Heat Modeling and Material Development of Mg-Based Nanomaterials Combined with Solid Oxide Fuel Cell for Stationary Energy Storage
Abstract
:1. Introduction
2. Experimental Section
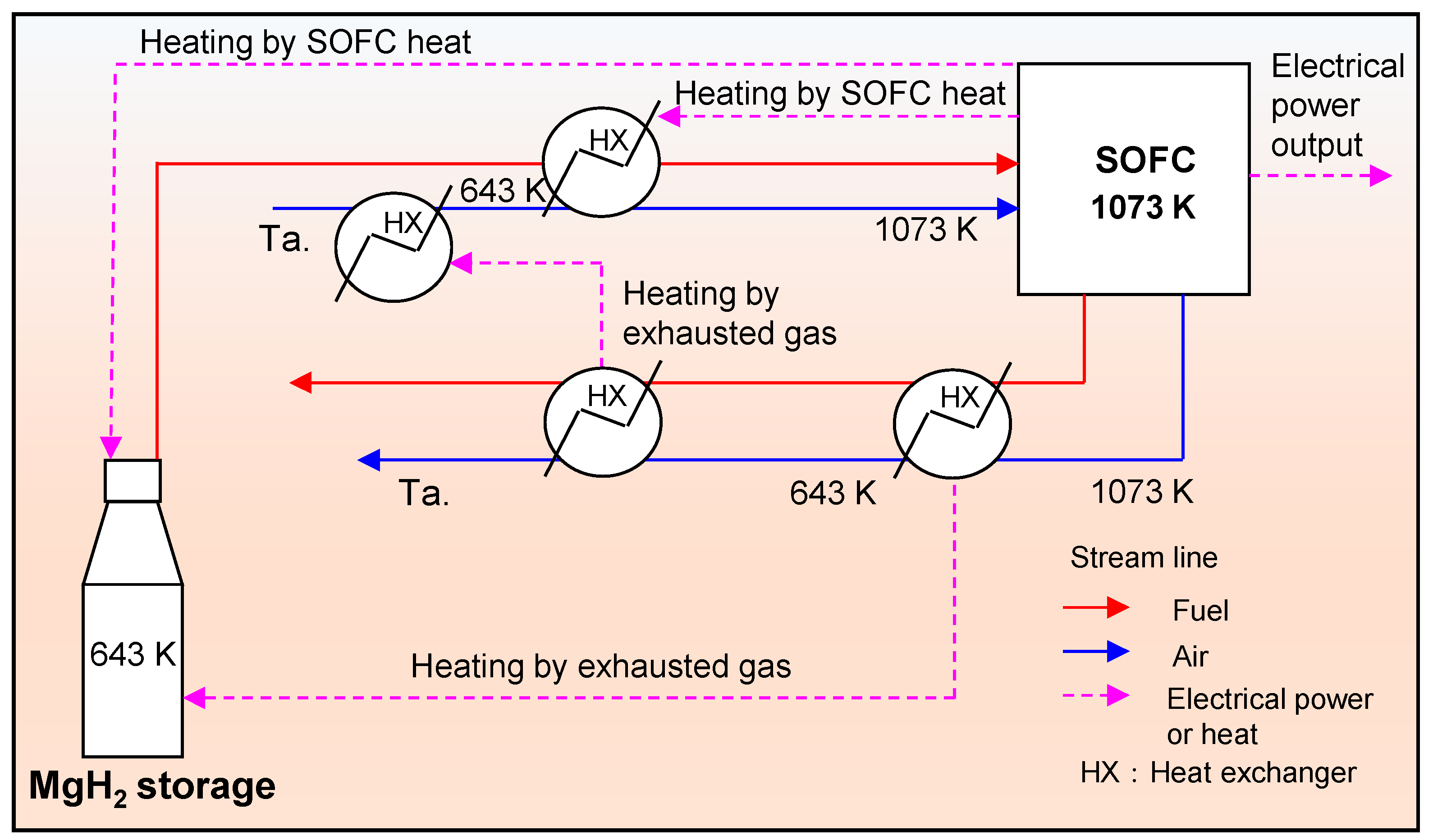
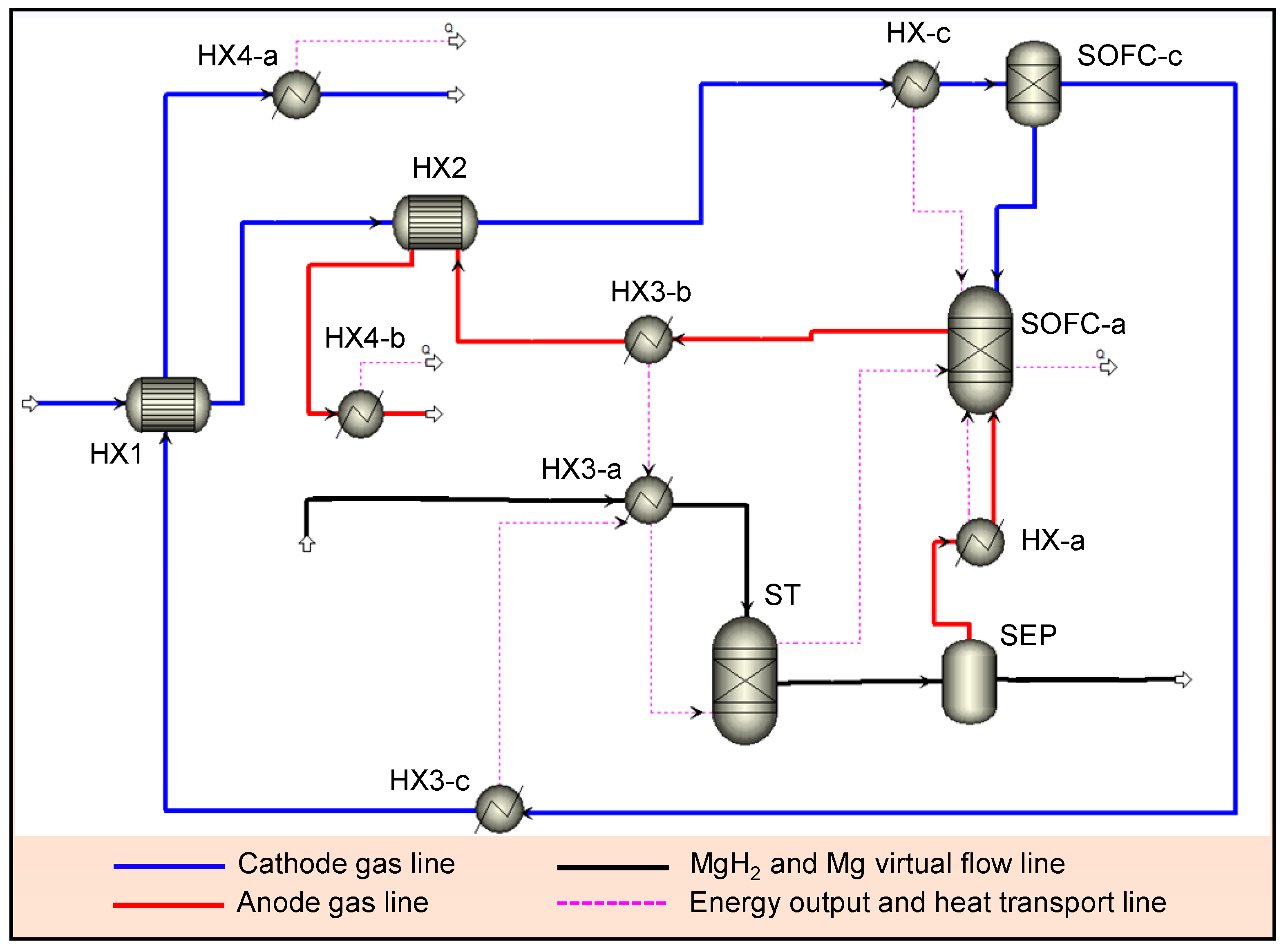
2.1. Simulation of MgH2-SOFC
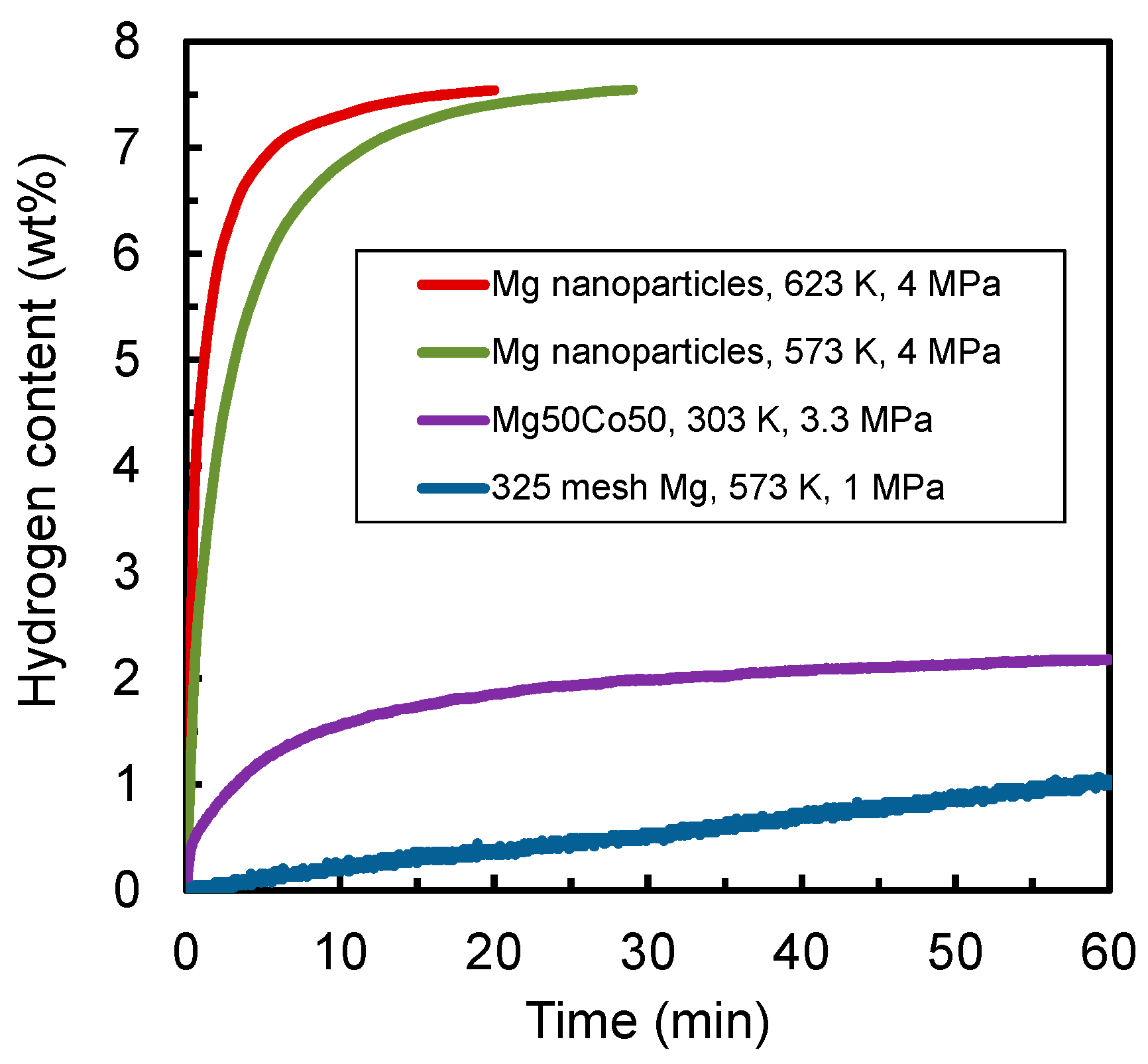
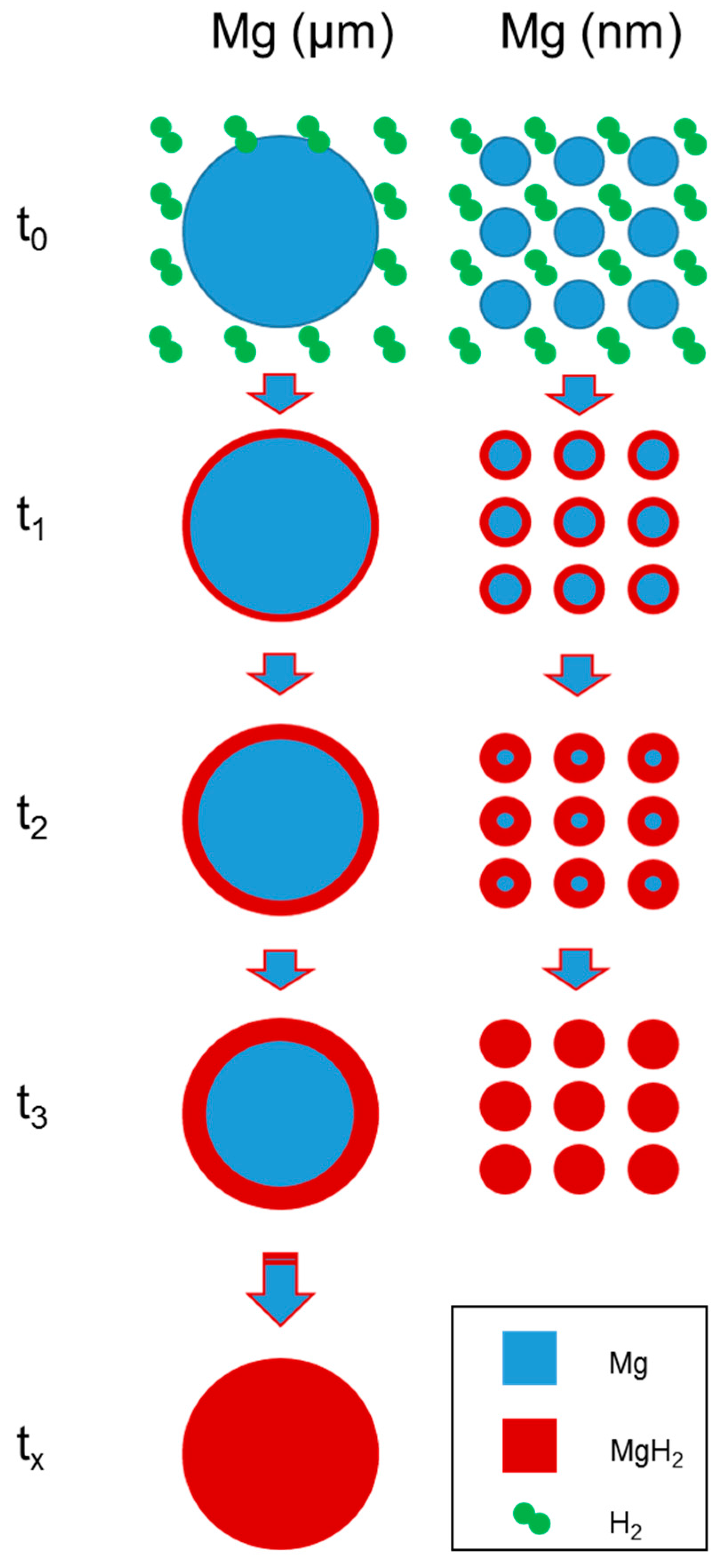
2.2. Experimental Details of Material Development
3. Results and Discussion
4. Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Shao, H.; Xin, G.; Zheng, J.; Li, X.; Akiba, E. Nanotechnology in mg-based materials for hydrogen storage. Nano Energy 2012, 1, 590–601. [Google Scholar] [CrossRef]
- Delhomme, B.; Lanzini, A.; Ortigoza-Villalba, G.A.; Nachev, S.; de Rango, P.; Santarelli, M.; Marty, P.; Leone, P. Coupling and thermal integration of a solid oxide fuel cell with a magnesium hydride tank. Int. J. Hydrog. Energy 2013, 38, 4740–4747. [Google Scholar] [CrossRef]
- Doyle, T.S.; Dehouche, Z.; Stankouic, S. Decentralized power and heat derived from an eco-innovative integrated gasification fuel cell combined cycle fuelled by waste. Int. J. Hydrog. Energy 2015, 40, 9013–9025. [Google Scholar] [CrossRef]
- Gkanas, E.I.; Makridis, S.S. Effective thermal management of a cylindrical MgH2 tank including thermal coupling with an operating SOFC and the usage of extended surfaces during the dehydrogenation process. Int. J. Hydrog. Energy 2016, 41, 5693–5708. [Google Scholar] [CrossRef]
- Yiotis, A.G.; Kainourgiakis, M.E.; Kosmidis, L.I.; Charalambopoulou, G.C.; Stubos, A.K. Thermal coupling potential of solid oxide fuel cells with metal hydride tanks: Thermodynamic and design considerations towards integrated systems. J. Power Sources 2014, 269, 440–450. [Google Scholar] [CrossRef]
- Shao, H.; He, L.; Lin, H.; Li, H.-W. Progress and trends in mg-based materials for energy-storage research: A review. Energy Technol. 2017. [Google Scholar] [CrossRef]
- Lithium-Ion Battery. Available online: http://en.Wikipedia.Org/wiki/lithium-ion_battery (accessed on 4 July 2017).
- Mahato, N.; Banerjee, A.; Gupta, A.; Omar, S.; Balani, K. Progress in material selection for solid oxide fuel cell technology: A review. Prog. Mater Sci. 2015, 72, 141–337. [Google Scholar] [CrossRef]
- Bhattacharyya, D.; Rengaswamy, R. A review of solid oxide fuel cell (SOFC) dynamic models. Ind. Eng. Chem. Res. 2009, 48, 6068–6086. [Google Scholar] [CrossRef]
- Malzbender, J.; Steinbrech, R.W.; Singheiser, L. A review of advanced techniques for characterising SOFC behaviour. Fuel Cells 2009, 9, 785–793. [Google Scholar] [CrossRef]
- Patakangas, J.; Ma, Y.; Jing, Y.F.; Lund, P. Review and analysis of characterization methods and ionic conductivities for low-temperature solid oxide fuel cells (LT-SOFC). J. Power Sources 2014, 263, 315–331. [Google Scholar] [CrossRef]
- Perng, S.W.; Chen, C.R. Numerical investigation of anode thickness on the performance and heat/mass transport phenomenon for an anode-supported SOFC button cell. J. Nanomater. 2015, 2015, 172876. [Google Scholar] [CrossRef]
- Cimenti, M.; Hill, J.M. Direct utilization of liquid fuels in SOFC for portable applications: Challenges for the selection of alternative anodes. Energies 2009, 2, 377–410. [Google Scholar] [CrossRef]
- Nguyen, X.V.; Chang, C.T.; Jung, G.B.; Chan, S.H.; Huang, W.C.W.; Hsiao, K.J.; Lee, W.T.; Chang, S.W.; Kao, I.C. Effect of sintering temperature and applied load on anode-supported electrodes for SOFC application. Energies 2016, 9, 701. [Google Scholar] [CrossRef]
- Rabbani, A.; Rokni, M. Modeling and analysis of transport processes and efficiency of combined SOFC and pemfc systems. Energies 2014, 7, 5502–5522. [Google Scholar] [CrossRef] [Green Version]
- JXTG Company Website. Available online: http://www.Noe.Jx-group.Co.Jp (accessed on 4 July 2017).
- Powell, M.; Meinhardt, K.; Sprenkle, V.; Chick, L.; McVay, G. Demonstration of a highly efficient solid oxide fuel cell power system using adiabatic steam reforming and anode gas recirculation. J. Power Sources 2012, 205, 377–384. [Google Scholar] [CrossRef]
- Satyapal, S.; Petrovic, J.; Read, C.; Thomas, G.; Ordaz, G. The u.S. Department of energy’s national hydrogen storage project: Progress towards meeting hydrogen-powered vehicle requirements. Catal. Today 2007, 120, 246–256. [Google Scholar] [CrossRef]
- Nielsen, T.K.; Manickam, K.; Hirscher, M.; Besenbacher, F.; Jensen, T.R. Confinement of MgH2 nanoclusters within nanoporous aerogel scaffold materials. ACS Nano 2009, 3, 3521–3528. [Google Scholar] [CrossRef] [PubMed]
- Ruminski, A.M.; Bardhan, R.; Brand, A.; Aloni, S.; Urban, J.J. Synergistic enhancement of hydrogen storage and air stability via mg nanocrystal-polymer interfacial interactions. Energy Environ. Sci. 2013, 6, 3267–3271. [Google Scholar] [CrossRef]
- Cui, J.; Wang, H.; Liu, J.; Ouyang, L.; Zhang, Q.; Sun, D.; Yao, X.; Zhu, M. Remarkable enhancement in dehydrogenation of MgH2 by a nano-coating of multi-valence ti-based catalysts. J. Mater. Chem. A 2013, 1, 5603–5611. [Google Scholar] [CrossRef]
- Lu, J.; Choi, Y.J.; Fang, Z.Z.; Sohn, H.Y.; Ronnebro, E. Hydrogen storage properties of nanosized MgH2–0.1TiH2 prepared by ultrahigh-energy-high-pressure milling. J. Am. Chem. Soc. 2009, 131, 15843–15852. [Google Scholar] [PubMed]
- Anastasopol, A.; Pfeiffer, T.V.; Middelkoop, J.; Lafont, U.; Canales-Perez, R.J.; Schmidt-Ott, A.; Mulder, F.M.; Eijt, S.W.H. Reduced enthalpy of metal hydride formation for Mg–Ti nanocomposites produced by spark discharge generation. J. Am. Chem. Soc. 2013, 135, 7891–7900. [Google Scholar] [CrossRef] [PubMed]
- Shao, H.Y.; Ma, W.G.; Kohno, M.; Takata, Y.; Xin, G.B.; Fujikawa, S.; Fujino, S.; Bishop, S.; Li, X.G. Hydrogen storage and thermal conductivity properties of Mg-based materials with different structures. Int. J. Hydrog. Energy 2014, 39, 9893–9898. [Google Scholar] [CrossRef]
- Shao, H.; Wang, Y.; Xu, H.; Li, X. Hydrogen storage properties of magnesium ultrafine particles prepared by hydrogen plasma-metal reaction. Mater. Sci. Eng. B 2004, 110, 221–226. [Google Scholar] [CrossRef]
- Shao, H.; Asano, K.; Enoki, H.; Akiba, E. Fabrication, hydrogen storage properties and mechanistic study of nanostructured Mg50Co50 body-centered cubic alloy. Scr. Mater. 2009, 60, 818–821. [Google Scholar] [CrossRef]
- Cadoret, L.; Yu, C.C.; Huang, H.P.; Lee, M.J. Effects of physical properties estimation on process design: A case study using aspenplus. Asia-Pac. J. Chem. Eng. 2009, 4, 729–734. [Google Scholar] [CrossRef]
- Zhang, W.; Croiset, E.; Douglas, P.L.; Fowler, M.W.; Entchev, E. Simulation of a tubular solid oxide fuel cell stack using AspenPlusTM unit operation models. Energy Convers. Manag. 2005, 46, 181–196. [Google Scholar] [CrossRef]
- Shao, H.; Akiba, E. Catalyzed nanostructured Mg-based materials for energy storage. In Proceedings of the 2nd Asian Symposium on Hydrogen Storage Materials, Jeju, South Korea, 22–25 April 2012. [Google Scholar]
- Iwata, M.; Hikosaka, T.; Morita, M.; Iwanari, T.; Ito, K.; Onda, K.; Esaki, Y.; Sakaki, Y.; Nagata, S. Performance analysis of planar-type unit SOFC considering current and temperature distributions. Solid State Ion. 2000, 132, 297–308. [Google Scholar] [CrossRef]
- Wang, Y.; Yoshiba, F.; Watanabe, T.; Weng, S. Numerical analysis of electrochemical characteristics and heat/species transport for planar porous-electrode-supported SOFC. J. Power Sources 2007, 170, 101–110. [Google Scholar] [CrossRef]
- Lin, Y.; Beale, S.B. Performance predictions in solid oxide fuel cells. Appl. Math. Model. 2006, 30, 1485–1496. [Google Scholar] [CrossRef]
- Guo, H.A.; Iqbal, G.; Kang, B.S. Effects of PH3 contaminant on solid oxide fuel cells performance and related anode surface temperature measurements. Int. J. Appl. Ceram. Technol. 2011, 8, 68–73. [Google Scholar] [CrossRef]
- Eveloy, V.; Karunkeyoon, W.; Rodgers, P.; Al Alili, A. Energy, exergy and economic analysis of an integrated solid oxide fuel cell—Gas turbine—Organic rankine power generation system. Int. J. Hydrog. Energy 2016, 41, 13843–13858. [Google Scholar] [CrossRef]
- Ferrero, D.; Lanzini, A.; Leone, P.; Santarelli, M. Reversible operation of solid oxide cells under electrolysis and fuel cell modes: Experimental study and model validation. Chem. Eng. J. 2015, 274, 143–155. [Google Scholar] [CrossRef]
- Shao, H.; Asano, K.; Enoki, H.; Akiba, E. Correlation study between hydrogen absorption property and lattice structure of Mg-based BCC alloys. Int. J. Hydrog. Energy 2009, 34, 2312–2318. [Google Scholar] [CrossRef]
- Braun, R.J.; Klein, S.A.; Reindl, D.T. Evaluation of system configurations for solid oxide fuel cell-based micro-combined heat and power generators in residential applications. J. Power Sources 2006, 158, 1290–1305. [Google Scholar] [CrossRef]
- Rokni, M. Plant characteristics of an integrated solid oxide fuel cell cycle and a steam cycle. Energy 2010, 35, 4691–4699. [Google Scholar] [CrossRef]
- Bloom Energy Company Website. Available online: http://www.Bloomenergy.Com (accessed on 4 July 2017).
- CeresPower Company Website. Available online: http://www.Cerespower.Com (accessed on 4 July 2017).
- CFCL Company Website. Available online: http://www.Cfcl.Com.Au (accessed on 4 July 2017).
- Shao, H.; Cheng, C.; Liu, T.; Li, X. Phase, microstructure and hydrogen storage properties of mg-ni materials synthesized from metal nanoparticles. Nanotechnology 2014, 25, 135704. [Google Scholar] [CrossRef] [PubMed]
- Sadhasivam, T.; Kim, H.T.; Jung, S.; Roh, S.H.; Park, J.H.; Jung, H.Y. Dimensional effects of nanostructured Mg/MgH2 for hydrogen storage applications: A review. Renew. Sustain. Energy Rev. 2017, 72, 523–534. [Google Scholar] [CrossRef]
- Zhu, M.; Lu, Y.S.; Ouyang, L.Z.; Wang, H. Thermodynamic tuning of mg-based hydrogen storage alloys: A review. Materials 2013, 6, 4654–4674. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.S.; Rosa, L.; Wang, K.L.; Endo, M.; Juodkazis, S.; Ohtani, B.; Kowalska, E. Size-controlled gold nanoparticles on octahedral anatase particles as efficient plasmonic photocatalyst. Appl. Catal. B Environ. 2017, 206, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, L.Z.; Ma, M.L.; Huang, M.H.; Duan, R.M.; Wang, H.; Sun, L.X.; Zhu, M. Enhanced hydrogen generation properties of MgH2-based hydrides by breaking the magnesium hydroxide passivation layer. Energies 2015, 8, 4237–4252. [Google Scholar] [CrossRef]
- Shen, C.Q.; Aguey-Zinsou, K.F. Electrodeposited magnesium nanoparticles linking particle size to activation energy. Energies 2016, 9, 1073. [Google Scholar] [CrossRef]
- Shao, H.Y.; Wang, Y.T.; Li, X.G. Synthesis and properties of nanostructured Mg2Ni-based compounds. Mater. Sci. Forum 2005, 475–479, 2445–2448. [Google Scholar] [CrossRef]
- Bhouri, M.; Burger, I. Numerical investigation of H2 absorption in an adiabatic high-temperature metal hydride reactor based on thermochemical heat storage: MgH2 and Mg(OH)2 as reference materials. Int. J. Hydrog. Energy 2017, 42, 16632–16644. [Google Scholar] [CrossRef]
- Shen, D.; Zhao, C.Y. Thermal analysis of exothermic process in a magnesium hydride reactor with porous metals. Chem. Eng. Sci. 2013, 98, 273–281. [Google Scholar] [CrossRef]


| Item | Q (W) | Fraction of Total Energy Generated (%) |
|---|---|---|
| Total energy generated by SOFC | 68,970 | 100 |
| Necessary heat to sustain operation | 21,633 | 31.4 |
| Remaining energy (electrical power output) | 47,337 | 68.6 |
| Available heat recovery | 9237 | 13.4 |
| Available electrical power output + heat recovery | 56,574 | 82.0 |
| Total energy generated by SOFC | 68,970 | 100 |
| Electricity Generation | Heat Recovery | System Configuration | Source |
|---|---|---|---|
| 68.6% LHV | 13.4% LHV | SOFC + MgH2 storage unit | This study * |
| 56.6% LHV | - | SOFC + gas recirculation unit | Powell et al. [17] |
| 45% LHV | 43% LHV | SOFC + micro CHP unit | Braun et al. [37] |
| 51% LHV | 71% LHV | SOFC + steam turbine combined | Rokni [38] |
| 42% LHV | 45% LHV | SOFC + Heat recovery unit | ENE-FARM [16] |
| 50% LHV | - | SOFC electrical power generation | Bloom Energy [39] |
| 40–55% LHV | 40% LHV | 1 kW to MW CHP system | Ceres Power [40] |
| 60% LHV | 29% LHV | Small scale (1.5 kW) CHP system | Ceramic Fuel Cells [41] |
| Sample | Hydrogen Capacity (wt %) | Hydrogen Absorption Kinetics | Thermal Conductivity (W/(m·K)) |
|---|---|---|---|
| 325 mesh Mg | 7.7 | poor | 10.42 |
| Mg nanoparticles | 7.7 | good | 4.985 |
| Mg50Co50 bcc alloy | 2–3 | superior | 0.432 |
© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shao, H. Heat Modeling and Material Development of Mg-Based Nanomaterials Combined with Solid Oxide Fuel Cell for Stationary Energy Storage. Energies 2017, 10, 1767. https://doi.org/10.3390/en10111767
Shao H. Heat Modeling and Material Development of Mg-Based Nanomaterials Combined with Solid Oxide Fuel Cell for Stationary Energy Storage. Energies. 2017; 10(11):1767. https://doi.org/10.3390/en10111767
Chicago/Turabian StyleShao, Huaiyu. 2017. "Heat Modeling and Material Development of Mg-Based Nanomaterials Combined with Solid Oxide Fuel Cell for Stationary Energy Storage" Energies 10, no. 11: 1767. https://doi.org/10.3390/en10111767





