Influence of Combustion Parameters on Fouling Composition after Wood Pellet Burning in a Lab-Scale Low-Power Boiler
Abstract
:1. Introduction
2. Experimental Section
2.1. Combustion Facility
2.2. Fuel Burned in the Experimental Plant

| Proximate analysis (wt.% of dry fuel) | |
| Moisture | 6.18 |
| Volatile Matter | 70.57 |
| Fixed Carbon | 26.11 |
| Ash | 3.32 |
| Ultimate analysis (wt.% of dry fuel) | |
| C | 46.03 |
| H | 5.95 |
| N | 1.88 |
| O * | 42.82 * |
| Analysis of ash using XRF (wt.% of ash) | |
| Na2O | 0.36 |
| MgO | 4.85 |
| Al2O3 | 6.97 |
| SiO2 | 35.46 |
| P2O5 | 2.75 |
| SO3 | 6.04 |
| Cl | 0.95 |
| K2O | 6.46 |
| CaO | 28.03 |
| TiO2 | 3.14 |
| MnO | 0.29 |
| Fe2O3 | 3.52 |
| NiO | 0.02 |
| CuO | 0.04 |
| ZnO | 0.27 |
| Heating value (MJ/kg in wet basis) | |
| HV | 16.72 |
2.3. Combustions Performed and Samples Collected
| Experiment | Total time of stable combustion | Primary–secondary airflow distribution | Total airflow supplied | Deposition probe temperature |
|---|---|---|---|---|
| 1 | 3 h | 25%–75% | 11 m3/h | ≈20 °C–25 °C |
| 2 | 3 h | 25%–75% | 14 m3/h | ≈20 °C–25 °C |
| 3 | 3 h | 25%–75% | 17 m3/h | ≈20 °C–25 °C |
| 4 | 3 h | 25%–75% | 20 m3/h | ≈20 °C–25 °C |
| 5 | 3 h | 25%–75% | 23 m3/h | ≈20 °C–25 °C |
| 6 | 3 h | 25%–75% | 25 m3/h | ≈20 °C–25 °C |
| 7 | 3 h | 25%–75% | 11 m3/h | ≈60 °C–65 °C |
| 8 | 3 h | 25%–75% | 17 m3/h | ≈60 °C–65 °C |
| 9 | 3 h | 25%–75% | 25 m3/h | ≈60 °C–65 °C |

2.4. Theoretical Deposition Indices
| Name | Equation | Equation number | Approximate ranges |
|---|---|---|---|
| Base-to-acid ratio | (1) | (*) | |
| Base-to-acid ratio (simplified) | (2) | ||
| Base-to-acid ratio (+P) | (3) | ||
| Slagging index | (4) | RS < 0.6 low slagging
RS = 0.6–2.0 medium RS = 2.0–2.6 high RS > 2.6 extremely high | |
| Slag ratio | (5) | SR > 72 low slagging
72 ≥ SR > 65 medium SR < 65 high | |
| Fouling index | (6) | FU ≤ 0.6 low fouling
0.6 < FU ≤ 40 high FU > 40 extremely high (tendency to sintering of deposits) |
2.5. Thermal and Chemical Analysis Performed to the Samples
2.5.1. Thermogravimetry and Differential Scanning Calorimetry (TG-DSC)
2.5.2. Scanning Electron Microscopy with Energy Dispersive X-Ray Spectroscopy (SEM-EDS)
3. Results and Discussion
3.1. Theoretical Deposition Indices Predicted from Inorganic Elements
| Index | Result | Interpretation |
|---|---|---|
| B/A | 0.95 | Low-Medium |
| B/A(simp) | 0.86 | |
| B/A(+P) | 1.01 | |
| RS | 0.05 | Low |
| RS(+P) | 0.05 | Low |
| SR | 49.35 | Medium |
| FU | 6.47 | High |
| FU(+P) | 6.88 | High |

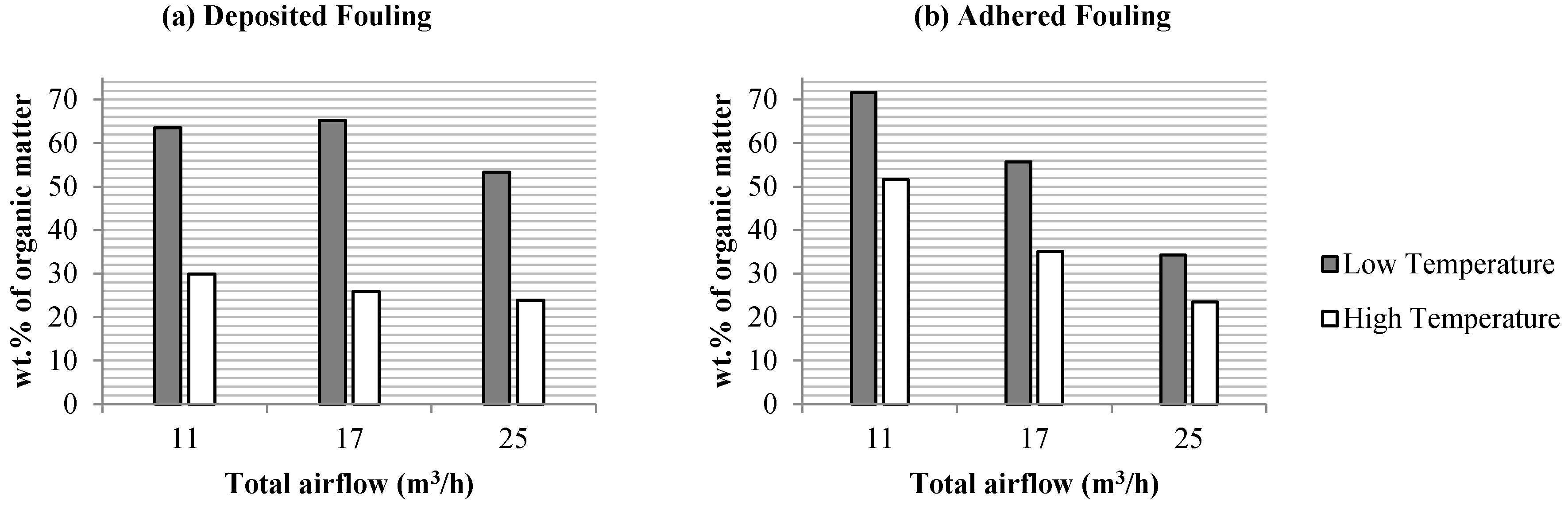
3.2. Influence on Fouling of the Total Airflow Supplied
3.2.1. Thermal Analysis: Characterization of the Organic Matter in the Fouling Deposits
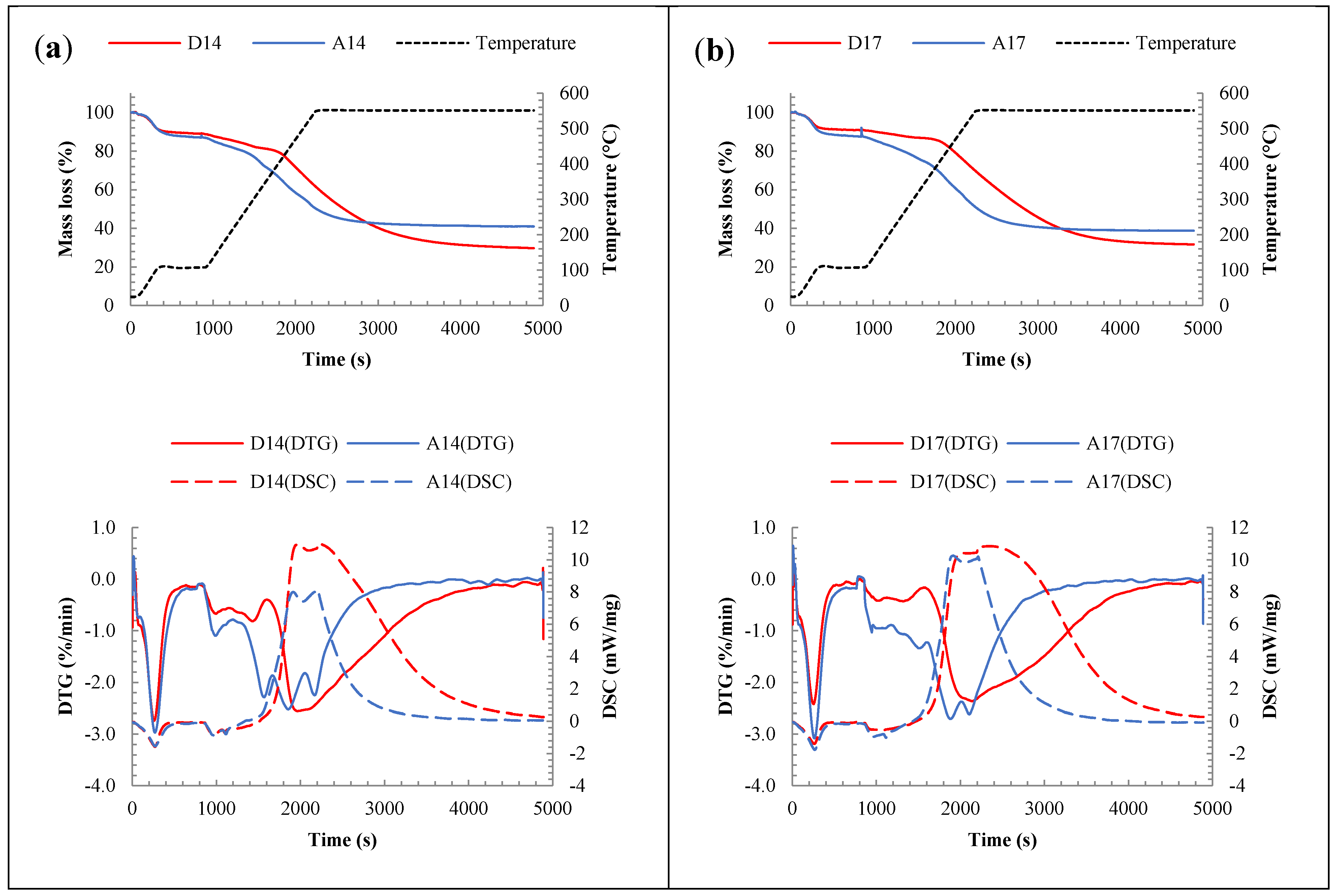
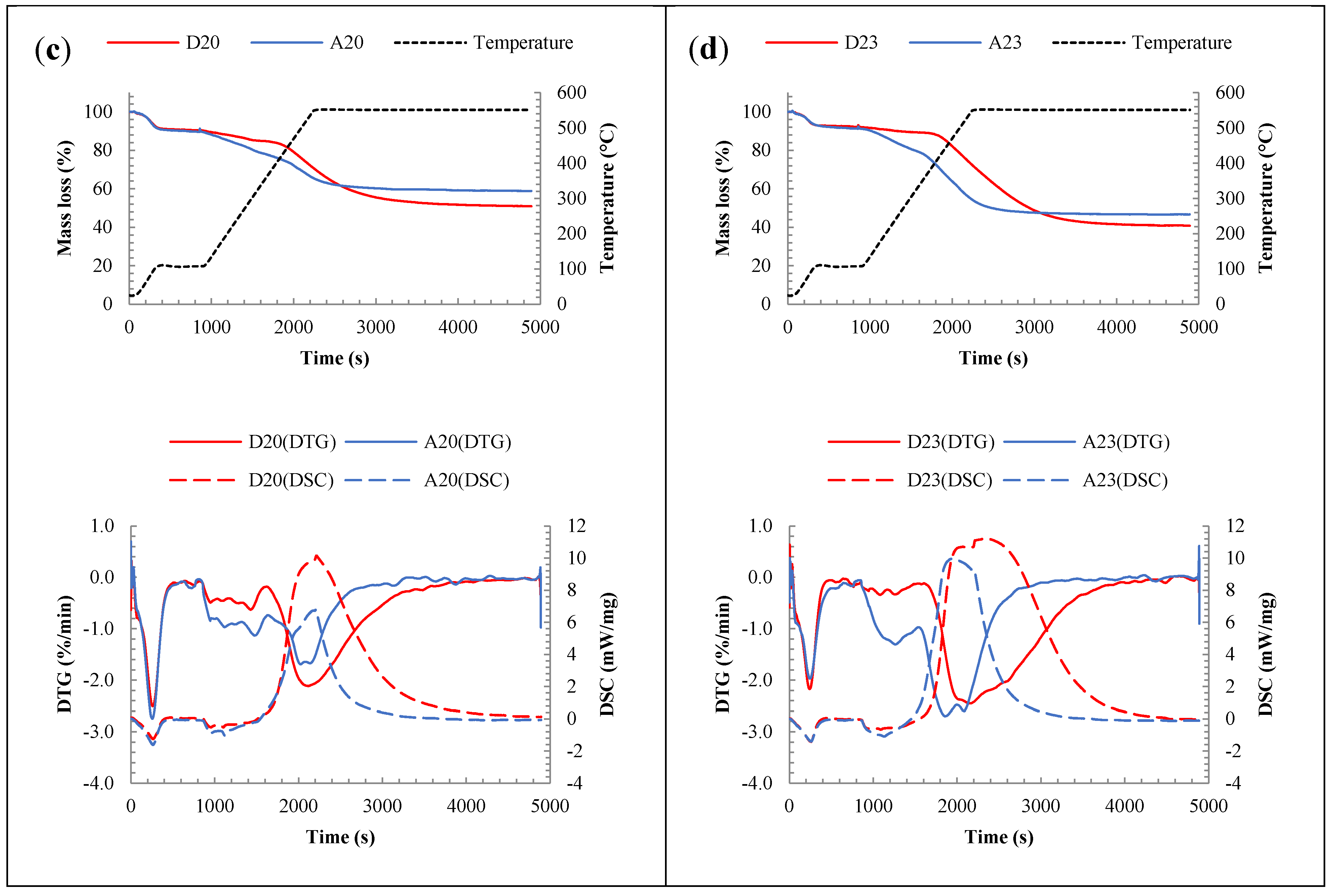
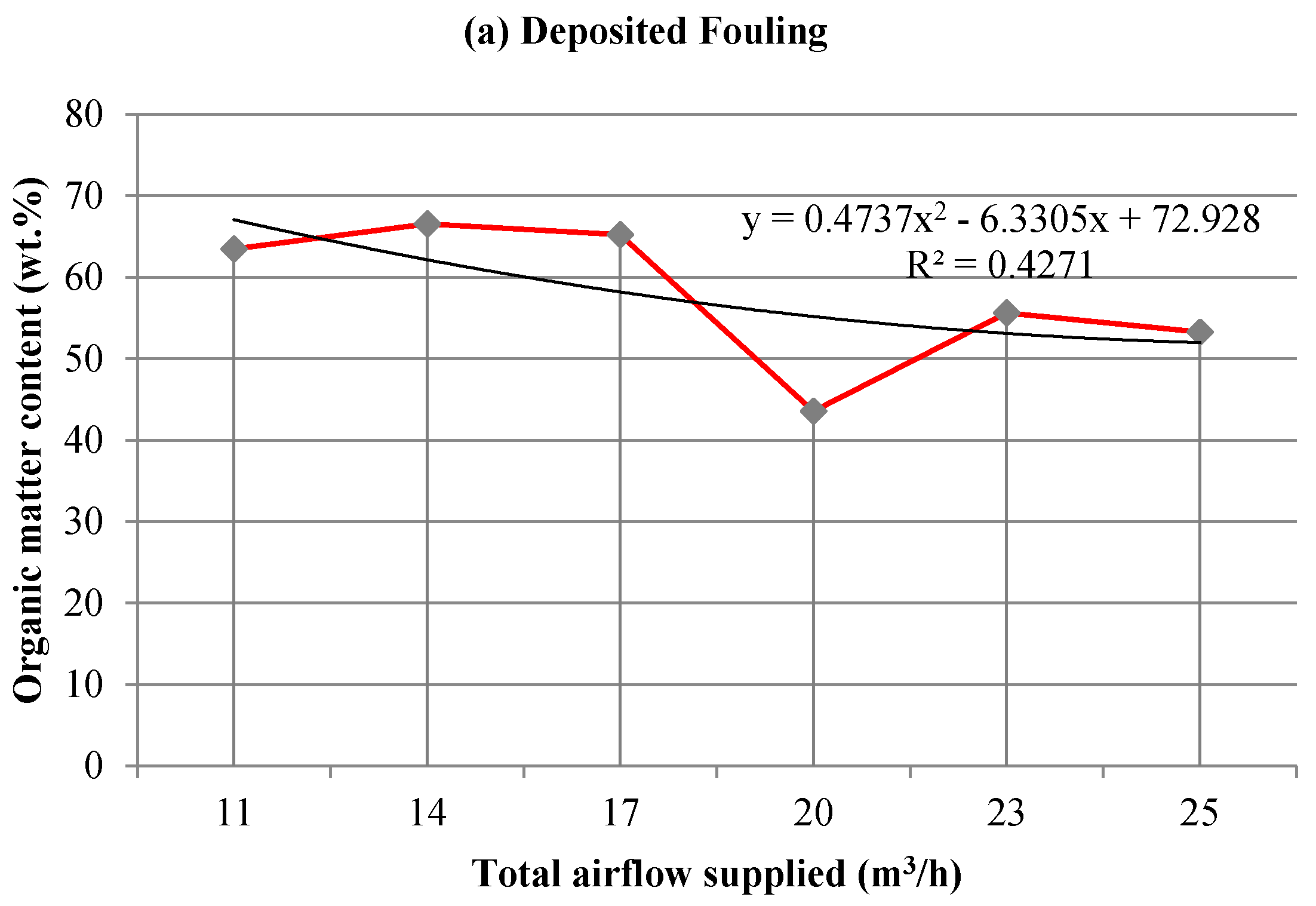
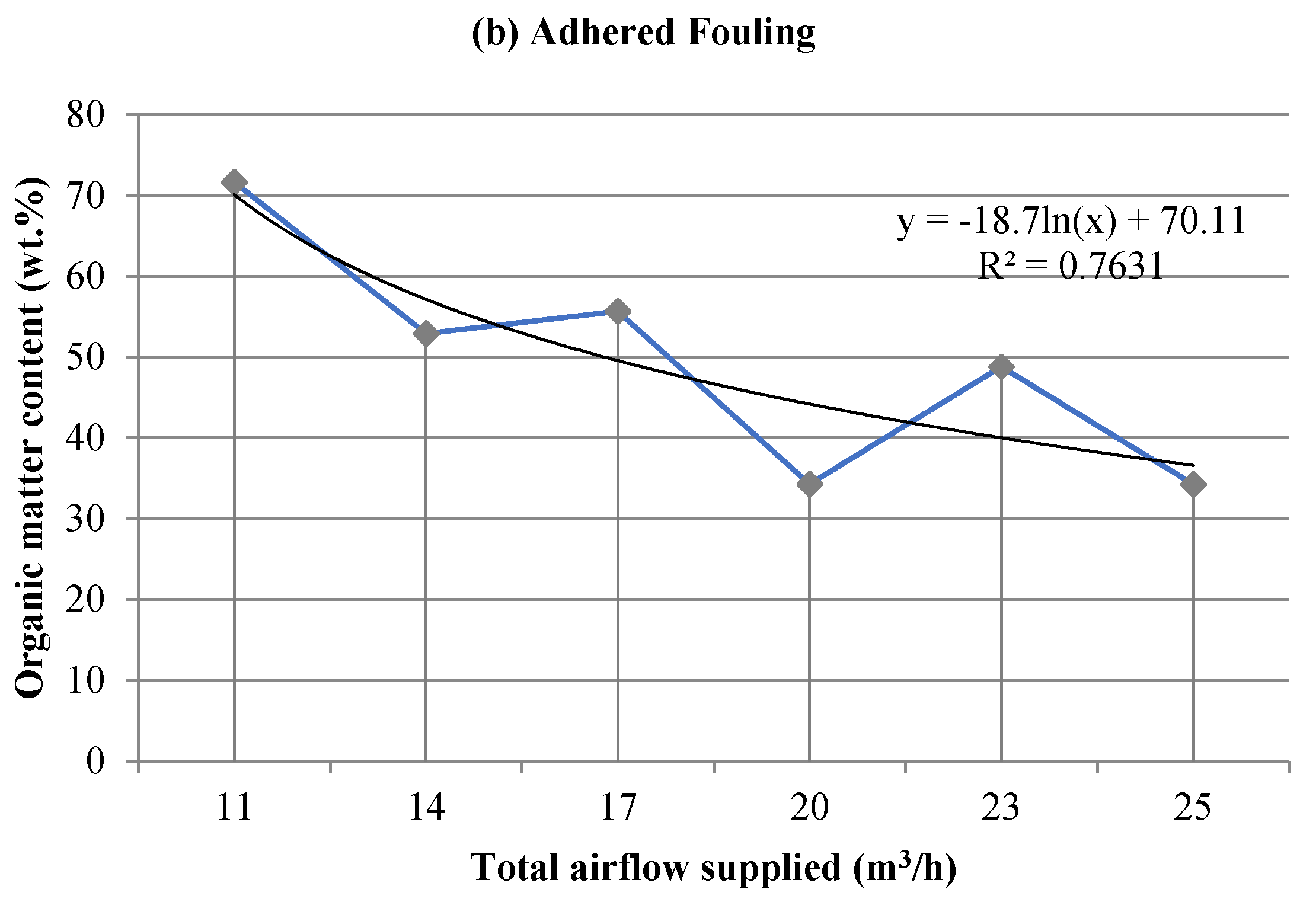
3.2.2. Chemical Analysis: Characterization of the Inorganic Matter in the Fouling Deposits
| D11LT (wt.%) | A11LT (wt.%) | D25LT (wt.%) | A25LT (wt.%) | |
|---|---|---|---|---|
| C | 75.58 | 69.38 | 53.47 | 31.59 |
| O | 9.17 | 11.24 | 16.08 | 15.27 |
| Na | 0.52 | 0.74 | 1.15 | 1.74 |
| Mg | 0.27 | 0.24 | 1.06 | 0.75 |
| Al | 0.23 | 0.38 | 0.46 | 0.65 |
| Si | 0.54 | 0.73 | 1.91 | 0.88 |
| P | 0.14 | 0.26 | 0.39 | 0.46 |
| S | 1.30 | 2.60 | 3.48 | 1.61 |
| Cl | 7.72 | 6.36 | 8.65 | 16.43 |
| K | 1.51 | 1.99 | 3.58 | 3.49 |
| Ca | 1.73 | 3.89 | 6.51 | 2.65 |
| Ti | 0.16 | 0.28 | 0.35 | 0.35 |
| Cr | 0.00 | 0.00 | 0.18 | 3.83 |
| Mn | 0.30 | 0.23 | 0.41 | 0.44 |
| Fe | 0.60 | 0.68 | 1.71 | 16.78 |
| Ni | 0.00 | 0.00 | 0.00 | 2.13 |
| Zn | 0.22 | 1.00 | 0.61 | 0.92 |
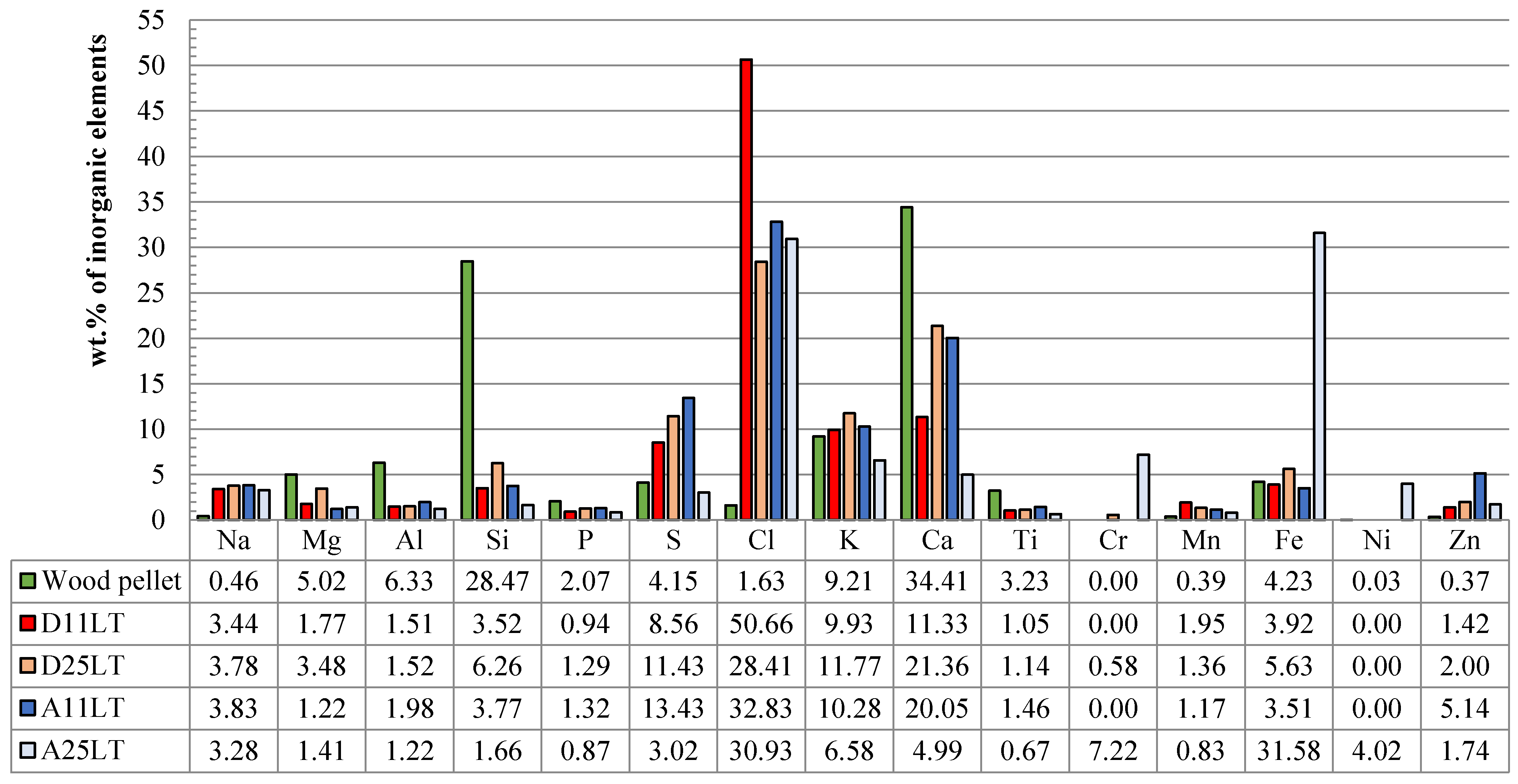
3.3. Influence on Fouling of the Deposition Probe Temperature
3.3.1. Thermal Analysis: Characterization of the Organic Matter in the Fouling Deposits
- T∞,1: water temperature (293 K or 333 K)
- T∞,3: gases temperature (773 K)
- r1: inner radio of the tube (0.0105 m)
- r2: outer radio of the tube (0.0125 m)
- r3: outer radio of the tube plus fouling thickness
- L: length of the tube (0.12 m)
- h1: convection coefficient of water (2500 W/m2K)
- kA: conduction coefficient of steel (14.9 W/mK stainless steel AISI 304 [44])
- kB: conduction coefficient of fouling (1.95 W/mK [45])
- h3: convection coefficient of gases (100 W/m2K [46])

3.3.2. Chemical Analysis: Characterization of the Inorganic Matter in the Fouling Deposits
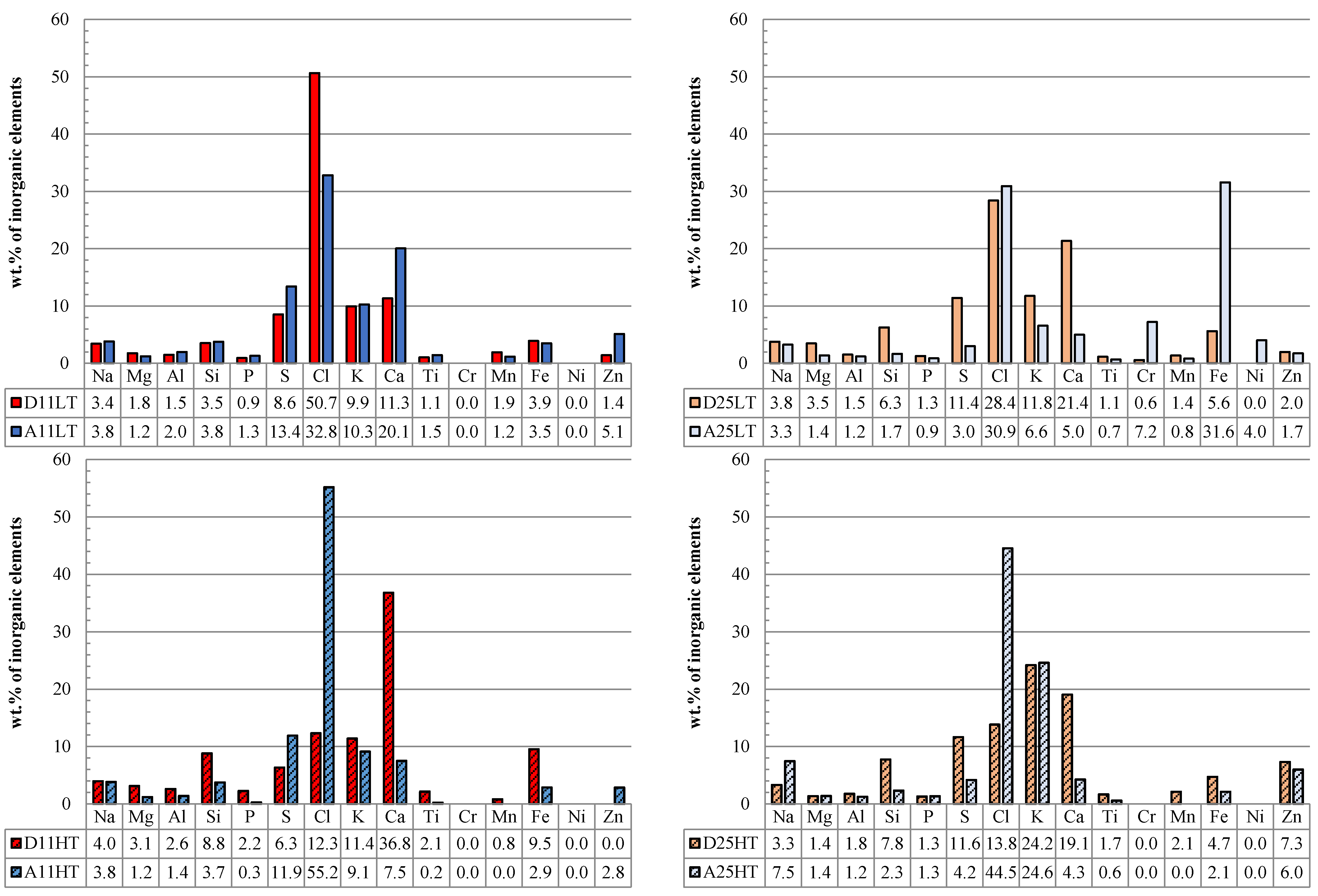
4. Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Demirbas, A. Potential applications of renewable energy sources, biomass combustion problems in boiler power systems and combustion related environmental issues. Prog. Energy Combust. Sci. 2005, 31, 171–192. [Google Scholar] [CrossRef]
- Fargione, J.; Hill, J.; Tilman, D.; Polasky, S.; Hawthorne, P. Land clearing and the biofuel carbon debt. Science 2008, 319, 1235–1238. [Google Scholar] [CrossRef] [PubMed]
- Bridgwater, A.V. Renewable fuels and chemicals by thermal processing of biomass. Chem. Eng. J. 2003, 91, 87–102. [Google Scholar] [CrossRef]
- Demirbas, M.F.; Balat, M.; Balat, H. Potential contribution of biomass to the sustainable energy development. Energ. Convers. Manag. 2009, 50, 1746–1760. [Google Scholar] [CrossRef]
- Tchapda, A.H.; Pisupati, S.V. A review of thermal co-conversion of coal and biomass/waste. Energies 2014, 7, 1098–1148. [Google Scholar] [CrossRef]
- Saidur, R.; Abdelaziz, E.A.; Demirbas, A.; Hossain, M.S.; Mekhilef, S. A review on biomass as a fuel for boilers. Renew. Sust. Energ. Rev. 2011, 15, 2262–2289. [Google Scholar] [CrossRef]
- Khan, A.A.; de Jong, W.; Jansens, P.J.; Spliethoff, H. Biomass combustion in fluidized bed boilers: Potential problems and remedies. Fuel Process. Technol. 2009, 90, 21–50. [Google Scholar] [CrossRef]
- Vassilev, S.V.; Baxter, D.; Andersen, L.K.; Vassileva, C.G.; Morgan, T.J. An overview of the organic and inorganic phase composition of biomass. Fuel 2012, 94, 1–33. [Google Scholar] [CrossRef]
- McKendry, P. Energy production from biomass (part 1): Overview of biomass. Bioresource Technol. 2002, 83, 37–46. [Google Scholar] [CrossRef]
- Vassilev, S.V.; Baxter, D.; Andersen, L.K.; Vassileva, C.G. An overview of the chemical composition of biomass. Fuel 2010, 89, 913–933. [Google Scholar] [CrossRef]
- Demirbas, A. Combustion characteristics of different biomass fuels. Prog. Energy Combust. Sci. 2004, 30, 219–230. [Google Scholar] [CrossRef]
- Bryers, R.W. Fireside slagging, fouling, and high-temperature corrosion of heat-transfer surface due to impurities in steam-raising fuels. Prog. Energy Combust. Sci. 1996, 22, 29–120. [Google Scholar] [CrossRef]
- Teixeira, P.; Lopes, H.; Gulyurtlu, I.; Lapa, N.; Abelha, P. Evaluation of slagging and fouling tendency during biomass co-firing with coal in a fluidized bed. Biomass Bioenerg. 2012, 39, 192–203. [Google Scholar] [CrossRef]
- Pronobis, M. The influence of biomass co-combustion on boiler fouling and efficiency. Fuel 2006, 85, 474–480. [Google Scholar] [CrossRef]
- Gilbe, C.; Öhman, M.; Lindström, E.; Boström, D.; Backman, R.; Samuelsson, R.; Burvall, J. Slagging characteristics during residential combustion of biomass pellets. Energ. Fuel. 2008, 22, 3536–3543. [Google Scholar] [CrossRef]
- Jenkins, B.M.; Baxter, L.L.; Miles, T.R., Jr.; Miles, T.R. Combustion properties of biomass. Fuel Process. Technol. 1998, 54, 17–46. [Google Scholar] [CrossRef]
- Skrifvars, B.J.; Yrjas, P.; Laurén, T.; Kinni, J.; Tran, H.; Hupa, M. The fouling behavior of rice husk ash in fluidized-bed combustion. 2. Pilot-scale and full-scale measurements. Energ. Fuel. 2005, 19, 1512–1519. [Google Scholar] [CrossRef]
- James, A.K.; Thring, R.W.; Helle, S.; Ghuman, H.S. Ash management review-applications of biomass bottom ash. Energies 2012, 5, 3856–3873. [Google Scholar] [CrossRef]
- Theis, M.; Skrifvars, B.-J.; Hupa, M.; Tran, H. Fouling tendency of ash resulting from burning mixtures of biofuels. Part 1: Deposition rates. Fuel 2006, 85, 1125–1130. [Google Scholar] [CrossRef]
- Szemmelveisz, K.; Szucs, I.; Palotás, Á.B.; Winkler, L.; Eddings, E.G. Examination of the combustion conditions of herbaceous biomass. Fuel Process. Technol. 2009, 90, 839–847. [Google Scholar] [CrossRef]
- Yin, C.; Rosendahl, L.A.; Kær, S.K. Grate-firing of biomass for heat and power production. Prog. Energy Combust. Sci. 2008, 34, 725–754. [Google Scholar] [CrossRef]
- Shao, Y.; Wang, J.; Preto, F.; Zhu, J.; Xu, C. Ash deposition in biomass combustion or co-firing for power/heat generation. Energies 2012, 5, 5171–5189. [Google Scholar] [CrossRef]
- Johansen, J.M.; Aho, M.; Paakkinen, K.; Taipale, R.; Egsgaard, H.; Jakobsen, J.G.; Frandsen, F.J.; Glarborg, P. Release of k, cl, and s during combustion and co-combustion with wood of high-chlorine biomass in bench and pilot scale fuel beds. Proc. Combust. Inst. 2013, 34, 2363–2372. [Google Scholar] [CrossRef]
- Li, R.; Kai, X.; Yang, T.; Sun, Y.; He, Y.; Shen, S. Release and transformation of alkali metals during co-combustion of coal and sulfur-rich wheat straw. Energ. Convers. Manag. 2014, 83, 197–202. [Google Scholar] [CrossRef]
- Pronobis, M.; Kalisz, S.; Polok, M. The impact of coal characteristics on the fouling of stoker-fired boiler convection surfaces. Fuel 2013, 112, 473–482. [Google Scholar] [CrossRef]
- Tortosa Masiá, A.A.; Buhre, B.J.P.; Gupta, R.P.; Wall, T.F. Characterising ash of biomass and waste. Fuel Process. Technol. 2007, 88, 1071–1081. [Google Scholar]
- Viana, H.; Vega-Nieva, D.J.; Ortiz Torres, L.; Lousada, J.; Aranha, J. Fuel characterization and biomass combustion properties of selected native woody shrub species from central portugal and nw spain. Fuel 2012, 102, 737–745. [Google Scholar] [CrossRef]
- Theis, M.; Skrifvars, B.-J.; Zevenhoven, M.; Hupa, M.; Tran, H. Fouling tendency of ash resulting from burning mixtures of biofuels. Part 3. Influence of probe surface temperature. Fuel 2006, 85, 2002–2011. [Google Scholar] [CrossRef]
- Febrero, L.; Granada, E.; Pérez, C.; Patiño, D.; Arce, E. Characterisation and comparison of biomass ashes with different thermal histories using tg-dsc. J. Therm. Anal. Calorim. 2014, 118, 669–680. [Google Scholar] [CrossRef]
- Granada, E.; Patiño, D.; Míguez, J.L.; Morán, J. Analysis of deposition of fouled matter in a water-tube heat exchanger of a biomass combustor. In Proceedings of the 34th International Symposium on Combustion, Warsaw, Poland, 29 July–3 August 2012.
- Vamvuka, D.; Zografos, D. Predicting the behaviour of ash from agricultural wastes during combustion. Fuel 2004, 83, 2051–2057. [Google Scholar] [CrossRef]
- Darvell, L.I.; Jones, J.M.; Gudka, B.; Baxter, X.C.; Saddawi, A.; Williams, A.; Malmgren, A. Combustion properties of some power station biomass fuels. Fuel 2010, 89, 2881–2890. [Google Scholar] [CrossRef]
- Pronobis, M. Evaluation of the influence of biomass co-combustion on boiler furnace slagging by means of fusibility correlations. Biomass Bioenerg. 2005, 28, 375–383. [Google Scholar] [CrossRef]
- Bartolomé, C.; Gil, A. Ash deposition and fouling tendency of two energy crops (cynara and poplar) and a forest residue (pine chips) co-fired with coal in a pulverized fuel pilot plant. Energ. Fuel. 2013, 27, 5878–5889. [Google Scholar] [CrossRef]
- Coaltech. Common slagging and fouling indices. Available online: http://www.coaltech.com.au/LinkedDocuments/Slagging%20&%20Fouling.pdf (accessed on 6 May 2014).
- Vassilev, S.V.; Baxter, D.; Andersen, L.K.; Vassileva, C.G. An overview of the composition and application of biomass ash. Part 1. Phase-mineral and chemical composition and classification. Fuel 2013, 105, 40–76. [Google Scholar] [CrossRef]
- Obernberger, I.; Brunner, T.; Bärnthaler, G. Chemical properties of solid biofuels-significance and impact. Biomass Bioenerg. 2006, 30, 973–982. [Google Scholar] [CrossRef]
- Baxter, L.L. Ash deposition during biomass and coal combustion: A mechanistic approach. Biomass Bioenerg. 1993, 4, 85–102. [Google Scholar] [CrossRef]
- Umamaheswaran, K.; Batra, V.S. Physico-chemical characterisation of indian biomass ashes. Fuel 2008, 87, 628–638. [Google Scholar] [CrossRef]
- Pietro, M.; Paola, C. Thermal analysis for the evaluation of the organic matter evolution during municipal solid waste aerobic composting process. Thermochim. Acta 2004, 413, 209–214. [Google Scholar] [CrossRef]
- Batra, V.S.; Urbonaite, S.; Svensson, G. Characterization of unburned carbon in bagasse fly ash. Fuel 2008, 87, 2972–2976. [Google Scholar] [CrossRef]
- Vassilev, S.V.; Baxter, D.; Andersen, L.K.; Vassileva, C.G. An overview of the composition and application of biomass ash.: Part 2. Potential utilisation, technological and ecological advantages and challenges. Fuel 2013, 105, 19–39. [Google Scholar] [CrossRef]
- Broström, M.; Enestam, S.; Backman, R.; Mäkelä, K. Condensation in the kcl–nacl system. Fuel Process. Technol. 2013, 105, 142–148. [Google Scholar] [CrossRef]
- Incropera, F.P.; DeWitt, D.P. Fundamentos De Transferencia De Calor, 4th ed.; Prentice Hall: Zapopan, Mexico, 1999; p. 886. [Google Scholar]
- Quan, Z.; Chen, Y.; Ma, C. Experimental study of fouling on heat transfer surface during forced convective heat transfer. Chinese J. Chem. Eng. 2008, 16, 535–540. [Google Scholar] [CrossRef]
- Kreith, F.; Manglik, R.M.; Bohn, M.S. Principios De Transferencia De Calor, 7th ed.; Cengage Learning Editores S.A.: Santa Fe, Mexico, 2012; Chapter 8; p. 496. [Google Scholar]
- Jiménez, S.; Ballester, J. Influence of operating conditions and the role of sulfur in the formation of aerosols from biomass combustion. Combust. Flame 2005, 140, 346–358. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Febrero, L.; Granada, E.; Regueiro, A.; Míguez, J.L. Influence of Combustion Parameters on Fouling Composition after Wood Pellet Burning in a Lab-Scale Low-Power Boiler. Energies 2015, 8, 9794-9816. https://doi.org/10.3390/en8099794
Febrero L, Granada E, Regueiro A, Míguez JL. Influence of Combustion Parameters on Fouling Composition after Wood Pellet Burning in a Lab-Scale Low-Power Boiler. Energies. 2015; 8(9):9794-9816. https://doi.org/10.3390/en8099794
Chicago/Turabian StyleFebrero, Lara, Enrique Granada, Araceli Regueiro, and José Luis Míguez. 2015. "Influence of Combustion Parameters on Fouling Composition after Wood Pellet Burning in a Lab-Scale Low-Power Boiler" Energies 8, no. 9: 9794-9816. https://doi.org/10.3390/en8099794







