Multi-Stage Control of Waste Heat Recovery from High Temperature Slags Based on Time Temperature Transformation Curves
Abstract
: This paper presents a significant method and a basic idea of waste heat recovery from high temperature slags based on Time Temperature Transformation (TTT) curves. Three samples with a fixed CaO/SiO2 ratio of 1.05 and different levels of Al2O3 were designed and isothermal experiments were performed using a Single Hot Thermocouple Technique (SHTT). The TTT curves established through SHTT experiments described well the variation of slag properties during isothermal processes. In this study, we propose a multi-stage control method for waste heat recovery from high temperature slags, in which the whole temperature range from 1500 °C to 25 °C was divided into three regions, i.e., Liquid region, Crystallization region and Solid region, based on the TTT curves. Accordingly, we put forward an industrial prototype plant for the purpose of waste heat recovery and the potential of waste heat recovery was then calculated. The multi-stage control method provided not only a significant prototype, but also a basic idea to simultaneously extract high quality waste heat and obtain glassy phases on high temperature slags, which may fill the gap between slag properties and practical waste heat recovery processes.1. Introduction
It is well known that the steel industry is energy intensive, consuming around 9% of anthropogenic energy [1] and emitting large quantities of CO2 into the atmosphere [2]. With the acceleration of global warming nowadays, energy saving and CO2 emission reduction in the steel industry is attracting more and more attention, although the energy efficiency has already been substantially improved by implementing extensive advanced technologies. According to the previous estimations [3,4], high temperature (1450–1550 °C) slags, carrying a substantial amount of high quality heat, represent the last potential source for energy reduction in the steel industry. In China, the steel industry's output of crude steel was more than 710 million tons in 2012 [5], and accordingly around 200 million tons of high temperature blast furnace slags (BF slags) and 70 million tons of steel slags were produced [6] and the total waste heat was more than 4.80 × 1019 J, equivalent to 16 million tons of standard coal, whereas less than 2% of that was recovered, according to the estimation of Cai et al. [7], so there is a great potential of waste heat recovery. BF slags alone accounted for more than 70% of the waste heat of the slags in the steel industry, and therefore the previous studies were mainly focused on BF slags [8].
Conventionally, BF slags can be treated through two methods: gradually cooled by air in a slag pit [9] or rapidly quenched by water [10]. Air-gradually-cooled slags have low utilization values because of their weak hydraulicity due to the high content of crystalline phases [11], while water quenched BF slags are increasingly utilized as cementitious materials because of the glassy phases [11] and the similarity between the components of slags and Portland cement, high content of calcium silicates [12]. However, the water quenched method faces a series of problems, such as water consumption and pollution, SO2 and H2S emissions and energy consumption and waste [13,14]. To solve the aforementioned problems and realize the recovery of waste heat, a dry granulation method has been proposed, through which the liquid slags are granulated into small droplets with the diameter of several millimeters using different waterless granulation technologies, such as rotary cup atomizer [15–18], rotating drum [19,20], air blasts [21,22] and so on. Recently the combination of dry granulation with other waste heat utilization methods, such as hydrogen production from biogas [18,23], coal gasification [24,25] and heat storage of phase change materials (PCM) [26] has been intensively studied, and is expected to be a promising method in the future. Whatever the utilization method applied, the understanding of the variation of slag properties during cooling processes and the control of heat transfer are fundamental for simultaneously realizing waste heat recovery and slag recycling. Considering the large amount of BF slags, the present study was focused on BF slags. Meanwhile, iron ore has been degraded and Al2O3 content in gangues has been increasing in the past decades [27], which has led to variations in the chemical composition of BF slags, such as an increase of Al2O3 content. With the variation of chemical compositions of slags, the slag properties, including crystallization properties could be changed, which could influence the waste heat recovery of high temperature slags. Three samples containing different levels of Al2O3 were therefore designed in this study.
The investigation was carried out using a Single Hot Thermocouple Technique (SHTT) for visualizing phase changes in the slag melts, through which Time Temperature Transformation (TTT) curves were established. TTT curves described well the variation of slag properties, based on which a multi-stage control method was proposed in this paper. The potential of waste heat recovery at different stages was accordingly calculated.
2. Materials and Methods
2.1. Sample Preparation
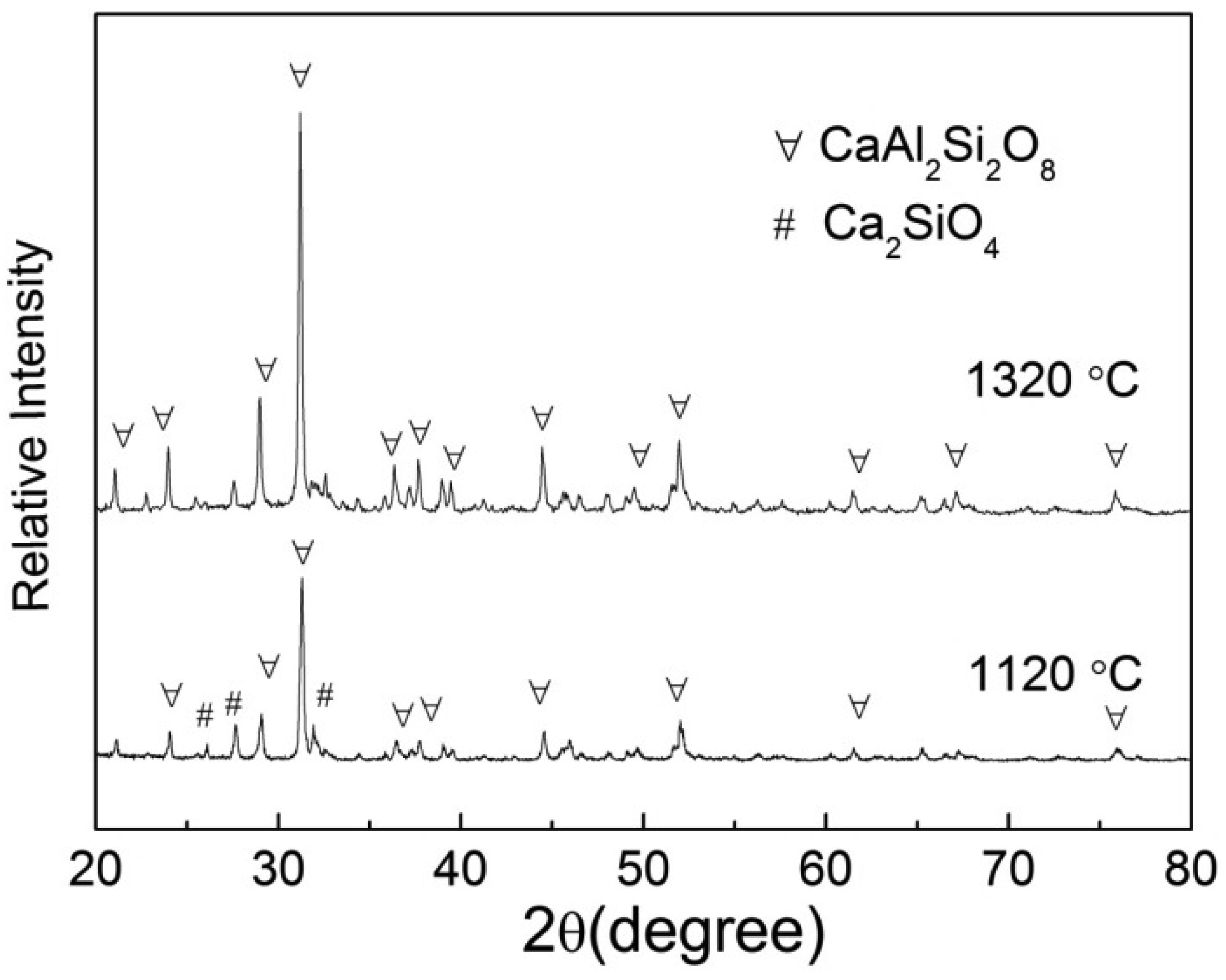
In the present study, three slag samples with a CaO/SiO2 ratio of 1.05 and Al2O3 contents of 10–25 wt% were prepared using analytically regent (AR) pure oxides. These oxides were mixed and pre-melted in a molybdenum (Mo) crucible (Φ40 mm × 45 mm × H40 mm) under an argon atmosphere at 1500 °C for 2 h to homogenize the chemical compositions. Then the liquid slags were rapidly poured into the cold water to obtain glassy phases. Subsequently, the solid slags were dried at 120 °C for 12 h, crushed and ground to 300 mesh size for compositional analysis and SHTT experiments. The chemical compositions were measured by the X-Ray fluoroscopy (XRF) technique and the results are listed in Table 1. To confirm the glassy phases of slags and the accuracy of SHTT experiments, X-ray diffraction (XRD) tests were carried out and the results are shown in Figure 1.
2.2. Apparatus and Procedure
Experiments in this study were carried out using SHTT for visualizing the phase transformations and measuring the incubation time of these slags. SHTT combines the advantages of in-situ optical observation and the low inertia of the system, with a maximum cooling rate of 200 °C/s. The work mechanism of SHTT has been described in detail elsewhere [28,29] and is only briefly outlined here. As schematically shown in Figure 2, a Pt-Rh thermocouple was used to heat the sample and simultaneously measure the temperature, which was controlled by a computer program. A microscope equipped with a video camera was used to observe and capture images of the slag melts. The isothermal experiments by SHTT were performed following several steps as below. Firstly, the temperature of thermocouple was calibrated using pure K2SO4 with a constant melting point 1067 °C. Secondly, about 10 mg sample was mounted on the top of the thermocouple, heated to 1500 °C, and held for 120 s to eliminate the bubbles and homogenize the chemical composition. Thirdly, the liquid slags were rapidly quenched at a cooling rate of 50 °C/s to a given temperature and held for a long time at this temperature. Then the evolution of crystallization with time in the slag melts was observed and the sample images were captured by the video camera, shown in Figure 3 as an example. The crystalline phases precipitated in the melts were identified by the XRD technique.
After the liquid slag was rapidly quenched to a given temperature, it may take some time to crystallize, i.e., there is a certain incubation time. To reduce measurement errors, the incubation time at each temperature point was measured at least three times and the average value was used. The obtained incubation time can help to design a waste heat recovery process in a reasonable time to avoid crystal formation. The variation of incubation time with isothermal experiments was presented in TTT curves. The crystalline phases precipitated in slags were confirmed by Factsage software calculation and the latent heat of phase transformation was calculated [30].
3. Results and Discussion
3.1. Multi-Stage Control of Waste Heat Recovery Based on TTT Curves
3.1.1. TTT Curves
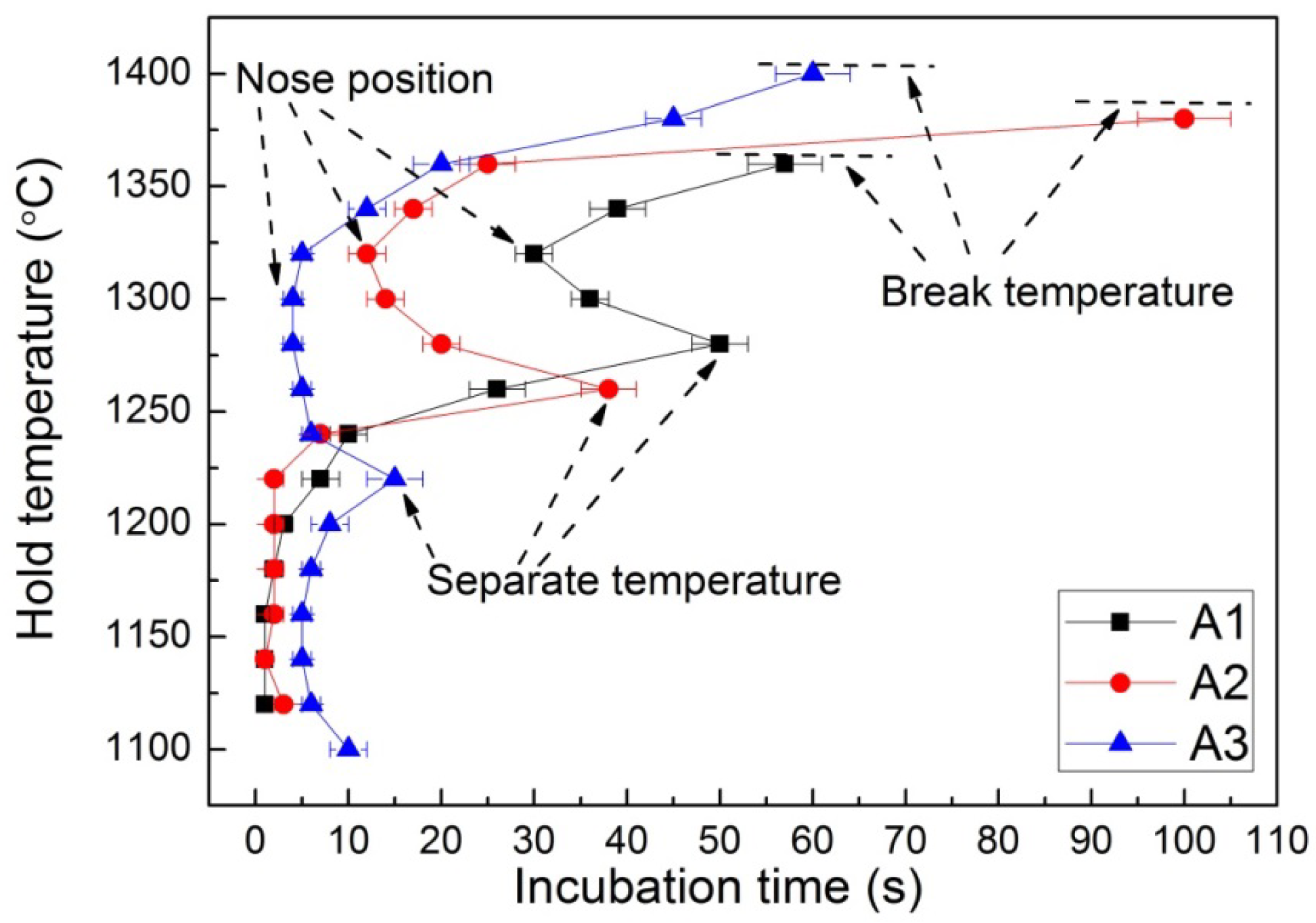
The TTT curves of different samples were presented in Figure 4. As can be seen, these TTT curves showed a similar shape, that is a double “C” from high temperature to low temperature, which suggested that two different crystallization events occurred. As the slag melt was quenched from 1500 °C, the crystallization was not observed when it was higher than the break temperature (the highest temperature at which crystallization could appear), indicating that the waste heat recovery from the slags can last a long time during this temperature interval. It can be seen that the break temperature increased from sample A1 to A3, which suggested that the crystallization was enhanced by Al2O3 addition.
As the temperature decreased lower than the break temperature, crystals may precipitate after an incubation time. The incubation time decreased with the decrease of isothermal temperature until the nose position, then it increased with further decreasing temperature to the separate temperature between these two “C” shapes. With the decrease of temperature, the undercooling degree was increased, which was beneficial for crystal formation, whereas the viscosity increased at the same time, which suppressed the crystal formation. These two factors caused by temperature decrease influenced the crystal precipitation in opposite directions and caused the appearance of a shortest incubation time, i.e., at the nose position. The shortest incubation time at the nose position suggested that the waste heat recovery time with medium must be very quick at this temperature in order to avoid crystallization.
With further decreasing temperature, a similar variation tendency as with the first “C” shape was observed in the crystallization temperature range. Finally the temperature was decreased to a low temperature zone where no crystallization was observed due to the high resistance of mass transfer caused by the higher viscosity. These results indicated that the waste heat recovery time from slags could be set up with temperature, and the cooling process was therefore determined based on the TTT curves. An apparent trend was observed in that the TTT curves moved to the left side in high temperature zones with increasing Al2O3 content, which indicated that the crystallization was enhanced by Al2O3. This suggested that an increasing Al2O3 content resulted in a lower waste heat recovery time in this temperature interval. It is also noted that the second “C” shape in low temperature zones did not show an apparent variation tendency for different samples, which might be because that the effect of undercooling on the crystallization was greater than that of the Al2O3 content [31].
3.1.2. Multi-Stage Control Method
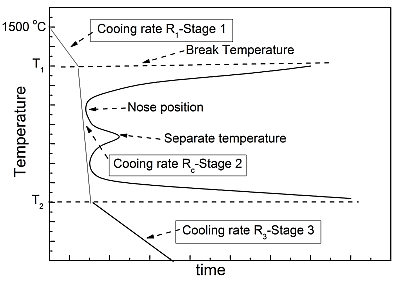
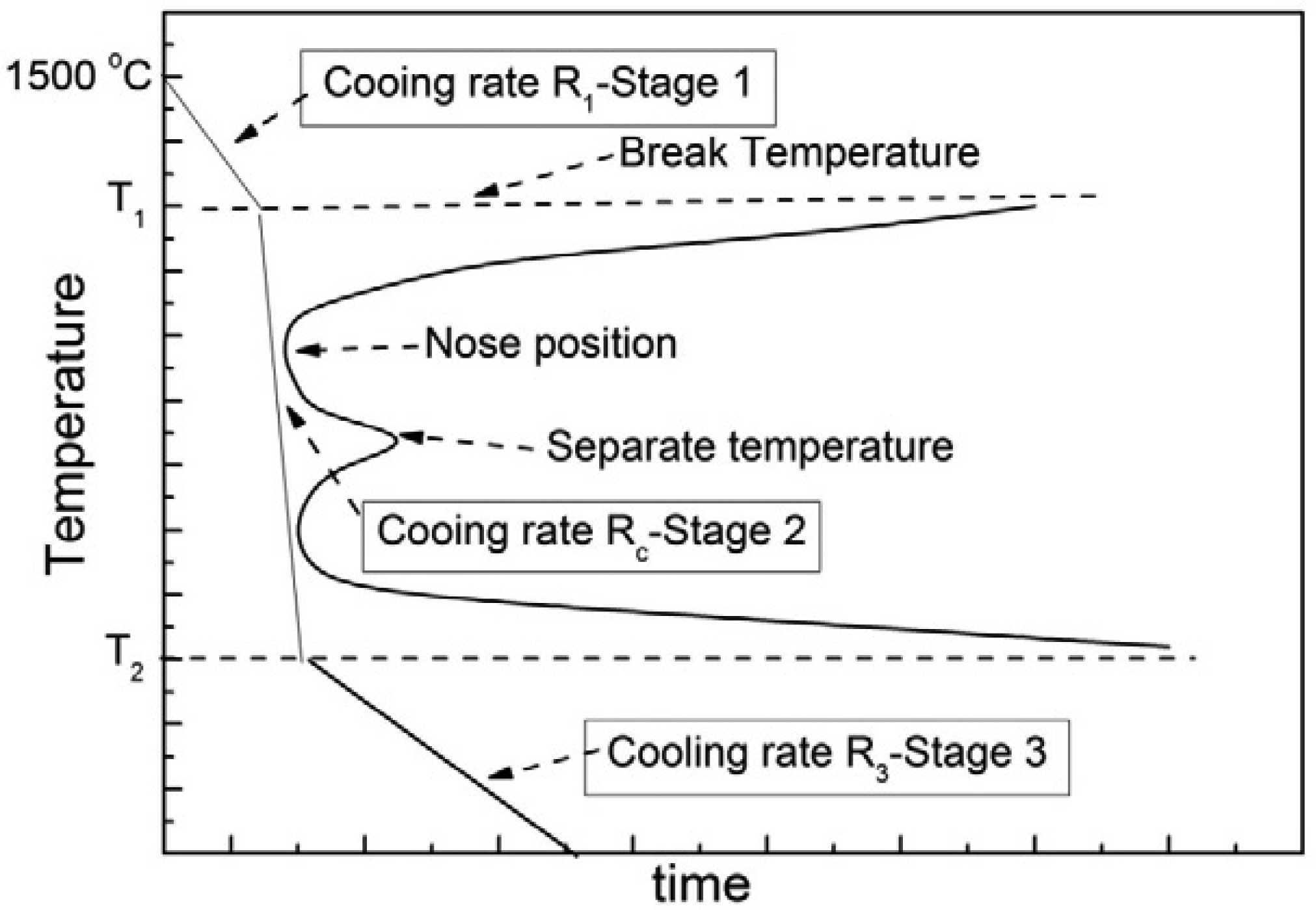
Based on the aforementioned findings, a multi-stage control method of waste heat recovery from high temperature slags was proposed here, a general schematic diagram of which is shown in Figure 5. During stage 1, as the temperature was higher than the break temperature, no crystallization was observed and the heat exchange could continue for a long time to fully extract the waste heat. Slags in this temperature zone could be slowly cooled and the high quality waste heat could be fully exchanged to PCM [26] and stored for further utilization. This stage was located in the temperature range where slags were liquid, defined as Liquid region. During stage 2, with the temperature decreasing to the first “C” and the second “C” shapes, the heat exchange time should be adjusted according to the incubation time. The cooling rate during this stage must be larger than the critical cooling rate, which was a cooling rate larger than which crystallization does not appear. The critical cooling rate can be calculated by the following expression [32]:
In this section, the whole temperature range during waste heat recovery process was divided into three regions, i.e., Liquid region, Crystallization region and Solid region, and the technological parameters of heat exchange can be therefore designed according to the TTT curves, as shown in Figure 5.
3.2. Fitting of TTT Curves
To realize a continuous control of waste heat recovery from high temperature slags, it is necessary to obtain a fitting of the TTT curves for a practical waste heat recovery process. The polynomial expressions could be used to fit the TTT curves, from which the incubation time at a given temperature could be deduced [33]. The following equation gave acceptable calculation results that fitted the measured incubation time for samples in this study:
For sample A1:
For sample A2:
For sample A3:
3.3. Phase Change of Slags
Conventionally, solidified slags containing less than 5% crystalline phase are applicable as raw materials for the cement manufacturing industry. The primary crystalline phase in these slag melts was examined by XRD tests and the results are shown in Figure 6. It can be seen that the primary phase was CaAl2Si2O8, and Ca2SiO4 was precipitated upon further decreasing the temperature, which agreed with the Factsage phase diagrams. The latent heat of phase change (CaAl2Si2O8) was around 421.81 KJ/mol referred to Factsage software data [30]. The small amount of crystals (5%) formed in the melts could result in the increase of waste heat recovery due to the heat release from phase transformation, but it was difficult to control the crystallization degree in practical operation processes. It is therefore proposed that the liquid slags should be rapidly cooled to avoid crystallization according to the TTT curves.
3.4. Industrial Prototype Plant
Based on the aforementioned analysis, an industrial prototype plant was proposed for the purpose of recovering the high quality waste heat from high temperature slags, and the process flow is shown in Figure 7. The whole process flow could be divided into several parts. Firstly, SHTT experiments were carried out for a slag sample and the TTT curves were obtained. Secondly, the waste heat of the high temperature slags produced in steel industry was extracted with the dry granulation based on a multi-stage control method and the integrated heat was used by coal gasification, hydrogen production or heat storage in PCM. Then the solidified glassy slags could be utilized as cementitious material and finally both the waste heat and slag resources were recovered.
The possible recovered energy of this prototype plant based on multi-stage control method was calculated and listed in Table 2, in which average heat capacity of slags was referred to Factsage software data [30], the crystal content was assumed as 1% and the slags were assumed to be cooled to ambient temperature (25 °C). As can be seen, the calculation was divided into three stages based on the multi-stage control method. Accordingly the extraction of the high quality energy of slags could be divided into three stages, during which several operational parameters were controlled including heat exchange time and cooling rates; and finally different quantities of waste heat was recovered. For example, the possible extracted waste heat for sample A1 was 160.94 MJ, 198.91 MJ and 1235.86 MJ per ton of slags in the temperature range of 1500–1360 °C (Stage 1), 1360–1100 °C (Stage 2) and 1100–25 °C (Stage 3), respectively, and the total extracted waste heat could be up to 1695.71 MJ per ton of slags without phase change. Accordingly, the cooling rate and heat exchange time during stage 1 and 3 could be adjusted and that during stage 2 should be controlled larger than 6 °C/s and less than 43.3 s. With the heat release of 151.73 MJ per ton of slags from 1% crystal precipitation, the potential of total extracted waste heat could be more than 1847 MJ per ton of slags. In a similar way, the potential extracted waste heat during different stages for samples A2 and A3 could be calculated and the total extracted waste heat for samples A2 and A3 without phase transformation was 1793.82 MJ and 1828.82 MJ per ton of slags, respectively, as listed in Table 2.
4. Conclusions
The present study provided an analysis of high temperature slags for the purpose of recovering both the high quality waste heat and the slag resources. Three samples were designed containing different levels of Al2O3 at a fixed CaO/SiO2 ratio of 1.05. A Single Hot Thermocouple Technique (SHTT) was used to investigate the slag properties during isothermal experiments and Time Temperature Transformation (TTT) curves were therefore obtained. The main conclusions were summarized as below:
A multi-stage control method of waste heat recovery from high temperature slags was proposed based on TTT curves, in which the whole temperature range was divided into three regions, i.e., Liquid region, Crystallization region and Solid region. Some significant technological parameters, including heat exchange time and cooling rate should be effectively controlled during these stages;
The polynomial expressions fitted the TTT curves well and could be used to deduce the incubation time at any temperature;
An industrial prototype plant was put forward aiming to recover the high quality waste heat of high temperature slags, and accordingly the potential of the extracted waste heat was calculated.
Acknowledgments
The authors gratefully acknowledge financial support by the Common Development Fund of Beijing and the National Natural Science Foundation of China (51074009, and 51172001). Supports by the National High Technology Research and Development Program of China (863 Program, 2012AA06A114) and Key Projects in the National Science & Technology Pillar Program (2011BAB03B02) are also acknowledged.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Milford, R.L.; Pauliuk, S.; Allwood, J.M.; Muller, D.B. The roles of energy and material efficiency in meeting steel industry CO2 targets. Environ. Sci. Technol. 2013, 47, 3455–3462. [Google Scholar]
- Allwood, J.M.; Cullen, J.M.; Milford, R.L. Options for achieving a 50% cut in industrial carbon emissions by 2050. Environ. Sci. Technol. 2010, 44, 1888–1894. [Google Scholar]
- Bisio, G. Energy recovery from molten slag and exploitation of the recovered energy. Energy 1997, 22, 501–509. [Google Scholar]
- Barati, M.; Esfahani, S.; Utigard, T.A. Energy recovery from high temperature slags. Energy 2011, 36, 5440–5449. [Google Scholar]
- November 2012 Crude Steel Production. Available online: http://www.worldsteel.org/media-centre/press-releases/2012/11-2012-crude-steel.html (accessed on 7 January 2014).
- Zhang, H.; Wang, H.; Zhu, X.; Qiu, Y.J.; Li, K.; Chen, R.; Liao, Q. A review of waste heat recovery technologies towards molten slag in steel industry. Appl. Energy 2013, 112, 956–966. [Google Scholar]
- Cai, J.J.; Wang, J.J.; Chen, C.X.; Lu, Z.W. Recovery of residual heat integrated steelworks. Iron Steel 2007, 42, 1–6. [Google Scholar]
- China Iron and Steel Association. Available online: http://www.chinaisa.org.cn (accessed on 7 January 2014).
- Pandelaers, L.; D'alfonso, A.; Jones, P.T.; Blanpain, B. A quantitative model for slag yard cooling. ISIJ Int. 2013, 53, 1106–1111. [Google Scholar]
- Li, Q.H.; Meng, A.H.; Zhang, Y.G. Recovery Status and Prospect of Low-Grade Waste Energy in China. Proceedings of the International Conference on Sustainable Power Generation and Supply, Nanjing, China, 6–7 April 2009; pp. 1–6.
- Van Oss, H.G.; Slag-iron and steel. US Geological Survey Minerals Yearbook; U.S. Department of the Interior and U.S. Geological Survey: Reston, VA, USA, 2010; pp. 69.1–69.10. Available online: http://minerals.usgs.gov/minerals/pubs/commodity/iron_&_steel_slag/index.html#myb (accessed on 7 January 2014).
- Nakada, T.; Nakayama, H.; Fujii, K.; Iwahashi, T. Heat recovery in dry granulation of molten blast furnace slag. Energy Dev. Jpn. 1983, 55, 287–309. [Google Scholar]
- Guo, H.; Zhou, S.H. Discussion about Heat Recovery Technology of Blast Furnace Slag. Proceedings of the Ironmaking Technology Conference and Ironmaking Academic Annual Meeting, Beijing, China, 26–28 May 2010; pp. 1141–1175.
- Mizuochi, T.; Akiyama, T.; Shimada, T.; Kasai, E.; Yagi, J.I. Feasibility of rotary cup atomizer for slag granulation. ISIJ Int. 2001, 41, 1423–1428. [Google Scholar]
- Pickering, S.J.; Hay, N.; Roylance, T.F.; Thomas, G.H. New process for dry granulation and heat recovery from molten blast furnace slag. Ironmak. Steelmak. 1985, 12, 14–21. [Google Scholar]
- Akiyama, T.; Oikawa, K.; Shimada, T.; Kasai, E.; Yagi, J.I. Thermodynamic analysis of thermochemical recovery of high temperature wastes. ISIJ Int. 2000, 40, 286–291. [Google Scholar]
- Shimada, T.; Kochura, V.; Akiyama, T.; Kasai, E.; Yagi, J.I. Effects of slag compositions on the rate of methane-steam reaction. ISIJ Int. 2001, 41, 111–115. [Google Scholar]
- Kasai, E.; Kitajima, T.; Akiyama, T.; Yagi, J.I.; Saito, F. Rate of methane-steam reforming reaction on the surface of molten BF Slag: For heat recovery from molten slag by using a chemical reaction. ISIJ Int. 1997, 37, 1031–1036. [Google Scholar]
- Yoshinaga, M.; Fujii, K.; Shigematsu, T.; Nakata, T. Method of dry granulation and solidification of molten blast furnace slag. Tetsu-To-Hagane 1981, 67, 917–924. [Google Scholar]
- Sieverding, F. Heat recovery by dry granulation of blast furnace slags. Steel Times 1980, 208, 469–472. [Google Scholar]
- Yoshida, H.; Nara, Y.; Nakatani, G.; Anazi, T.; Sato, H. The Technology of Slag Heat Recovery. Proceedings of the NKK SEAISI (Nippon Kokan KK-Southeast Asia Iron and Steel Institute) Conference of Energy Utilization in the Iron and Steel Industry, Singapore, 10–13 September 1984; pp. 1–20.
- Xie, D.; Jahanshahi, S. Waste Heat Recovery from Molten Slags. Proceedings of the 4th International Congress on Science and Technology of Steelmaking, Gifu, Japan, 6–8 October 2008; pp. 674–677.
- Purwanto, H.; Akiyama, T. Hydrogen production from biogas using hot slag. Int. J. Hydrog. Energy 2006, 31, 491–495. [Google Scholar]
- Li, P.; Yu, Q.B.; Qin, Q.; Lei, W. Kinetics of CO2/Coal gasification in molten blast furnace slag. Ind. Eng. Chem. Res. 2012, 51, 15872–15883. [Google Scholar]
- Li, P.; Yu, Q.B.; Xie, H.Q.; Qin, Q.; Wang, K. CO2 gasification rate analysis of Datong coal using slag granules as heat carrier for heat recovery from blast furnace slag by using a chemical reaction. Energy Fuel 2013, 27, 4810–4817. [Google Scholar]
- Nomura, T.; Okinaka, N.; Akiyama, T. Technology of latent heat storage for high temperature application. ISIJ Int. 2010, 50, 1229–1239. [Google Scholar]
- Yajima, K.; Matsuura, H.; Tsukihashi, F. Effect of simultaneous addition of Al2O3 and MgO on the liquidus of the CaO–SiO2–FeOx system with various oxygen partial pressures at 1573 K. ISIJ Int. 2010, 50, 191–194. [Google Scholar]
- Kashiwaya, Y.; Cicutti, C.E.; Cramb, A.W.; Ishii, K. Development of double and dingle hot thermocouple technique for in site observation and measurement of mold slag crystallization. ISIJ Int. 1998, 38, 357–365. [Google Scholar]
- Kashiwaya, Y.; Cicutti, C.E.; Cramb, A.W.; Ishii, K. An investigation of the crystallization of a continuous casting mold slag using the single hot thermocouple technique. ISIJ Int. 1998, 38, 348–356. [Google Scholar]
- Bale, C.W.; Chartrand, P.; Degterov, S.A.; Eriksson, G.; Hack, K.; Ben Mahfoud, R. FactSage thermochemical software and databases. Calphad 2002, 26, 189–228. [Google Scholar]
- Liu, H.; Wen, G.H.; Tang, P. Crystallization behaviors of mold fluxes containing Li2O using single hot thermocouple technique. ISIJ Int. 2009, 49, 843–850. [Google Scholar]
- Kashiwaya, Y.; Nakauchi, T.; Pham, K.S.; Akiyama, S.; Ishii, K. Crystallization behaviors concerned with TTT and CCT diagrams of blast furnace slag using hot thermocouple technique. ISIJ Int. 2007, 47, 44–52. [Google Scholar]
- Zhou, L.J.; Wang, W.L.; Liu, R.; Brian, G.; Thomas, B.G. Computational modeling of temperature, flow, and crystallization of mold slag during double hot thermocouple technique experiments. Metall. Mater. Trans. B 2013, 44, 1264–1279. [Google Scholar]




| Samples | Basicity (B) | CaO | SiO2 | MgO | Al2O3 |
|---|---|---|---|---|---|
| A1(XRF) | 1.05 | 40.7% | 38.9% | 9.2% | 11.3% |
| A2(XRF) | 1.02 | 35.8% | 35.1% | 9.1% | 20.0% |
| A3(XRF) | 1.02 | 34.3% | 33.5% | 9.0% | 23.1% |
| Sample | Stage | Temperature range (°C) | Heat exchange time (s) | Cooling rate (°C/s) | Heat capacity (J/mol/K) | Possible recovered waste heat (MJ/t) | Phase change heat (MJ/t) | Total extracted heat (MJ/t) |
|---|---|---|---|---|---|---|---|---|
| A1 | 1 | 1500–1360 | - | - | 69.99 | 160.94 | Crystals: 1% | Non-Crystallization: 1695.71 |
| 2 | 1360–1100 | 43.3 | 6 | 298.91 | 151.73 | Crystallization: 1847.44 | ||
| 3 | 1100–25 | - | - | 1235.86 | ||||
| A2 | 1 | 1500–1380 | - | - | 78.32 | 145.94 | Crystals: 1% | Non-Crystallization: 1793.82 |
| 2 | 1380–1100 | 18.7 | 15 | 340.52 | 151.73 | Crystallization: 1945.55 | ||
| 3 | 1100–25 | - | - | 1307.36 | ||||
| A3 | 1 | 1500–1400 | - | - | 81.51 | 123.99 | Crystals: 1% | Non-Crystallization: 1828.82 |
| 2 | 1400–1080 | 6.4 | 50 | 396.76 | 151.73 | Crystallization: 1980.55 | ||
| 3 | 1080–25 | - | - | 1308.07 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sun, Y.; Zhang, Z.; Liu, L.; Wang, X. Multi-Stage Control of Waste Heat Recovery from High Temperature Slags Based on Time Temperature Transformation Curves. Energies 2014, 7, 1673-1684. https://doi.org/10.3390/en7031673
Sun Y, Zhang Z, Liu L, Wang X. Multi-Stage Control of Waste Heat Recovery from High Temperature Slags Based on Time Temperature Transformation Curves. Energies. 2014; 7(3):1673-1684. https://doi.org/10.3390/en7031673
Chicago/Turabian StyleSun, Yongqi, Zuotai Zhang, Lili Liu, and Xidong Wang. 2014. "Multi-Stage Control of Waste Heat Recovery from High Temperature Slags Based on Time Temperature Transformation Curves" Energies 7, no. 3: 1673-1684. https://doi.org/10.3390/en7031673