Double Biocatalysis Signal Amplification Glucose Biosensor Based on Porous Graphene
Abstract
:1. Introduction
2. Experimental
2.1. Reagents
2.2. Apparatus and Measurements
2.3. Biosynthesized Porous Graphene
2.4. Preparation of GOx-Modified Electrode
2.5. Growth of AuNPs
3. Results and Discussion
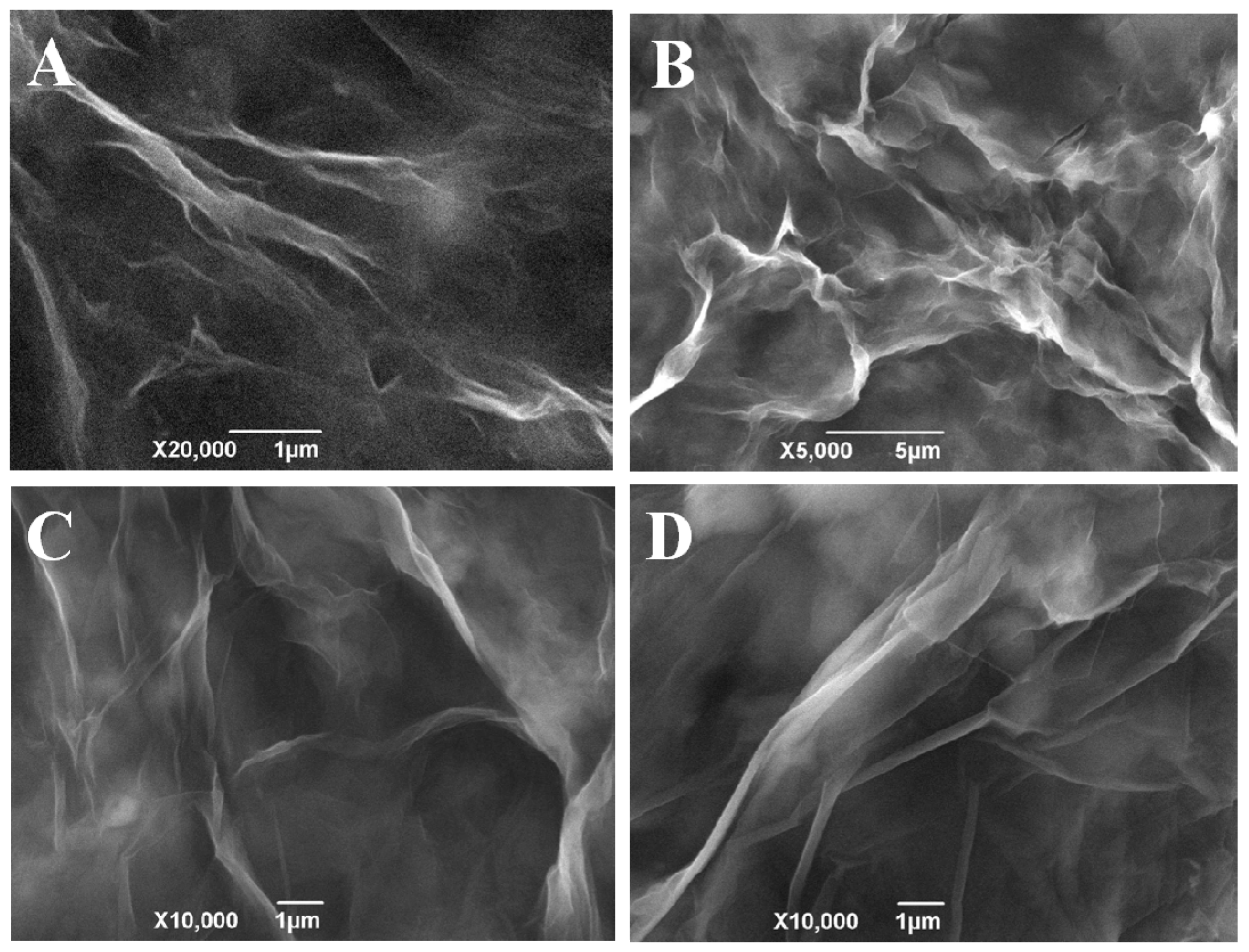
3.1. The Controllable Biosynthesized Porous Graphene
3.2. The Mechanisms of Biocatalytically–Grown AuNPs
3.3. Optimization of Sensor Performance
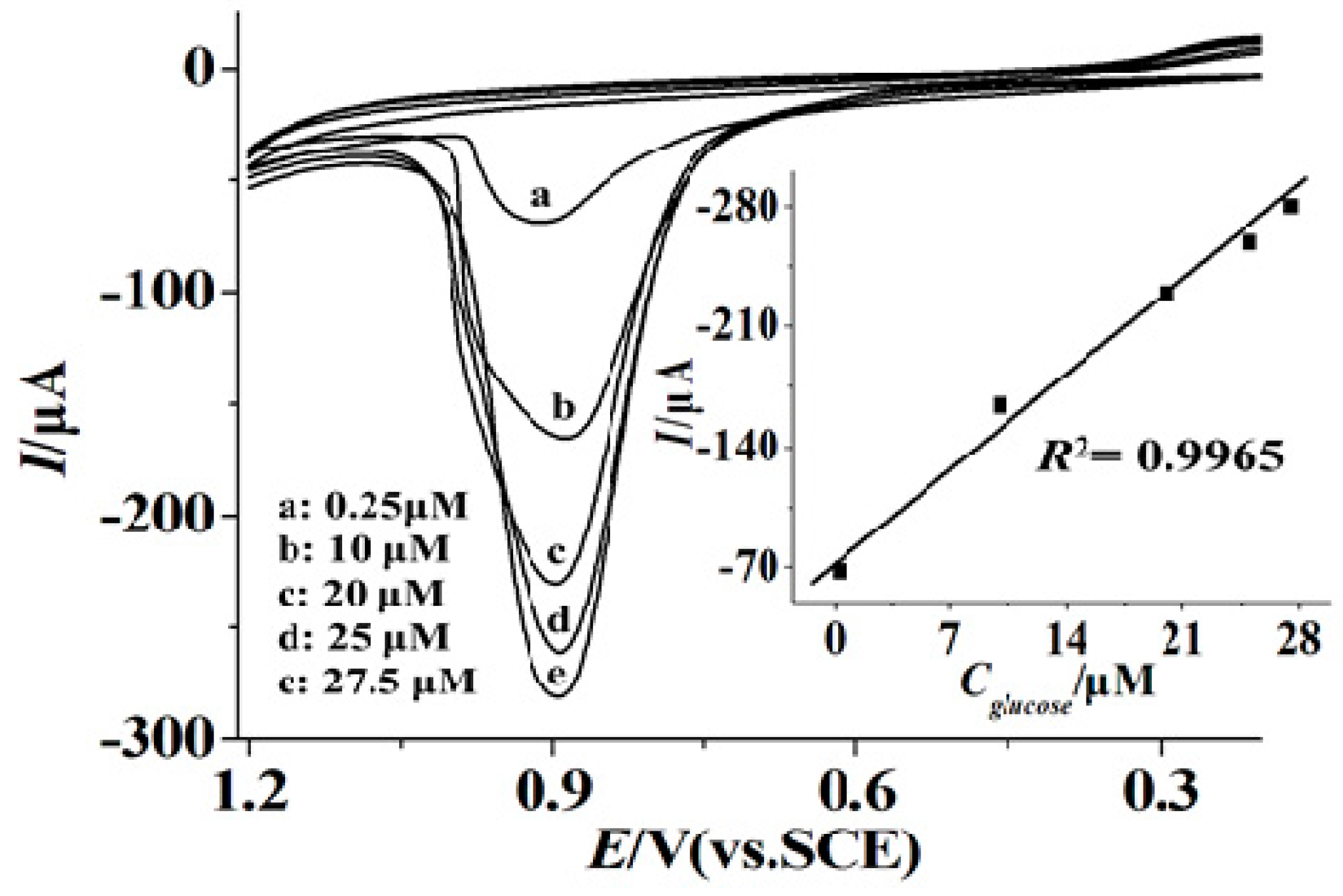
3.4. The Determination of Glucose Level
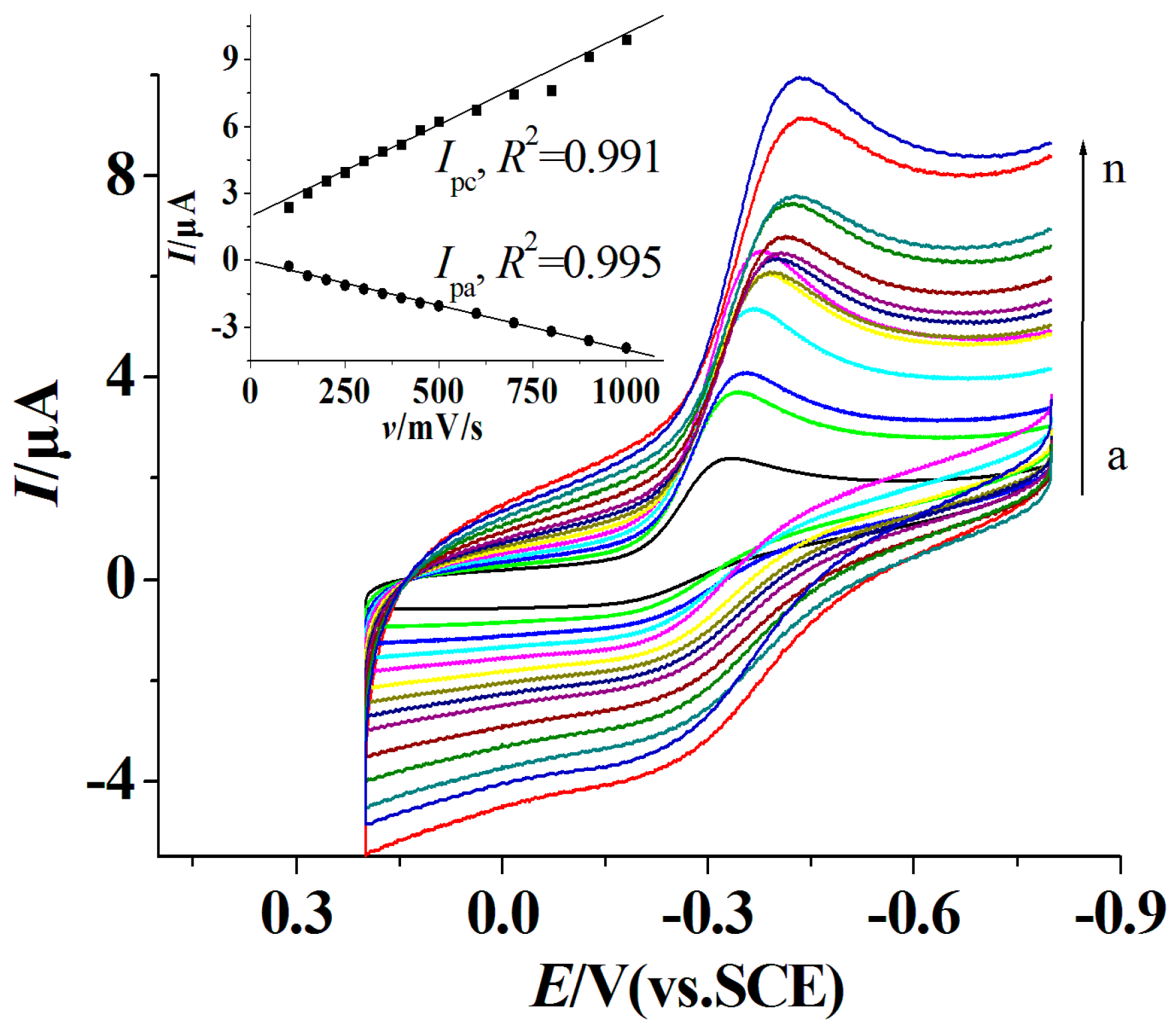
3.5. Direct Electrochemistry
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chandran, G.; Li, X.; Ogata, A.; Penner, R.M. Electrically transduced sensors based on nanomaterials (2012–2016). Anal. Chem. 2017, 89, 249–275. [Google Scholar] [CrossRef] [PubMed]
- Lizandara-Pueyo, C.; Siroky, S.; Wagner, M.R.; Hoffmann, A.; Reparaz, J.S.; Lehmann, M.; Polarz, S. Shape anisotropy influencing functional properties: Trigonal prismatic ZnO nanoparticles as an example. Adv. Funct. Mater. 2011, 21, 295–304. [Google Scholar] [CrossRef]
- Rathod, D.; Dickinson, C.; Egan, D. Platinum nanoparticle decoration of carbon materials with applications in non–enzymatic glucose sensing. Sens. Actuators B Chem. 2010, 143, 547–554. [Google Scholar] [CrossRef]
- Meng, H.; Xie, F.Y.; Chen, J. Morphology Controllable Growth of Pt Nanoparticles/nanowires on carbon powders and its application as novel electrocatalyst for methanol oxidation. Nanoscale 2011, 3, 5041–5048. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Yuan, R.; Chai, Y.Q. A high-sensitive amperometric hydrogen peroxide biosensor based on the immobilization of hemoglobin on gold colloid/l–cysteine/gold colloid/nanoparticles Pt–chitosan composite film–modified platinum disk electrode. Colloids Surf. B Biointerfaces 2008, 61, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Davis, M.E. Ordered Porous Materials for Emerging Applications. Nature 2002, 417, 813–821. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.X.; Wang, A.L.; Li, G.R.; Liu, Z.Q.; Zhao, W.X.; Su, C.Y.; Tong, Y.X. Porous Pt–Ni–P composite nanotube arrays: Highly electroactive and durable catalysts for methanol electrooxidation. J. Am. Chem. Soc. 2012, 134, 5730–5733. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.H.; Han, J.H.; Chung, T.D. Electrochemistry at nanoporous interfaces: New opportunity for electrocatalysis. Phys. Chem. Chem. Phys. 2012, 14, 448–463. [Google Scholar] [CrossRef] [PubMed]
- Dergunov, S.A.; Durbin, J.; Pattanaik, S.; Pinkhassik, E. pH–Mediated catch and release of charged molecules with porous hollow nanocapsules. J. Am. Chem. Soc. 2014, 136, 2212–2215. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.; Radhakrishnan, L.; Zhao, B.; Uppalapati, B.; Daniels, R.C.; Ward, K.R.; Collinson, M.M. Electrochemical properties of nanostructured porous gold electrodes in biofouling solutions. Anal. Chem. 2013, 85, 11610–11618. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Friedman, A.K.; Baker, L.A. Nanopore sensing. Anal. Chem. 2017, 89, 157–188. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.B.; Xiao, Y.; Lei, Z.B.; Zhang, Q. A promising electrochemical biosensing platform based on graphitized ordered mesoporous carbon. J. Mater. Chem. 2009, 19, 4707–4714. [Google Scholar] [CrossRef]
- Malhotra, R.; Papadimitrakopoulos, F.; Rusling, J.F. Sequential layer analysis of protein immunosensors based on single wall carbon nanotube forests. Langmuir 2010, 26, 15050. [Google Scholar] [CrossRef] [PubMed]
- He, Y.P.; Sheng, Q.L.; Zheng, J.B.; Liu, B. Magnetite–Graphene for the direct electrochemistry of hemoglobin and its biosensing application. Electrochim. Acta 2011, 56, 2471–2476. [Google Scholar] [CrossRef]
- Bertoncini, P.; Chauvet, O. Conformational Structural Changes of Bacteriorhodopsin Adsorbed onto Single–Walled Carbon Nanotubes. J. Phys. Chem. B 2010, 114, 4345–4350. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Guo, J.D.; Guo, L.P.; Bai, J. Electrochemical sensing platform based on the highly ordered mesoporous carbon–fullerene system. Anal. Chem. 2008, 80, 4642–4650. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.X.; He, Q.Y.; Tan, C.L.; Zhang, H. Graphene–Based electrochemical sensors. Small 2013, 9, 1160–1172. [Google Scholar] [CrossRef] [PubMed]
- Qiu, W.Z.; Nguyen, P.; Skafidas, E. Graphene nanopores: Electronic transport properties and design methodology. Phys. Chem. Chem. Phys. 2014, 16, 1451–1459. [Google Scholar] [CrossRef] [PubMed]
- Merchant, C.A.; Healy, K.; Wanunu, M.; Ray, V.; Peterman, N.; Bartel, J.; Fischbein, M.D.; Venta, K.; Luo, Z.T.; Johnson, A.T.; et al. DNA translocation through graphene nanopores. Nano Lett. 2010, 10, 2915–2921. [Google Scholar] [CrossRef] [PubMed]
- Siwy, Z.S.; Davenport, M. Nanopores: Graphene opens up to DNA. Nat. Nanotechnol. 2010, 5, 697–698. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, H.X.; Kong, J.L. Facile Preparation of mesocellular graphene foam for direct glucose oxidase electrochemistry and sensitive glucose sensing. Sens. Actuators B Chem. 2014, 193, 708–714. [Google Scholar] [CrossRef]
- Kim, T.Y.; Jung, G.; Yoo, S.; Kwang, S.S.; Rodney, S.R. Activated graphene–based carbons as supercapacitor electrodes with macro– and mesopores. ACS Nano 2013, 7, 6899–6905. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.Y.; Ning, Y.; Yang, Y.; Wang, C.Y. One–Step synthesis of porous graphene–based hydrogels containing oil droplets for drug delivery. RSC Adv. 2014, 4, 3211–3218. [Google Scholar] [CrossRef]
- Wilson, P.; Mbah, G.N.; Smith, T.G.; Schmidt, D.; Lai, R.Y.; Hofmann, T.; Sinitskii, A. Three–Dimensional periodic graphene nanostructures. J. Mater. Chem. C 2014, 2, 1751–1752. [Google Scholar] [CrossRef]
- Gregg, P.K.; Brett, L.A.; Harindra, V.; Yanamala, N.; Kapralov, A.A.; Tyurina, Y.Y.; Klein-Seetharaman, J.; Kagan, V.E.; Star, A. The enzymatic oxidation of graphene oxide. ACS Nano 2011, 5, 2098–2108. [Google Scholar]
- Liu, T.; Yu, K.; Gao, L.; Chen, H.; Wang, N.; Hao, L.H.; Li, T.X.; He, H.C.; Guo, Z.H. Graphene quantum dots decorated SrRuO3 mesoporous film as an efficient counter electrode for high-performance dye-sensitized solar cells. J. Mater. Chem. A 2017, 5, 17848–17855. [Google Scholar] [CrossRef]
- Liu, Z.; Huang, Z.; Cheng, F.; Guo, Z.H.; Wang, G.; Chen, X.; Wang, Z. Efficient dual-site carbon monoxide electro-catalysts via interfacial nano-engineering. Sci. Rep. 2016, 6, 33127–33137. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; He, Q.; Wei, H.; Guo, J.; Ding, K.Q.; Wang, Q.; Wei, S.Y.; Guo, Z.H. Optimal Electrocatalytic Pd/MWNTs nanocatalysts toward formic acid oxidation. Electrochim. Acta 2015, 184, 452–465. [Google Scholar] [CrossRef]
- Alippi, C. A unique timely moment for embedding intelligence in applications. CAAI Trans. Intell. Technol. 2016, 1, 1–3. [Google Scholar] [CrossRef]
- Jin, H.Y.; Chen, Q.; Chen, Z.X.; Hu, Y.; Zhang, J.W. Multi-Leap motion sensor based demonstration for robotic refine tabletop object manipulation task. CAAI Trans. Intell. Technol. 2016, 1, 104–113. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Gao, H.B.; Guo, M.; Li, G.P.; Liu, Y.C.; Li, D.Y. A study on key technologies of unmanned driving. CAAI Trans. Intell. Technol. 2016, 1, 4–13. [Google Scholar] [CrossRef]
- Padhy, S.; Panda, S. A hybrid stochastic fractal search and pattern search technique based cascade PI-PD controller for automatic generation control of multi-source power systems in presence of plug in electric vehicles. CAAI Trans. Intell. Technol. 2017, 2, 12–25. [Google Scholar] [CrossRef]
- Zheng, J.B.; He, Y.P.; Sheng, Q.L.; Zhang, H.F. DNA as a linker for biocatalytic deposition of Au nanoparticles on graphene and its application in glucose detection. J. Mater. Chem. 2011, 21, 12873–12879. [Google Scholar] [CrossRef]
- Zhang, H.F.; Liu, R.X.; Sheng, Q.L.; Zheng, J.B. Enzymatic deposition of Au nanoparticles on the designed electrode surface and its application in glucose detection. Colloids Surf. B 2011, 82, 532–535. [Google Scholar] [CrossRef] [PubMed]
- Li, K.T.; Liu, B.; Zheng, J.B.; Sheng, Q.L.; Liu, R.X. Direct electrochemistry of glucose oxidase on nail–like carbon and its biosensing for glucose. Electroanalysis 2010, 22, 701–706. [Google Scholar] [CrossRef]
- Liu, J.P.; Guo, C.X.; Li, C.M.; Li, Y.Y.; Chi, Q.B.; Huang, X.T.; Liao, L.; Yu, T. Carbon–decorated ZnO nanowire array: A novel platform for direct electrochemistry of enzymes and biosensing applications. Electrochem. Commun. 2009, 11, 202–205. [Google Scholar] [CrossRef]
- Laviron, E. The use of linear potential sweep voltammetry and of a.c. voltammetry for the study of the surface electrochemical reaction of strongly adsorbed systems and of redox modified electrodes. J. Electroanal. Chem. Interfacial Electrochem. 1979, 100, 263–270. [Google Scholar] [CrossRef]


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, Y.; Zheng, J.; Wang, B.; Ren, H. Double Biocatalysis Signal Amplification Glucose Biosensor Based on Porous Graphene. Materials 2017, 10, 1139. https://doi.org/10.3390/ma10101139
He Y, Zheng J, Wang B, Ren H. Double Biocatalysis Signal Amplification Glucose Biosensor Based on Porous Graphene. Materials. 2017; 10(10):1139. https://doi.org/10.3390/ma10101139
Chicago/Turabian StyleHe, Yaping, Jianbin Zheng, Bini Wang, and Hongjiang Ren. 2017. "Double Biocatalysis Signal Amplification Glucose Biosensor Based on Porous Graphene" Materials 10, no. 10: 1139. https://doi.org/10.3390/ma10101139




