed. Mater. Res A 2016, 104, 866–878. [Google Scholar] [CrossRef] [PubMed]
- Clarke, B. Normal Bone Anatomy and Physiology. Clin. J. Am. Soc. Nephrol. 2008, 3, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Boonrungsiman, S.; Gentleman, E.; Carzaniga, R.; Evans, N.D.; McComb, D.W.; Porter, A.E.; Stevens, M.M. The role of intracellular calcium phosphate in osteoblast-mediated bone apatite formation. Proc. Natl. Acad. Sci. USA 2012, 109, 14170–14175. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Yin, Q.; Zhang, Y.; Li, M.; Lan, G.; Lin, X.; Tan, L.; Yang, K. Biocompatibility of silicon containing micro-arc oxidation coated magnesium alloy ZK60 with osteoblasts cultured in vitro. Chin. J. Repar. Reconstr. Surg. 2013, 27, 612–618. [Google Scholar]
- Lund, S.A.; Giachelli, C.M.; Scatena, M. The role of osteopontin in inflammatory processes. J. Cell Commun. Signal. 2009, 3, 311–322. [Google Scholar] [CrossRef] [PubMed]
- Kläning, E.; Christensen, B.; Sørensen, E.S.; Vorup-Jensen, T.; Jensen, J.K. Osteopontin binds multiple calcium ions with high affinity and independently of phosphorylation status. Bone 2014, 66, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Carlsen, B.; Rudkin, G.; Berry, M.; Ishida, K.; Yamaguchi, D.T.; Miller, T.A. Osteopontin is a negative regulator of proliferation and differentiation in MC3T3-E1 pre-osteoblastic cells. Bone 2004, 34, 799–808. [Google Scholar] [CrossRef] [PubMed]



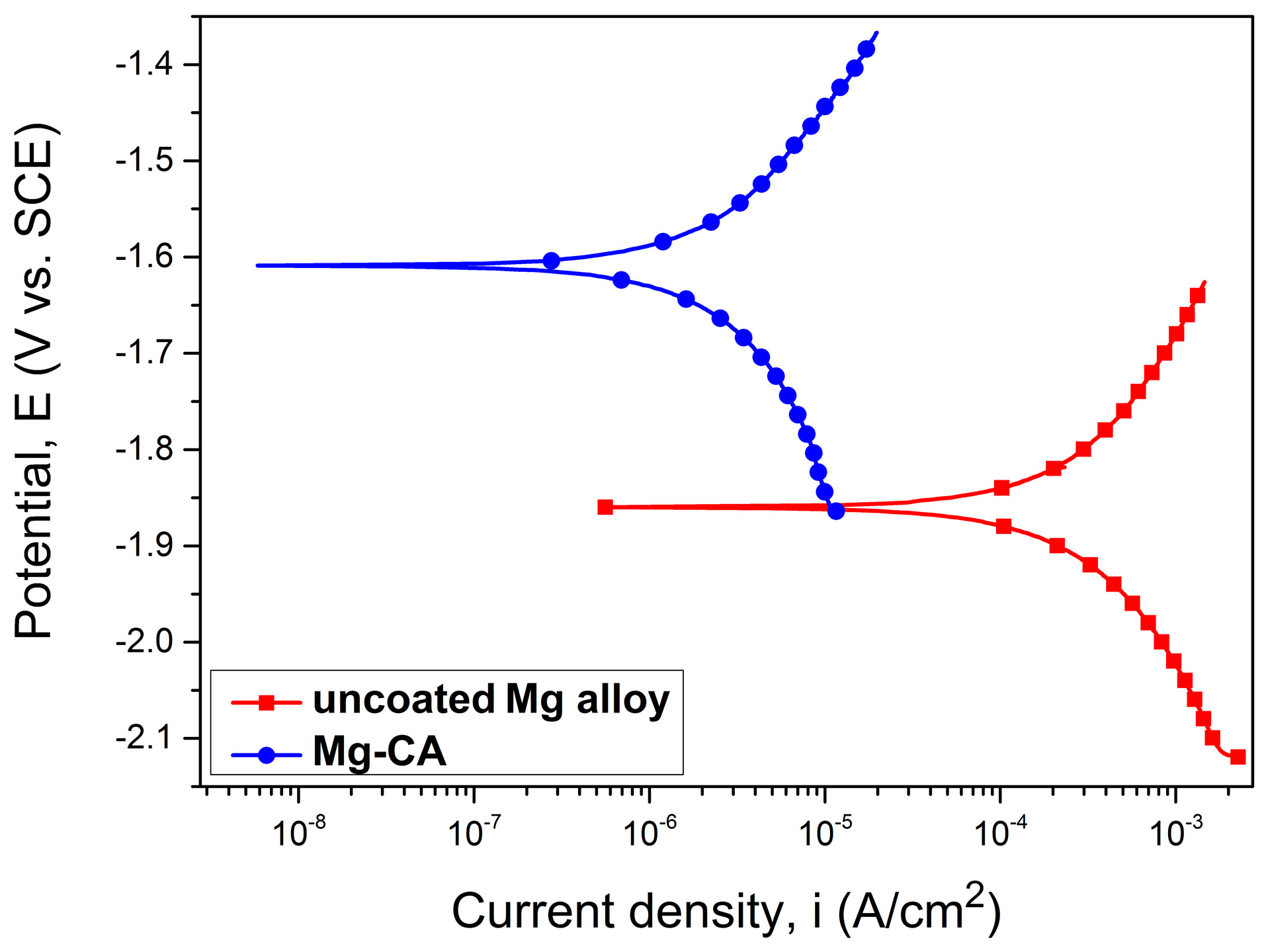

| Sample | Ecorr (V) | icorr (µA/cm2) | −βc (mV) | βa (mV) | Rp (kΩxcm2) | Pe (%) |
|---|---|---|---|---|---|---|
| uncoated Mg alloy | −1.85 | 497.96 | 422.92 | 475.14 | 0.195 | - |
| Mg-CA | −1.60 | 4.88 | 614.56 | 391.57 | 21.28 | 99.02 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neacsu, P.; Staras, A.I.; Voicu, S.I.; Ionascu, I.; Soare, T.; Uzun, S.; Cojocaru, V.D.; Pandele, A.M.; Croitoru, S.M.; Miculescu, F.; et al. Characterization and In Vitro and In Vivo Assessment of a Novel Cellulose Acetate-Coated Mg-Based Alloy for Orthopedic Applications. Materials 2017, 10, 686. https://doi.org/10.3390/ma10070686
Neacsu P, Staras AI, Voicu SI, Ionascu I, Soare T, Uzun S, Cojocaru VD, Pandele AM, Croitoru SM, Miculescu F, et al. Characterization and In Vitro and In Vivo Assessment of a Novel Cellulose Acetate-Coated Mg-Based Alloy for Orthopedic Applications. Materials. 2017; 10(7):686. https://doi.org/10.3390/ma10070686
Chicago/Turabian StyleNeacsu, Patricia, Adela Ioana Staras, Stefan Ioan Voicu, Iuliana Ionascu, Teodoru Soare, Seralp Uzun, Vasile Danut Cojocaru, Andreea Madalina Pandele, Sorin Mihai Croitoru, Florin Miculescu, and et al. 2017. "Characterization and In Vitro and In Vivo Assessment of a Novel Cellulose Acetate-Coated Mg-Based Alloy for Orthopedic Applications" Materials 10, no. 7: 686. https://doi.org/10.3390/ma10070686










