Ecophysiological and Climatological Effects on Distribution of Vector Species and Malaria Incidence in India
Abstract
:1. Introduction
2. Methods and Data
2.1. Outline of the Coupled Model for Ecophysiological and Climatological Distribution of Mosquito Generation (ECD-mg)

2.2. Epidemiological Data, and Geographical Characteristics of Analysis Sites
3. Results and Discussion
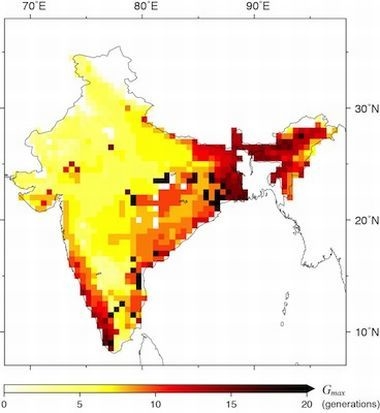
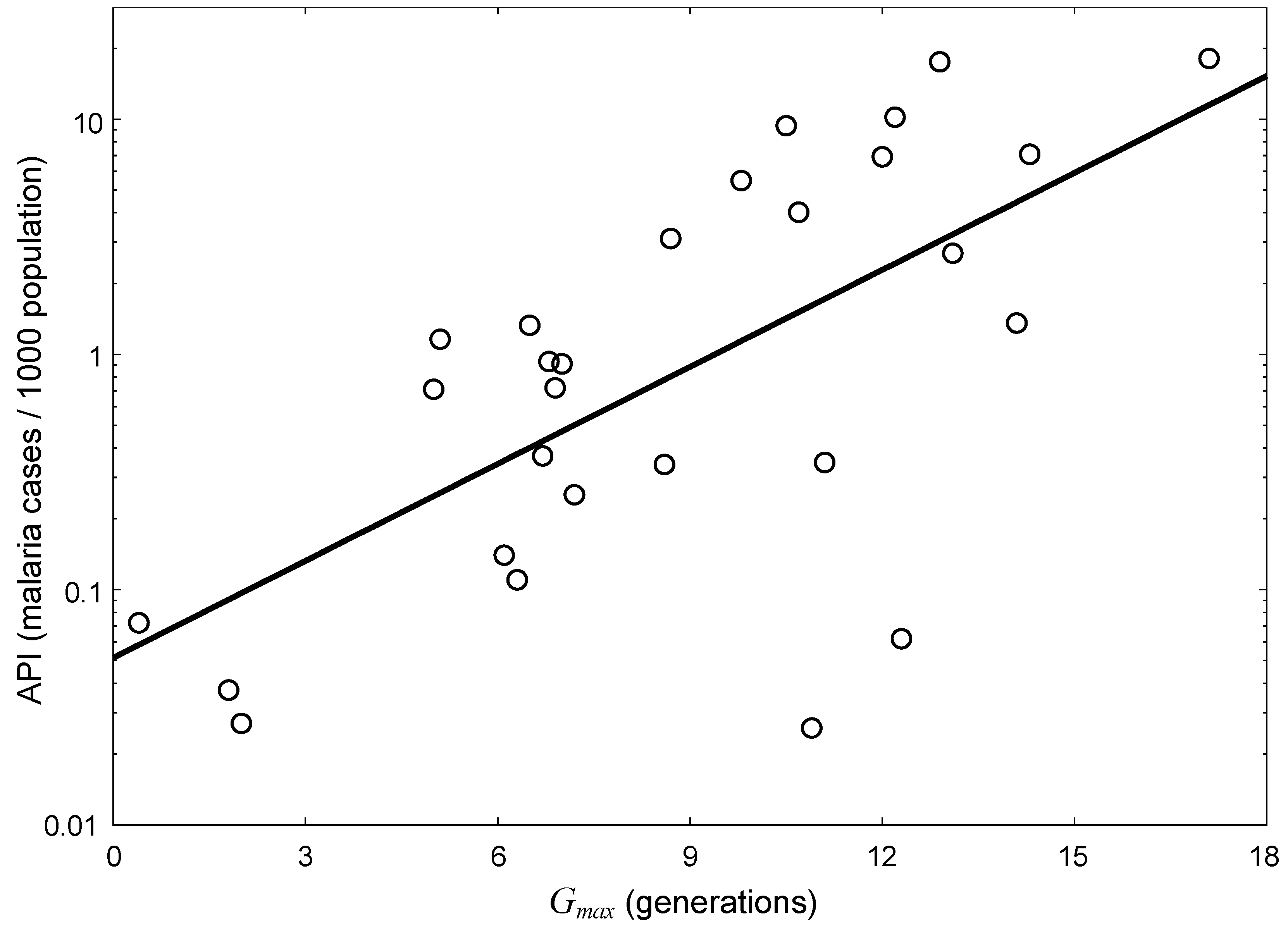
3.1. Spatial Relationship between Malaria Incidences and Potential Distribution of Vector Species

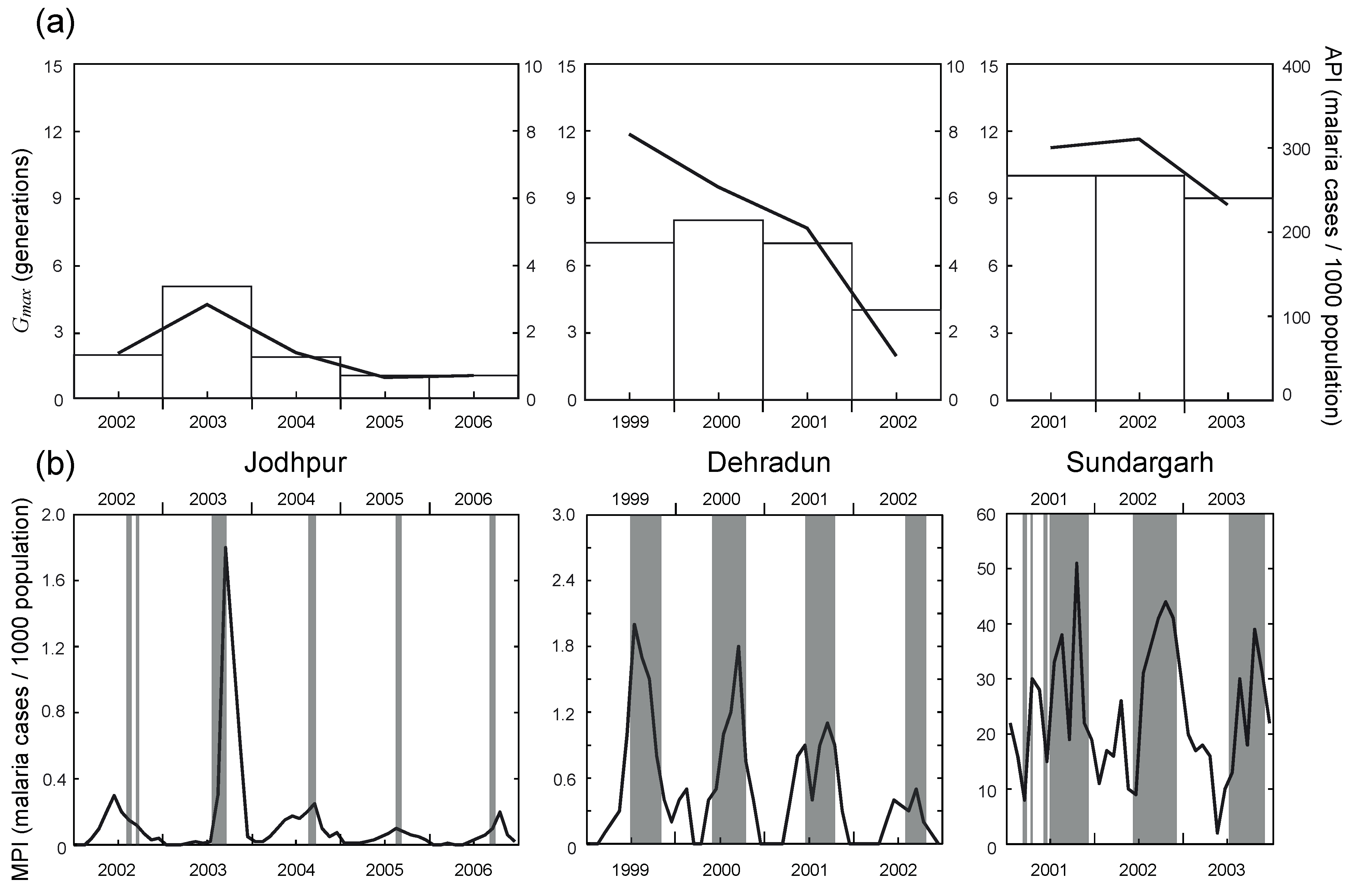
3.2. Temporal Relationship between Malaria Incidences and Potential Distribution of Vector Species
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Lal, S.; Sonal, G.S.; Phukan, P.K. Status of malaria in India. J. Indian Acad. Clin. Med. 2000, 5, 19–23. [Google Scholar]
- Kumar, A.; Valecha, N.; Jain, T.; Dash, A.P. Burden of malaria in India: Retrospective and prospective view. Am. J. Trop. Med. Hyg. 2007, 77, 69–78. [Google Scholar]
- World Health Organization (WHO), World Malaria Report 2010; World Health Organization: World Health Organization, 2010.
- National Vector Borne Disease Control Programme (NVBDCP). Available online: nvbdcp.gov.in/ (accessed on 8 July 2012).
- Devi, N.P.; Jauhari, R.K. Relationship between Anopheles fluviatilis & A. stephensi (Diptera: Culicidae) catches & the prevalence of malaria cases at Kalsi area in Dehradun district (Uttaranchal). Indian J. Med. Res. 2006, 123, 151–158. [Google Scholar]
- Devi, N.P.; Jauhari, R.K. Climatic variables and malaria incidence in Dehradun, Uttaranchal, India. J. Vector Borne Dis. 2006, 43, 21–28. [Google Scholar]
- Sharma, S.K.; Tyagi, P.K.; Padhan, K; Upadhyay, A.K.; Haque, M.A.; Nanda, N.; Joshi, H.; Biswas, S.; Adak, T.; Das, B.S.; et al. Epidemiology of malaria transmission in forest and plain ecotype villages in Sundargarh District, Orissa, India. Trans. R. Soc. Trop. Med. Hyg. 2006, 10, 917–925. [Google Scholar]
- Martens, W.J.M.; Niessen, L.W.; Rotmans, J.; Jetten, T.H.; McMichael, A.J. Potential impact of global climate change on malaria risk. Environ. Health Perspect. 1995, 103, 458–464. [Google Scholar]
- Martin, P.H.; Lefebvre, M.G. Malaria and climate: Sensitivity of malaria potential transmission to climate. Ambio 1995, 24, 200–207. [Google Scholar]
- Craig, M.H.; Snow, R.W.; le Sueur, D. A climate-based distribution model of malaria transmission in sub-Saharan Africa. Parasitol. Today 1999, 15, 105–111. [Google Scholar] [CrossRef]
- Githeko, A.K.; Lindsay, S.W.; Confalonieri, U.E.; Patz, J.A. Climate change and vector-borne diseases: A regional analysis. Bull. World Health Organ. 2000, 78, 1136–1147. [Google Scholar]
- Patz, J.A.; Lindsay, S.W. New challenges, new tools: The impact of climate change on infectious diseases. Curr. Opin. Microbiol. 1999, 2, 445–451. [Google Scholar] [CrossRef]
- Reid, C. Implications of Climate Change on Malaria in Karnataka, India. Available online: www.brown.edu/ Research/EnvStudies_Theses/full9900/creid/ (accessed on 22 June 2012).
- Bhattacharya, S.; Sharma, C.; Dhiman, R.C.; Mitra, A.P. Climate change and malaria in India. Curr. Sci. 2006, 90, 369–375. [Google Scholar]
- Sinka, M.E.; Bangs, M.J.; Manguin, S.; Chareonviriyaphap, T., Patil; Temperley, W.H.; Gething, P.W.; Elyazar, I.R.; Kabaria, C.W.; Harbach, R.E.; et al. The dominant Anopheles vectors of human malaria in the Asia-Pacific region: Occurrence data, distribution maps and bionomic précis. Parasit. Vectors 2011. [Google Scholar] [CrossRef]
- Ohta, S.; Kaga, T. Effect of climate on malarial vector distribution in Monsoon Asia: Coupled model for Ecophysiological and Climatological Distribution of mosquito generations (ECD-mg). Clim. Res. 2012, 53, 77–88. [Google Scholar] [CrossRef]
- Patz, J.A.; Strzepek, K.; Lele, S.; Hedden, M.; Greene, S.; Noden, B.; Hay, S.I.; Kalkstein, L.; Beier, J.C. Predicting key malaria transmission factors, biting and entomological inoculation rates, using modeled soil moisture in Kenya. Trop. Med. Int. Health 1998, 3, 818–827. [Google Scholar] [CrossRef]
- Kearney, M.; Porter, W.P.; Williams, C.; Ritchie, S.; Hoffmann, A.A. Integrating biophysical models and evolutionary theory to predict climatic impacts on species’ ranges: The dengue mosquito Aedes aegypti in Australia. Funct. Ecol. 2009, 23, 528–538. [Google Scholar] [CrossRef]
- Sharma, V.P. Fighting malaria in India. Curr. Sci. 1998, 75, 1127–1140. [Google Scholar]
- National Oceanic and Atmospheric Administration (NOAA). NNDC Climate Data Online. Available online: www.ncdc.noaa.gov/oa/ncdc.html (accessed on 2 June 2012).
- New, M.; Hulme, M.; Jones, P. Representing twentieth-century space-time climate variability. Part 1: Development of a 1961–90 mean monthly terrestrial climatology. J. Clim. 1999, 12, 829–856. [Google Scholar]
- Dunne, K.A.; Willmott, C.J. Global distribution of plant-extractable water capacity of soil. Int. J. Climatol. 1996, 16, 841–859. [Google Scholar] [CrossRef]
- Office of the Registrar General and Census of Commissionar, India. Census of India. Available online: censusindia.gov.in (accessed on 9 July 2012).
- Anand, P.K.; Swarn, L.; Yadav, S.P.; Singh, H. Disease dynamics, distribution and surveillance of malaria in arid ecology of Jodhpur, Rajasthan, India during 2002 to 2006. J. Public Health Epidemiol. 2011, 3, 301–307. [Google Scholar]
- Tyagi, B.K.; Yadav, S.P. Bionomics of malaria vectors in two physiographically different areas of the epidemic-prone Thar Desert, north-western Rajasthan (India). J. Arid Environ. 2001, 47, 161–172. [Google Scholar] [CrossRef]
- Lehmann, T.; Hawley, W.A.; Grebert, H.; Collins, F.H. The effective population size of Anopheles gambiae in Kenya: Implications for population structure. Mol. Biol. Evol. 1998, 15, 264–276. [Google Scholar] [CrossRef]
- Yamamura, K.; Kiritani, K. A simple method to estimate the potential increase in the number of generations under global warming in temperate zones. Appl. Entomol. Zool. 1998, 33, 289–298. [Google Scholar]
- World Health Organization (WHO). Malaria Country Profile India (1995–2007). Available online: www.whoindia.org/LinkFiles/Malaria_Country_Profile-Malaria.pdf (accessed on 29 May 2010).
- Sharma, S.K.; Pradhan, P.; Padhi, D.M. Socio-economic factors associated with malaria in a tribal area of Orissa, India. Indian J. Public Health 2001, 45, 93–98. [Google Scholar]
- Sandeep, K. Control, eradication and resurgence of malaria in Kerala during the past 50 years. Kerala Med. J. 2008, 3, 5–9. [Google Scholar]
- Government of India. Bihar State Report. Available online: mohfw.nic.in/NRHM/ Documents/ High_Focus_Reports/Bihar_report.pdf (accessed on 3 July 2012).
- Bhatt, R.M.; Sharma, R.C.; Gautam, A.S.; Gupta, D.K. Seasonal prevalence of anophelines in Kheda district, Gujarat. Indian J. Malariol. 1991, 28, 9–18. [Google Scholar]
- Konradsen, F.; Stobberup, K.A.; Sharma, S.K.; Gulati, O.T.; van der Hoek, W. Irrigation water releases and Anopheles culicifacies abundance in Gujarat, India. Acta. Trop. 1998, 71, 195–197. [Google Scholar] [CrossRef]
- Baeza, A.; Bouma, M.J.; Dobson, A.P.; Dhiman, R.; Srivastava, H.C.; Pascual, M. Climate forcing and desert malaria: The effect of irrigation. Malar. J. 2011. [Google Scholar] [CrossRef]
- Ohta, S.; Kaga, T. Effect of irrigation systems on temporal distribution of malaria vectors in semi-arid regions. Int. J. Biometeorol. 2012. submitted for publication. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kaga, T.; Ohta, S. Ecophysiological and Climatological Effects on Distribution of Vector Species and Malaria Incidence in India. Int. J. Environ. Res. Public Health 2012, 9, 4704-4714. https://doi.org/10.3390/ijerph9124704
Kaga T, Ohta S. Ecophysiological and Climatological Effects on Distribution of Vector Species and Malaria Incidence in India. International Journal of Environmental Research and Public Health. 2012; 9(12):4704-4714. https://doi.org/10.3390/ijerph9124704
Chicago/Turabian StyleKaga, Takumi, and Shunji Ohta. 2012. "Ecophysiological and Climatological Effects on Distribution of Vector Species and Malaria Incidence in India" International Journal of Environmental Research and Public Health 9, no. 12: 4704-4714. https://doi.org/10.3390/ijerph9124704