1. Introduction
Hydrogen fuel cells (H
2-FC) are an attractive solution for supplying electricity due to their high efficiency compared to other technologies. The fuel cell operation and maintenance cost (O & M cost) has been reported to be reasonably lower (0.0019
$ kW
−1) than that for other technologies such as turbine, photo voltaic, and wind turbine (the turbine O & M cost was 2.63 times higher than that of FC), with efficiencies of approximately 40–60% [
1,
2]. Moreover, a H
2-FC avoids greenhouse gas (GHG) emissions when renewable H
2 is employed [
2].
H
2 could be obtained from biomass by coupling a biological process that involves bioethanol production from a biomass and a catalytic process, which includes bioethanol steam reforming and purification of the H
2 stream. In this process, it is possible to achieve higher H
2 yields than other processes such as direct glucose steam reforming and H
2 production from fermentative pathways, due to the ease of yeasts to transform sugars into ethanol and the use of stable and selective catalysts that can efficiently transform ethanol into H
2 [
3,
4].
The biomass
Cachaza, a type of non-centrifugal sugarcane press-mud, is an agroindustrial waste obtained from sugar mills and non-centrifugal sugar (NCS) facilities. This waste is produced after sugarcane juice clarification, and it was reported to be a relevant source of carbohydrates (32–56 wt %) [
5,
6,
7,
8]. In Colombia,
Cachaza production is approximately 700 MTon year
−1, and it is mainly employed as fertilizer and compost. However, such applications could have several drawbacks such as (i) health effects due to the presence of possible pathogens; (ii) water pollution; (iii) soil eutrophication; and (iv) GHG emissions due to improper disposal [
9]. Rather than being used as a fertilizer or compost, up-grading
Cachaza is more attractive in terms of environmental impact and the possibility of boosting NCS facilities. Due to NCS being one of the most relevant agroindustrial sectors in Colombia, the furtherance of new products from this sector could increase its performance, thus improving the farmers’ economic and social development.
Cachaza has been employed as an alternative source of valuable products throughout different conversion pathways. For instance, López-González et al. [
6] assessed the methane (CH
4) production from sugar cane press mud, achieving a yield of 337 mL CH
4 per gram of volatile solids. Similarly, Kuruti et al. [
10] evaluated the bioethanol and volatile fatty acids production throughout an acidogenic pathway by using a microbial consortium. Results showed that 40 kg ethanol and 270 kg volatile fatty acids could be obtained per ton of sugar press mud by this pathway. In addition, Ansari et al. [
5] employed sugarcane press mud to produce chemicals through thermochemical pathways such as pyrolysis; they obtained a wide variety of valued by-products like gas and liquid hydrocarbons, alcohols, phenols, acids, aldehydes, ketones, and biochar. Radjaram et al. [
11] developed a process to obtain H
2 by the anaerobic decomposition of this waste, achieving 59 mol% H
2 and 890 mL H
2 per kg of press mud. Therefore,
Cachaza has been identified as an alternative feedstock to produce a wide spectrum of products. Nevertheless, there are no reports of the
Cachaza harness throughout a process that combines a biological and a catalytic process to produce H
2. Therefore, this research paper highlights the production of bioethanol from
Cachaza by a fermentative pathway, for its further conversion into H
2 by a catalytic pathway.
Bioethanol concentration is a critical factor to produce electricity from biomass because higher bioethanol production represents a downstream energy savings and the lower amount of by-products represents a better catalytic performance. Thus, several authors have shown that an increase in the substrate concentration leads to a higher ethanol production. A lower water concentration in the bioethanol stream corresponds to a lower energy consumption for bioethanol purification [
12]. For instance, Lu et al. [
13] concluded that a proportional increase of final ethanol concentration was achieved between 10 wt % and 30 wt % solids, when corn stover was used as feedstock. Similarly, Siqueira et al. [
14] assessed the effect of the initial soluble solids concentration (Brix) on the ethanol concentration from the fermentation of soybean molasses; they found that the highest ethanol concentration (i.e., 61.35 g·L
−1 ethanol) was achieved under the highest amount of initial total sugars (i.e., 311 g·L
−1 total sugar). Likewise, they found that there was a proportional increase of the ethanol concentration regarding sugar concentration. Alvira et al. [
15] showed similar results. They found that there was also a proportional increase in the ethanol concentration, when the amount of steam-exploded wheat straw concentration increased during fermentation.
However, increasing solid content in the fermentation culture should be done carefully to not over-increase the osmotic pressure and it is not enough to achieve a proper fermentation performance. Hence, other nutrients such as peptone and yeast extract must be added to improve bioethanol production [
16,
17]. For instance, Hu et al. [
18] optimized a glucose culture to improve ethanol production under high amount of glucose content (i.e., 300 g·L
−1). They added peptone, yeast extract, Mg
2+, and Ca
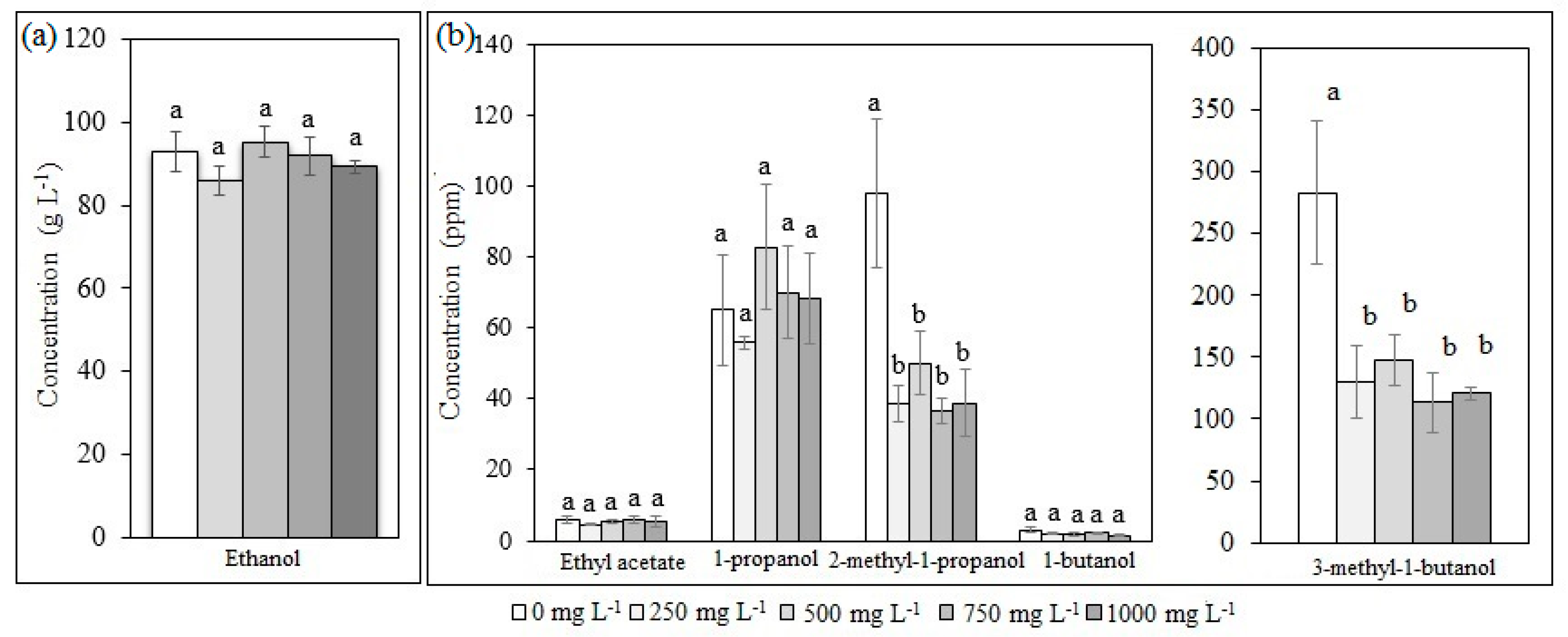
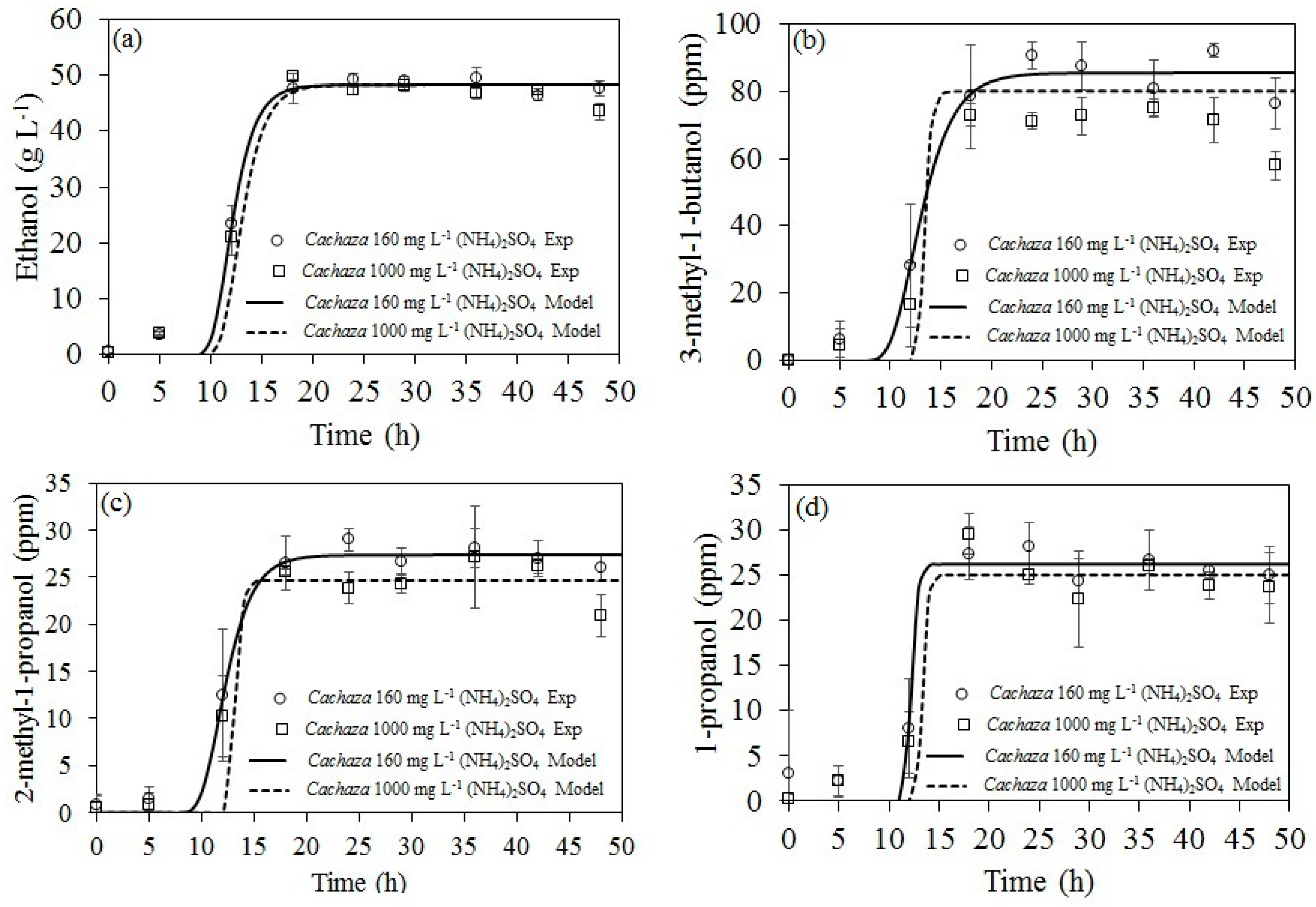
2+ and produced 17.1 vol % ethanol. However, aliphatic components such as organic acids, aldehydes, ketones, and higher, branched alcohols such as 2-methyl-1-propanol (2M1P) and 3-methyl-1-butanol (3M1B) can be enhanced and they are undesirable for further bioethanol steam reforming when acid catalysts are employed [
19,
20,
21,
22]. Among those components, Le Valant et al. [
20] reported that those branched alcohols caused an important effect on Rh/MgAl
2O
4 catalyst deactivation. Conversely, we reported that those alcohols can increase H
2 production but in the presence of a very active, resistant RhPt/CeO
2 catalyst [
3]. Other authors have assessed the bioethanol steam reforming to produce H
2 by employing bioethanol obtained from different agroindustrial matrices, such as wheat [
21], wood [
23], and sugarcane [
24]. However, to the best of our knowledge, there are no studies focused on the bioethanol production with suitable characteristics (i.e., low branched alcohols) for its further conversion into H
2 by catalytic steam reforming. Therefore, it is relevant to assess some strategies to control the formation of those branched alcohols during fermentation. Nitrogen addition could be a possible strategy to control their formation.
Nitrogen is an essential nutrient during fermentation because it influences both yeast growth and metabolism. In addition, nitrogen deficiency is the main cause of sluggish fermentation [
25]. The fermentation rate, ethanol tolerance, yeast cell growth [
26], and by-product formation are affected by variations in the amount and the source of nitrogen in the culture media.
Saccharomyces cerevisiae (
S. cerevisiae) cannot assimilate certain nitrogen sources such as nitrate, nitrites, proteins, and polypeptides because it is a non-diazotrophic and non-proteolytic organism [
27]. Thus, other nitrogen sources such as ammonia and amino acids should be employed as nitrogen sources in fermentation [
26]. The addition of ammonium salts to the culture media can induce a decrease in the production of higher alcohols. Vidal et al. [
28] found that the addition of (NH
4)
2SO
4 influenced the gene expression related to the Ehrlich Metabolic Pathway (EMP), producing an exhausting higher alcohol generation during the production of industrial
Cachaça (i.e., Brazilian white spirit beverage). Hernadez-Orte et al. [
29] suggested that the presence of ammonia during the fermentation of a synthetic media, which simulates a standard grape juice, decreased the amount of higher alcohol formation. Beltran et al. [
30] stated that higher alcohols could also be produced by the biosynthetic pathway of amino acids during synthetic grape must fermentation. Both pathways, Ehrlich and de novo amino acids, produce α-keto-acids, which are mainly responsible for the formation of higher alcohols, as demonstrated by Nördstrom using brewer’s yeast [
31]. (NH
4)
2SO
4 has been widely employed to control the formation of aliphatic compounds for beer and wine production [
28,
29,
32]. However, there are no reports of the use of (NH
4)
2SO
4 to control those components in bioethanol for energy applications.
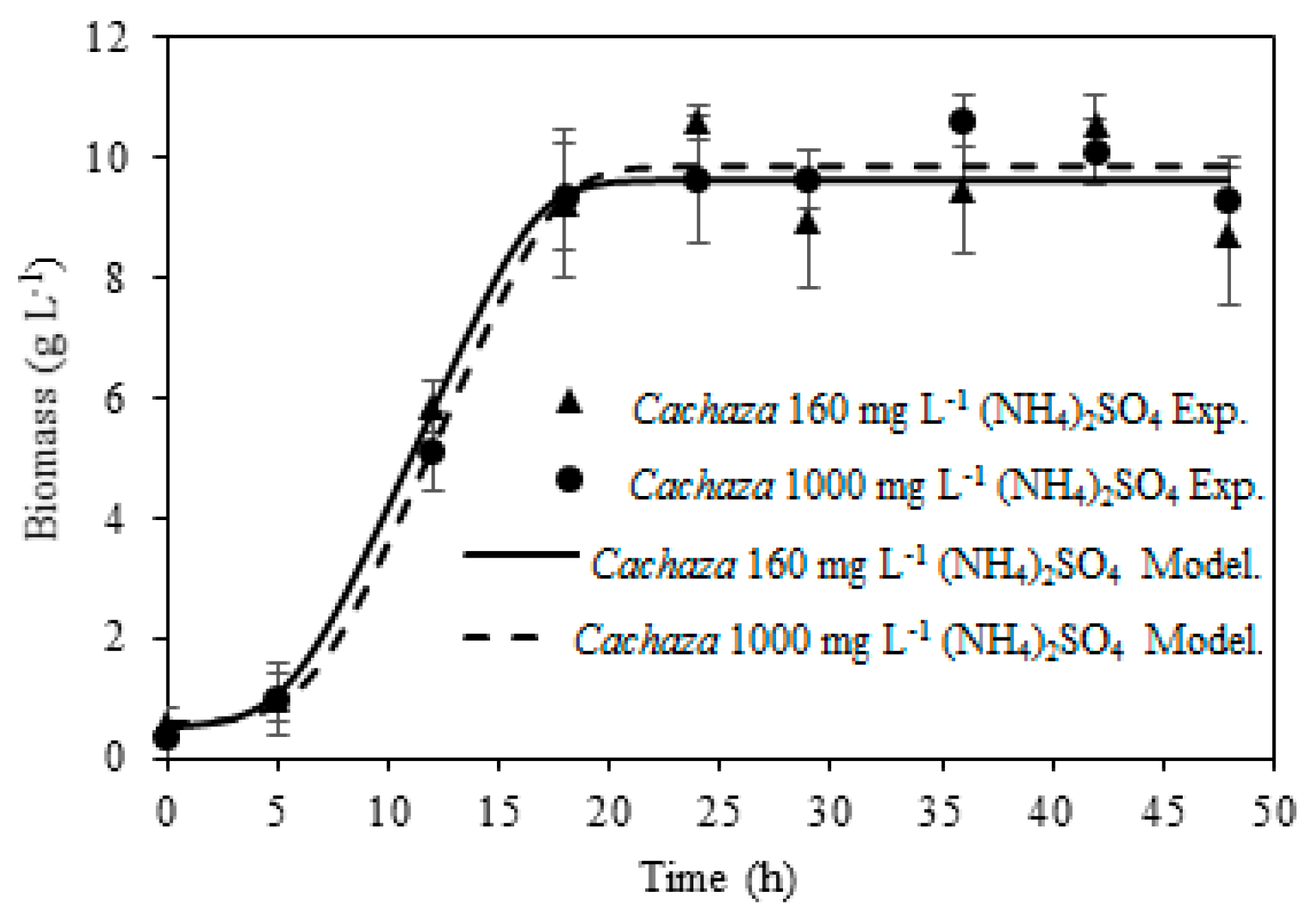
Accordingly, the aim of this work was to assess the fermentation under different (NH
4)
2SO
4 loadings for bioethanol production from
Cachaza, employing a
S. cerevisiae strain. The kinetic parameters for the production of ethanol and aliphatic compounds were estimated to determine the conditions that increased ethanol and reduced aliphatic compounds [
33]. Those parameters were fit to the modified Gompertz model for ethanol, 2M1P, 3M1B, and 1-propanol production. As there are no reports of bioethanol production from
Cachaza, this study is pertinent for providing new added value to this waste for energy applications.