A Self-Supported Direct Borohydride-Hydrogen Peroxide Fuel Cell System
Abstract
:Introduction
2. Results and Discussion
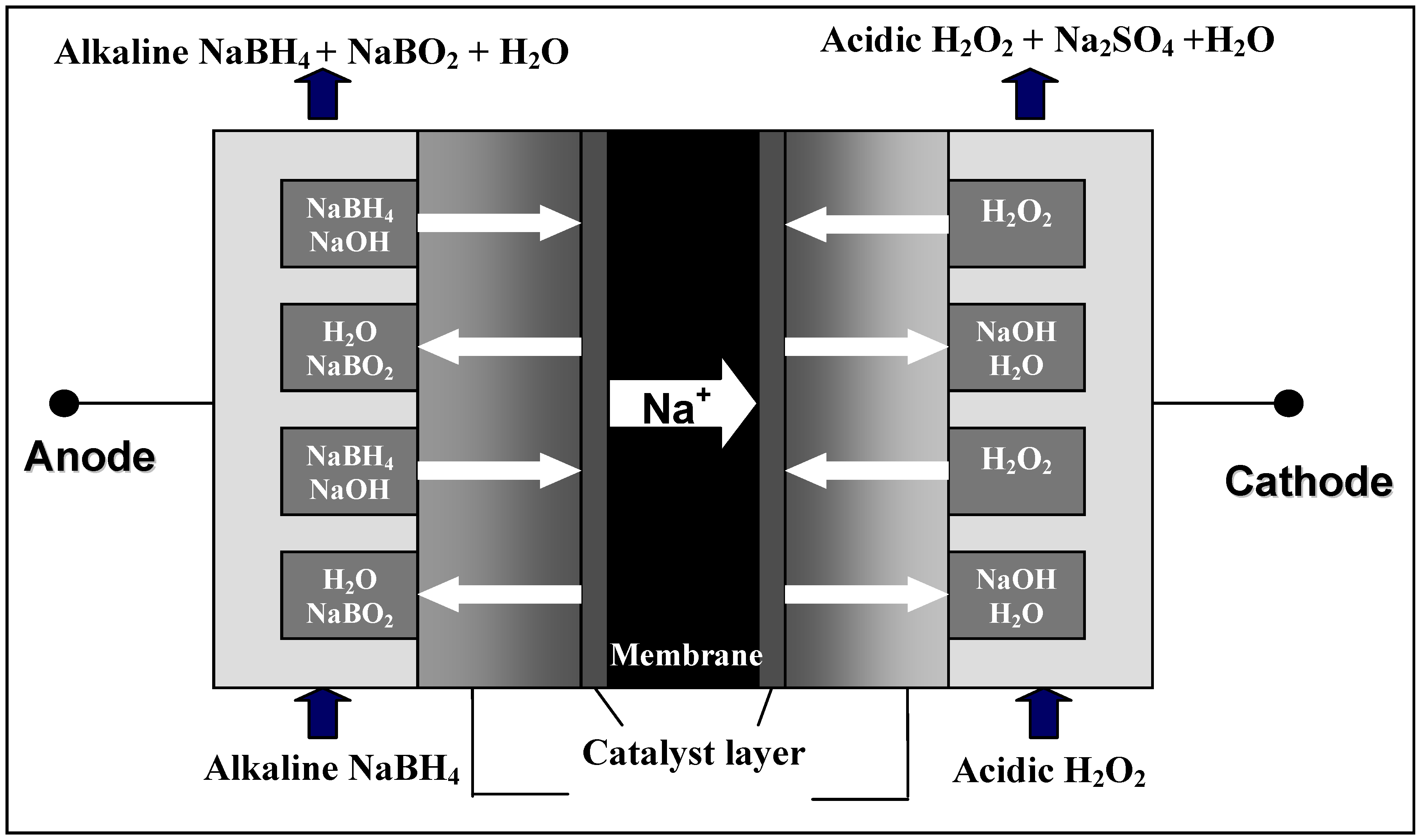
2.1. The operating principle of DBFCs
2.2. Single cell studies
| Reactant | NaBH4 | NaOH | H2O2 | H2SO4 | Total |
|---|---|---|---|---|---|
| Stoichometric (g) | x | 8.46x | 3.6x | 10.37x | 23.43 x |
| Experimental (g) | x | 9.47x | 4x | 11.61x | 26.08x + H2O weight |
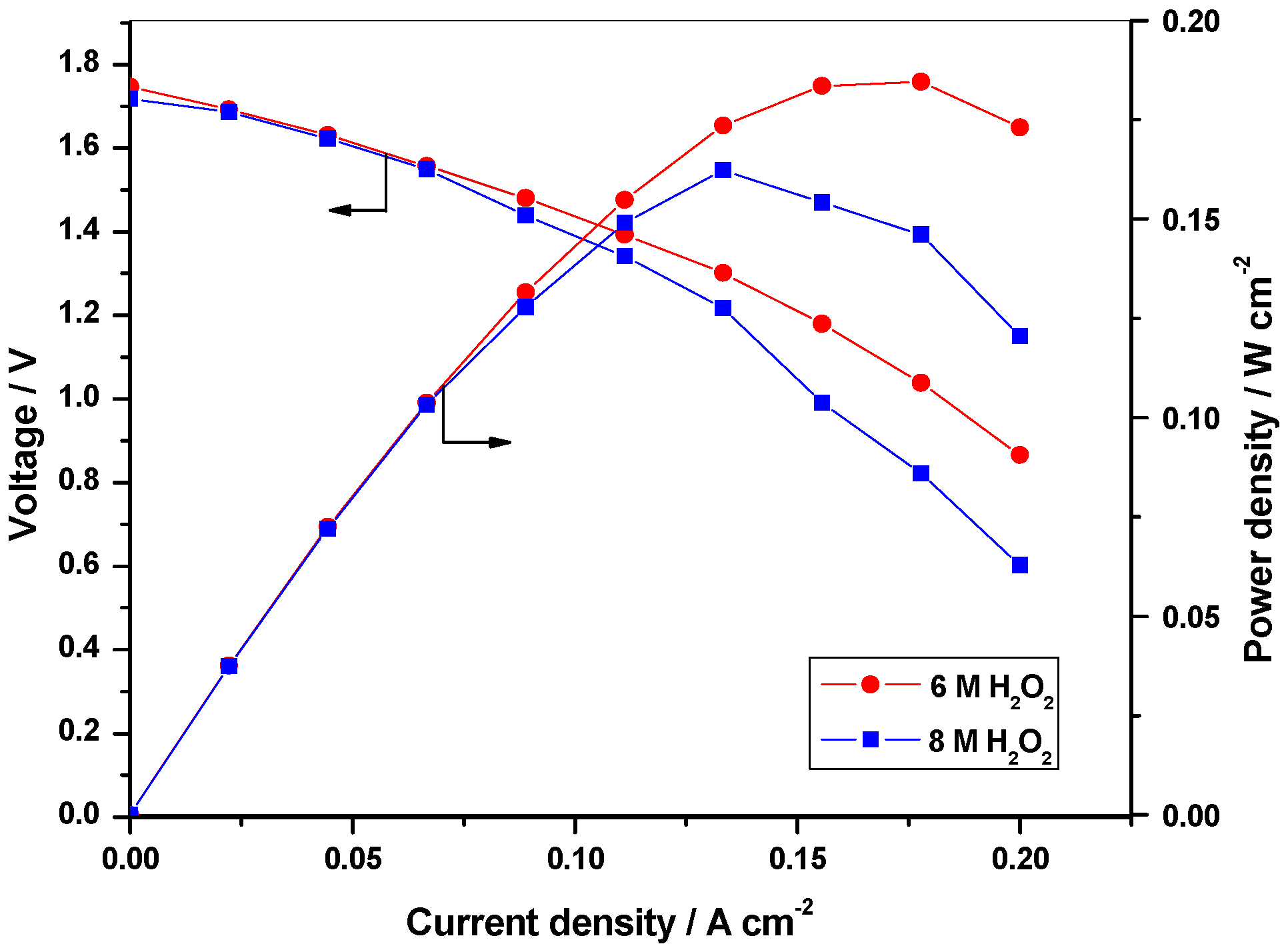
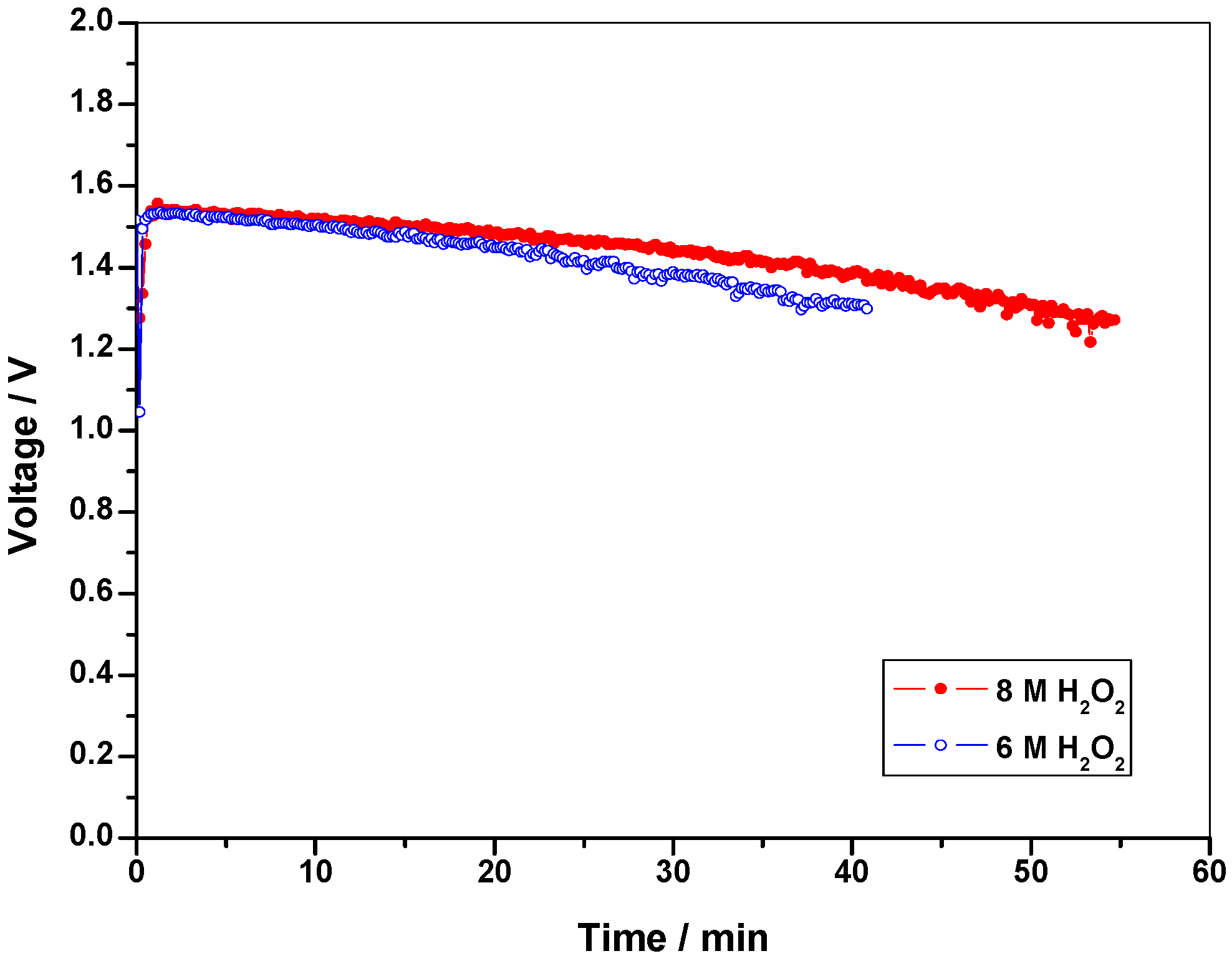
2.2.1. Optimization of oxidant concentration
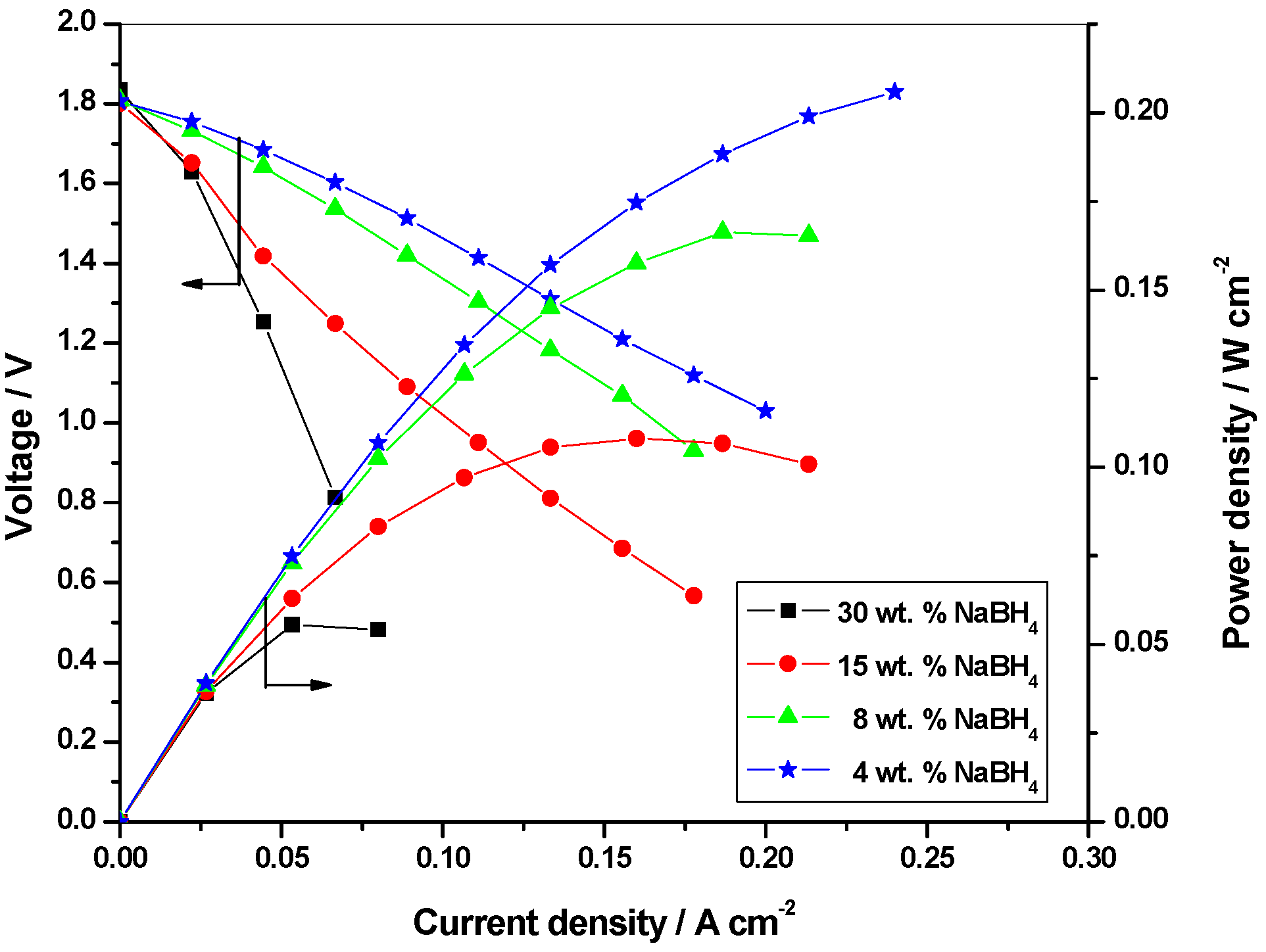
2.2.2. Optimization of fuel concentration

2.3. Studies on the DBFC stack
2.3.1. Optimization of oxidant concentration
| H2O2 (M) in 1.5 M H2SO4 | 8 | 6 | 4 | 2 | 1 |
| Operating time (min.) | 37 | 41 | 47 | 52 | 36 |
2.3.2. Optimization of fuel concentration
| NaBH4 (wt. %) in 11 wt. % NaOH | 30 | 15 | 8 | 4 |
| Operating time (min) | - | - | 52 | 40 |
2.3.3. Stack polarization
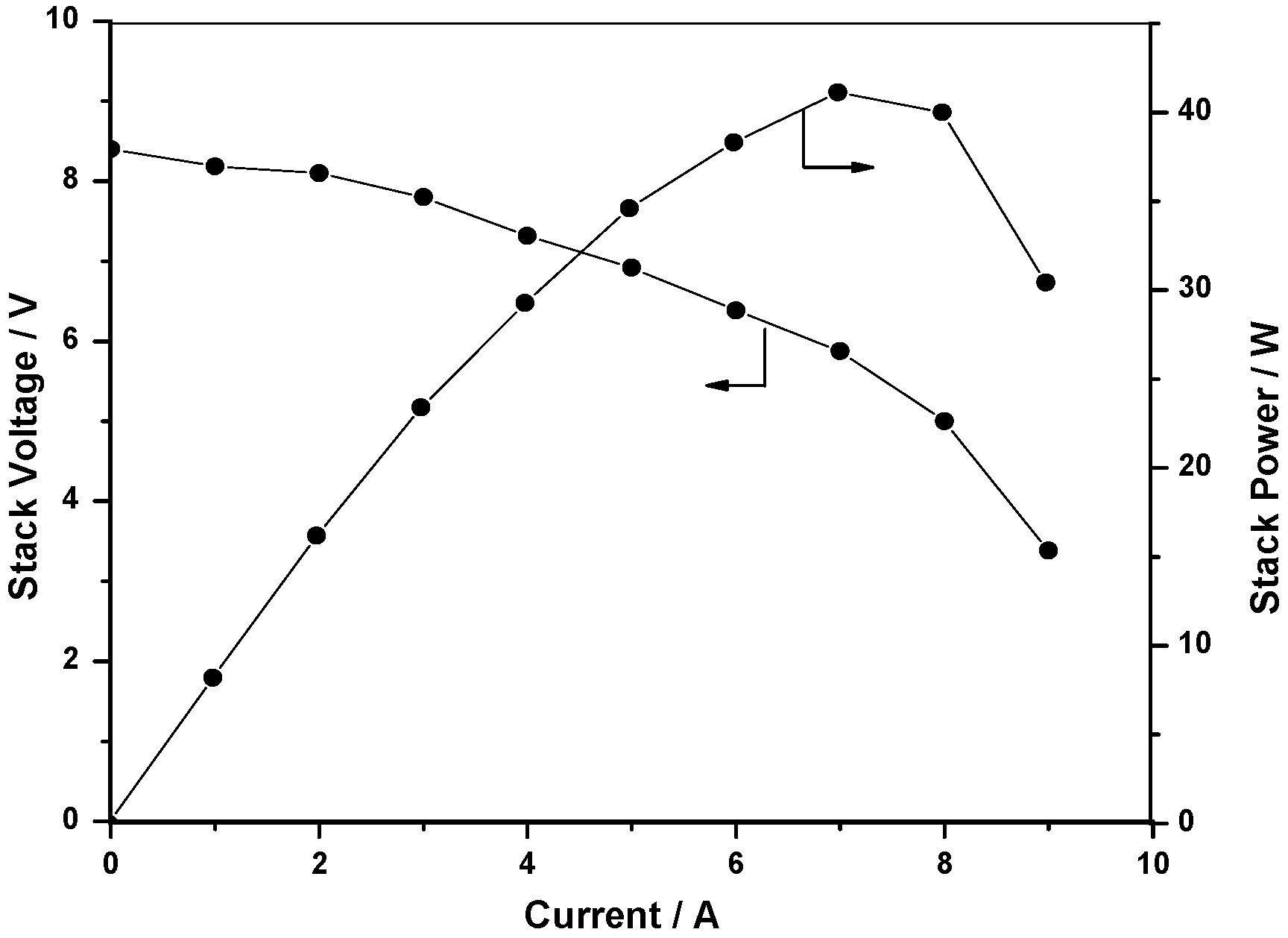
2.4. Self-supported DBFC system
2.5. Operating-time enhancement for the DBFC system
2.6. Safety
3. Experimental
3.1. Membrane electrode assemblies (MEAs)
3.2. Bipolar plate

3.3. Stack assembly

3.4. Construction and working of self- supported DBFC system
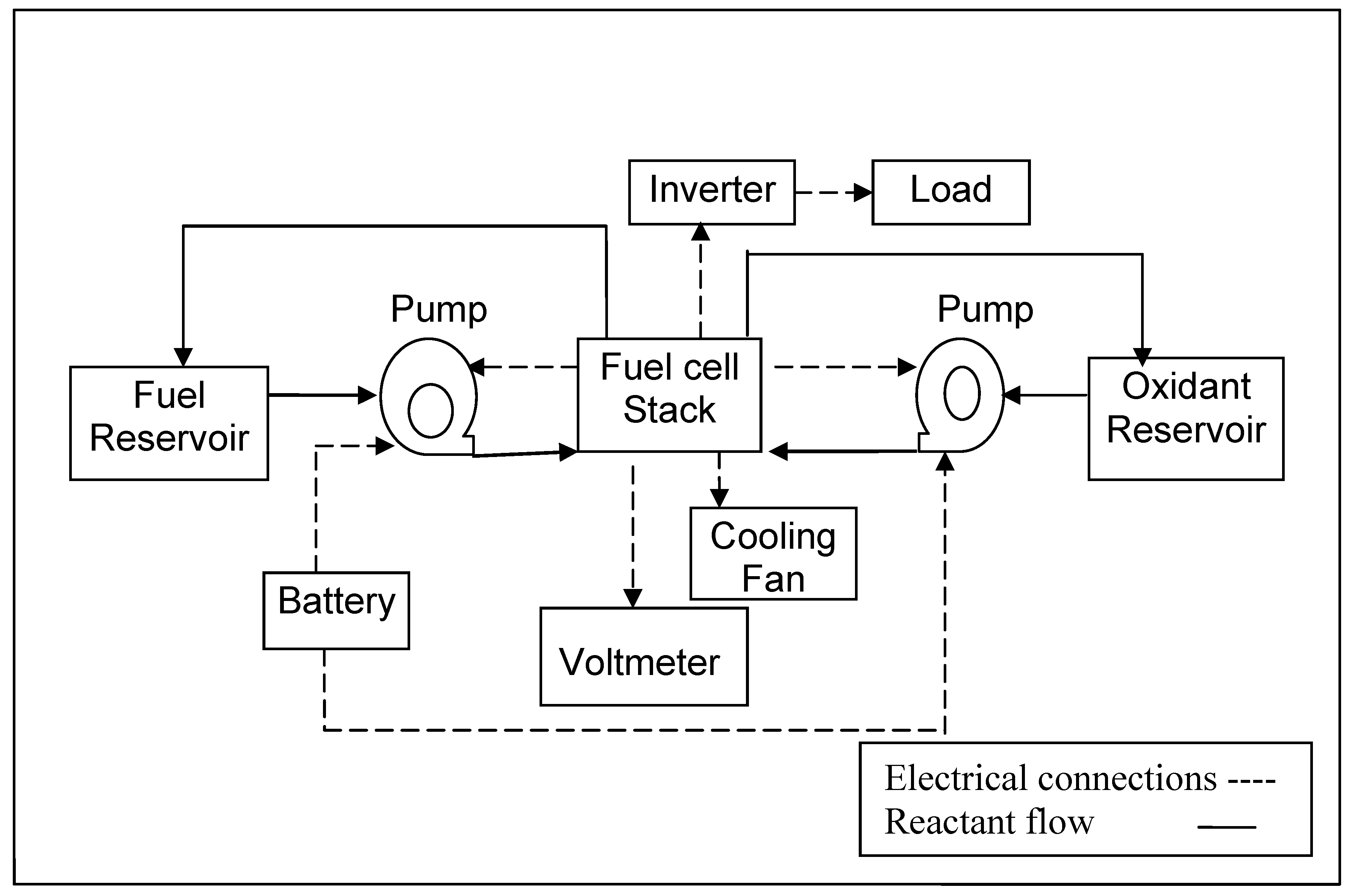
3.5. Electrochemical characterization of single cell and multi-cell stack
4. Conclusions
Acknowledgements
References and Notes
- Larminie, J.; Dicks, A. Fuelling fuel cell. In Fuel Cell System Explained; 2nd Ed.; John Wiley & Sons Ltd: Chichester, West Sussex, England, 2003; pp. 229–308. [Google Scholar]
- Amendola, S.C.; Sharp-Goldman, S.L.; Janjua, M.S.; Spencer, N.C.; Kelly, M.T.; Petillo, P.J.; Binder, M. A safe, portable, hydrogen gas generator using aqueous borohydride solution and Ru catalyst. Int. J. Hydrogen Energy 2000, 25, 969–975. [Google Scholar] [CrossRef]
- Wu, C.; Zhang, H.; Yi, B. Hydrogen generation from catalytic hydrolysis of sodium borohydride for proton exchange membrane fuel cells. Catal. Today 2004, 93-95, 477–483. [Google Scholar] [CrossRef]
- Shukla, A.K.; Jackson, C.L.; Scott, K.; Raman, R.K. An improved-performance liquid-feed solid-polymer-electrolyte direct methanol fuel cell operating at near-ambient conditions. Electrochim. Acta. 2002, 47, 3401–3407. [Google Scholar] [CrossRef]
- Shukla, A.K.; Raman, R.K. Methanol-resistant oxygen-reduction catalysts for direct methanol fuel cells. Annu. Rev. Mater. Res. 2003, 33, 155–168. [Google Scholar] [CrossRef]
- Li, Z.P.; Liu, B.H.; Arai, K.; Asaba, K.; Suda, S. Evaluation of alkaline borohydride solutions as the fuel for fuel cell. J. Power Sources 2004, 126, 28–33. [Google Scholar] [CrossRef]
- Choudhury, N.A.; Raman, R.K.; Sampath, S.; Shukla, A.K. An alkaline direct borohydride fuel cell with hydrogen peroxide as oxidant. J. Power Sources 2005, 143, 1–8. [Google Scholar] [CrossRef]
- Raman, R.K.; Choudhury, N.A.; Shukla, A.K. A high output voltage direct borohydride fuel cell. Electrochem. Solid-State Lett. 2004, 7, A488–A491. [Google Scholar] [CrossRef]
- Raman, R.K.; Prashant, S.K.; Shukla, A.K. A 28-W portable direct borohydride–hydrogen peroxide fuel-cell stack. J. Power Sources 2006, 162, 1073–1076. [Google Scholar] [CrossRef]
- Ponce de León, C.; Walsh, F.C.; Rose, A.; Lakeman, J.B.; Browning, D.J.; Reeve, R.W. A direct borohydride—acid peroxide fuel cell. J. Power Sources 2007, 164, 441–448. [Google Scholar] [CrossRef]
- Miley, G.H.; Luo, N.; Mather, J.; Burton, R.; Hawkins, G.; Gu, L.; Byrd, E.; Gimlin, R.; Shrestha, P.J.; Benavides, G.; Laystrom, J.; Carroll, D. Direct NaBH4/H2O2 Fuel Cells. J. Power Sources 2007, 165, 509–516. [Google Scholar] [CrossRef]
- Cheng, H.; Scott, K. Influence of operation conditions on direct borohydride fuel cell performance. J. Power Sources 2006, 160, 407–412. [Google Scholar] [CrossRef]
- Yang, W.; Yang, S.; Sun, W.; Sun, G.; Xin, Q.J. Nanostructured palladium-silver coated nickel foam cathode for magnesium–hydrogen peroxide fuel cells. Electrochim. Acta 2006, 52, 9–14. [Google Scholar] [CrossRef]
- www.h2o2.com/intro/properties/thermodynamic.html.
- Luo, N.; Miley, G.H.; Mather, J.; Burton, R.; Hawkins, G.; Byrd, E.; Holcomb, F.; Rusek, J. Engineering of the bipolar stack of a direct NaBH4 fuel cell. J. Power Sources 2008, 185, 356–362. [Google Scholar] [CrossRef]
- Lyttle, D.A.; Jensen, E.H.; Struck, W.A. Simple volumetric assay for sodium borohydride. Anal. Chem. 1952, 24, 1843–1844. [Google Scholar] [CrossRef]
- Jeffery, G.H.; Bassett, J.; Mendham, J.; Denney, R.C. Titrimetric analysis. In Vogel’s Textbook of Quantitative Chemical Analysis; 5th Ed.; John Wiley & Sons Inc: New York, NY, USA, 1989; pp. 372–373. [Google Scholar]
- Jeffery, G.H.; Bassett, J.; Mendham, J.; Denney, R.C. Gravimetry. In Vogel’s Textbook of Quantitative Chemical Analysis; John Wiley & Sons Inc: New York, NY, USA, 1989; pp. 490–493. [Google Scholar]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Khadke, P.S.; Sethuraman, P.; Kandasamy, P.; Parthasarathi, S.; Shukla, A.K. A Self-Supported Direct Borohydride-Hydrogen Peroxide Fuel Cell System. Energies 2009, 2, 190-201. https://doi.org/10.3390/en20200190
Khadke PS, Sethuraman P, Kandasamy P, Parthasarathi S, Shukla AK. A Self-Supported Direct Borohydride-Hydrogen Peroxide Fuel Cell System. Energies. 2009; 2(2):190-201. https://doi.org/10.3390/en20200190
Chicago/Turabian StyleKhadke, Prashant S., Pitchumani Sethuraman, Palanivelu Kandasamy, Sridhar Parthasarathi, and Ashok K. Shukla. 2009. "A Self-Supported Direct Borohydride-Hydrogen Peroxide Fuel Cell System" Energies 2, no. 2: 190-201. https://doi.org/10.3390/en20200190




