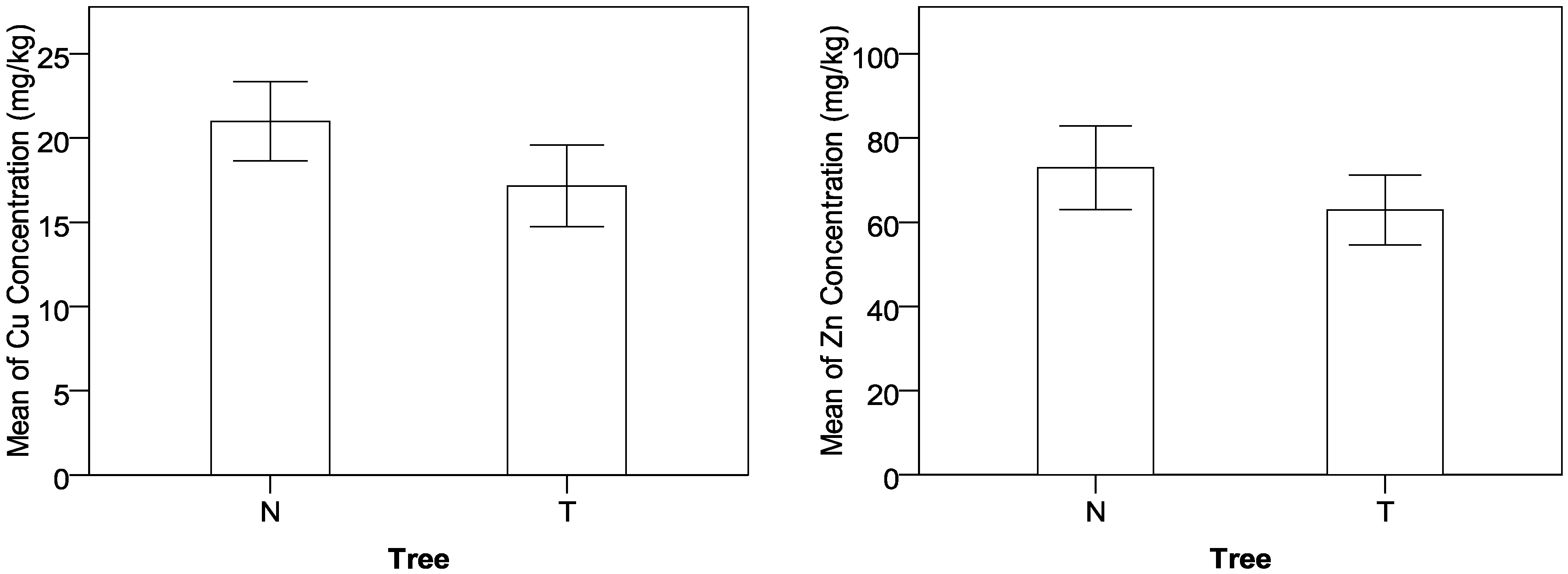1. Introduction
High concentrations of heavy metals may affect the ecosystem and human health. Heavy metals present in the roadside soils and grasses may be transported through the food chain to the human body and have a significant toxicity to people. The environmental issues related to heavy-metal contamination are becoming serious in developing countries [
1]. With the rapid industrialization and urbanization trend, the increment of traffic activities substantially contributes to the accumulations of heavy metals discharged by vehicles in roadside environments. Heavy-metal pollution in agricultural areas owing to traffic emissions may contaminate the crops growing near the roadways [
2]. In agricultural areas, uptake of heavy metals through the soil-crop system could play a predominant role in human exposure to heavy metals [
3].
Cu, Zn, Cd, and Pb are the typical metal pollutants due to traffic activities [
4]. At low doses some heavy metals are essential micronutrients for plants, but in higher doses, they may cause metabolic disorders and growth inhabitation in most plant species [
5]. Cu and Zn are trace elements that are essential for human health, but in high doses Cu can cause health problems such as anaemia, liver and kidney damage, and stomach and intestinal irritation [
6], and very high levels of zinc can damage the pancreas and disturb the protein metabolism, and cause arteriosclerosis. On the other hand, Cd and Pb, even at extremely low concentrations, are toxic and lead to many diseases, including increased risk of cancer [
7].
The sources of heavy-metal emissions from vehicles include fuel combustion, lubricating oil consumption, tire wear, brake wear, road abrasion,
etc. [
8,
9,
10,
11,
12]. Cd emission is mainly from lubricating oil consumption and tire wear. Zn comes from tire wear and galvanized parts such as fuel tanks [
13]. Brake wear is the most important source for Cu and Pb emissions. Pb comes also from exhaust gas and worn metal alloys in the engine [
8]. Through the atmospheric deposit or road runoff, heavy metals can be transported into the roadside soils [
14,
15], where the roadside grasses absorb these heavy-metal elements from the soils through their roots. The grasses’ leaves or stems may also absorb heavy metals from atmospheric particles [
16]. On average, the heavy metals’ concentrations in roadside grasses are significantly lower than those in roadside soils [
17,
18,
19].
Observation studies have been conducted in many countries to investigate the heavy-metal contamination of roadside soils. It was found that the heavy-metal concentrations were influenced by multiple factors, such as traffic volume [
20], highway characteristics [
21], road and roadside terrain [
22], roadside distance, wind direction [
23], rainfall [
20], seeded strip [
17,
18,
19,
20,
21,
22,
23,
24], local economy [
25,
26],
etc. High traffic volume can heighten the heavy-metal content in the roadside soils [
20]. Correspondingly, the roadside heavy-metal concentrations in developed areas are higher than those in underdeveloped areas [
22]. It has been reported that the heavy-metal content has a belt-shaped distribution in terms of roadside distance, decreasing exponentially with the distance [
27]. The influential scope of traffic pollution in roadside soil can be up to 100 m from the road edge [
28], and most of the deposited metal particles remain in the 0–5 cm of the roadside topsoil [
24]. Through absorption from roadside soils, the heavy-metal elements are further transferred into the roots, stem, and leaves of grasses. Therefore, the factors influencing the soil’s heavy-metal content have nearly the same effect on the heavy-metal concentrations in roadside grass.
In order to investigate the relationship between the heavy-metal content of soils and corresponding grasses, the Transfer Factor (TF), defined as the ratio of heavy-metal concentration in grass to that in soil, has been applied to assess plants’ capability to absorb heavy metals from the soil [
18,
19,
20,
21,
22,
23,
24,
25,
26,
27,
28,
29]. TF is also called bio-concentration factor (BCF), calculated as the ratio of element concentration in plant tissues to the element concentration in the soil [
30]. It should be noticed that only when a linear relationship is observed between the concentrations of grass and soil for a given element, TF can be an appropriate measure for the assessment [
31]. It was found that soil properties, grass absorption ability, and both the form and concentration of heavy metals in the soil comprehensively impact the uptake capability of heavy metals from the soil by the corresponding local grass [
29,
31]. It was observed that Zn has the highest TF value, followed by Cu and Cd, and Pb has the lowest TF value. Even when the samples are mixed with different types of grasses, a similar pattern can still be observed [
29]. However, few studies were focused on the roadside farmland environments to explore the relationship between the heavy-metal contaminations in soil and corresponding grass resulting from traffic activities.
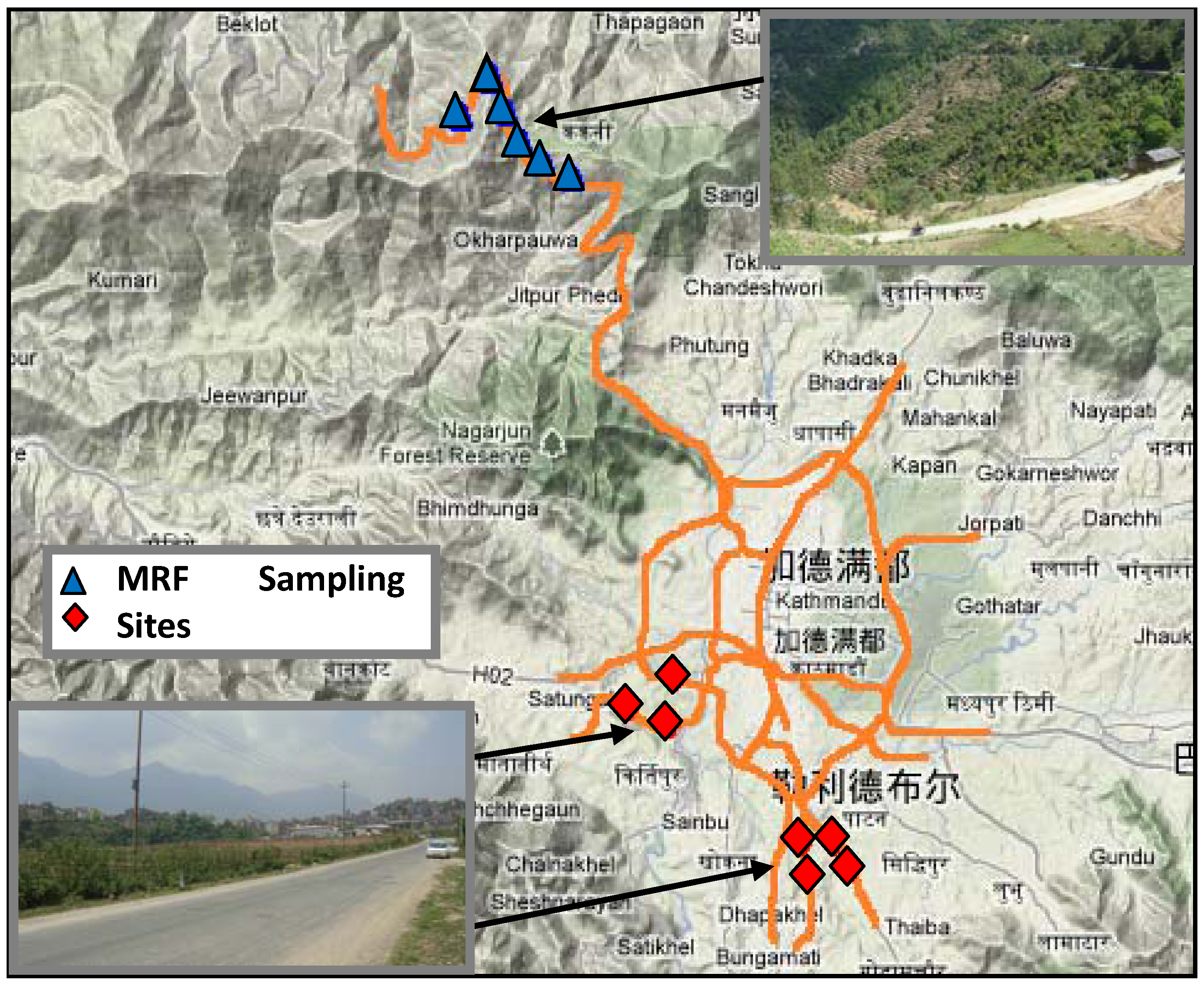
Previous environmental studies in the Kathmandu area have indicated that the air pollutants and heavy metal contaminants in tree leaves are substantially due to the city’s traffic emissions [
32,
33]. The authors of this paper have conducted a study to investigate the influence of transportation activities on farmland soils along a highway across mountainous areas around Kathmandu in Nepal [
34]. It was found that concentration patterns of the heavy metals in soil proved their homology with the traffic pollution source. Furthermore, some spots with peak concentrations may be severely polluted although average accumulations of heavy metals pose no hazard in the region. To ensure food safety and ecosystem security, the forms and behavior of heavy metals in farmland need to be dynamically monitored and assessed [
35]. The heavy-metal transport process in roadside grasses is the same as that in crops. Therefore, this research aims at characterizing the effect of location, roadside distance, and tree protection on heavy-metal distributions in roadside farmland soils and corresponding grasses, using the MANOVA experimental design method. Especially, the objective of this study is to explore the relationship between heavy-metal concentrations in the roadside soils on the heavy-metal uptake capabilities of the corresponding local grasses.
4. Conclusions
This study conducted a MANOVA experiment to investigate the heavy-metal concentrations in roadside soils and corresponding grasses in roadside farmland around Kathmandu, Nepal. The sampling location, roadside distance, tree protection, and the type of samples are selected as the tested factors.
Based on the correlation analyses of heavy metals in both grass and soil samples, it was found that more significant correlations among the heavy metals can be identified in soil samples than those in grass samples. This is because the uptake capabilities from soil to grass for different heavy metals are significantly different.
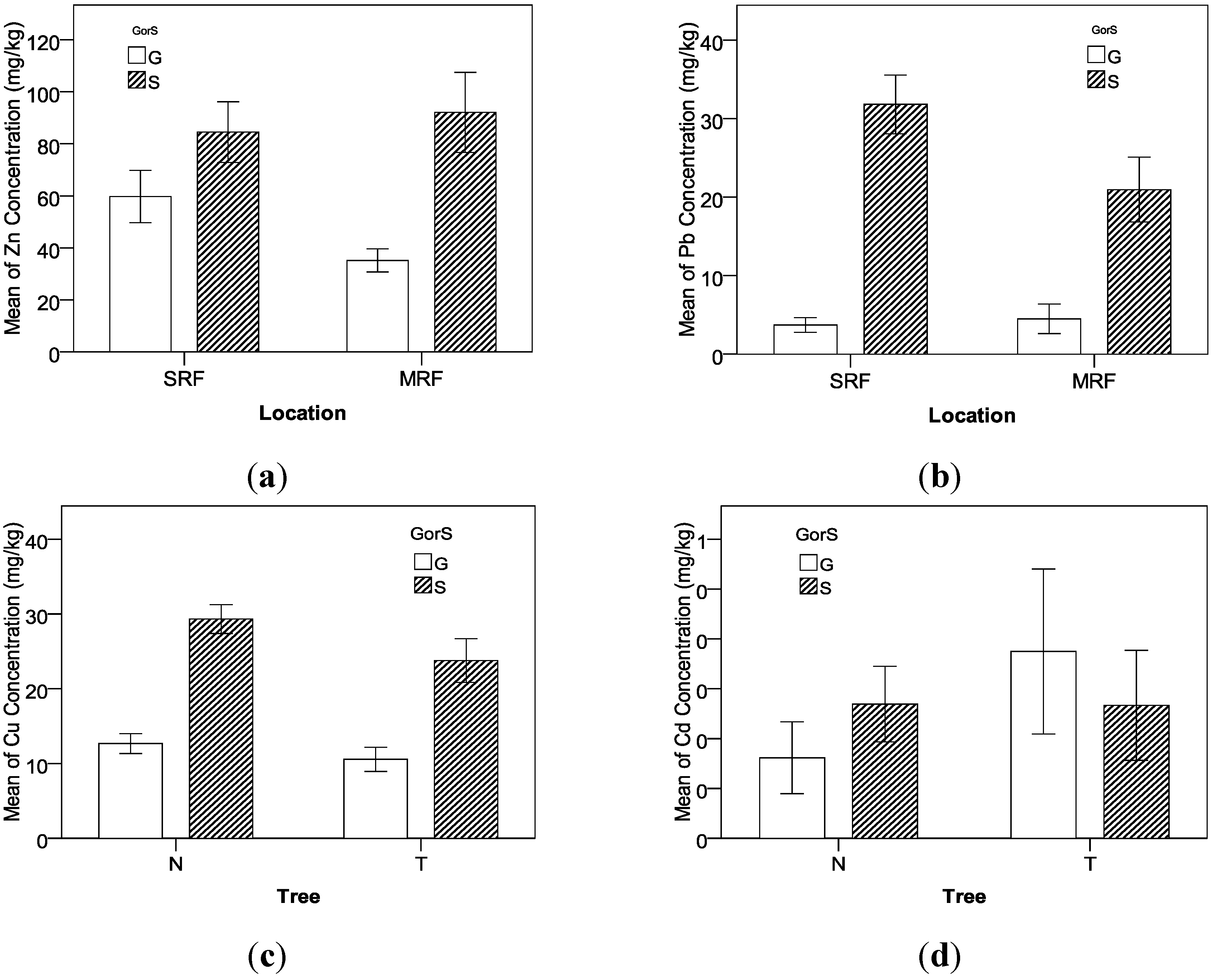
The MANOVA results indicate that the concentrations of Cu, Zn, and Pb in the soil samples are significantly higher than those in the grass samples. The TF order indicating uptake capability from soil to grass is Zn > Cu > Pb. Because the order is the same as the previous studies for the unique plants, it implies that the TF order of Cu, Zn, and Pb would be applicable for different types of grasses. It was also found that the concentrations of Cu and Pb in the suburban roadside farmland are higher than those in the rural mountainous roadside farmland because of the difference in traffic volume between the two types of sampling locations. Additionally, the factor of Tree influences the concentrations of Cu and Zn. The analysis indicates that the trees growing linearly along the roadways can effectively reduce the heavy metals’ concentrations in the roadside farmland. Therefore, planting trees may be considered as an effective remedy countermeasure for the existing crop plots that are very close to roadways.
The most interesting finding in this study is that the transfer factor influencing the capability that grass absorbs heavy metal from soil is related to the soil’s heavy-metal concentrations. There is a monotone decreasing relationship between transfer factor and heavy-metal concentrations in soil: as the soil’s heavy-metal concentrations increase, the grass’s capability of heavy-metal absorption decreases. The exponential regression form is the best model that can characterize this relationship statistically. The finding implies that the grass’s self-protection mechanism would mitigate grasses’ uptake of heavy metals from soil at high concentration sites.
The results of this study would be useful for understanding how the heavy-metal accumulations in farmland roadside soils and grasses owning to traffic activities are influenced by roadside attributes, which is helpful in making policies for avoiding heavy-metal contaminants in agricultural areas. Furthermore, the relationship between transfer factor and the ratio of heavy-metal concentration in grass to that in soil deserves a further lab experiment study under well-controlled settings to accurately describe the influence mechanism of heavy-metal concentration in the roadside soil on the heavy-metal uptake capabilities of the corresponding local grasses.