Oxidative Response and Antioxidative Mechanism in Germinating Soybean Seeds Exposed to Cadmium
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
2.1.1. Cd Concentration in Germinating Seeds
| Cd concentration (mg/L) | Exposure time (h) | |||
|---|---|---|---|---|
| 24 | 48 | 72 | 96 | |
| 0 | 1.67 ± 0.25a | 1.67 ± 0.25a | 1.67 ± 0.25a | 1.67 ± 0.25a |
| 50 | 6.88 ± 0.64b | 8.52 ± 3.06ab | 11.94 ± 2.07a | 26.28 ± 11.4b |
| 100 | 7.64 ± 1.36bc | 16.58 ± 6.22b | 40.56 ± 13.2b | 46.46 ± 7.05c |
| 200 | 11.9 ± 2.31c | 31.86 ± 5.82c | 72.19 ± 13.9c | 33.94 ± 2.76bc |
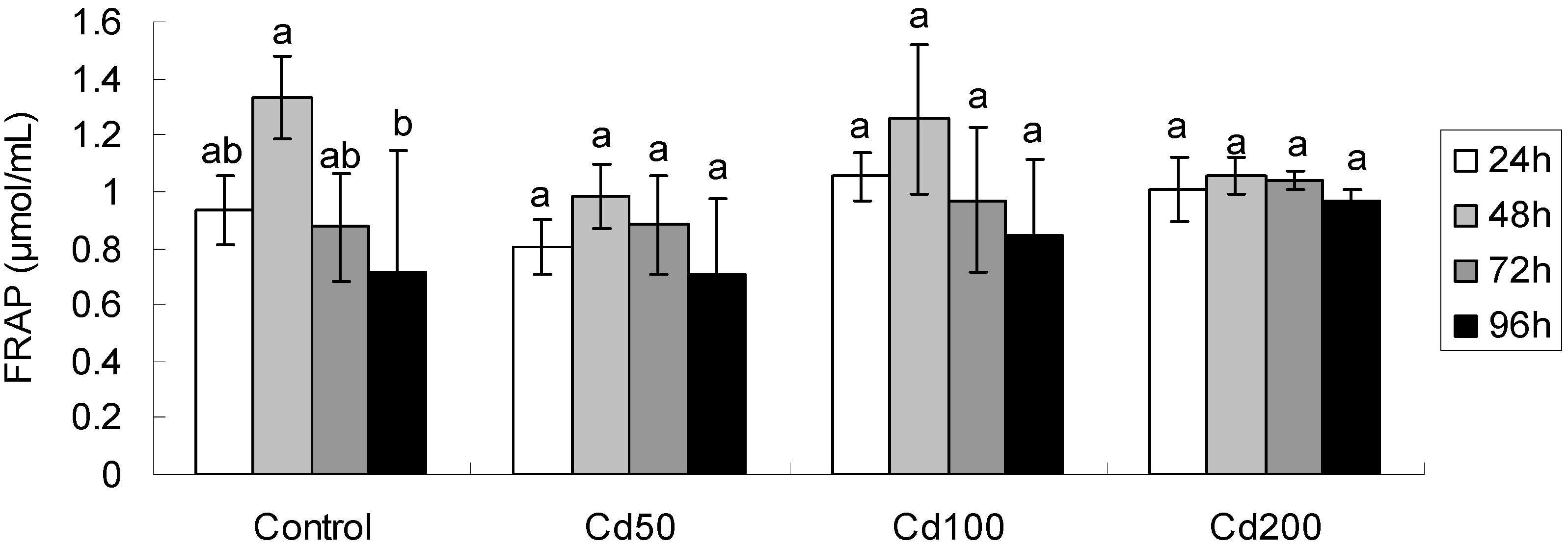
2.1.2. Effects of Cd on Total Antioxidant Capacity
| Effect | FRAP | MDA | GSH | Proline | GST |
|---|---|---|---|---|---|
| Time | F3,48 = 9.15 ** | F3,48 = 52.55 ** | F3,48 = 59.67 ** | F3,48 = 113.8 ** | F3,48 = 4.40 ** |
| Conc | F3,48 = 2.51 | F3,48 = 17.72 ** | F3,48 = 2.02 | F3,48 = 9.31 ** | F3,48 = 3.43 * |
| Time × Conc | F9,48 = 1.75 | F9,48 = 10.39 ** | F9,48 = 1.83 | F9,48 = 2.57 * | F9,48 = 1.69 |
2.1.3. Effects of Cd on MDA Content

2.1.4. Effects of Cd on GSH/hGSH Concentration

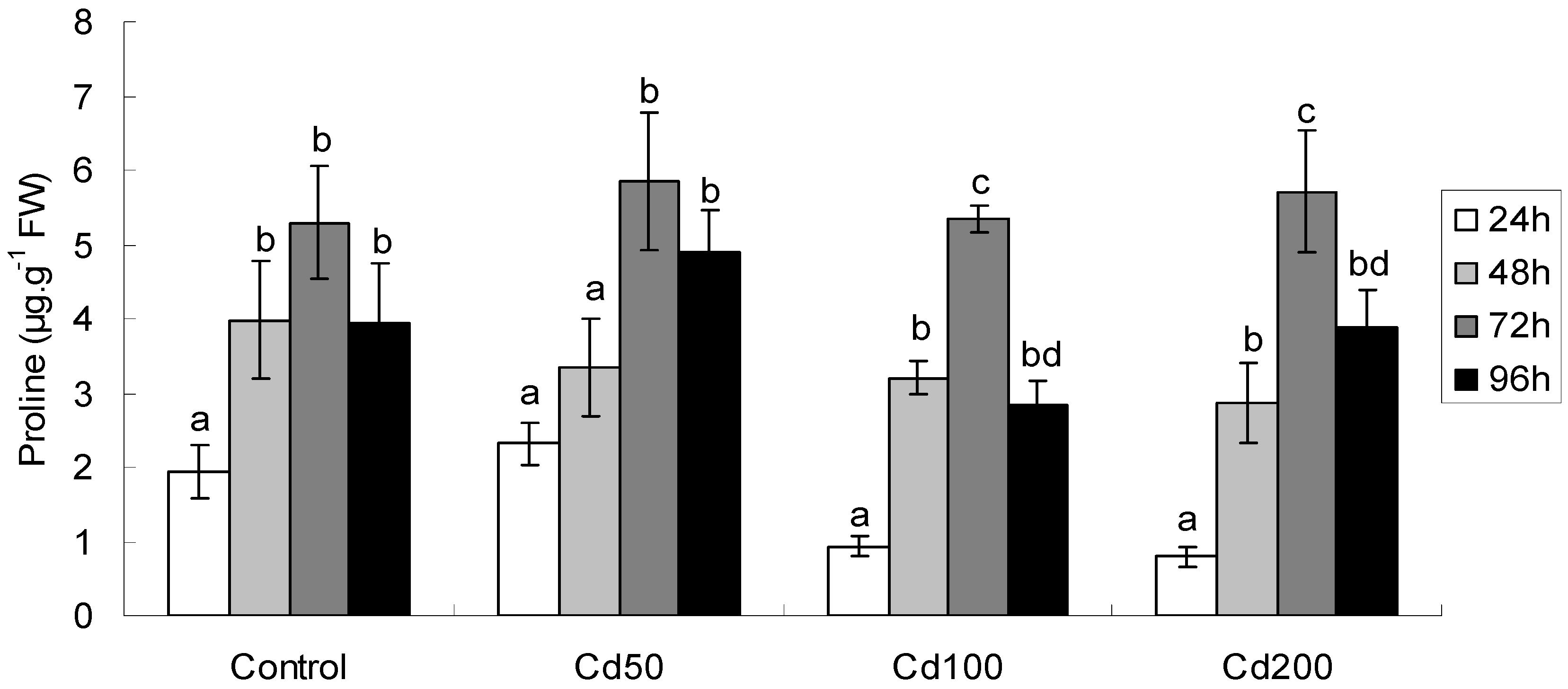
2.1.5. Effects of Cd on Proline Content
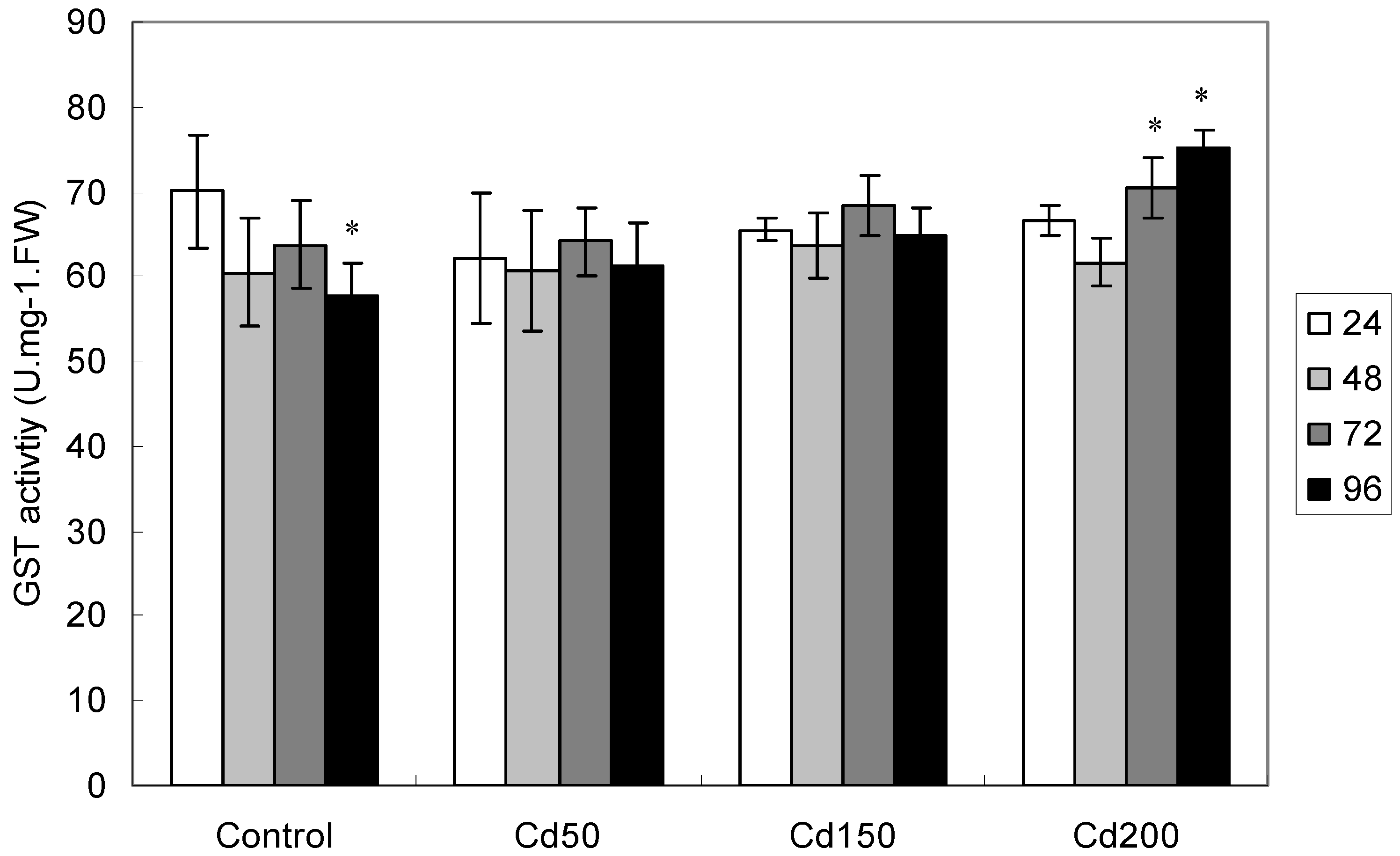
2.1.6. Effects of Cd on GST Activity
2.1.7. Effects of Dosage, Exposure Time and Dosage × Exposure Interaction
2.2. Discussion
3. Experimental Section
3.1. Seed Germination and Cadmium Exposure
3.2. Cd Content in Seeds
3.3. Samples Preparation
3.4. Determination of Total Antioxidant Capacity
3.5. Determination of Lipid Peroxidation
3.6. Glutathione/Homoglutathione (GSH/hGSH)
3.7. Proline Content
3.8. Determination of GST Activity
3.9. Statistic Analysis
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Moreno, J.L.; Hernandez, T.; Garcia, C. Effects of a cadmium-contaminated sewage sludge compost on dynamics of organic matter and microbial activity in an arid soil. Biol. Fertil. Soils 2009, 28, 230–237. [Google Scholar]
- Qadir, A.M.; Ghafoor, G.; Murtaza, G. Cadmium concentration in vegetables grown on urban soils irrigated with untreated municipal sewage. Environ. Dev. Sustain. 2000, 2, 11–19. [Google Scholar]
- Cho, U.H.; Seo, N.H. Oxidative stress in Arabidopsis thaliana exposed to cadmium is due to hydrogen peroxide accumulation. Plant Sci. 2005, 168, 113–120. [Google Scholar] [CrossRef]
- Aina, R.; Labra, M.; Fumagalli, P.; Vannini, C.; Marsoni, M.; Cucchi, U.; Bracale, M.; Sgorbati, S.; Cittrio, S. Thiol-peptide level and proteomic changes in response to cadmium toxicity in Oryza sativa L. roots. Environ. Exp. Bot. 2007, 59, 381–392. [Google Scholar] [CrossRef]
- Rahoui, S.; Chaoui, A.; Ferjani, E.E. Membrane damage and solute leakage from germinating pea seed under cadmium stress. J. Hazard. Mater. 2010, 178, 1128–1131. [Google Scholar] [CrossRef]
- Smiri, M.; Chaoui, A.; Rouhier, N.; Celhaye, E.; Jacquot, J.P.; Ferjani, E.I. Oxidative damage and redox change in pea seeds treated with cadmium. C. R. Biol. 2010, 333, 801–807. [Google Scholar] [CrossRef]
- Smiri, M.; Chaoui, A.; Rouhier, N.; Gelhaye, E.; Jacquot, J.P.; Ferjani, E.E. Cadmium affects the glutathione/glutaredoxin system in germinating pea seeds. Bio. Trace Elem. Res. 2011, 142, 93–105. [Google Scholar] [CrossRef]
- Srivastava, R.; Khan, R.; Manzoor, N.; Mahmooduzzafar. Effects of cadmium exposures on growth, physio-biochemical characteristics and the antioxidative defence system of soybean (Glycine max L.). J. Phycol. 2011, 3, 20–25. [Google Scholar]
- Balestrasse, K.B.; Gallego, S.M.; Tomaro, M.L. Cadmium-induced senescence in nodules of soybean (Glycine max L.) plants. Plant Soil 2004, 262, 373–381. [Google Scholar] [CrossRef]
- Noriega, G.O.; Balestrasse, K.B.; Batlle, A.; Tomaro, M.L. Heme oxygenase exterts a protective role against oxidative stress in soybean leaves. Biochem. Biophys. Res. Commun. 2004, 323, 1003–1008. [Google Scholar] [CrossRef]
- Liu, T.T.; Wu, P.; Wang, L.H.; Zhou, Q. Response of soybean seed germination to cadmium and acid rain. Biol. Trace Elem. Res. 2011, 144, 1186–1196. [Google Scholar] [CrossRef]
- Ghiselli, A.; Serafini, M.; Natella, F.; Saccini, C. Total antioxicant capacity as a tool to assess redox status: Critical view and experiment data. Free Radical. Biol. Med. 2000, 29, 1106–1114. [Google Scholar] [CrossRef]
- Schützendübel, A.; Polle, A. Plant responses to abiotic stresses: Heavy metal-induced oxidative stress and protection by mycorrhization. J. Exp. Bot. 2002, 53, 1351–1365. [Google Scholar] [CrossRef]
- Tan, S.H.; He, D.Y.; Yan, F.; Fang, L. Effects of NaNO2 on malondialdehyde content and total antioxidative capacity in the liver of Carassius auratus. J. Agro. Environ. Sci. 2005, 24, 21–24. [Google Scholar]
- Szőllősi, R.; Varga, I.Z.; Erdei, L.; Mihalik, E. Cadmium-induced oxidative stress and antioxidative mechanisms in germinating Indian mustard (Brassica juncea L.) seeds. Ecotoxicol. Environ. Saf. 2009, 72, 1337–1342. [Google Scholar] [CrossRef]
- Mendoza-Cόzatl, D.G.; Moreno-Sánchez, R. Control of glutathione and phytochelatin synthesis under cadmium stress. Pathway modeling for plants. J. Theor. Biol. 2006, 238, 919–936. [Google Scholar] [CrossRef]
- Rüegsegger, A.; Schmutz, D.; Brunold, C. Regulationof glutathione synthesis by cadmium in Pisum sativum L. Plant Physiol. 1990, 93, 1579–1584. [Google Scholar]
- Szabados, L.; Savouré, A. Proline: A multifunctional amino acid. Trends Plant Sci. 2009, 15, 89–97. [Google Scholar] [CrossRef]
- Theriappan, P.; Gupta, A.K.; Dhasarathan, P. Accumulation of proline under salinity and heavy metal stress in Cauliflower seedlings. J. Appl. Sci. Environ. Manag. 2011, 15, 251–255. [Google Scholar]
- Seregin, I.V.; Ivanov, V.B. Physiological aspects of cadmium and lead toxic effects in higher plants. Russ. J. Plant Phys. 2001, 48, 423–544. [Google Scholar]
- Dong, J.; Wu, F.; Zhang, C. Influence of cadmium on antioxidant capacity and four microelement concentrations in tomato seedlings (Lycopersicon esculentum). Chemosphere 2006, 64, 1659–1666. [Google Scholar] [CrossRef]
- Sun, R.L.; Zhou, Q.X.; Sun, F.H.; Jin, C.X. Antioxidative defense and proline/phytocheletin accumulation in a newly discovered Cd-hyperaccumulator, Solanum nigrum L. Environ. Exp. Bot. 2007, 60, 468–476. [Google Scholar] [CrossRef]
- Noctor, G.; Mhamdi, A.; Chaouch, S.; Han, Y.; Neukermans, J.; Marquez-Garcia, B.; Queval, G.; Foyer, C.H. Glutathion in plant: An integrated overview. Plant Cell Environ. 2012, 35, 454–484. [Google Scholar] [CrossRef]
- Gallego, S.M.; Benavides, M.P.; Tomaro, M.L. Effect of cadmium ions on antioxidant defense system in sunflower cotyledons. Biol. Plant 1999, 42, 49–55. [Google Scholar]
- Islam, M.M.; Hoque, M.A.; Okuma, E.; Banu, M.N.K.; Shimoishi, Y.; Nakamura, Y.; Murata, Y. Exogenous proline and glycinebetaine increase antioxidant enzyme activities and confer tolerance to cadmium stress in cultured tobacco cells. J. Plant Physiol. 2009, 166, 1587–159. [Google Scholar] [CrossRef]
- Sharma, S.S.; Schat, H.; Vooijs, R. In vitro alleviation of heavy metal induced enzyme inhibition by proline. Phytochemistry 1998, 46, 1531–1535. [Google Scholar]
- Matysik, J.; Alia Bhalu, B.; Mohanty, P. Molecular mechnanism of quenching of reactive oxygen species by proline under stress in plants. Curr. Sci. 2002, 82, 525–532. [Google Scholar]
- Siripornadulsil, S.; Traina, S.; Verma, D.P.S.; Sayre, R.T. Molecular mechanisms of proline-mediated tolerance to toxic heavy metals in transgenic microalgae. Plant Cell 2000, 14, 2837–2847. [Google Scholar]
- Edwards, R.; Dixon, D.P.; Walbot, V. Plant glutathione S-transferases: Enzymes with multiple functions in sickness and in health. Trends Plant Sci. 2000, 5, 193–198. [Google Scholar] [CrossRef]
- Cummins, I.; Cole, D.J.; Ewards, R. A role for glutathione transferases functioning as glutathione peroxidases in resistance to multiple herbicides in black-grass. Plant J. 1999, 18, 285–292. [Google Scholar] [CrossRef]
- Dixit, V.; Pandey, V.; Shyam, R. Differential antioxidative responses to cadmium in roots and leaves of pea (Pisum sativum L. cv. Azad). J. Exp. Bot. 2001, 52, 1101–1109. [Google Scholar] [CrossRef]
- Marrs, K.A. The function and regulation of glutathione-S-transferase in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1996, 47, 127–158. [Google Scholar] [CrossRef]
- Iannelli, M.A.; Pietrini, F.; Fiore, L.; Petrilli, L.; Massacci, A. Antioxidant response to cadmium in Phragmites australis plants. Plant Physiol. Biochem. 2002, 40, 977–982. [Google Scholar] [CrossRef]
- Benzie, F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar]
- Ellman, G.L. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 1959, 82, 70–77. [Google Scholar] [CrossRef]
- Ringel, C.; Siebert, S.; Wienhaus, O. Photometric determination of proline in quartz microplats: Remarks on specificity. Anal. Biochem. 2003, 313, 167–169. [Google Scholar]
- Cui, J.; Zhang, R.; Wu, G.L.; Zhu, H.M.; Yang, H. Salicylic acid reduces napropamide toxicity by preventing its accumulation in rapeseed (Brassica napus L.). Biohazard. Mate 2010, 59, 100–108. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yang, S.; Xie, J.; Li, Q. Oxidative Response and Antioxidative Mechanism in Germinating Soybean Seeds Exposed to Cadmium. Int. J. Environ. Res. Public Health 2012, 9, 2827-2838. https://doi.org/10.3390/ijerph9082827
Yang S, Xie J, Li Q. Oxidative Response and Antioxidative Mechanism in Germinating Soybean Seeds Exposed to Cadmium. International Journal of Environmental Research and Public Health. 2012; 9(8):2827-2838. https://doi.org/10.3390/ijerph9082827
Chicago/Turabian StyleYang, Shiyong, Jianchun Xie, and Quanfa Li. 2012. "Oxidative Response and Antioxidative Mechanism in Germinating Soybean Seeds Exposed to Cadmium" International Journal of Environmental Research and Public Health 9, no. 8: 2827-2838. https://doi.org/10.3390/ijerph9082827




