Screening and Evaluation of Some Green Algal Strains (Chlorophyceae) Isolated from Freshwater and Soda Lakes for Biofuel Production
Abstract
:1. Introduction
2. Results and Discussion
2.1. Sampling, Isolation and Morphological Identification of Microalgae
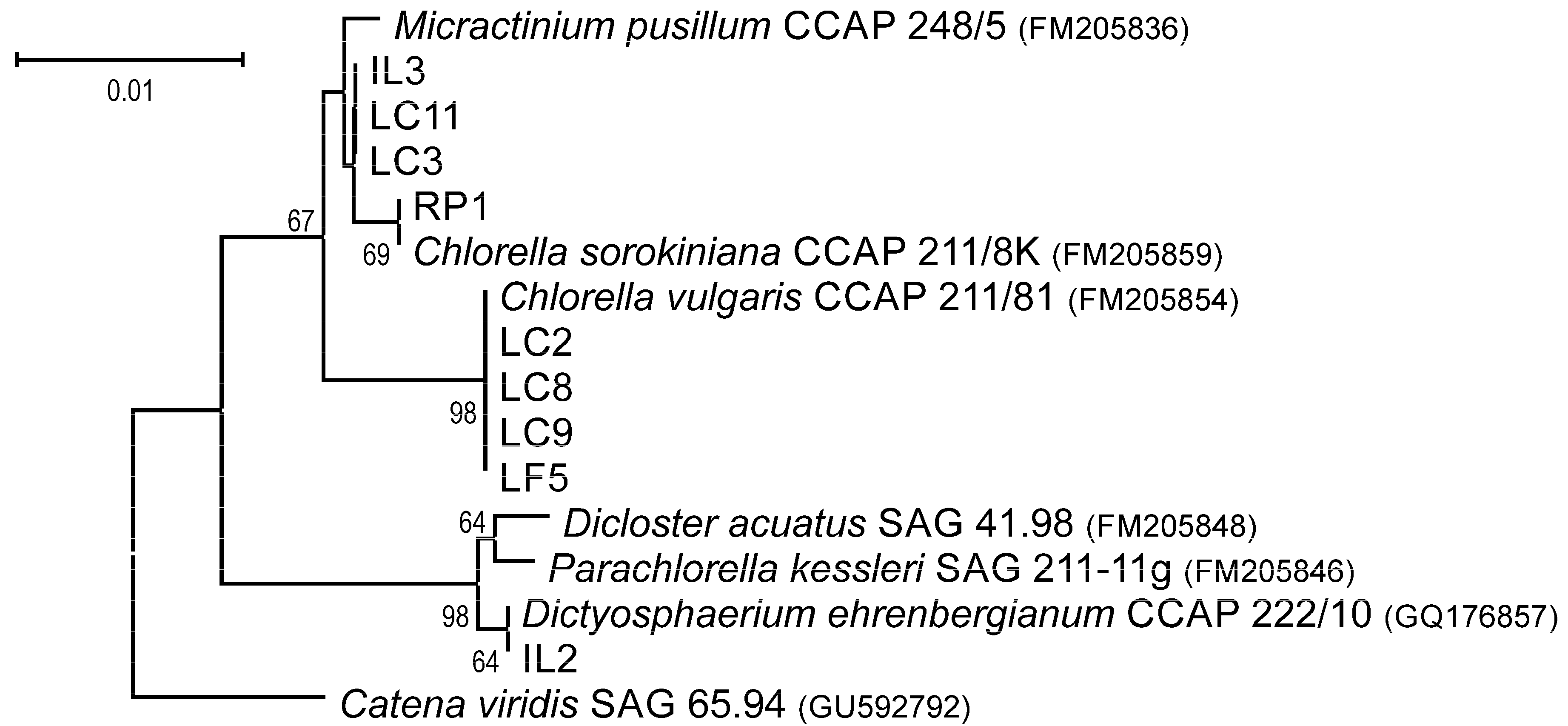
2.2. Molecular Identification and Phylogenetic Analysis
| Genus | Sampling Site Characteristics | Strain No. | Species | Morphological Characters | Cell Dimensions | Microscopic Image |
|---|---|---|---|---|---|---|
| Chlorella | Lake Feneketlen (LF) 47°28′36″ N; 19°2′29″ E; Temperature: 12.3 °C; pH: 8.6; Electrical Conductivity: 325 μS/cm | LF5 | Chlorella vulgaris | Cells unicellular, circular, pyrenoids present, cup shaped chloroplast present, mucilagenous sheath absent | 4.7 μm (length) × 4.2 μm (width) |  |
| Lacul Ciucas (LC) 46°08′44.7″ N; 25°51′25.0″ E; Temperature: 4.1 °C; pH: 8.1; Electrical Conductivity: 804 μS/cm | LC2 | Chlorella vulgaris | Unicellular, circular shaped, pyrenoids present, cup shaped chloroplast, mucilagenous sheath absent | 3.4 μm (length) × 3.0 μm (width) |  | |
| Lacul Ciucas (LC) see above | LC8 | Chlorella vulgaris | Unicellular cells, cup- to girdle shaped chloroplast seen in some cells, pyrenoids present | 4.0 μm (length) × 4.2 μm (width) |  | |
| Lacul Ciucas (LC) see above | LC9 | Chlorella vulgaris | Cells unicellular, circular, pyrenoids present, cup shaped chloroplast present, no mucilagenous sheath observed | 5.4 μm (length) × 4.9 μm (width) |  | |
| Lake Velence-Reed Pond (RP) 47°12′40.6″ N; 18°34′10.5″ E; Temperature: 15.0 °C; pH: 9.1; Electrical Conductivity: 1845 μS/cm | RP1 | Chlorella sorokiniana | Cells unicellular, circular, pyrenoids present, cup shaped chloroplast present | 5.6 μm (length) × 6.1 μm (width) |  | |
| Dictyosphaerium | Inner Lake, (IL) Tihany 46°54′33.9″ N; 17°53′09.7″ E; Temperature: 16.1 °C; pH: 8.4; Electrical Conductivity: 860 μS/cm | IL2 | Dictyosphaerium ehrenbergianum | Unicellular cells perpendicular to colony surface, attached to the ends of thin stalks emerging from centre of colony and branching tetrachotomously, spines are absent | 4.9 μm |  |
| Micractinium | Inner Lake (IL) Tihany see above | IL3 | Micractinium sp. | Solitary cells without mucilaginous sheath, planktonic, spherical, Chloroplast single parietal, cup shaped with an ellipsoidal pyrenoid | 6.5 μm (length) × 5.8 μm (width) |  |
| Lacul Ciucas (LC) see above | LC3 | Micractinium sp. | Cells are solitary without mucilaginous sheath, spherical, pyrenoid present within the chloroplast | 3.9 μm (length) × 4.0 μm (width) |  | |
| Lacul Ciucas (LC) see above | LC11 | Micractinium sp. | Cells are solitary without mucilaginous sheath, spherical, cup-shaped chloroplast with an ellipsoidal pyrenoid | 6.0 μm (length) × 6.2 μm (width) |  |
| Strain Code | Sequence Length (nt) | Closest Relative | Similarity (%) | Accession Number |
|---|---|---|---|---|
| LF5 | 925 | Chlorella vulgaris SAG 211-11b | 100 | KF569724 |
| LC2 | 926 | Chlorella vulgaris SAG 211-11b | 100 | KF569728 |
| LC8 | 907 | Chlorella vulgaris SAG 211-11b | 100 | KF569734 |
| LC9 | 924 | Chlorella vulgaris SAG 211-11b | 100 | KF569735 |
| RP1 | 919 | Chlorella sorokiniana CCAP 211/8K | 100 | KF569750 |
| IL2 | 926 | Dictyosphaerium ehrenbergianum CCAP 222/10 | 100 | KF569743 |
| IL3 | 924 | Micractinium sp. | 99.89 | KF569744 |
| LC3 | 925 | Micractinium pusillum CCAP 248/3 | 99.78 | KF569729 |
| LC11 | 879 | Micractinium pusillum CCAP 248/3 (and 248/1) | 99.66 | KF569742 |
2.3. Nile Red Staining Observations

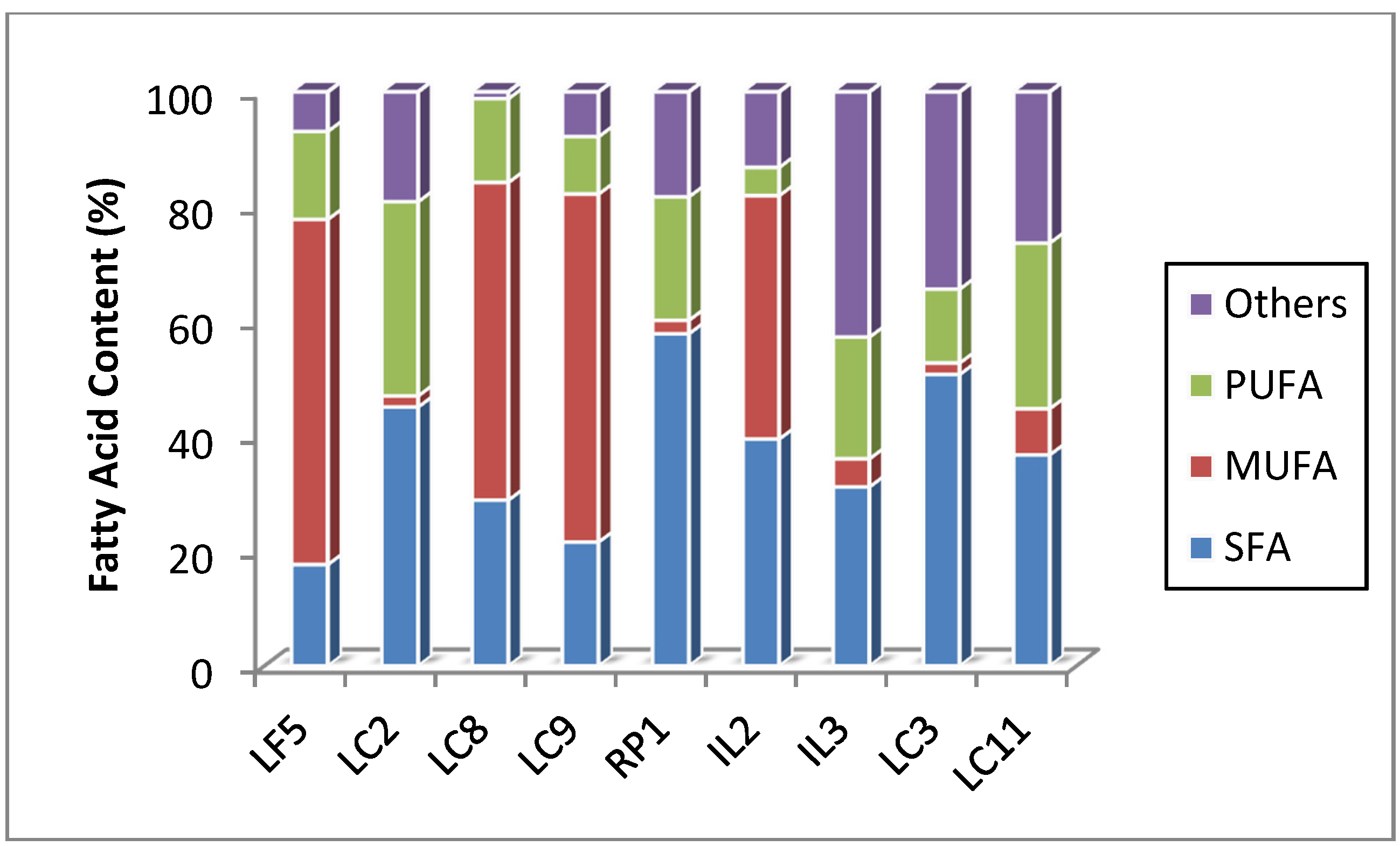
2.4. Biomass and Lipid Content of Screened Microalgae Strains
| Strain Code | Cell Dry wt (mg/L) | Protein (% dwt) | Carbohydrate (% dwt) | Lipid (% dwt) |
|---|---|---|---|---|
| LF5 | 443.8 ± 15.5 | 40.2 ± 8.2 | 19.3 ± 1.2 | 16.5 ± 0.1 |
| LC2 | 615.8 ± 10.5 | 42.6 ± 5.8 | 24.4 ± 0.9 | 8.9 ± 1.2 |
| LC8 | 396.0 ± 24.1 | 28.7 ± 1.3 | 20.1 ± 1.5 | 42.1 ± 2.6 |
| LC9 | 549.7 ± 17.2 | 22.3 ± 0.6 | 43.6 ± 2.3 | 14.2 ± 0.1 |
| RP1 | 571.5 ± 56.0 | 43.6 ± 3.5 | 10.0 ± 0.4 | 10.1 ± 1.0 |
| IL2 | 403.3 ± 49.7 | 17.3 ± 2.5 | 40.3 ± 6.1 | 34.1 ± 2.5 |
| IL3 | 414.3 ± 49.7 | 14.3 ± 3.5 | 41.5 ± 8.1 | 28.1 ± 2.5 |
| LC3 | 294.3 ± 13.4 | 27.5 ± 2.8 | 38.2 ± 2.3 | 23.6 ± 1.8 |
| LC11 | 428.1 ± 1.0 | 21.3 ± 1.5 | 13.9 ± 0.9 | 32.3 ± 6.7 |
2.5. Carbohydrate Composition
2.6. Protein Composition
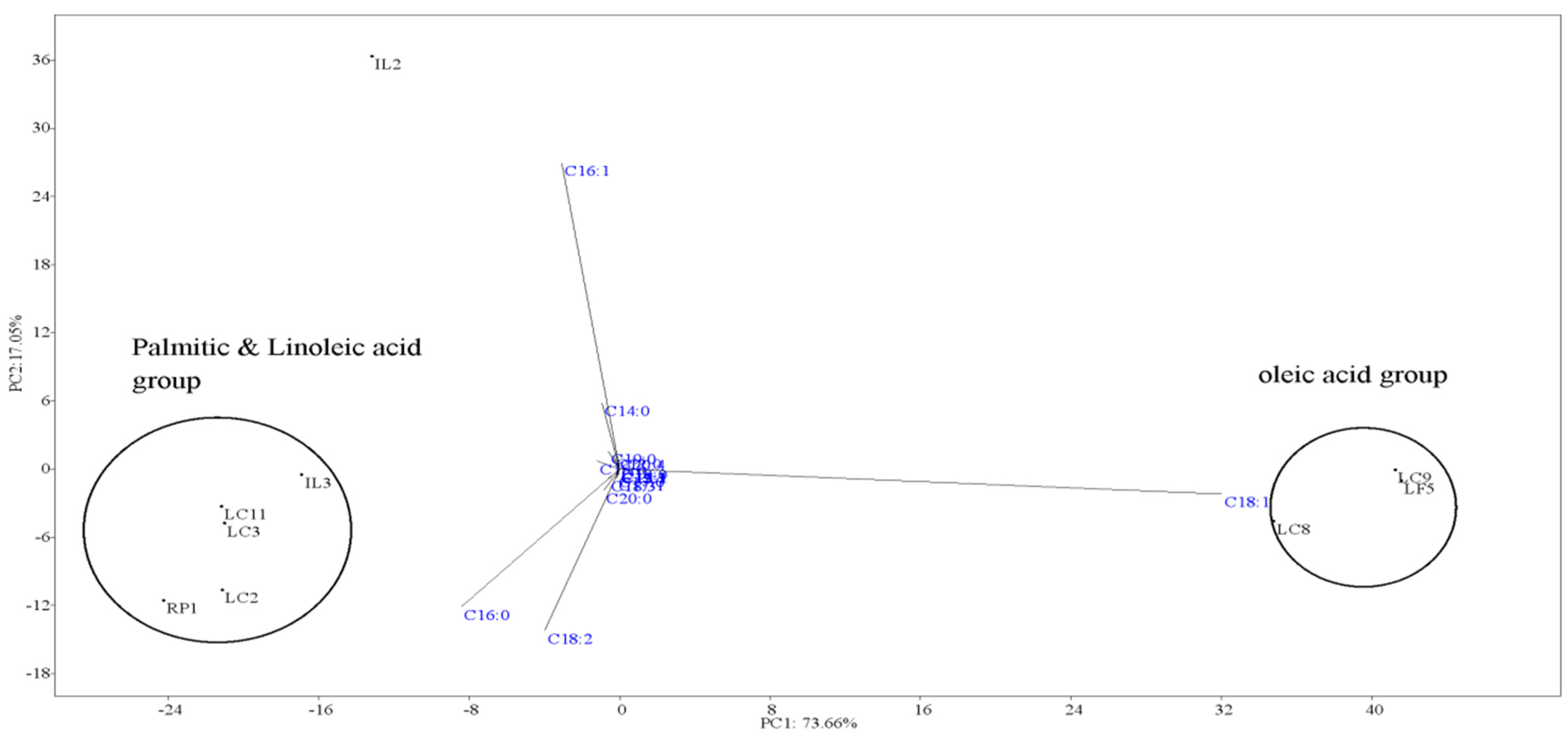
2.7. Fatty Acid Profiles
| Fatty Acids | LF5 | LC2 | LC8 | LC9 | RP1 | IL2 | IL3 | LC3 | LC11 |
|---|---|---|---|---|---|---|---|---|---|
| C9:0 | nd | nd | nd | nd | 0.03 | nd | nd | nd | 0.03 |
| C10:0 | 0.01 | nd | 0.02 | 0.03 | 0.04 | 0.33 | nd | 0.21 | 0.05 |
| C12:0 | 0.08 | nd | 0.07 | nd | 0.1 | 1.73 | 1.53 | 1.07 | nd |
| C14:0 | 0.15 | 0.61 | 0.15 | 0.16 | 0.82 | 8.46 | 1.34 | 1.6 | 0.6 |
| C16:0 | 16.2 | 31.85 | 27.71 | 19.73 | 52.07 | 22.63 | 21.28 | 42.65 | 34.1 |
| C17:0 | 0.22 | 0.43 | 0.2 | 0.28 | 0.77 | nd | 0.61 | 0.36 | 0.8 |
| C18:0 | 0.92 | 4.11 | 0.8 | 1.34 | 3.14 | 3.81 | 4.38 | 3.04 | 0.88 |
| C19:0 | 0.04 | 0.39 | 0.02 | 0.04 | 0.28 | 2.64 | 2.13 | 1.91 | 0.38 |
| C20:0 | 0.09 | 7.78 | nd | 0.06 | 0.67 | nd | nd | nd | nd |
| C12:1 | 0.05 | nd | nd | nd | 0.12 | nd | nd | 0.86 | 0.09 |
| C14:1 ω5c | nd | nd | nd | nd | nd | nd | nd | nd | 0.05 |
| C15:1 ω5c | nd | nd | nd | nd | nd | nd | nd | nd | 0.07 |
| C15:1 ω6c | 0.05 | nd | 0.04 | 0.05 | nd | nd | nd | nd | nd |
| C15:1 ω8c | 0.08 | nd | nd | nd | nd | nd | nd | nd | nd |
| C16:1 ω7c | 0.04 | 0.4 | 0.04 | nd | 0.09 | 36.66 | 0.83 | nd | 6.56 |
| C17:1 ω6c | nd | nd | nd | nd | nd | nd | 2.99 | nd | nd |
| C17:1 ω8c | 1.04 | 1.23 | 0.52 | 1.69 | 0.71 | nd | 1.08 | 1.13 | 1.26 |
| C18:1 ω5c | 9.99 | nd | 11.63 | nd | nd | nd | nd | nd | nd |
| C18:1 ω9c | 48.86 | nd | 43.04 | 58.8 | nd | 5.69 | nd | nd | nd |
| C19:1 ω11c | nd | 0.3 | nd | 0.05 | nd | nd | nd | nd | nd |
| C20:1 ω9c | nd | nd | nd | nd | 1.42 | 0 | nd | nd | nd |
| C18:2 ω6c | 15.31 | 33.79 | 14.58 | 9.99 | 18.98 | 3.47 | 21.17 | 11.5 | 26.94 |
| C18:3 ω6c | nd | nd | nd | nd | 2.51 | nd | nd | 1.36 | 1.79 |
| C20:4 ω6c | nd | nd | nd | nd | nd | 1.45 | nd | nd | 0.09 |
| C16-C18 | 81.29 | 69.75 | 86.13 | 89.86 | 76.7 | 35.6 | 46.83 | 58.55 | 63.71 |
2.8. Predicted Fuel Properties
3. Materials and Methods
3.1. Sampling
| Property | LF5 | LC2 | LC8 | LC9 | RP1 | IL2 | IL3 | LC3 | LC11 | Biodiesel | US (ASTM D6751-08) | Europe (EN 14214) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Kinematic viscosity 40 °C (mm2 s−1) | 4.63 | 4.77 | 4.67 | 4.69 | 4.90 | 4.91 | 5.04 | 5.07 | 4.95 | 4–5 | 1.9–6.0 | 3.5–5.0 |
| Specific gravity (kg−1) | 0.878 | 0.876 | 0.877 | 0.877 | 0.875 | 0.875 | 0.874 | 0.873 | 0.875 | 0.87–0.89 | 0.85–0.9 | - |
| Cloud point (°C) | 7.84 | 10.77 | 8.78 | 9.18 | 13.58 | 13.85 | 16.66 | 17.19 | 14.65 | - | - | - |
| Cetane number | 56.81 | 58.27 | 57.27 | 57.47 | 59.68 | 59.81 | 61.21 | 61.48 | 60.21 | 45–55 | Minimum 47 | Minimum 51–Maximum 120 |
| Iodine value (g Iodine/100 g) | 80.39 | 64.03 | 75.18 | 72.95 | 48.41 | 46.92 | 31.30 | 28.33 | 42.46 | - | - | Maximum 120 |
| HHV (MJ/kg) | 40.14 | 39.75 | 40.01 | 39.96 | 39.38 | 39.34 | 38.97 | 38.90 | 39.24 | - | - | - |
| Average Unsaturation | 0.91 | 0.69 | 0.84 | 0.81 | 0.48 | 0.46 | 0.25 | 0.20 | 0.40 | - | - | - |
| Long-chain saturated factor (LCFF) | 2.17 | 13.02 | 3.171 | 2.703 | 7.447 | 4.188 | 4.318 | 5.785 | 3.85 | - | - | - |
| Cold Filter Plugging Point (CFPP) | −9.66 | 2.43 | −6.51 | −7.98 | 6.91 | −3.31 | −2.91 | 1.69 | −4.38 | - | - | −5 to −13 |
3.2. Isolation and Cultivation of Microalgal Strains
3.3. Morphological Investigations
3.4. Molecular Characterization
3.5. Nile Red Staining of Neutral Lipid Droplets
3.6. Determination of Total Lipids
3.7. Fatty Acid Methyl Esters (FAME) Determination
3.8. Calorimetric Determination of Total Protein
3.9. Determination of Total Carbohydrates
3.10. Bioprospecting of Biodiesel Properties Based on FAME Profiles
3.11. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Chen, F.; Wei, D.; Zhang, X.; Chen, G. Biodiesel production by microalgal biotechnology. Appl. Energy 2010, 87, 38–46. [Google Scholar] [CrossRef]
- Chen, F. Microalgae and their biotechnological potential. J. Biotechnol. 2008, 136, S521. [Google Scholar] [CrossRef]
- Buijks, J. Future of Algae Based Biodiesel Production in The Netherlands. Master’s Thesis, University of Utrecht, Utrecht, The Netherlands, July 2013. [Google Scholar]
- Scott, S.A.; Davey, M.P.; Dennis, J.S.; Horst, I.; Howe, C.J.; Lea-Smith, D.J.; Smith, A.G. Biodiesel from algae: Challenges and prospects. Curr. Opin. Biotechnol. 2010, 21, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Knothe, G. Biodiesel and renewable diesel: A comparison. Prog. Energy Combust. Sci. 2010, 36, 364–373. [Google Scholar] [CrossRef]
- Lundquist, T.; Woertz, I.; Quinn, N.; Benemann, J. A Realistic Technology and Engineering Assessment of Algae Biofuel Production; University of California: Berkeley, CA, USA, 2010. [Google Scholar]
- Woertz, I.; Feffer, A.; Lundquist, T.; Nelson, Y. Algae grown on dairy and municipal wastewater for simultaneous nutrient removal and lipid production for biofuel feedstock. J. Environ. Eng. 2009, 135, 1115–1122. [Google Scholar] [CrossRef]
- Rawat, I.; Kumar, R.R.; Mutanda, T.; Bux, F. Dual role of microalgae: Phycoremediation of domestic wastewater and biomass production for sustainable biofuels production. Appl. Energy 2011, 88, 3411–3424. [Google Scholar] [CrossRef]
- Yun, Y.-S.; Lee, S.B.; Park, J.M.; Lee, C.-I.; Yang, J.-W. Carbon Dioxide Fixation by Algal Cultivation Using Wastewater Nutrients. J. Chem. Technol. Biotechnol. 1997, 69, 451–455. [Google Scholar] [CrossRef]
- Pittman, J.K.; Dean, A.P.; Osundeko, O. Bioresource technology the potential of sustainable algal biofuel production using wastewater resources. Bioresour. Technol. 2011, 102, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Park, D.H.; Ruy, H.W.; Lee, K.Y.; Kang, C.H.; Kim, T.H.; Lee, H.Y. The production of hydrocarbons from photoautotrophic growth of dunaliella salina 1650. Appl. Biochem. Biotechnol. 1998, 70, 739–746. [Google Scholar] [CrossRef] [PubMed]
- Spolaore, P.; Joannis-cassan, C.; Duran, E.; Isambert, A. Commercial Applications of Microalgae. J. Biosci. Bioeng. 2006, 101, 87–96. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mata, T.M.; Martins, A.A.; Caetano, N.S. Microalgae for biodiesel production and other applications: A review. Renew. Sustain. Energy Rev. 2010, 14, 217–232. [Google Scholar] [CrossRef]
- Abomohra, A.E.F.; Wagner, M.; El-Sheekh, M.; Hanelt, D. Lipid and total fatty acid productivity in photoautotrophic fresh water microalgae: Screening studies towards biodiesel production. J. Appl. Phycol. 2013, 25, 931–936. [Google Scholar] [CrossRef]
- Selvarajan, R.; Velu, P.; Sanniyasi, E. Molecular Characterization and Fatty Acid Profiling Of Indigenous Microalgae Species with Potential For Biofuel Production In Tamil Nadu India. Golden Res. Thoughts 2014, 3, 1–9. [Google Scholar]
- Abou-shanab, R.A.I.; Hwang, J.; Cho, Y.; Min, B.; Jeon, B. Characterization of microalgal species isolated from fresh water bodies as a potential source for biodiesel production. Appl. Energy 2011, 88, 3300–3306. [Google Scholar] [CrossRef]
- Sivakumar, G.; Jeong, K.; Lay, J.O. Bioprocessing of stichococcus bacillaris strain siva 2011. Biotechnol. Biofuels 2014, 7, 62. [Google Scholar] [CrossRef] [PubMed]
- Sivakumar, G.; Xu, J.; Thompson, R.W.; Yang, Y.; Randol-Smith, P.; Weathers, P.J. Integrated green algal technology for bioremediation and biofuel. Bioresour. Technol. 2012, 107, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Yun, H.S.; Lee, H.; Park, Y.T.; Ji, M.K.; Kabra, A.N.; Jeon, C.; Jeon, B.H.; Choi, J. Isolation of novel microalgae from acid mine drainage and its potential application for biodiesel production. Appl. Biochem. Biotechnol. 2014, 173, 2054–2064. [Google Scholar] [CrossRef] [PubMed]
- Talebi, A.F.; Mohtashami, S.K.; Tabatabaei, M.; Tohidfar, M.; Bagheri, A.; Zeinalabedini, M.; Hadavand Mirzaei, H.; Mirzajanzadeh, M.; Malekzadeh Shafaroudi, S.; Bakhtiari, S. Fatty acids profiling: A selective criterion for screening microalgae strains for biodiesel production. Algal Res. 2013, 2, 258–267. [Google Scholar] [CrossRef]
- Hoekman, S.K.; Broch, A.; Robbins, C.; Ceniceros, E.; Natarajan, M. Review of biodiesel composition, properties, and specifications. Renew. Sustain. Energy Rev. 2012, 16, 143–169. [Google Scholar] [CrossRef]
- Sydney, E.B.; da Silva, T.E.; Tokarski, A.; Novak, A.C.; de Carvalho, J.C.; Woiciecohwski, A.L.; Larroche, C.; Soccol, C.R. Screening of microalgae with potential for biodiesel production and nutrient removal from treated domestic sewage. Appl. Energy 2011, 88, 3291–3294. [Google Scholar] [CrossRef]
- Rawat, I.; Ranjith Kumar, R.; Mutanda, T.; Bux, F. Biodiesel from microalgae: A critical evaluation from laboratory to large scale production. Appl. Energy 2013, 103, 444–467. [Google Scholar] [CrossRef]
- Do Nascimento, M.; Ortiz-Marquez, J.C.F.; Sanchez-Rizza, L.; Echarte, M.M.; Curatti, L. Bioprospecting for fast growing and biomass characterization of oleaginous microalgae from South-Eastern Buenos Aires, Argentina. Bioresour. Technol. 2012, 125, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Abou-shanab, R.A.I.; Matter, I.A.; Kim, S.; Oh, Y.; Choi, J.; Jeon, B. Characterization and identification of lipid-producing microalgae species isolated from a freshwater lake. Biomass Bioenergy 2011, 35, 3079–3085. [Google Scholar] [CrossRef]
- Hu, Q.; Sommerfeld, M.; Jarvis, E.; Ghirardi, M.; Posewitz, M.; Seibert, M.; Darzins, A. Microalgal triacylglycerols as feedstocks for biofuel production: Perspectives and advances. Plant J. 2008, 54, 621–639. [Google Scholar] [CrossRef] [PubMed]
- Pereira, H.; Barreira, L.; Custódio, L.; Alrokayan, S.; Mouffouk, F.; Varela, J.; Abu-Salah, K.M.; Ben-Hamadou, R. Isolation and fatty acid profile of selected microalgae strains from the red sea for biofuel production. Energies 2013, 6, 2773–2783. [Google Scholar] [CrossRef]
- Pereira, H.; Barreira, L.; Mozes, A.; Florindo, C.; Polo, C.; Duarte, C.V.; Custódio, L.; Varela, J. Microplate-based high throughput screening procedure for the isolation of lipid-rich marine microalgae. Biotechnol. Biofuels 2011, 4, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, T.; Matsumoto, M.; Maeda, Y.; Sugiyama, H.; Sato, R.; Tanaka, T. Characterization of marine microalga, Scenedesmus sp. strain JPCC GA0024 toward biofuel production. Biotechnol. Lett. 2009, 31, 1367–1372. [Google Scholar] [CrossRef] [PubMed]
- Moazami, N.; Ranjbar, R.; Ashori, A.; Tangestani, M. Biomass and lipid productivities of marine microalgae isolated from the persian gulf and the qeshm island. Biomass Bioenergy 2011, 35, 1935–1939. [Google Scholar] [CrossRef]
- Tripathi, R.; Singh, J.; Thakur, I.S. Characterization of microalga Scenedesmus sp. ISTGA1 for potential CO2 sequestration and biodiesel production. Renew. Energy 2015, 74, 774–781. [Google Scholar] [CrossRef]
- Doan, T.T.Y.; Obbard, J.P. Improved nile red staining of Nannochloropsis sp. J. Appl. Phycol. 2011, 23, 895–901. [Google Scholar] [CrossRef]
- Brennan, L.; Owende, P. Biofuels from microalgae—A review of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sustain. Energy Rev. 2010, 14, 557–577. [Google Scholar] [CrossRef]
- Liu, J.; Huang, J.; Chen, F. Microalgae as Feedstocks for Biodiesel Production. In Biodiesel-Feedstocks and Processing Technologies; Stoytcheva, M.C., Ed.; INTECH: Rijeka, Croatia, 2011; pp. 58–78. [Google Scholar]
- Phukan, M.M.; Chutia, R.S.; Konwar, B.K.; Kataki, R. Microalgae Chlorella as a potential bio-energy feedstock. Appl. Energy 2011, 88, 3307–3312. [Google Scholar] [CrossRef]
- Song, M.; Pei, H.; Hu, W.; Ma, G. Evaluation of the potential of 10 microalgal strains for biodiesel production. Bioresour. Technol. 2013, 141, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Illman, A.M.; Scragg, A.H.; Shales, S.W. Increase in Chlorella strains calorific values when grown in low nitrogen medium. Enzyme Microb. Technol. 2000, 27, 631–635. [Google Scholar] [CrossRef]
- Feng, D.; Chen, Z.; Xue, S.; Zhang, W. Increased lipid production of the marine oleaginous microalgae Isochrysis zhangjiangensis (Chrysophyta) by nitrogen supplement. Bioresour. Technol. 2011, 102, 6710–6716. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Zhao, X.Q.; Yen, H.W.; Ho, S.H.; Cheng, C.L.; Lee, D.J.; Bai, F.W.; Chang, J.S. Microalgae-based carbohydrates for biofuel production. Biochem. Eng. J. 2013, 78, 1–10. [Google Scholar] [CrossRef]
- Trzcinski, A.P.; Hernandez, E.; Webb, C. A novel process for enhancing oil production in algae biorefineries through bioconversion of solid by-products. Bioresour. Technol. 2012, 116, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Guo, W.; Lo, Y.; Chang, J.; Ren, N. Characterization and kinetics of bio-butanol production with Clostridium acetobutylicum ATCC824 using mixed sugar medium simulating microalgae-based carbohydrates. Biochem. Eng. J. 2014, 91, 220–230. [Google Scholar] [CrossRef]
- Pulz, O.; Gross, W. Valuable products from biotechnology of microalgae. Appl. Microbiol. Biotechnol. 2004, 65, 635–648. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Wang, Z.; Yu, C.; Yin, Y.; Zhou, G. Evaluation of the potential of 9 Nannochloropsis strains for biodiesel production. Bioresour. Technol. 2014, 167, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Knothe, G. Analyzing biodiesel: Standards and other methods. J. Am. Oil Chem. Soc. 2006, 83, 823–833. [Google Scholar] [CrossRef]
- Knothe, G. “Designer” biodiesel: Optimizing fatty ester composition to improve fuel properties. Energy Fuels 2008, 22, 1358–1364. [Google Scholar] [CrossRef]
- Stournas, S.; Lois, E.; Serdari, A. Effects of fatty acid derivatives on the ignition quality and cold flow of diesel fuel. J. Am. Oil Chem. Soc. 1995, 72, 433–437. [Google Scholar] [CrossRef]
- Mutanda, T.; Ramesh, D.; Karthikeyan, S.; Kumari, S.; Anandraj, A.; Bux, F. Bioprospecting for hyper-lipid producing microalgal strains for sustainable biofuel production. Bioresour. Technol. 2011, 102, 57–70. [Google Scholar] [CrossRef] [PubMed]
- Georgianna, D.R.; Mayfield, S.P. Exploiting diversity and synthetic biology for the production of algal biofuels. Nature 2012, 488, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Mandotra, S.K.; Kumar, P.; Suseela, M.R.; Ramteke, P.W. Fresh water green microalga Scenedesmus abundans: A potential feedstock for high quality biodiesel production. Bioresour. Technol. 2014, 156, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Francisco, É.C.; Neves, D.B.; Jacob-Lopes, E.; Franco, T.T. Microalgae as feedstock for biodiesel production: Carbon dioxide sequestration, lipid production and biofuel quality. J. Chem. Technol. Biotechnol. 2010, 85, 395–403. [Google Scholar]
- Stanier, R.Y.; Kunisawa, R.; Mandel, M.; Cohen-Bazire, G. Purification and properties of unicellular blue-green algae (order Chroococcales). Bacteriol. Rev. 1971, 35, 171–205. [Google Scholar] [PubMed]
- Rippka, R.; Deruelles, J.; Waterbury, J.B.; Herdman, M.; Stanier, R.Y. Generic assignments, strain histories and properties of pure cultures of cyanobacteria. J. Gen. Microbiol. 1979, 111, 1–61. [Google Scholar] [CrossRef]
- Whitton, P.B.A.; John, D.M.; Kelly, M.G.; Haworth, E.Y.; Tindall, C.I. The Freshwater Algal Flora of the British Isles an Identification Guide to Freshwater and Terrestrial Algae; Cambridge University Press: Cambridge, UK, 2003. [Google Scholar]
- Zhu, F.; Massana, R.; Not, F.; Marie, D.; Vaulot, D. Mapping of picoeucaryotes in marine ecosystems with quantitative PCR of the 18S rRNA gene. FEMS Microbiol. Ecol. 2005, 52, 79–92. [Google Scholar] [CrossRef] [PubMed]
- Moon-van der Staay, S.Y.; van der Staay, G.W.M.; Guillou, L.; Vaulot, D.; Claustre, H.; Medlin, L.K. Abundance and diversity of prymnesiophytes in the picoplankton community from the equatorial Pacific Ocean inferred from 18S rDNA sequences. Limnol. Oceanogr. 2000, 45, 98–109. [Google Scholar] [CrossRef]
- Somogyi, B.; Felföldi, T.; Solymosi, K.; Flieger, K.; Márialigeti, K.; Böddi, B.; Vörös, L. One step closer to eliminating the nomenclatural problems of minute coccoid green algae: Pseudochloris wilhelmii, gen. et sp. nov. (Trebouxiophyceae, Chlorophyta). Eur. J. Phycol. 2013, 48, 427–436. [Google Scholar] [CrossRef] [Green Version]
- Altschul, S.F.; Madden, T.L.; Schäffer, A.A.; Zhang, J.; Zhang, Z.; Miller, W.; Lipman, D.J. Gapped BLAST and PSI-BLAST: A new generation of protein database search programs. Nucleic Acids Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011, 28, 2731–2739. [Google Scholar] [CrossRef] [PubMed]
- Pruesse, E.; Peplies, J.; Glöckner, F.O. SINA: Accurate high-throughput multiple sequence alignment of ribosomal RNA genes. Bioinformatics 2012, 28, 1823–1829. [Google Scholar] [CrossRef] [PubMed]
- Elsey, D.; Jameson, D.; Raleigh, B.; Cooney, M.J. Fluorescent measurement of microalgal neutral lipids. J. Microbiol. Methods 2007, 68, 639–642. [Google Scholar] [CrossRef] [PubMed]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef] [PubMed]
- Freedman, B.; Butterfield, R.O.; Pryde, E.H. Transesterification kinetics of soybean oil. J. Am. Oil Chem. Soc. 1986, 63, 1375–1380. [Google Scholar] [CrossRef]
- Sahu, A.; Pancha, I.; Jain, D.; Paliwal, C.; Ghosh, T.; Patidar, S.; Bhattacharya, S.; Mishra, S. Fatty acids as biomarkers of microalgae. Phytochemistry 2013, 89, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [PubMed]
- DuBois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Nascimento, I.A.; Marques, S.S.I.; Cabanelas, I.T.D.; Pereira, S.A.; Druzian, J.I.; de Souza, C.O.; Vich, D.V.; de Carvalho, G.C.; Nascimento, M.A. Screening microalgae strains for biodiesel production: Lipid productivity and estimation of fuel quality based on fatty acids profiles as aelective criteria. Bioenergy Res. 2013, 6, 1–13. [Google Scholar] [CrossRef]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 9–18. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Selvarajan, R.; Felföldi, T.; Tauber, T.; Sanniyasi, E.; Sibanda, T.; Tekere, M. Screening and Evaluation of Some Green Algal Strains (Chlorophyceae) Isolated from Freshwater and Soda Lakes for Biofuel Production. Energies 2015, 8, 7502-7521. https://doi.org/10.3390/en8077502
Selvarajan R, Felföldi T, Tauber T, Sanniyasi E, Sibanda T, Tekere M. Screening and Evaluation of Some Green Algal Strains (Chlorophyceae) Isolated from Freshwater and Soda Lakes for Biofuel Production. Energies. 2015; 8(7):7502-7521. https://doi.org/10.3390/en8077502
Chicago/Turabian StyleSelvarajan, Ramganesh, Tamás Felföldi, Tamás Tauber, Elumalai Sanniyasi, Timothy Sibanda, and Memory Tekere. 2015. "Screening and Evaluation of Some Green Algal Strains (Chlorophyceae) Isolated from Freshwater and Soda Lakes for Biofuel Production" Energies 8, no. 7: 7502-7521. https://doi.org/10.3390/en8077502







