Prevalence and Risk Factors for Poor Nutritional Status among Children in the Kilimanjaro Region of Tanzania
Abstract
:1. Introduction
2. Methods
2.1. Study Sites
2.2. Sampling Procedures
2.3. Measures
2.3.1. Anthropometry
2.3.2. Hb Levels
2.3.3. Social, Economic and Demographic Indicators
| Background variables ** | Number (Percentage) | |
|---|---|---|
| Maternal Educational Level | ||
| No schooling | 11 (2.6) | |
| Primary not completed | 25 (5.9) | |
| Primary completed | 352 (83.2) | |
| Secondary and above | 35 (8.3) | |
| Paternal Educational Level | ||
| No schooling | 6 (1.5) | |
| Primary not completed | 20 (4.9) | |
| Primary completed | 334 (81.3) | |
| Secondary and above | 51 (12.4) | |
| Maternal Occupational level | ||
| Skilled | 17 (4.1) | |
| Unskilled | 389 (92.1) | |
| No occupation | 9 (2.1) | |
| Student | 7 (1.7) | |
| Paternal Occupational level | ||
| Skilled | 47 (11.0) | |
| Unskilled | 365 (85.7) | |
| No occupation | 1 (0.2) | |
| Student | 5 (1.2) | |
| Marital Status | ||
| Married | 242(57.5) | |
| Single | 67 (15.9) | |
| Divorced | 10 (2.4) | |
| Widowed | 3 (0.7) | |
| Cohabiting | 99 (23.5) | |
| Total number of maternal pregnancies | ||
| 1 | 80 (19.2) | |
| 2 | 98 (23.5) | |
| 3 | 75 (18.0) | |
| 4 | 52 (12.5) | |
| 5 | 41 (9.8) | |
| 6 and more | 71 (17.0) | |
| Is the child breastfeeding | ||
| Yes | 332 (81.9) | |
| No | 77 (18.4) | |
| House Ownership | ||
| Yes | 338 (83.9) | |
| No | 65 (16.1) | |
| Material used to build house | ||
| Mud | 117(28.4) | |
| All other materials | 295 (71.6) | |
| Distance to water source | ||
| More than 15 minutes | 141(34.9) | |
| Less than 15 minutes | 263 (65.1) | |
| Characteristic | (N = 423) |
|---|---|
| Mean Age in months (SD) | 13.04 (7.78) |
| Boys (%) | 209 (49.4%) |
| Incomplete immunization (%) | 4 (0.9%) |
| Low birth weight ** | 16 (3.8%) |
| Concerns about child growth and development (%) | 65 (15.4%) |
| Mother alive | 422 (100%) |
| Father alive | 420 (98.6%) |
| Mean Maternal age (SD) | 28.67 (6.92) |
| Mean Paternal age (SD) | 34.36 (8.11) |
2.4. Data Management and Statistical Analysis
2.5. Ethical Considerations
3. Results
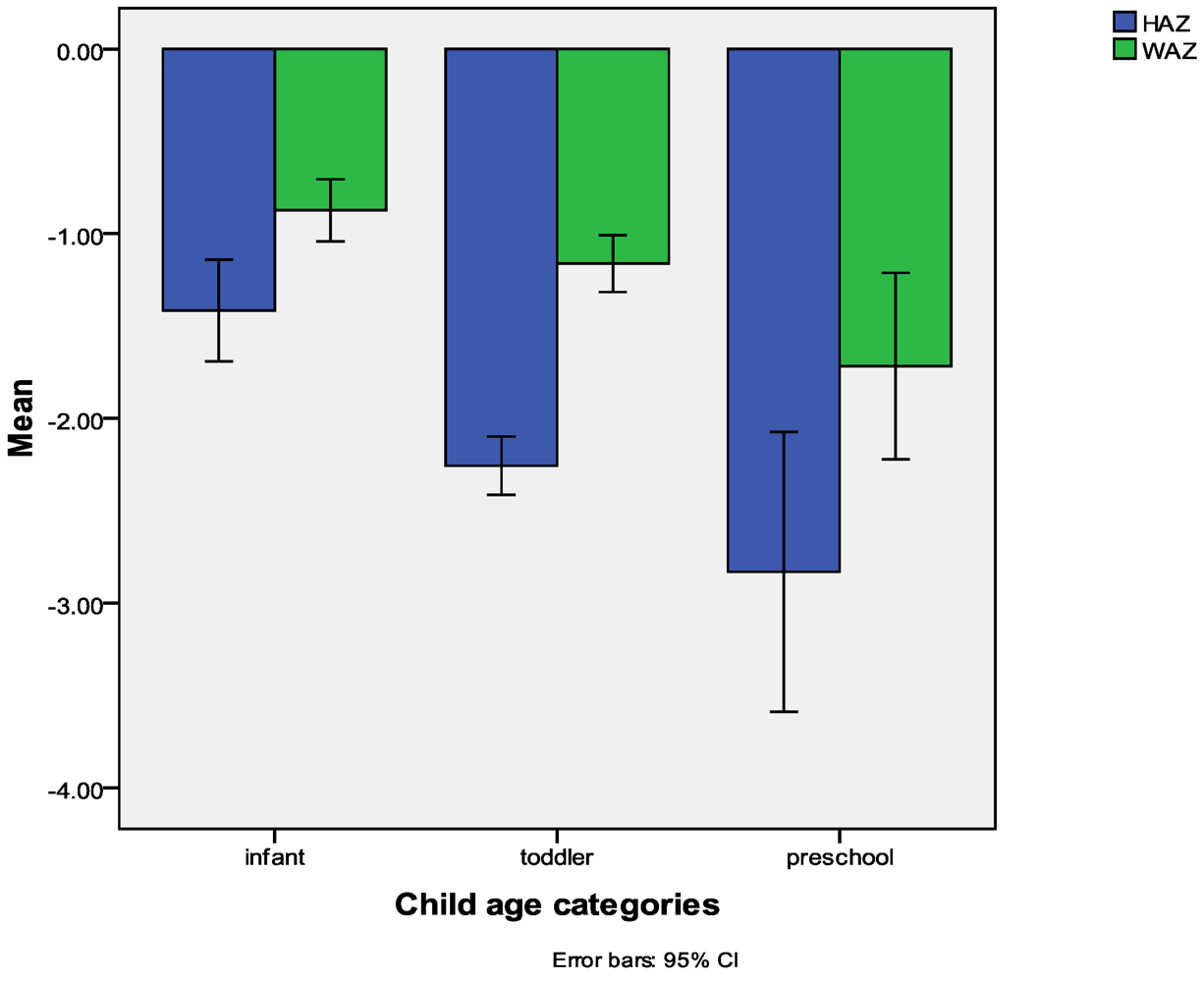
3.1. Nutritional Status of the Sample
3.2. Predictors of Stunting
| Factors | B (SE) | Odds Ratio (95% C.I) |
|---|---|---|
| Materials used for building house | 0.29 (0.25) | 1.34 (0.82–2.12) |
| Concerns on child growth | –1.55 (0.37) *** | 0.22 (0.10–0.44) |
| Child’s ill health | –0.34 (0.37) | 0.71 (0.35–1.46) |
| Child age | –1.11 (0.22) *** | 0.33 (0.21–0.51) |
| Maternal education | 0.66 (0.25) ** | 1.94 (1.18–3.18) |
| Breastfed | 0.28 (0.33) | 1.32 (0.69–2.52) |
| Constant | 0.79 (0.74) |
3.3. Predictors of Being Underweight
| Factors | B (SE) | Odds Ratio (95% C.I) |
|---|---|---|
| Concerns on child growth | –2.76 (0.39) *** | 0.06 (0.03–0.14) |
| Child’s ill health | -0.44 (0.45) | 0.64 (0.27–1.55) |
| Maternal education | 0.07 (0.32) | 1.07 (0.56–2.01) |
| Child age | –0.26 (0.26) | 0.76 (0.45–1.28) |
| Maternal age | –0.05 (0.03) | 0.94 (0.89–1.01) |
| Distance to water source | –1.18 (0.37) ** | 0.31 (0.15–0.64) |
| Paternal education | 0.29 (0.32) | 1.32 (0.68–2.54) |
| Number of pregnancies | –0.05 (0.13) | 0.94 (0.74–1.22) |
| Constant | 4.16 (1.26) |
3.4. Predictors of Being Anaemic
4. Discussion
5. Conclusions
Acknowledgments
Conflict of Interest
References
- Black, R.E.; Allen, L.H.; Bhutta, Z.A.; Caulfield, L.E.; de Onis, M.; Ezzati, M.; Mathers, C.; Rivera, J. Maternal and child undernutrition: Global and regional exposures and health consequences. Lancet 2008, 371, 243–260. [Google Scholar]
- de Onis, M.; Blossner, M.; Borghi, E.; Morris, R.; Frongillo, E.A. Methodology for estimating regional and global trends of child malnutrition. Int. J. Epidemiol. 2004, 33, 1260–1270. [Google Scholar] [CrossRef]
- de Onis, M.; Blossner, M.; Borghi, E.; Frongillo, E.A.; Morris, R. Estimates of global prevalence of childhood underweight in 1990 and 2015. JAMA 2004, 291, 2600–2606. [Google Scholar] [CrossRef]
- Agbere, A.D.; Eklu-Avlasu, E.; Tene Tchele, L.; Parent-Bassuka, A.; Gnamey, K. Slight to moderate malnutrition and hospital morbidity-mortality in children 6-36 months of age in Kara (North Togo). Arch. Pediatr. 1998, 5, 341. [Google Scholar]
- Murray, C.J.; Lopez, A.D. Global mortality, disability, and the contribution of risk factors: Global burden of disease study. Lancet 1997, 349, 1436–1442. [Google Scholar] [CrossRef]
- Victora, C.G.; Adair, L.; Fall, C.; Hallal, P.C.; Martorell, R.; Richter, L.; Sachdev, H.S. Maternal and child undernutrition: Consequences for adult health and human capital. Lancet 2008, 371, 340–357. [Google Scholar]
- Brabin, B.J.; Kalanda, B.F.; Verhoeff, F.H.; Chimsuku, L.H.; Broadhead, R.L. Risk factors for fetal anaemia in a malarious area of Malawi. Ann. Trop. Paediatr. 2004, 24, 311–321. [Google Scholar] [CrossRef]
- Anumudu, C.; Afolami, M.; Igwe, C.; Nwagwu, M.; Keshinro, O. Nutritional anaemia and malaria in preschool and school age children. Ann. Afr. Med. 2008, 7, 11–17. [Google Scholar] [CrossRef]
- Nyakeriga, A.M.; Troye-Blomberg, M.; Chemtai, A.K.; Marsh, K.; Williams, T.N. Malaria and nutritional status in children living on the coast of Kenya. Am. J. Clin. Nutr. 2004, 80, 1604–1610. [Google Scholar]
- Angyo, I.A.; Yakubu, A.M. Lack of association between some risk factors and hepatitis B surface antigenaemia in children with sickle cell anaemia. West Afr. J. Med. 2001, 20, 214–218. [Google Scholar]
- Hokama, T.; Gushi Ken, M.; Nosoko, N. Iron deficiency anaemia and child development. Asia Pac. J. Public Health 2005, 17, 19–21. [Google Scholar] [CrossRef]
- Berkman, D.S.; Lescano, A.G.; Gilman, R.H.; Lopez, S.L.; Black, M.M. Effects of stunting, diarrhoeal disease, and parasitic infection during infancy on cognition in late childhood: A follow-up study. Lancet 2002, 359, 564–571. [Google Scholar] [CrossRef]
- Abubakar, A.; Van Vijver, F.J.R.; Van Baar, A.L.; Kalu, R.; Mbonani, L.; Newton, C.J.R.; Holding, P.A. Socioeconomic status, athropometric status and psychomotor development in a resource limited setting in Kenya: A path analytic study. Early Hum. Dev. 2008, 84, 613–621. [Google Scholar] [CrossRef]
- Grantham-McGregor, S. Linear growth and cognition. Lancet 2002, 359, 542. [Google Scholar] [CrossRef]
- Hasanbegovic, E.; Sabanovic, S. Effects of iron therapy on motor and mental development of infants and small children suffering from iron deficiency anaemia (in Bosnian). Med. Arh. 2004, 58, 227–229. [Google Scholar]
- Semba, R.D.; de Pee, S.; Sun, K.; Sari, K.; Akhter, N.; Bloem, M.W. Effects of parental formal education on risk of child stunting in Indonesia and Bangladesh: A cross-sectional study. Lancet 2008, 371, 322–328. [Google Scholar]
- Walka, H.; Pollitt, E. A preliminary test of a developmental model for the study of undernourished children in Indonesia. Eur. J. Clin. Nutr. 2000, 54, S21–S27. [Google Scholar]
- Agueh, V.D.; Makoutode, M.; Diallo, P.; Soton, A.; Ouendo, E.M. Infant malnutrition and associated maternal factors in a secondary city south of Benin, Ouidah (in French). Rev. Epidemiol. Sante Publique 1999, 47, 219–228. [Google Scholar]
- Sakisaka, K.; Wakai, S.; Kuroiwa, C.; Cuadra Flores, L.; Kai, I.; Mercedes Aragon, M.; Hanada, K. Nutritional status and associated factors in children aged 0-23 months in Granada, Nicaragua. Public Health 2006, 120, 400–411. [Google Scholar] [CrossRef]
- Owusu, W.B.; Lartey, A.; de Onis, M.; Onyango, A.W.; Frongillo, E.A. Factors associated with unconstrained growth among affluent ghanaian children. Acta Paediatr. 2004, 93, 1115–1119. [Google Scholar] [CrossRef]
- Engebretsen, I.M.; Tylleskar, T.; Wamani, H.; Karamagi, C.; Tumwine, J.K. Determinants of infant growth in eastern Uganda: A community-based cross-sectional study. BMC Public Health 2008, 8. [Google Scholar] [CrossRef]
- Getaneh, T.; Assefa, A.; Tadesse, Z. Protein-energy malnutrition in urban children: Prevalence and determinants. Ethiop Med J. 1998, 36, 153–166. [Google Scholar]
- Benson, T.; Shekar, M. Trends and issues in child undernutrition. In Disease and Mortality in Sub-Saharan Africa, 2nd; Jamison, D.T., Feachem, R.G., Makgoba, M.W., Bos, E.R., Baingana, F.K., Hofman, K.J., Rogo, K.O., Eds.; World Bank: Washington, DC, USA, 2006; pp. 87–106. [Google Scholar]
- Zere, E.; McIntyre, D. Inequities in under-five child malnutrition in South Africa. Int. J. Equity Health 2003, 2. [Google Scholar] [CrossRef] [Green Version]
- District Health Management Team, Same District. Comprehensive Council Health Plan 2010/2011; Government Report; Government of Tanzania: Dodoma, Tanzania, 2010.
- Cogill, B. Anthropometric Indicators Measurement Guide; Food and Nutrition Technical Assistance Project, Academy for Educational Development: Washington, DC, USA, 2003. [Google Scholar]
- World Health Organization. WHO anthro v3.01, 2009. WHO: Geneva, Switzerland. Available online: http://www.who.int/childgrowth/en/ (accessed on 7 September 2010).
- Filmer, D.; Pritchet, L.H. Estimating wealth effects without expenditure data-Or tears: An application to educational enrollments in states of india. Demography 2001, 38, 115–132. [Google Scholar]
- Vyas, S.; Kumaranayake, L. Constructing socio-economic status indices: How to use principal components analysis. Health Pol. Plann. 2006, 21, 459–468. [Google Scholar] [CrossRef]
- de Onis, M.; Garza, C.; Victora, C.G.; Onyango, A.W.; Frongillo, E.A.; Martines, J. The WHO multicentre growth reference study: Planning, study design, and methodology. Food Nutr. Bull. 2004, 25, S15–S26. [Google Scholar]
- de Onis, M.; Monteiro, C.; Akre, J.; Clugston, G. The worldwide magnitude of protein-energy malnutrition: An overview from the who global database on child growth. Bull. World Health Org. 1993, 71, 703–712. [Google Scholar]
- World Health Organization, Haemoglobin Concentrations for the Diagnosis of Anaemia and Assessment of Severity. Vitamin and Mineral Nutrition Information Cystem; WHO: Geneva, Switzerland, 2011.
- Bloss, E.; Wainaina, F.; Bailey, R.C. Prevalence and predictors of underweight, stunting, and wasting among children aged 5 and under in western Kenya. J. Trop. Pediatr. 2004, 50, 260–270. [Google Scholar] [CrossRef]
- Magalhães, R.J.S.; Clements, A.C.A. Mapping the risk of anaemia in preschool-age children: The contribution of malnutrition, malaria, and helminth infections in west Africa. PLoS Med 2011, 8. [Google Scholar] [CrossRef]
- Ali, N.S.; Zuberi, R.W. The relationship of socio-demographic factors with iron deficiency anaemia in children of 1-2 years of age. J. Pak. Med. Assoc. 2001, 51, 130–132. [Google Scholar]
- Choi, H.J.; Lee, H.J.; Jang, H.B.; Park, J.Y.; Kang, J.H.; Park, K.H.; Song, J. Effects of maternal education on diet, anaemia, and iron deficiency in Korean school-aged children. BMC Public Health. 2011, 16. [Google Scholar] [CrossRef]
- Glascoe, F.P. Parents’ concerns about children’s development: Prescreening technique or screening test? Pediatrics 1997, 99, 522–528. [Google Scholar] [CrossRef]
- Glascoe, F.P.; Dworkin, P.H. The role of parents in the detection of developmental and behavioral problems. Pediatrics 1995, 95, 829–836. [Google Scholar]
- Cochrane, S.H.; Leslie, J.; O’Hara, D.J. Parental education and child health: Intracountry evidence. Health Policy Educ. 1982, 2, 213–250. [Google Scholar] [CrossRef]
- Gurung, G. Investing in mother’s education for better maternal and child health outcomes. Rural Remote Health 2010, 10, 1352. [Google Scholar]
- Tatala, S.R.; Kihamia, C.M.; Kyungu, L.H.; Svanberg, U. Risk factors for anaemia in schoolchildren in Tanga Region, Tanzania. Tanzan. J. Health Res. 2008, 10, 189–202. [Google Scholar]
- Kahigwa, E.; Schellenberg, D.; Sanz, S.; Aponte, J.J.; Wigayi, J.; Mshinda, H.; Alonso, P.; Menendez, C. Risk factors for presentation to hospital with severe anaemia in Tanzanian children: A case-control study. Trop. Med. Int. Health 2002, 7, 823–830. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Abubakar, A.; Uriyo, J.; Msuya, S.E.; Swai, M.; Stray-Pedersen, B. Prevalence and Risk Factors for Poor Nutritional Status among Children in the Kilimanjaro Region of Tanzania. Int. J. Environ. Res. Public Health 2012, 9, 3506-3518. https://doi.org/10.3390/ijerph9103506
Abubakar A, Uriyo J, Msuya SE, Swai M, Stray-Pedersen B. Prevalence and Risk Factors for Poor Nutritional Status among Children in the Kilimanjaro Region of Tanzania. International Journal of Environmental Research and Public Health. 2012; 9(10):3506-3518. https://doi.org/10.3390/ijerph9103506
Chicago/Turabian StyleAbubakar, Amina, Jacqueline Uriyo, Sia E. Msuya, Mark Swai, and Babill Stray-Pedersen. 2012. "Prevalence and Risk Factors for Poor Nutritional Status among Children in the Kilimanjaro Region of Tanzania" International Journal of Environmental Research and Public Health 9, no. 10: 3506-3518. https://doi.org/10.3390/ijerph9103506




