Quantitative Variations of Intracellular Microcystin-LR, -RR and -YR in Samples Collected from Four Locations in Hartbeespoort Dam in North West Province (South Africa) During the 2010/2011 Summer Season
Abstract
:1. Introduction
- Extract and identify intracellular microcystin congeners from algal blooms collected from four sites on the dam accessible by the public either directly or indirectly (by boat).
- Quantify intracellular microcystin congeners using the LC-ESI-MS technique and determine their spatial distribution with respect to locations (sites), time and MC type (congener).
- Establish an MC congener dominance profile for water-quality assessment with respect to the use of Hartbeespoort Dam water resources.
2. Materials and Methods
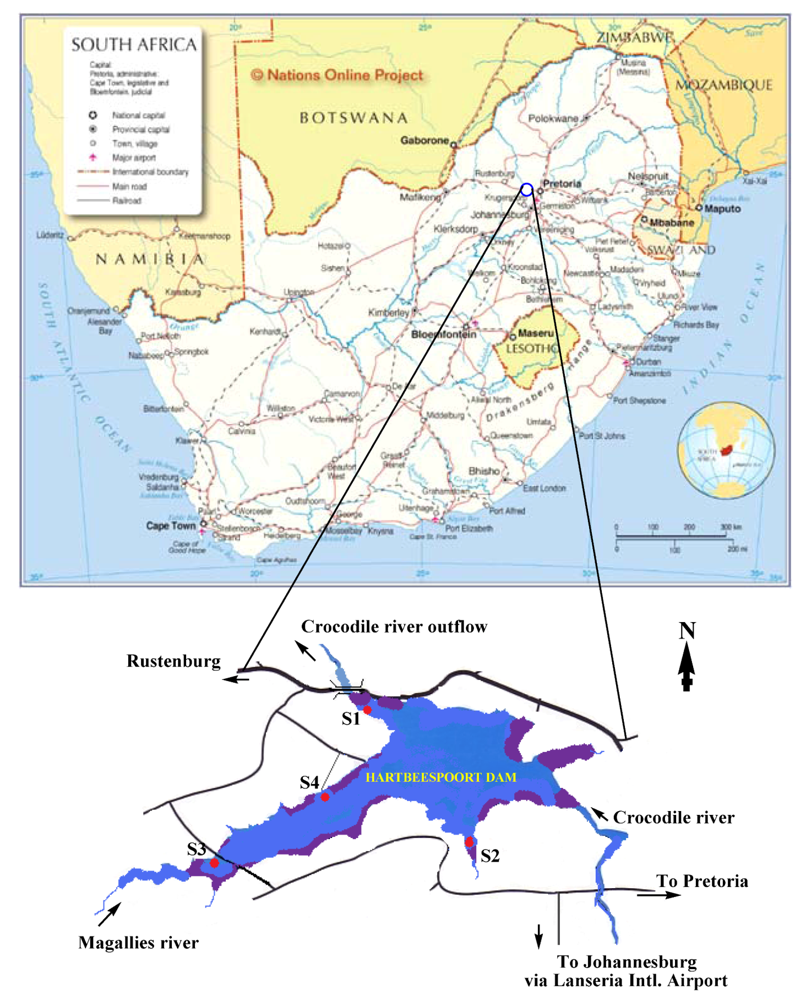
2.1. Sample Collection
- Areas that can easily be reached either by boat or by walking along the banks of the dam (S1–S4).
- Relative distance to fishing/conservation area hotspots (S2–S4).
- Proximity to Magalies River inflow and Crocodile River outflow to/from the dam (S3 and S1, respectively).
- Proximity to the animal conservation area (zoo) and water purification station (S1).
2.2. Sample Pre-Treatment for Microcystin Extraction
2.3. Physicochemical Parameters and Species Identification
2.4. Analytical Standards, Reagents and Laboratory Materials
2.5. LC-ESI-MS Instrumentation and Conditions
2.6. Microcystin Analysis and Quantification
3. Results and Discussion
3.1. Site Selection
3.2. Microcystis spp. Identification
3.3. On-Site Environmental Conditions
3.4. Isolation, Separation and Identification of MCs using LC-ESI-MS
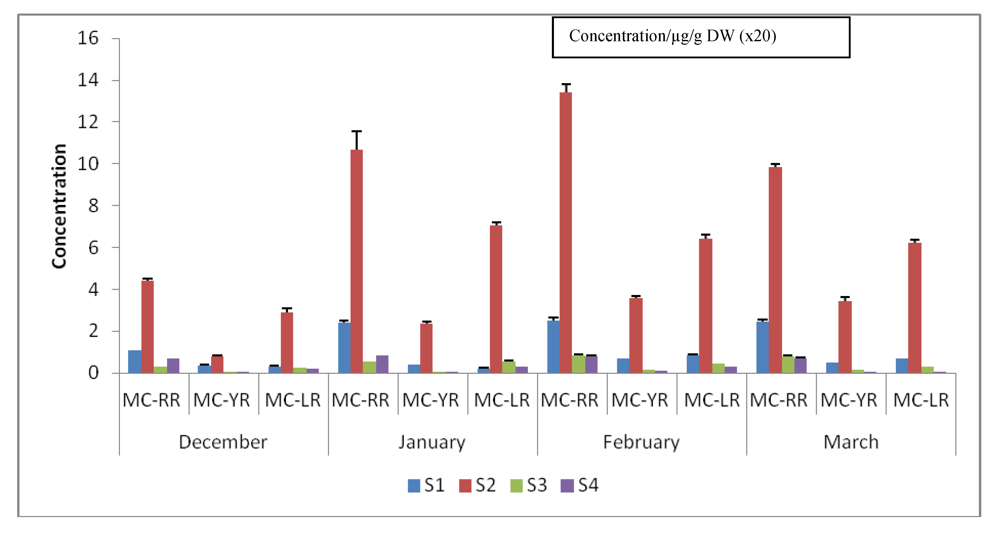
3.5. Quantification and Quantitative Variations of Intracellular MC-LR, -RR and -YR
| Month | Site | Temp(°C) | Surface water temperature (avg. °C) | Conductivity (µS/cm) | Dissolved oxygen (mg/L) | pH |
|---|---|---|---|---|---|---|
| S1 | 24.6 | 578.9 ± 0.77 | 8.6 ± 0.35 | 8.4 ± 0.07 | ||
| December | S2 | 25.2 | 24.8 | 586.3 ± 0.92 | 8.4 ± 0.28 | 8.2 ± 0.07 |
| 2010 | S3 | 24.8 | 568.3 ± 1.55 | 7.7 ± 0.21 | 8.7 ± 0.07 | |
| S4 | 24.7 | 561.9 ± 0.56 | 7.3 ± 0.21 | 8.5 ± 0.14 | ||
| S1 | 26.2 | 562.6 ± 2.47 | 7.3 ± 0.28 | 8.2± 0.07 | ||
| January | S2 | 25.8 | 25.9 | 559.7 ± 1.13 | 7.8 ± 0.21 | 7.9 ± 0.07 |
| 2011 | S3 | 25.4 | 558.9 ± 0.98 | 7.0 ± 0.14 | 8.4 ± 0.14 | |
| S4 | 26.2 | 578.3 ± 1.2 | 7.3 ± 0.21 | 8.0 ± 0.07 | ||
| S1 | 25.2 | 555.3 ± 2.05 | 6.0 ± 0.35 | 7.8 ± 0.14 | ||
| February | S2 | 25.7 | 25.3 | 549.3 ± 0.64 | 3.4 ± 0.35 | 7.4 ± 0.07 |
| 2011 | S3 | 25.1 | 561.9 ± 1.69 | 3.8 ± 0.14 | 8.5 ± 0.14 | |
| S4 | 25.3 | 554.9 ± 2.62 | 5.1 ± 0.28 | 8.3 ± 0.21 | ||
| S1 | 24.7 | 564.3 ± 0.64 | 6.2 ± 0.21 | 8.1 ± 0.14 | ||
| March | S2 | 24.2 | 24.5 | 559.2 ± 1.48 | 3.3 ± 0.21 | 8.2 ± 0.14 |
| 2011 | S3 | 24.9 | 560.5 ± 0.98 | 3.2 ± 0.14 | 8.7 ± 0.07 | |
| S4 | 24.4 | 559.7 ± 1.13 | 3.5 ± 0.14 | 8.8 ± 0.21 |
| MC-Congener | Site S1 | Site S2 | ||||||
|---|---|---|---|---|---|---|---|---|
| Dec | Jan | Feb | Mar | Dec | Jan | Feb | Mar | |
| MC-RR | 21.96 ± 0.16 | 45.7 ± 2.9 | 50.46 ± 2.66 | 49.56 ± 1.62 | 88.56 ± 1.84 | 213.78 ± 17.76 | 268.16 ± 8.06 | 197.2 ± 2.87 |
| MC-YR | 7.24 ± 0.48 | 8.02 ± 0.4 | 14.12 ± 0.13 | 9.74 ± 0.58 | 15.78 ± 0.72 | 47.2 ± 1.58 | 71.22 ± 2.74 | 69.12 ± 1.4 |
| MC-LR | 6.46 ± 0.86 | 4.16 ± 0.54 | 17.26 ± 0.28 | 13.48 ± 0.52 | 57.58 ± 3.86 | 140.68 ± 2.88 | 128.66 ± 3.36 | 124.78 ± 3.02 |
| TOTAL MC (µg/g DW) | 35.66 ± 1.5 | 57.88 ± 3.84 | 81.84 ± 3.07 | 72.78 ± 2.72 | 161.92 ± 6.42 | 401.68 ± 22.42 | 468.04 ± 13.16 | 391.1 ± 7.29 |
| % MC-RR | 61.58 | 78.95 | 61.65 | 68.09 | 54.69 | 53.22 | 57.29 | 50.42 |
| % MC-LR | 18.11 | 7.18 | 21.08 | 18.52 | 35.56 | 35.02 | 27.49 | 31.9 |
| % MC-YR | 20.3 | 13.86 | 17.25 | 13.38 | 9.75 | 11.76 | 15.22 | 17.67 |
| MC-Congener | Site S3 | Site S4 | ||||||
| Dec | Jan | Feb | Mar | Dec | Jan | Feb | Mar | |
| MC-RR | 5.94 ± 0.16 | 10.68 ± 0.34 | 16.78 ± 1.1 | 15.9 ± 0.94 | 13.78 ± 0.38 | 17.12 ± 0.2 | 15.98 ± 0.56 | 14.04 ± 0.64 |
| MC-YR | 0.22 ± 0.002 | 0.68 ± 0.04 | 3.34 ± 0.22 | 2.97 ± 0.28 | 0.14 ± 0.02 | 1.54 ± 0.02 | 1.72 ± 0.04 | 1.34 ± 0.06 |
| MC-LR | 5.24 ± 0.1 | 11.34 ± 0.18 | 9.12± 0.08 | 6.002 ± 0.2 | 3.78 ± 0.18 | 6.32 ± 0.08 | 5.82 ± 0.46 | 0.38 ± 0.04 |
| TOTAL MC (µg/g DW) | 11.4 ± 0.26 | 22.7 ± 0.56 | 29.24 ± 1.4 | 24.87 ± 1.42 | 17.7 ± 0.58 | 24.98 ± 0.015 | 23.52 ± 1.06 | 15.76 ± 0.74 |
| % MC-RR | 52.10 | 47.05 * | 57.38 | 63.93 | 77.85 | 68.53 | 67.94 | 89.08 |
| % MC-LR | 45.96 | 49.95 * | 31.19 | 24.13 | 21.56 | 25.3 | 24.74 | 2.41 |
| % MC-YR | 1.93 | 2.99 | 11.23 | 11.94 | 0.79 | 6.16 | 7.31 | 8.5 |

| Entity | DF | Sum | Sq mean | Sq F value | Pr (>F) |
|---|---|---|---|---|---|
| MC | 2 | 50.145 | 25.072 | 15.0204 | 1.806e−05 * |
| Site | 3 | 261.023 | 87.008 | 52.1246 | 3.511e−13 * |
| MC:Site | 6 | 58.292 | 9.715 | 5.8203 | 0.0002569 * |
| Residuals | 36 | 60.092 | 1.669 | - | - |
3.6. Health Implications of MC-RR Dominance and the Occurrence of (D-Asp3, Dha7)MC-RR Congener
4. Conclusions
Acknowledgements
References
- Carmichael, W.W. Cyanobacteria secondary metabolites—The cyanotoxins. J. Appl. Bacteriol. 1992, 72, 445–459. [Google Scholar] [CrossRef]
- Sivonen, K.; Jones, G. Cyanobacterial Toxins. Reference. In Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences; Monitoring and Management; Chorus, I., Bartram, J., Eds.; E&FN Spon: London, UK, 1999; pp. 55–124, Chapter 3. [Google Scholar]
- WHO, Toxic Cyanobacteria in Water: A Guide to their Public Health Consequences; Monitoring and Management; Chorus, I.; Bartram, J. (Eds.) E&FN Spon: London, UK, 1999; pp. 1–400.
- Anderson, D.M.; Glibert, P.M.; Burkholder, J.M. Harmful algal blooms and eutrophication nutrient sources: Composition and consequences. Estuaries 2002, 25, 704–726. [Google Scholar] [CrossRef]
- Watanabe, M.F.; Oishi, S.; Matsuura, K.; Kawai, H.; Suzuki, M. Toxins contained in Microcystis species of cyanobacteria (blue-green algae). Toxicon 1988, 26, 1017–1025. [Google Scholar] [CrossRef]
- Rinehart, K.L.; Harada, K.; Namikoshi, M.; Chen, C.; Harvis, C.A.; Munro, M.H.G.; Blunt, J.W.; Mulligan, P.; Beasley, E.V.R.; Dahlem, A.M.; Carmichael, W.W. Nodularin, microcystin and the configuration of Adda. J. Am. Chem. Soc. 1988, 110, 8557–8558. [Google Scholar]
- Maizels, M.; Budde, W.A. LC/MS method for the determination of cyanobacteria toxins in water. Anal. Chem. 2004, 76, 1342–1351. [Google Scholar] [CrossRef]
- Sivonen, K.; Namikoshi, M.; Evans, W.R.; Carmichael, W.W.; Sun, F.; Ruhiainen, L.; Luukkainen, R.; Rinehart, K.L. Isolation and characterization of a variety of microcystins from seven strains of cyanobacterial genus Anabaena. Appl. Environ. Microbiol. 1992, 58, 2495–2500. [Google Scholar]
- Rinehart, K.L.; Namikoshi, M.; Choi, B.W. Structure and biosynthesis of toxins from blue-green algae (cyanobacteria). J. Appl. Phycol. 1994, 6, 159–176. [Google Scholar] [CrossRef]
- Metcalf, J.S.; Bell, S.G.; Codd, G.A. Colorimetric immuno-protein phosphatase inhibition assay for specific detection of microcystins and nodularins of cyanobacteria. Appl. Environ. Microbiol. 2001, 67, 904–909. [Google Scholar] [CrossRef]
- Choi, W.B.; Noh, H.Y.; Lee, J.-S. Studies on the structure and biological activity of microcystins produced from Korean cyanobacteria; Microcystis species. J. Korean Ind. Eng. Chem. 1997, 8, 610–616. [Google Scholar]
- Hoeger, S.J.; Schmid, D.; Blom, J.F.; Ernst, B.; Dietrich, D.R. Analytical and Functional Characterization of Microcystins [Asp3]MC-RR and [Asp3, Dhb7]MC-RR: Consequences for Risk Assessment? Environ. Sc. Technol. 2007, 41, 2609–2616. [Google Scholar] [CrossRef]
- Msagati, T.A.M.; Siame, B.A.; Shushu, D.D. Evaluation of methods for isolation, detection and quantification of cyanobacterial hepatotoxins. Aquat. Toxicol. 2006, 78, 282–297. [Google Scholar]
- Fischer, W.J.; Gartwaite, I.; Mile, C.O.; Ross, K.M.; Aggen, J.B.; Chamberlin, A.R.; Towers, N.R.; Dietrich, D.D. Congener-independent immunoassay for microcystins and nodularins. Environ Sci. Technol. 2001, 35, 4849–4856. [Google Scholar] [CrossRef]
- Meriluoto, J.A.O.; Eriksson, J.E.; Harada, K.; Dahlem, A.M.; Sivonen, K.; Carmichael, W. Internal surface reversed-phase high performance liquid-chromatographic separation of the cyanobacterial peptide toxins microcystin-LA; -LR; -YA; -RR and nodularin. J. Chromatogr. 1990, 509, 390–395. [Google Scholar] [CrossRef]
- Lawton, L.A.; Edwards, C.; Codd, G.A. Extraction and high-perfomance liquid chromatography method for the determination of microcystins in raw and treated waters. Analyst 1994, 119, 1525–1530. [Google Scholar] [CrossRef]
- Ward, C.J.; Beattie, K.A.; Lee, E.Y.C.; Codd, G.A. Colorimetric protein phosphatase inhibition assay of laboratory strains and natural blooms of cyanobacteria: Comparisons with high-performance liquid chromatographic analysis for microcystins. FEMS Microbiol. Lett. 1997, 153, 465–473. [Google Scholar] [CrossRef]
- Nicholson, B.C.; Burch, M.D. Evaluation of Analytical Methods for Detection and Quantification of Cyanotoxins in Relation to Australian Drinking Water Guidelines. Available online: http://www.nhmrc.gov.au/_files_nhmrc/publications/attachments/eh22.pdf?q=publications/synopses/_files/eh22.pdf (accessed on 14 May 2012).
- Hummert, C.; Dahlmann, J.; Reinhardt, K.; Dang, H.P.H.; Luckas, B. Liquid chromatography-mass spectrometry identification of microcystins in Microcystis aeruginosa strain from Lake Thanh Cong, Hanoi, Vietnam. Chromatography 2001, 54, 569–575. [Google Scholar] [CrossRef]
- Lawrence, J.F.; Menard, C. Determination of microcystins in blue-green algae, fish and water using liquid chromatography with ultraviolet detection after sample clean-up employing immunoaffinity chromatography. J. Chromatogr. A 2001, 922, 111–117. [Google Scholar] [CrossRef]
- Trojanowicz, M. Chromatographic and capillary electrophoretic determination of microcystins. J. Sep. Sci. 2010, 33, 337–359. [Google Scholar] [CrossRef]
- Harding, W.R.A. Research Strategy for the Detection and Management of Algal Toxins in Water Sources. In WRC Report No. TT 277/06; Development of a Research Strategy for the Detection and Management of Algal Toxins in Water Sources in South Africa (WRC Consultancy K8/576); Water Research Commission: Pretoria; South Africa, 2006; pp. 1–60. [Google Scholar]
- Song, L.; Chen, W.; Peng, L.; Wan, N.; Gan, N.; Zhang, X. Distribution and bioaccumulation of microcystins in water columns: A systematic investigation into the environmental fate and the risks associated with microcystins in Meiliang Bay; Lake Taihu. Water Res. 2007, 41, 2853–2864. [Google Scholar] [CrossRef]
- Van Ginkel, C.E. A National Survey of the Incidence of Cyanobacterial Blooms and Toxin Production in Major Impoundments. In Internal Report No. N/0000/00/DEQ/0503. Resource Quality Services; Department of Water Affairs and Forestry: Pretoria, South Africa, 2004. [Google Scholar]
- Oberholster, P.J.; Botha, A.-M. Use of remote sensing and molecular markers to detect toxic cyanobacterialhyperscum crust: A case study on Lake Hartbeespoort; South Africa. Afr. J. Biotechnol. 2010, 9, 8791–8799. [Google Scholar]
- Jayatissa, L.P.; Silva, E.I.L.; McElhineynd, J.; Lawton, L.A. Occurrence of toxigeniccyanobacterial blooms in freshwaters of Sri Lanka. System. Appl. Microbiol. 2006, 29, 156–164. [Google Scholar] [CrossRef]
- Von Sperling, E.; Ferreira, A.C.S.; Gomes, L.N.L. Comparative eutrophication development in two Brazilian water supply reservoirs with respect to nutrient concentrations and bacteria growth. Desalination 2008, 226, 169–174. [Google Scholar] [CrossRef]
- Sivonen, K. Effects of light, temperature, nitrate, orthophosphate, and bacteria on growth of and hepatotoxin production by Oscillatoria agardhii strains. Appl. Environ. Microbiol. 1990, 56, 2658–2666. [Google Scholar]
- Rapala, J.; Sivonen, K.; Lyra, C.; Niemela, S.I. Variation of microcystins, cyanobacterial hepatotoxins, in Anabaena spp. as a function of growth stimuli. Appl. Environ. Microbiol. 1997, 63, 2206–2212. [Google Scholar]
- Tsuji, K.; Watanuki, T.; Kondo, F.; Watanabe, M.F.; Nakazawa, H.; Sujuki, M.; Uchida, H.; Harada, K.I. Stability of microcystins from cyanobacteria-IV. Effect of chlorination on decomposition. Toxicon. 1997, 35, 1033–1041. [Google Scholar] [CrossRef]
- Butler, N.; Carlisle, J.C.; Linville, R.; Washburn, B. Microcystins: A Brief Overview of Their Toxicity and Effects, with Special Reference to Fish, Wildlife, and Livestock; Ecotoxicology Program Integrated Risk Assessment Branch Office of Environmental Health Hazard Assessment California Environmental Protection Agency: Sacramento, CA, USA, 2009; pp. 1–17. [Google Scholar]
- Azevedo, S.M.F.O.; Carmichael, W.W.; Jochimsen, E.M.; Rinehart, K.L.; Lau, S.; Shaw, G.R.; Eaglesham, G.K. Human intoxication by microcystins during renal dialysis treatment in Caruaru-Brazil. Toxicol. 2002, 181-182, 441–446. [Google Scholar] [CrossRef]
- Mur, L.R.; Skulberg, O.M.; Utkilen, H. Cyanobacteria in the Environment. In Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences; Monitoring and Management; Chorus, I., Bartram, J., Eds.; E&FN Spon: London, UK, 1999; pp. 25–54. [Google Scholar]
- Arto, A.; Lehtimaki, J.; Mattilai, K.; Eriksson, J.E.; Sivonen, K.; Tapio, T.R.; Drakenberg, T. Solution structure of nodularin. An inhibitor of serine/threonine-specific protein phosphatases. J. Biol. Chem. 1996, 271, 16695–16702. [Google Scholar]
- Metcalf, J.S.; Codd, G.A. Microwave oven and boiling waterbath extraction of hepatotoxins from cyanobacterial cells. FEMS Microbiol. Lett. 2000, 184, 241–246. [Google Scholar] [CrossRef]
- Park, H.D.; Watanabe, M.F.; Harada, K.I.; Suzuki, M.; Hayashi, H.; Okino, T. Seasonal variations of Microcystis species and toxic heptapeptidemicrocystins in Lake Suwa. Environ. Toxicol. Water Qual. 1993, 8, 425–435. [Google Scholar] [CrossRef]
- Fitzgeorge, R.B.; Clatk, S.A.; Keevil, C.W. Reference. In Detection Methods for Cyanobacterial Toxins; Codd, G.A., Jefferies, T.M., Keevil, C.W., Potter, E., Eds.; The Royal Society of Chemistry: London, UK, 1994; pp. 69–74. [Google Scholar]
- Harding, W.R.; Paxton, B.R. Cyanobacteria in South Africa. A Review. In WRC Report No. TT 153/01; Water Research Commission: Pretoria, South Africa, 2001. [Google Scholar]
- Ibelings, B.W.; Chorus, I. Accumulation of cyanobacterial toxins in freshwater “seafood” and its consequences for public health: A review. Environ. Pollut. 2007, 150, 177–192. [Google Scholar]
- Papadimitriou, T.; Kagalou, I.; Stalikas, C.; Pilidis, G.; Leonardos, I.D. Assessment of microcystin distribution and biomagnifications in tissues of aquatic food web compartments from a shallow lake and evaluation of potential risks to public health. Ecotoxicol. 2012, 2, 1155–1166. [Google Scholar]
- Pflugmacher, S.; Wiegand, C.; Beattie, K.A.; Codd, G.A.; Steinberg, C.E.W. Uptake of the cyanobacterialhepatotoxinmicrocystin-LR by aquatic macrophytes. J. Appl. Bot. 1998, 72, 228–232. [Google Scholar]
- Lance, E.; Brient, L.; Bormans, M.; G’erard, C. Interactions between cyanobacteria and gastropods I. Ingestion of toxic Planktothrix agardhii by Lymnaea stagnalis and the kinetics of microcystin bioaccumulation and detoxification. Aquat. Toxicol. 2006, 79, 140–148. [Google Scholar] [CrossRef]
- Prakash, S.; Lawton, L.A.; Edwards, C. Stability of toxigenic Microcystis blooms. Harmful Algae. 2009, 8, 377–384. [Google Scholar] [CrossRef]
- Dai, R.; Liu, H.; Qu, J.; Ru, J.; Hou, Y. Cyanobacteria and their toxins in Guanting reservoir of Beijing, China. J. Haz. Mat. 2007, 153, 470–477. [Google Scholar]
- Oberholster, J.P.; Myburg, G.J.; Govender, D.; Bengis, R.; Botha, A.-M. Identification of toxigenic Microcystis strains after incidents of wild animal mortalities in the Kruger National Park, South Africa. Ecotoxicol. Environ. Saf. 2009, 72, 1177–1182. [Google Scholar] [CrossRef]
- Wicks, R.J.; Thiel, P.G. Environmental factors affecting the production of peptide toxins in floating scums of the cyanobacterium Microcystis aeruginosa in a hypertrophic African reservoir. Environ. Sci. Technol. 1990, 24, 1413–1418. [Google Scholar] [CrossRef]
- Van Ginkel, C.E.; Silberbauer, M.J.; Vermaak, E. The seasonal and spatial distribution of cyanobacteria in South African surface waters. Verh. Internat. Verein. Limnol. 2000, 27, 871–878. [Google Scholar]
- Downing, T.G.; Van Ginkel, C.E. Cyanobacterial Monitoring 1990–2000: Evaluation of SA data. In WRC Report No. 1288/1/04; Water Research Commission: Pretoria, South Africa, 2004; pp. 1–44. [Google Scholar]
- Park, H.-D.; Iwami, C.; Watanabe, M.F.; Harada, K.-I.; Okino, T.; Hayashi, H. Temporal variabilities of the concentrations of intra- and extracellular microcystin and toxic Microcystis species in a hypertrophic lake, Lake Suwa, Japan (1991–1994). Environ. Toxicol. Water Qual. 1998, 13, 61–72. [Google Scholar] [CrossRef]
- Msango, M.G. A Comparative Analysis of the Cytotoxicity of Cyanotoxins using In-Vitro (Cell Culture) and In-Vivo (Mouse) Assays.
- Conti, R.A.L.; Guerrero, J.M.; Regueira, J.M. Levels of microcystins in two Argentinean reservoirs used for water supply and recreation: Differences in the implementation of safe levels. Environ. Toxicol. 2005, 20, 263–269. [Google Scholar] [CrossRef]
- Moreno, M.I.; Herrador, A.; Atencio, L.; Puerto, M.; Gonzalez, G.; Camean, M.A. Differentiation between microcystin contaminated and uncontaminated fish by determination of un-conjugated MCs using an ELISA anti-Adda test based on receiver-operating characteristic curves threshold values: Application to Tinca tinca from natural ponds. Environ. Toxicol. 2009, 26, 45–56. [Google Scholar]
- An, J.; Carmichael, W.W. Use of a colorimetric protein phosphatase inhibition assay and enzyme linked immunosorbent assay for the study of microcystins and nodularins. Toxicon 1994, 32, 1495–1507. [Google Scholar] [CrossRef]
- Fischer, W.J.; Garthwaite, I.; Miles, C.O.; Ross, K.M.; Aggen, J.B.; Chamberlin, A.R.; Towers, N.R.; Dietrich, D.R. Congener-independent immunoassay for microcystins and nodularins. Environ. Sci. 2001, 35, 4849–4856. [Google Scholar] [CrossRef]
- Metcalf, J.S.; Beattie, K.A.; Ressler, J.; Gerbersdorf, S.; Pflugmacher, S.; Codd, G.A. Cross-reactivity and performance assessment of four microcystin immunoassays with detoxication products of the cyanobacterial toxin, microcystin-LR. J. Water Res. Tech-Aqua. 2002, 51, 145–151. [Google Scholar]
- Hoeger, S.J.; Hitzfeld, B.C.; Dietrich, D.R. Occurrence and elimination of cyanobacterial toxins in drinking water treatment plants. Toxicol. Appl. Pharmacol. 2005, 203, 231–242. [Google Scholar] [CrossRef]
- Hoeger, S.J.; Shaw, G.; Hitzfeld, B.C.; Dietrich, D.R. Occurrence and elimination of cyanobacterial toxins in two Australian drinking water treatment plants. Toxicon 2004, 43, 639–649. [Google Scholar] [CrossRef]
- Cardozo, K.H.M.; Carvalho, V.M.; Pinto, E.; Colepicolo, P. Fragmentation of mycosporine-like amino acids by hydrogen/deuterium exchange and electrosprayionisation tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2006, 20, 253–258. [Google Scholar]
- Cameán, A.; Moreno, I.M.; Ruiz, M.J.; Pico, Y. Determination of microcystins in natural blooms and cyanobacterial strain cultures by matrix solid-phase dispersion and liquid chromatography-mass spectrometry. Anal. Bioanal. Chem. 2004, 380, 537–544. [Google Scholar] [CrossRef]
- Kondo, F.; Ikai, Y.; Oka, H.; Ishikawa, N.; Watanabe, M.F.; Watanabe, M.; Harada, K.-I.; Suzuki, M. Separation and identification of microcystins in cyanobacteria by frit-fast atom bombardment liquid chromatography-mass spectrometry. Toxicon 1992, 30, 227–237. [Google Scholar] [CrossRef]
- Bateman, K.P.; Thibault, P.; Douglas, D.J.; White, R.L. Mass spectral analyses of microcystins from toxic cyanobacteria using on-line chromatographic and electrophoretic separations. J. Chromatogr. A 1995, 712, 253–268. [Google Scholar] [CrossRef]
- Messineo, V.; Bogialli, S.; Melchiorre, S.; Sechi, N.; Luglie, A.; Cassidu, P.; Marian, A.M. Cyanobacterial toxins in Italian freshwaters. Limnol. 2009, 39, 95–106. [Google Scholar] [CrossRef]
- R Development Core Team, Reference. In R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2011.
- Department of Water Affairs, Hartbeespoort Government Water Scheme; Department of Water Affairs: Pretoria, South Africa, 1991; pp. 1–6.
- Harding, W.R.; Downing, T.G.; Van Ginkel, C.E.; Moolman, A.P.M. An overview of cyanobacterial research and management in South Africa post-2000. Water SA 2009, 35, 479–484. [Google Scholar]
- Harding, W.R.; Thornton, J.A.; Steyn, G.; Panuska, J.; Morrison, I.R. Hartbeespoort Dam Remediation Project (Phase I). Action Plan (Volume I) Final Report; Department of Agriculture, Conservation, Environment and Tourism Project 58/2003; Department of Agriculture, Conservation, Environment and Tourism of the North West Province Government (DACET, NWP): Mmabatho, South Africa, 2004; pp. 1–158. [Google Scholar]
- Steyn, D.J.; Toerien, D.F. Eutrophication levels of some South African impoundments. II. Hartbeespoort Dam. Water SA 1975, 1, 93–101. [Google Scholar]
- . Department of Water Affairs and Forestry, HartbeespoortDam Integrated Biological Remediation Programme; Department of Water Affairs and Forestry: Pretoria, South Africa, 2007; pp. 1–18.
- Falconer, I.R.; Humpage, A.R. Health risk assessment of cyanobacterial (blue-green algal) toxins in drinking water. Int. J. Environ. Res. Public Health 2005, 2, 43–50. [Google Scholar] [CrossRef]
- Hötzel, G.; Croome, R. A Phytoplankton Methods Manual for Australian Freshwaters; LWRRDC Occasional Paper 22/99; Land and Water Resources Research and Development Cooperation: Canberra, Australia, 1999; pp. 1–58. [Google Scholar]
- Owuor, K.; Okonkwo, J.; Van Ginkel, C.; Scott, W. Environmental Factors Affecting the Persistence of Toxic Phytoplankton in the Hartbeespoort Dam; WRC Report No. 1401/3/07; Water Research Commission: Pretoria, South Africa, 2007. [Google Scholar]
- Haarhoff, J.; Langenegger, O.; Van der Merwe, P.J. Practical aspects of water treatment plant design for a hypertrophic impoundment. Water SA 1992, 18, 27–36. [Google Scholar]
- Jacoby, J.M.; Collier, D.C.; Welch, E.B.; Hardy, J.; Crayton, M. Environmental factors associated with a toxic bloom of Microcystis aeruginosa. Can. J. Fish. Aquat. Sci. 2000, 57, 231–240. [Google Scholar] [CrossRef]
- Jones, S.B.; Jones, J.R. Seasonal variation in cyanobacterial toxin production in two Nepalese lakes. Verh. Internat. Verein. Limnol. 2002, 28, 1017–1022. [Google Scholar]
- Dziallas, C.; Grossart, H.-P. Microbial interactions with the cyanobacterium Microcystis aeruginosa and their dependence on temperature. Mar. Biol. 2012. [Google Scholar]
- Lee, S.J.; Jang, M.-H.; Kim, H.-S.; Yoon, B.-D.; Oh, H.-M. Variation of microcystin content of Microcystis aeruginosa relative to the N:P ratio and growth stage. J. Appl. Microbiol. 2000, 89, 323–329. [Google Scholar] [CrossRef]
- Greenwald, I. The Solubility of Calcium Phosphate I. The Effect of pH and of Amount of Solid Phase. Available online: http://www.jbc.org/content/143/3/703.full.pdf (accessed on 10 June 2012).
- Shapiro, J. Blue-green algae in lakes: The role and management significance of pH and CO2. Int. Rev. Gesamten. Hydrobiol. 1990, 69, 765–780. [Google Scholar] [CrossRef]
- Kardinaal, W.E.A.; Visser, P.M. Dynamics of cyanobacterial toxins: Sources of variability in microcystin concentrations. In Harmful Cyanobacteria; Huisman, J., Matthijs, H.C.P., Visser, P.M., Eds.; Springer-Verlag: Berlin, Germany, 2005; pp. 41–63. [Google Scholar]
- Imai, H.; Chang, K.-H.; Kusaba, M.; Nakano, S.-I. Temperature dependent dominance of Microcystis (Cyanophyceae) species: M. aeruginosa and M. wesenbergii. J. Plank. Res. 2009, 31, 171–178. [Google Scholar]
- Amé, M.V.; del Pilar Díaz, M.; Wunderlin, D.A. Occurrence of toxic cyanobacteria bloom in San Roque reservoir (Córdoba, Argentina): A field and chemometric study. Environ. Toxicol. 2003, 18, 192–201. [Google Scholar] [CrossRef]
- Department of Water Affairs. Hartbeespoort Dam Remediation Program—Clear Water State and Algae Concentration. Available online: www.196.3.165.92/hartiesdev/algaesitrep.aspx (accessed on 11 June 2012).
- Namikoshi, M.; Sun, F.; Choi, W.B.; Rinehart, L.K. Seven more microcystins from Homer lake cells: Application of the general method for structure assignment of peptides containing α-β-dehydroamino acid unit(s). J. Org. Chem. 1995, 60, 3671–3679. [Google Scholar] [CrossRef]
- Antoniou, M.G.; Delacruz, A.; Dionysiou, D. Intermediates and reaction pathways from the degradation of microcystin-LR with sulfate radicals. Environ. Sci. Technol. 2010, 44, 7238–7244. [Google Scholar] [CrossRef]
- Spoof, L.; Vesterkvist, P.; Lindholm, T.; Meriluoto, J. Screening for cyanobacterialhepatotoxins, microcystins and nodularin in environmental water samples by reversed-phase liquid chromatography-electrospray ionization mass spectrometry. J. Chromatogr. A 2003, 1020, 105–119. [Google Scholar] [CrossRef]
- Zohary, T. Hyperscum of the cyanobacterium Microcystis aeruginosa in a hypertrophic lake (Hartbeespoort Dam, South Africa). J. Plankton Res. 1985, 7, 399–409. [Google Scholar] [CrossRef]
- Krishnamyrthy, T.; Szafraniec, L.; Hunt, D.F.; Shabanowitz, J.; Yates, J.R.; Hauer, C.R.; Carmichael, W.W.; Skulberg, O.; Codd, G.A.; Missler, S. Structural characterization of toxic cyclic peptides from blue-green algae by tandem mass spectrometry. Proc. Nat. Acad. Soc. USA 1989, 86, 770–774. [Google Scholar] [CrossRef]
- Poon, K.F.; Lam, M.H.W.; Lam, P.K.S.; Wong, B.S.F. Determination of microcystins in cyanobacterial blooms by solid-phase microextraction-high-performance liquid chromatography. Environ. Toxicol. Chem. 2001, 20, 1648–1655. [Google Scholar]
- Phelan, R.R.; Downing, T.G. Optimization of laboratory scale production and purification of microcystin-LR from pure cultures of Microcystis aeruginosa. Afr. J. Biotechnol. 2007, 6, 2451–2457. [Google Scholar]
- Downing, T.G. A Model for Environmental Regulation of Microcystin Production by Microcystis. In WRC Report No. 1401/1/07; Water Research Commission: Pretoria, South Africa, 2007; pp. 1–69. [Google Scholar]
- Briand, E.; Gugger, M.; François, J.-C.; Bernard, C.; Humbert, J.-F; Quiblier, C. Temporal variations in the dynamics of potentially microcystin-producing strains in a bloom-forming Planktothrix agardhii (cyanobacterium) population. Appl. Environ. Microbiol. 2008, 74, 3839–3849. [Google Scholar] [CrossRef]
- Pawlik-Skowrońska, B.; Pirszel, J.; Kornijów, R. Spatial and temporal variations in microcystin concentrations during perennial bloom of Planktothrix agardhii in a hypertrophic lake. Ann. Limnol. Int. J. Lim. 2008, 44, 145–150. [Google Scholar] [CrossRef]
- Indabawa, I.I. Detection of variants of microcystin produced by Microcystis aeruginosa in some burrow pits of Kano, Nigeria. Bajopas. 2009, 2, 189–197. [Google Scholar]
- Sabart, M.; Pobel, D.; Briand, E.; Combourieu, B.; Salençon, M.J.; Humbert, J.F.; Latour, D. Spatiotemporal variations in microcystin concentrations and in the proportions of microcystin-producing cells in several Microcystis aeruginosa populations. Appl. Environ. Microbiol. 2010, 76, 4750–4759. [Google Scholar] [CrossRef] [Green Version]
- Graham, J.L.; Jones, J.R.; Jones, S.B.; Clevenger, T.E. Spatial and temporal dynamics of microcystin in a Missouri Reservoir. Lake Reserve. Manage. 2006, 22, 59–68. [Google Scholar] [CrossRef]
- Latour, D.; Giraudet, H.; Berthon, J.-L. Frequency of dividing cells and viability of Microcystis aeruginosa in sediments of a eutrophic reservoir. Aquat. Microbiol. Ecol. 2004, 36, 117–122. [Google Scholar] [CrossRef]
- Preston, T.; Stewart, W.D.P.; Reynolds, C.S. Bloom-forming Microcystis aeruginosa overwinters on sediment surface. Nature 1980, 288, 365–367. [Google Scholar] [CrossRef]
- Scott, W.E. Occurrence and significance of toxic cyanobacteria in Southern Africa. Water Sci. Technol. 1991, 23, 175–180. [Google Scholar]
- Cazenave, J.; Bistoni, M.A.; Pesce, S.F.; Wunderlin, D.A. Differential detoxification and antioxidant response in diverse organs of Corydora paleatus exposed to microcystin-RR. Aquat. Toxicol. 2006, 76, 1–12. [Google Scholar] [CrossRef]
- Sivonen, K. Cyanobacterial toxins and toxin production. Phycologia 1996, 35, 12–24. [Google Scholar] [CrossRef]
- Ito, E.; Takai, A.; Kondo, F.; Masui, H.; Imanishi, S.; Harada, K. Comparison of protein phosphatase inhibitory activity and apparent toxicity of microcystins and related compounds. Toxicon 2002, 40, 1017–1025. [Google Scholar] [CrossRef]
- Cazenave, J.; Wunderlin, D.A.; Bistoni, M.A.; AME, M.V.; Krause, E.; Pflugmacher, S.; Wiegand, C. Uptake, tissue accumulation and distribution of microcystin-RR in Corydora paleatus, Jenynsia multidentata and Odontesthes bonariensis field and laboratory study. Aquat. Toxicol. 2005, 75, 178–190. [Google Scholar] [CrossRef]
- Osswald, J.; Carvalho, A.P.; Claro, J.; Vasconcelos, V. Effects of cyanobacterial extracts containing anatoxin-a and pure anatoxin-a on early developmental stages of carp. J. Ecotoxicol. Environ. Saf. 2009, 72, 473–478. [Google Scholar] [CrossRef]
- Blom, J.F.; Robinson, J.A.; Jüttner, F. High grazer toxicity of [D-Asp3, (E)-Dha7]MC-RR of Planktothrix rubescens as compared to different microcystins. Toxicon 2001, 39, 1923–1932. [Google Scholar] [CrossRef]
- World Health Organisation (WHO), Guidelines for Safe Recreational Waters. Volume1: Coastal and Fresh Waters; WHO Publishing: Geneva, Switzerland, 2003; p. 136.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mbukwa, E.A.; Msagati, T.A.M.; Mamba, B.B. Quantitative Variations of Intracellular Microcystin-LR, -RR and -YR in Samples Collected from Four Locations in Hartbeespoort Dam in North West Province (South Africa) During the 2010/2011 Summer Season. Int. J. Environ. Res. Public Health 2012, 9, 3484-3505. https://doi.org/10.3390/ijerph9103484
Mbukwa EA, Msagati TAM, Mamba BB. Quantitative Variations of Intracellular Microcystin-LR, -RR and -YR in Samples Collected from Four Locations in Hartbeespoort Dam in North West Province (South Africa) During the 2010/2011 Summer Season. International Journal of Environmental Research and Public Health. 2012; 9(10):3484-3505. https://doi.org/10.3390/ijerph9103484
Chicago/Turabian StyleMbukwa, Elbert A., Titus A.M. Msagati, and Bhekie B. Mamba. 2012. "Quantitative Variations of Intracellular Microcystin-LR, -RR and -YR in Samples Collected from Four Locations in Hartbeespoort Dam in North West Province (South Africa) During the 2010/2011 Summer Season" International Journal of Environmental Research and Public Health 9, no. 10: 3484-3505. https://doi.org/10.3390/ijerph9103484




